July 2020 LTCF Newsletter
Welcome to NHSN’s LTCF Component!
Now that you’ve been briefly introduced to NHSN, let’s take a brief tour through the NHSN LTCF Component. The LTCF component of NHSN was launched in 2012 to provide LTCFs with a resource to systematically collect data regarding infections, communicable diseases, influenza vaccination among healthcare personnel safety, and prevention process measures.
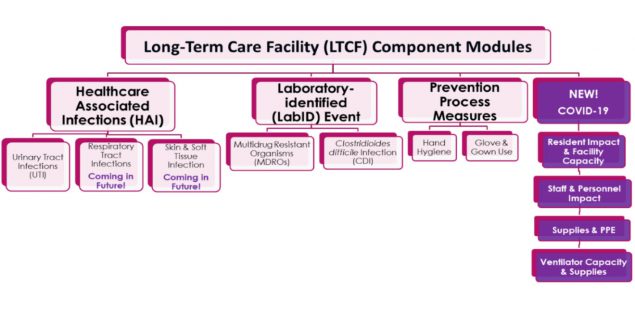
The Component consists of four modules, including the healthcare-associated infections (HAI) module, laboratory-identified (LabID) event module, prevention process measures module, and most recently, the COVID-19 module. The below diagram provides a snapshot of current and future reporting options for NHSN LTCF Component users. If you are interested in learning more about the Component, please visit our LTCF webpage.
In April 2020, in support the nation’s COVID-19 response, NHSN released the LTCF COVID-19 Module within the LTCF Component to supplement COVID-19 reporting. This module includes four pathways for understanding and prioritizing public health action in relation to resident impact and facility capacity, staff and personnel impact, supplies and personal protective equipment, and ventilator usage and supplies.
With over 14,000 newly enrolled LTCFs since late April, the NHSN LTCF team recognizes an increased commitment to support our users. The goal of the LTCF newsletter is to provide an additional channel for communication and support. Newsletters are released quarterly, with each providing important updates and other information critical for the successful use of NHSN.
Did You Receive A COVID-19 Noncompliance Letter From CMS?
The reporting requirements for nursing homes became effective on May 8, 2020, when CMS published new requirements [PDF – 400 KB] for long-term care facilities (LTCFs) to report COVID-19 facility data to the Centers for Disease Control and Prevention (CDC) National Healthcare Safety Network (NHSN) on a weekly basis (at least every 7 days).
If you have received a non-compliance letter from CMS, we encourage you to verify the following:
- First, verify that your CMS Certified Number (CCN), also referred to as federal provider number listed in NHSN is correct. CMS identifies data submitted with the CCN listed in the facility profile. Please refer to this look-up tool and select “Basic Search” to the left under “Tools” to look up the correct CCN for the facility. If you find that your CCN in NHSN is incorrect, please use this guidance document [PDF – 1 MB] to either add or correct your CCN in the NHSN application
- Second, verify that you enrolled your LTCF using the correct Facility Type (skilled nursing/nursing home, assisted living, or developmentally disabled). Your facility type will show when using the CCN look-up tool described in step 2.
- Be sure to manually enter or submit (if CSV upload) data for the week by Sunday at 11:59 pm.
- Please visit the Frequently Asked Questions [PDF – 200 KB] document
- If you have specific questions about CMS compliance, fines, etc., please contact CMS enforcement directly DNH_Enforcement@cms.hhs.gov. Questions regarding to how the data is displayed on the CMS website should be directed to CMS at NH_COVID_Data@cms.hhs.gov
As a reminder when viewing your data…
Be sure you are looking at the correct time frame since data on the NHSN online application are current and there is an approximate 11-day delay on the CMS website.
What’s New to the LTCF COVID-19 Module?
The NHSN team appreciates all of the feedback we’ve received from our users. We are working hard making updates to incorporate your feedback, while also continuously improving the usability of the NHSN application.
Here’s what’s new since the launch of the COVID-19 Module, beginning with the most recent updates:
- The NEWLY developed NHSN COVID-19 Dashboard for LTCFs is NOW available for groups and facilities. The dashboard provides a summary of data entered into the COVID-19 Module and includes an interactive chart display where a user may customize data views. While this first edition is focused on the Resident Impact and Facility Capacity pathway, we will continue to add additional pathways and analysis options. Guidance documents for groups and facilities to assist with navigating and understanding the features are available on the NHSN LTCF COVID-19 website
- You asked, and we listened! We recognize that many SNFs are setting up COVID-19 units to receive transfers from other LTCFs, such as Assisted Living Residences. These transfers result in increased demand on the receiving LTCFs. Therefore, the count for “Admissions” now includes resident admissions and readmissions who were previously diagnosed with COVID-19 from another facility. For additional information, please review the form [PDF – 100 KB] and accompanying instructions for the Resident Impact and Facility Capacity pathway [PDF – 350 KB]. LTCFs may update previously entered data but are not required.
