FAQs: Pediatric Ventilator-Associated Events (PedVAE)
- Lower respiratory tract events
- Pneumonia present on admission or prior to initiation of ventilation and PedVAE surveillance
- Brain injury/ brain death in ventilated patients
- Excluded ventilator modes
- Weaning/mechanical ventilation liberation trials and PedVAE
- Daily minimum values
- Secondary BSI to lower respiratory events in locations performing PedVAE surveillance
- Episode of mechanical ventilation
- Ventilator data
- Home ventilators
- Location of mechanical ventilation
- Date of mechanical ventilation initiation
Lower respiratory tract events
We know this can be an area of confusion. We need to consider events occurring in patients on mechanical ventilation and events occurring in patients NOT on mechanical ventilation, and we have to consider events that occur in adults and events that occur in neonates and in children. Let’s review what is available for in-plan or off-plan surveillance of lower respiratory tract events in NHSN. Keep in mind that “in-plan” surveillance means that you/your facility have committed to following the NHSN surveillance protocol for that particular event in your NHSN monthly reporting plan. “Off-plan” surveillance is surveillance that is done because you/your facility have decided to track a particular event for internal use. Data that are entered into NHSN “off-plan” are not used or reported in NSHN annual reports or other NHSN publications. A facility makes no commitment to follow the protocol for “off-plan” events.
What lower respiratory tract event surveillance can be done “in-plan”?
- VAE: This event is available for in-plan surveillance of mechanically ventilated inpatients in adult locations only (regardless of the age of the patient). Pediatric and neonatal locations are excluded from VAE surveillance (even in circumstances where a pediatric unit may occasionally care for adult patients).
- PedVAP: This event uses the PNEU protocol and is available for in-plan surveillance of mechanically ventilated patients in pediatric locations only (regardless of the age of the patient). In-plan surveillance for ventilator-associated PNEU (VAP) is not available for inpatients in adult or neonatal locations.
- PedVAE: This event is available for in-plan surveillance of mechanically ventilated inpatients in pediatric and neonatal locations only. Adult locations are excluded from PedVAE surveillance (even in circumstances where an adult unit may occasionally care for pediatric patients).
NOTE: When conducting CLABSI surveillance, the PNEU definition is available for use as a site-specific infection to which a bloodstream infection (BSI) can be attributed as a secondary BSI for all patients, all locations, regardless of use of mechanical ventilation. This includes ventilated or non-ventilated adults, children, or neonates in any patient location.
What lower respiratory tract event surveillance can be done “off-plan”?
- VAE: VAE surveillance can be done “off-plan” in mechanically ventilated inpatients in adult locations only.
- PedVAP or PNEU/VAP: Surveillance using the PNEU protocol is available for “off-plan” surveillance in all mechanically ventilated inpatients (adults, children, or neonates) in any inpatient location.
- PedVAE: PedVAE surveillance can be done “off-plan” in mechanically ventilated inpatients in neonatal and pediatric locations only.
- PNEU: PNEU surveillance is available for “off-plan” surveillance in non-mechanically ventilated adults, children, and neonates in any inpatient location.
- LRI: Surveillance for non-pneumonia lower respiratory infections (using LUNG definition) is available for “off-plan” surveillance in mechanically ventilated or non-mechanically ventilated adults, children, and neonates.
Yes. PedVAE and PedVAP are two separate protocols and detect different types of events reported to NHSN.
A pediatric location can conduct simultaneous in-plan PedVAE and in-plan PedVAP (using the PNEU protocol) surveillance, or a neonatal location can conduct simultaneous in-plan PedVAE and off-plan PedVAP surveillance, and a patient can meet one and not the other, meet both, or meet neither. In other words, detection of one type of event (such as a PedVAE) in a particular patient would have no bearing on the conduct of surveillance for the other event type (PedVAP) in the same patient. Patients who meet a PedVAE definition and a PedVAP definition would have two events identified in units where surveillance for multiple respiratory events is occurring.
Pneumonia present on admission or prior to initiation of ventilation and PedVAE surveillance
No. Tracking of daily minimum MAP and FiO2 should be done for all patients who are eligible for PedVAE surveillance in units in which in-plan PedVAE surveillance is being conducted, regardless of the reason for which the patient was admitted or the reason for initiation of mechanical ventilation. Additionally, PedVAE and PNEU (PedVAP) detect two separate events. If a patient meets a PNEU definition, while this establishes a 14-day RIT for subsequent PNEU events, the PNEU RIT does not apply to or account for the identification of PedVAEs.
