3.1 Population Coverage
Once the purposes of surveillance have been established, the next steps are to define the population under surveillance and identify the area of coverage. The programme may be population based or facility or hospital based (see Fig. 3.1). The coverage (geographical area) for a population-based surveillance programme can be a city, a region or an entire country. A population-based programme has a defined source population (typically defined by maternal residence), and all identified congenital anomalies occurring within that source population are ascertained and included, regardless of delivery site.
In contrast, the source population for a hospital-based programme typically cannot be defined accurately. The coverage for a hospital-based surveillance programme is usually at least a few hospitals or clinics in one geographic region. However, there generally are no distinct catchment areas for specific hospitals and, thus, no defined denominator of the entire source population from which all cases are ascertained. Hospital-based programmes work best when they capture most of the population of interest in a geographic region.
Owing to lack of resources or other restrictions, some countries may not find it feasible to start a surveillance programme as a population-based programme and might therefore choose to begin with the development of a facility-based or hospital-based programme. However, it is critical to understand the limitations of a hospital-based programme, and interpret any findings from such a system within those limitations.
Population-based surveillance programmes
Population-based congenital anomalies surveillance programmes collect data from an entire source population (fetuses or neonates with a congenital anomaly and the total number of births) born to resident mothers living in a defined catchment area (geographical area), within a defined time period.
Thus, the denominator used to calculate prevalence in a population-based programme consists of births to resident mothers. The corresponding numerator consists of fetuses or neonates with congenital anomalies born to resident mothers. Because of this definition, all births are collected in a population-based programme, meaning not only births occurring in hospitals or maternity hospitals but also those occurring at home.
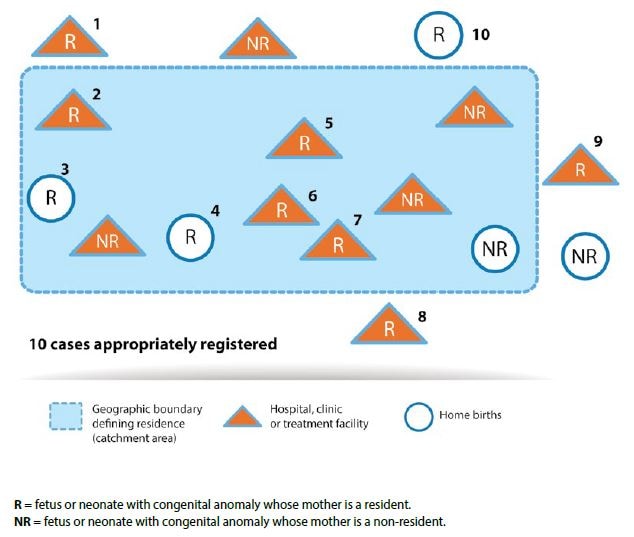
Most congenital anomalies surveillance programmes use the mother’s primary residence at the time of delivery or pregnancy termination to define the source population among which the cases occur. For example, residence can be defined as the mother’s primary address during the 3 months prior to pregnancy and the first trimester of pregnancy. However, the crucial issue is that the definition of resident status used for cases (the numerator) must be the same as that used for all births (the denominator). Fig. 3.2 illustrates an example of a population-based programme. All fetuses or neonates identified with a congenital anomaly born to mothers residing within the catchment area (dashed area) are included in the programme (labels 2 through 7 in Fig. 3.2 ). Similarly, a fetus or neonate with a congenital anomaly who is born outside of the defined catchment area (including one who is born at home while the mother is visiting a family member living outside of the catchment area, for example) would still be included if the mother is herself a resident of the catchment area ( labels 1, 8, 9 and 10 in Fig. 3.2 ). Fetuses or neonates identified with congenital anomalies and born to non-resident mothers are not included. Data sources include all health facilities within the catchment area where births occur, vital records (e.g. birth and death registries), referral treatment centres for individuals with congenital anomalies (up to the defined age period for inclusion), administrative databases, and any health-care facility that identifies a fetus or neonate with a congenital anomaly.
The steps for calculating prevalence, and information on how to define the denominator, are described later on in this chapter.
Hospital-based surveillance programmes
Hospital-based congenital anomalies surveillance programmes capture all pregnancy outcomes with congenital anomalies that occur in selected hospitals in a defined geographic area (e.g. a state, province or county).
The denominator used to estimate prevalence in a hospital-based programme consists of births occurring in the participating hospitals. The numerator (cases) typically consists of affected live births and stillbirths occurring in these hospitals. Fetuses or neonates with congenital anomalies who are delivered at home are not included, even if they are identified and captured in participating hospitals (because they are not part of the denominator).