- Pop-up alerts to remind users of potential data entry errors. For example, if a user attempts to enter more COVID-19 Deaths than Total Deaths, a pop-up alert will appear to remind user of the definitions. As a reminder, COVID-19 Deaths counts must be included in the Total Deaths count.
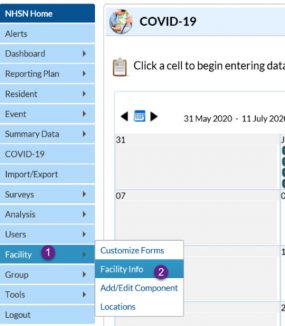
Non-modifiable fields have been added to each of the four pathway screens. You will notice that each pathway now has three additional pieces of information displayed at the top of each pathway: Create Date, CCN, and Facility Type. The purpose of Create Date is to display the first date of data entry (manual or CSV file upload) for a selected calendar date and pathway. The date will automatically save and cannot be modified by the user.
The CCN and Facility Type will also populate for each facility, allowing users to quickly verify the information associated with the data being submitted to CMS. These two variables may be edited by a facility user with NHSN Administrative rights; however, edits must be done outside of the COVID-19 Module by accessing Facility Info on the left navigation panel and following the instructions for editing CCN [PDF – 1 MB] and changing facility type [PDF – 300 KB]. These guidance documents are available under Facility Resources on the LTCF COVID-19 webpage.
- In early May, the question for assessing ventilator availability in the LTCF was updated to include “ventilator dependent units” and “ventilator dependent beds.” This change was in recognition that some LTCFs have ventilators available, but not necessarily a ventilator only unit or dedicated bed.
NOTE: LTCFs should only answer “yes” to “Do you have ventilator dependent unit(s) and/or beds in your facility?” if the LTCF has ventilators that are available for use on residents while remaining in the LTCF.
- Taking into consideration the additional burden associated with collecting and reporting retrospective counts, another change in early May was how retrospective counts from January 1, 2020 to April 30, 2020 were reported. Instead of including those retrospective, aggregate counts in with the first date of data entry in the COVID-19 module, users were instructed to enter those counts by selecting a date on the calendar prior to May 1. Reporting of these retrospective counts are encouraged, but not required. There is not a deadline for reporting these retrospective counts, so users may still enter counts that occurred prior to May 1.
What’s Coming…
As we continue to receive feedback from our LTCF users and learn more about COVID-19 and its impact on vulnerable populations and healthcare workers, the COVID-19 module will evolve.
Although, there may be additional changes, here are a few we know are in the horizon over the next couple of months.
- Expansion of the COVID-19 Dashboard for LTCFs, to include additional pathways and analysis options.
- Additional analysis options, such as line lists, that for facility and group users to allow detailed review of data entered in the COVID-19 Module that will allow correction of errors prior to CMS submission.
- Soft alerts and reminders will be added to the application as a reminder for users entering potentially erroneous counts. The purpose of these alerts is to improve the quality of data being entered and analyzed.
- Additional lab questions will be added to the Facility Impact and Facility Capacity Pathway
The rationale behind these additional questions include:- To become better informed of the limitations of COVID-19 testing capacity in LTCFs so that public health can allocate resources
- Allow public health departments to better interpret facility-specific data on new cases (for example, new confirmed cases in facilities that are not performing point prevalence surveys are likely to underestimate true burden of infection)
- Improved estimate burden and facility comparisons by knowing how widespread testing is in facilities
- Frequently Asked Questions (FAQs) document will be posted on the LTCF COVID-19 webpage in early July.
Please visit our LTCF COVID-19 website often for updated and new guidance documents, training opportunities, and other important updates.
What if My Facility is Enrolled Twice?
A facility should not enroll in NHSN more than once. Each enrolled LTCF should have one NHSN Org ID and only have one CCN affiliated with that Org ID. Facilities enrolled more than once and share the same CCN will not have accurate data recorded and submitted to CMS.
Please follow these steps to withdraw any duplicate facilities:
- Choose one NHSN facility Org ID to submit data and ensure the CCN is correct.
Note: If one of the enrolled facilities is a level 3 facility, meaning level 1 facility users have access to all NHSN data and reporting options, this is the preferred facility to keep. Contact nhsn@cdc.gov and request to be added as a user to the facility with limited rights to the COVID-19 Module (if you do not have an active grid card) - Merge data from any duplicate facilities into the one designated facility from step #1.
- Withdraw duplicate facilities from NHSN by doing the following:
- Log into the duplicate enrolled facility in NHSN.
- On the left navigation pane, select facility->facility info.
- Scroll down to the component itemization and deselect the component that is a duplicate.
- Accept the alert indicating that you’ve deselected the facility.
- Select update to reflect changes.
- Email NHSN@cdc.gov with the subject line “LVL1 duplicate facilities” when the data has been merged into the facility chosen and the duplicate facility has been withdrawn. Be sure to include the following in the email:
- The NHSN Org IDs which you have withdrawn and the NHSN Org ID which you wish to continue using to report COVID-19 data.