Brain injury/ brain death in ventilated patients
Brain injury and/or brain death alone is not sufficient for exclusion from PedVAE surveillance. The requirement is that if the PedVAE date of event (date of onset of worsening oxygenation) is on or after the date of documentation in the medical record of evidence of consent for organ donation AND the patient is being supported for organ donation purposes, then the event should not be reported as a PedVAE. If the requirement is not met, the patient is not excluded from surveillance, and a PedVAE should be reported if criteria are met.
Excluded ventilator modes
In some cases, patients may be on ECLS or paracorporeal membrane oxygenation for a portion of a calendar day, but not for the entire calendar day, for example when the support is first initiated or when discontinued. In these instances, the patient is eligible for inclusion in PedVAE surveillance during the portion of the calendar day when the patient was being mechanically ventilated using a conventional mode of mechanical ventilation only (not on ECLS or paracorporeal membrane oxygenation). You should review the FiO2 and MAP data recorded for the portion of the calendar day when the patient was NOT on ECLS or paracorporeal membrane oxygenation to select the daily minimum FiO2 and MAP.
Once the patient has been switched to ECLS or paracorporeal membrane oxygenation they are no longer included in PedVAE surveillance. On calendar days when the patient was on ECLS or paracorporeal membrane oxygenation for the entire day (specifically, from midnight to 11:59 pm), you will not record a daily minimum FiO2 or MAP – you will enter “Not applicable” or “Not eligible for surveillance” in your worksheet column and you will not enter values in the PedVAE calculator for daily minimum FiO2 and MAP for that particular day.
Once the patient has been switched back to a conventional mode of mechanical ventilation, PedVAE surveillance may resume. If the patient has been on ECLS or paracorporeal membrane oxygenation for one or more calendar days (such that there is a gap in recording of the daily minimum FiO2 and MAP), then upon return to a conventional mode of mechanical ventilation you will essentially need to start over with PedVAE surveillance. The patient would need to have at least 2 days of stability or improvement and at least 2 days of worsening oxygenation on the ventilator identified before you can detect a PedVAE.
For example, if the patient was on conventional mechanical ventilation on January 10 until 10:00 am, switched to ECMO at 10:00 am, remained on ECMO until 1:00 pm on January 11 and was then placed back on a conventional mode of mechanical ventilation, you would be able to evaluate the FiO2 and MAP values recorded for the patient from midnight to 10:00 am on January 10 (period on conventional mechanical ventilation) and from 1:00 pm to 11:59 pm on January 11 (period on conventional mechanical ventilation) when looking for PedVAEs. In contrast, if a patient was on ECMO for the entire calendar day on January 10 and January 11, then you would exclude them from PedVAE surveillance during this period. Once the patient returns to conventional mechanical ventilation for some portion of each calendar day you could again include in PedVAE surveillance and once again begin daily assessment for the daily minimum FiO2 and MAP values obtained while the patient was on the conventional mode of ventilation.
Weaning/mechanical ventilation liberation trials and PedVAE
Yes. As long as the patient is receiving support from a mechanical ventilator and is eligible for PedVAE surveillance, then you should review all FiO2 and MAP data that are recorded each day to identify the daily minimum FiO2 and MAP values – including FiO2 and MAP values that are recorded during periods of time when the patient is undergoing spontaneous awakening or spontaneous breathing trials (or other forms of weaning from mechanical ventilation). The only periods of time that are not taken into consideration when identifying the daily minimum FiO2 and MAP values are times when the patient is on ECLS or paracorporeal membrane oxygenation, or times when the patient is not receiving mechanical ventilation support (for example, a T-piece trial or a trach collar trial where the patient continues to receive supplemental oxygen but is receiving no additional support from the mechanical ventilator).
Daily minimum values
Definitions of “daily minimum FiO2” and “daily minimum MAP” can be found in the PedVAE protocol. Please refer to the PedVAE protocol for details. There will be multiple FiO2 settings and MAP readings documented each calendar day on mechanically ventilated patients. These FiO2 and MAP values are typically recorded in the paper or electronic medical record, on respiratory therapy and/or nursing flow sheets, in the section of the flow sheet that pertains to respiratory status/mechanical ventilation. You will make daily minimum value determinations using documented settings and readings specific to the calendar day and independently of the FiO2 settings or MAP readings on the previous calendar day or the next calendar day. Please note that the PedVAE surveillance protocol specifies to use the daily minimum FiO2 and MAP values when assessing for both the period of stability or improvement and the period that indicates worsening oxygenation.