Because the inclusion in a hospital-based programme depends on where the birth occurred rather than on the residence at birth, the source population of cases is difficult to establish. This becomes an issue in the surveillance of congenital anomalies when referral patterns skew the likelihood that an affected fetus or neonate is delivered at a hospital in the system. Thus, a major concern in hospital-based programmes is referral bias of cases – that is, the selective delivery of affected pregnancies in hospitals participating in the hospital-based programme. This referral bias can also vary over time, either because referral patterns change or because hospitals are added or removed from the surveillance programme. This, in turn, adds to the problem of using these hospital-based data longitudinally for monitoring.
Such hospital-based programmes typically collect data on live births and stillbirths. Because neonates are discharged from maternity hospitals within days following birth, hospital-based programmes typically capture only those congenital anomalies that are evident during the hospital stay, unless those readmitted to the hospital for surgery or other procedures are captured. Note that fetuses or neonates diagnosed after delivery in a hospital participating in a hospital-based programme are not included for the purposes of surveillance, unless they were also delivered at a participating site in a hospital-based programme.
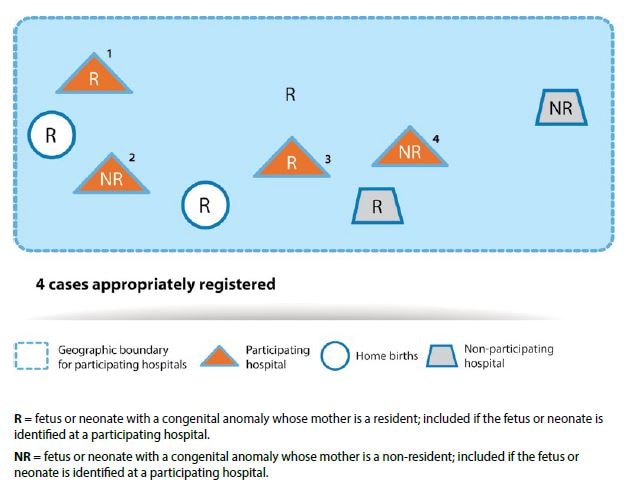
For illustration, Fig. 3.3 presents an example of participating and non-participating hospitals in a hospital-based surveillance programme. All fetuses or neonates with congenital anomalies born to mothers in participating hospitals, regardless of maternal residency, are included in the programme ( labels 1 through 4 in Fig. 3.3). Fetuses or neonates with congenital anomalies born to resident mothers but born outside of a participating hospital or at home are not included. Fetuses or neonates with congenital anomalies born to non-resident mothers are included if they are born in a participating hospital.
Ascertainment of fetuses or neonates identified with congenital anomalies in participating hospitals can vary. While some are primary hospitals, others may be specialized centres for certain conditions, or for prenatal diagnosis and care, and serve as referral hospitals for patients outside the catchment area. As discussed, such hospitals would disproportionately serve fetuses or neonates with congenital anomalies, thus introducing bias in the calculation of their birth prevalence.
The magnitude of bias may change over time, with fluctuations in referral patterns and the proportion of births occurring outside the hospital setting. This could lead to changes in rates that have nothing to do with the underlying prevalence, but are the result of referral patterns. Also, the bias will depend on how many hospitals or facilities are included – all, half or only a small percentage.
Estimates of birth outcomes with congenital anomalies in hospital-based surveillance programmes represent only those births at reporting hospitals in which data are collected. The prevalence estimates could, therefore, be biased, particularly if the hospital births are a minority of all births, if they receive a high proportion of difficult or complicated pregnancies, and/or if they are not representative of the population of interest. Bias limits the representativeness and usefulness of the data for surveillance. However, if nearly all hospitals in a country participate in the surveillance programme and nearly all births occur in hospitals, the surveillance programme may approximate a population-based surveillance programme.
Finally, a subset of facility-based or hospital-based surveillance known as sentinel surveillance is generally set up in key sites, to obtain rapid estimates of the occurrence of a birth outcome. Because congenital anomalies are relatively rare events, sentinel surveillance programmes may not be very effective for capturing them.
Although population-based and hospital-based surveillance programmes have clear differences, there are some characteristics that are common to both. These include:
- participating clinicians can be motivated “champions” – leaders who are committed to the programme;
- data collected are useful for documenting that a problem may exist;
- data collected are useful for alerting health and government officials to the need for investing further in possible causes and promoting prevention strategies;
- affected children can be referred to services; and
- high-quality case data – including data on potential risk factors, can be generated.