- Statement that you have successfully merged the data from the duplicate facility into the facility you will use.
NOTE: This step is very important and failure to follow the instructions will result in duplicate data shared with CMS for your facility.
Updated Guidance Document
Be sure to check-out the UPDATED Facility Guide to Using the COVID-19 Module [PDF – 1 MB], which can also be found on our NHSN LTCF COVID-19 Module Webpage. This guidance is a complete resource for using and reporting into the NHSN LTCF COVID-19 Module.
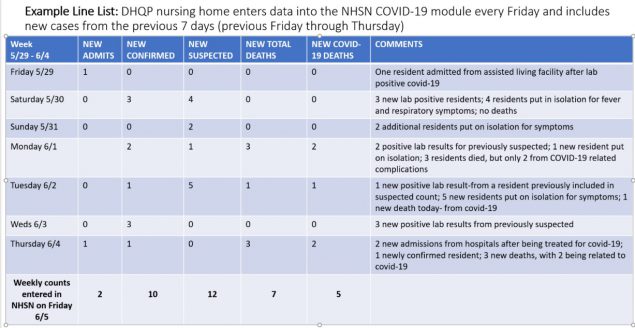
Example Line List for Weekly Reporting of Counts
As a reminder, counts should only include NEW counts. See the below line list as an example.
Frequently Asked Questions
Question: If a resident with COVID-19 is newly admitted to my facility, do I count the resident in the Admissions and Confirmed counts?
Response: NO. Residents with previous, known, or suspected COVID-19, admitted or readmitted from another facility should only be included in the ADMISSIONS count for your LTCF. The purpose of CONFIRMED is to assess potential LTCF onset cases.
Question: Is it ok to go back in and correct incorrect data or data errors?
Response: YES. Facilities are strongly encouraged to correct incorrect and/or data entry errors by clicking on the selected date from the calendar view and then making corrections, followed by clicking the SAVE button.
Question: Do I include new COVID-19 Death counts in the Total Deaths count?
Response: YES. TOTAL DEATHS count must include counts for all new deaths collected during the surveillance and/or reporting time period, including those related to COVID-19. TOTAL DEATHS count should always be greater than the COVID-19 DEATHS count.
Question: Do I have to enter data in the NHSN calendar every day?
Response: NO. LTCFs are encouraged to report data in NHSN as often as possible. However, facilities must report a minimum of once per week (7 days). If data are entered more than once a week, it’s important NOT to include duplicate counts (for example, do not roll over counts from the previous days).
Question: For residents who test positive for COVID-19, how long do I include them in my reported counts for NHSN?
Response: ONCE. CONFIRMED (laboratory positive) counts must represent incidence (new) counts since the last time confirmed counts were collected and/or reported to NHSN (depending if the facility reports weekly or more than once per week). CONFIRMED residents and/or staff should only be counted one time, to represent the date of the positive COVID-19 lab result. Additionally, reported CONFIRMED counts must not be removed from previous counts after a resident and/or staff recover.
Question: Should I enter cumulative counts from January 1, 2020 until the end of April?
Response: Yes. LTCFs are encouraged to enter cumulative counts from January 1, 2020 by selecting a date prior to May 1, 2020 to enter any known cumulative counts (for example, total laboratory positive cases, total COVID-19 death counts, etc.). Prior to June 5, NHSN had a business rule that prevented the user from submitting counts if the total counts were greater than the total census for the date entering the counts.
However, this business rule has been removed. If you attempted to enter retrospective counts and received the error message, please try again now. The only other question that will be required to answer when entering retrospective counts is Testing: Does your facility have access to COVID-19 testing while the resident is in the facility since this is a required question. Since you will not be able to submit the retrospective counts before answering this question, we encourage users to answer to the best of your ability, but do not let not knowing the answer prevent you from entering the facility total retrospective counts.
Question: We isolate all new admissions for 14 days to monitor for COVID-19 signs or symptoms. Should I include all these residents in the “Suspected” count?
Response: NO. Isolation alone does not meet NHSN definition for SUSPECTED. To be included in the SUSPECTED COVID-19 count, residents without laboratory positive results must be managed as though they have COVID-19 due to signs and/or symptoms compatible with COVID-19. See CDC’s Evaluating and Testing Persons for Coronavirus Disease 2019 (COVID-19). Examples include, but are not limited to fever, acute respiratory illness (cough, shortness of breath, difficulty breathing), chills, repeated shaking with chills, muscle pain, new loss of taste or smell, diarrhea, vomiting, headache or sore throat. The definition includes residents who have not been tested or those with pending test results.
Question: Do I still need to report if I have no new cases for the reporting week?
Response: YES. Continue to report in the module since questions include a combination of counts and yes/no responses. If your facility has no new cases to report for a specific question, enter a 0 for that question. This is important since a blank field is equivalent to “missing data” for a pathway.