From the multiple FiO2 settings that will be documented each calendar day, you will identify the minimum (lowest) FiO2 setting for that calendar day that is maintained for > 1 hour.
- If FiO2 settings are documented less frequently than every hour throughout the calendar day (for example, every 2 hours or every 4 hours), each setting will have been maintained for > 1 hour so the daily minimum FiO2 will be the lowest value documented on the calendar day.
- If FiO2 settings are documented hourly or more frequently you will need multiple consecutive recordings of that FiO2 setting.
- If FiO2 settings are monitored and recorded every 15 minutes, you would need 5 consecutive recordings of a particular FiO2 setting for that setting to be identified as the daily minimum value (for example, at 09:00, 09:15, 09:30, 09:45, and 10:00).
- If FiO2 settings are monitored and recorded every 30 minutes, you would need 3 consecutive recordings (for example, at 09:00, 09:30, and 10:00).
- If FiO2 settings are monitored and recorded hourly, you would need 2 consecutive recordings (for example, at 09:00 and 10:00).
- If there is no FiO2 setting that was maintained for > 1 hour, then select the lowest setting documented for that calendar day.
From the multiple MAP readings that will be documented each calendar day, you will identify lowest MAP reading for that calendar day regardless of how long the MAP value was maintained. MAP readings are to be rounded to the nearest whole number in the following manner: a MAP of 10.00 – 10.49 is rounded to 10 and a MAP of 10.50 – 10.99 is rounded to 11.
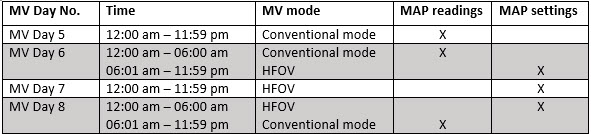
When patients are receiving support using HFOV, we understand that there may be both MAP readings as well as MAP settings documented in the medical record. In that instance the MAP settings are to be used to select the daily minimum MAP when the patient is on HFOV the entire calendar day. When switching from conventional to HFOV or HFOV to conventional ventilation during a calendar day, both MAP readings while on conventional mode and MAP settings while on HFOV are a part of the documented values from which you would select the lowest value (daily minimum value) for that calendar day. Once on HFOV the entire calendar day use the MAP settings only.
Secondary BSI to lower respiratory events in locations performing PedVAE surveillance
For purposes of NHSN, for a bloodstream infection to be determined to be secondary to a site-specific infection (specifically, related to an infection at another site, such that primary site of infection may have seeded the bloodstream secondarily), the patient must first meet one of the NHSN site-specific definitions. Please note, when conducting PedVAE surveillance, a secondary BSI cannot be attributed to PedVAE. While there are several optional data collection fields for PedVAE that pertain to identification of organisms in both respiratory and blood specimen sites, reporting of this information does not account for a secondary BSI.
When conducting PedVAE surveillance regardless of whether a PedVAE is detected or is not detected, identification of a BSI can be evaluated to see if it is secondary to any of the infection sites as defined in Chapter 17 [PDF – 686 KB] or PNEU [PDF – 703 KB], UTI [PDF – 913 KB], or SSI [PDF – 913 KB]event protocols. If another specific site infection to which the bloodstream infection can be attributed as a secondary BSI is not identified, it may need to be reported as a primary BSI/CLABSI.
For a secondary bloodstream infection to be deemed secondary to PNEU (PedVAP), the PNU2 or PNU3 definition must be met first. You cannot attribute a bloodstream infection secondary to PNEU (PedVAP) based on a clinical diagnosis of pneumonia or solely based on recovery of a matching pathogen from a lower respiratory tract specimen and blood specimen.
Episode of mechanical ventilation
Per the PedVAE surveillance protocol, the 14-day event period is to be observed even if a new episode of mechanical ventilation (MV) is established during that event period. The 14-day event period for PedVAE surveillance is governed by the date of event (date of onset of worsening oxygenation), not the date of initiation of mechanical ventilation. If a patient is removed from mechanical ventilator for one full calendar day or more and then is returned to the ventilator within the 14-day event period, a new episode of MV would begin, and the mechanical ventilation day count would start over again. The earliest a new PedVAE could be identified would be day 3 of the new MV episode; however, a new PedVAE cannot be reported until the 14 days have elapsed.
In the example presented in the table below, you will see that there is a PedVAE detected on hospital day 4 during the first episode of mechanical ventilation. The patient is extubated on hospital day 6 and remains off MV for one full calendar day (hospital day 7). On hospital day 8, the patient is re-intubated, thereby starting a second episode of MV. The patient is observed to meet PedVAE criteria, with a baseline period of stability or improvement on hospitals days 8 and 9 and a period of worsening on hospitals days 10 and 11 – but because the patient is still within the 14-day event period for the PedVAE detected on hospital day 4, a new PedVAE is not reported.
| Hosp Day No. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| MV Episode | 1 | 1 | 1 | 1 | 1 | 1 | —– | 2 | 2 | 2 | 2 | 2 |
| MV Day No. | 1
Intubated at noon |
2 | 3 | 4 | 5 | 6 Extubated at noon | —– | 1
Re-intubated at 0800 |
2 | 3 | 4 | 5 |
| PedVAE Criterion | — | Baseline Day 1 | Baseline Day 2 | Worsening Day 1 | Worsening Day 2 | —— | Baseline Day1 | Baseline Day 2 | Worsening Day 1 | Worsening Day 2 | ||
| PedVAE | PedVAE | No PedVAE | ||||||||||
| 14-day Event Period | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
Ventilator data
See Scenarios A through E below.
Scenario A:
Patient is intubated by the EMS in the field or is intubated in the ED. FiO2 and MAP data are available from the time the patient spent in the ED, prior to the patient being transferred to the PICU as an inpatient. Should I use the pre-hospital/ED ventilator data when making PedVAE determinations for that patient?
No. Ventilator data that is obtained from patients in the Emergency Department or other pre-hospital/pre-inpatient locations should not be included in PedVAE surveillance. Therefore, PedVAE surveillance begins for patients who are intubated in the pre-hospital or ED setting upon transfer to an inpatient location. Day 1 of ventilator data consists of data collected during the first calendar day of inpatient care.
Scenario B:
Patient is intubated and mechanically ventilated in an inpatient unit where PedVAE surveillance is not occurring. The patient is transferred to another inpatient unit in the same hospital where PedVAE surveillance is occurring. Do I use ventilator data from the transferring unit, even though PedVAE surveillance was not occurring in that unit?
Yes—to an extent. Since the transferring unit is in the same hospital, and since ventilator data from that transferring unit should be readily available, we advise that you go back 2 calendar days prior to transfer and utilize minimum daily MAP and FiO2 data from the transferring unit to determine whether a PedVAE has occurred during the first 2 days in the receiving unit. If a PedVAE is detected with an onset date on calendar day 1 or 2 (day of or day after transfer) in the receiving unit, that PedVAE would be attributable to the transferring unit and so would not be reported (since the transferring unit was not doing PedVAE surveillance).
Scenario C:
Patient is intubated and mechanically ventilated in an inpatient unit where PedVAE surveillance is occurring. The patient is transferred to another inpatient unit in the same hospital where PedVAE surveillance is also occurring. Do I use ventilator data from the transferring unit?
Yes. When transferring a patient between units that are both participating in PedVAE surveillance, surveillance should continue in an ongoing fashion. For example, if the patient had a PedVAE in the transferring unit on August 1, and was transferred to the receiving unit on August 4, a new PedVAE could not be detected in the receiving unit until the 14-day event period for the August 1 PedVAE had elapsed (so, August 15 in this case).
Scenario D:
Patient is intubated and mechanically ventilated in another hospital or healthcare facility and then transferred to my facility. It is unknown whether the transferring facility was performing PedVAE surveillance or not. Should I use ventilator data from the transferring facility (if available in my facility’s medical record) when making my PedVAE determinations?
When ventilator data are available from a transferring facility and documented in your facility’s medical record, you may use the ventilator data from the 2 calendar days prior to transfer to determine whether a PedVAE has occurred early in the course of the inpatient stay in your receiving facility. As in Scenario B, above, if a PedVAE is detected with onset date on calendar day 1 or 2 (day of or day after transfer) in your receiving facility, the PedVAE would be attributable to the transferring facility. If no ventilator data are available from the transferring facility, PedVAE surveillance begins on admission to your receiving facility/inpatient location where PedVAE surveillance is taking place.
Scenario E:
Patient is intubated and mechanically ventilated in another hospital or healthcare facility and then transferred to my facility. The transferring facility was performing PedVAE surveillance, and I have been informed that a PedVAE was detected in the transferring facility five days prior to transfer. Upon arrival in my receiving facility, does the 14-day event period apply, or do I need to “start fresh” with ventilator data available in my facility?
You should “start fresh,” although as noted above in Scenario D, you can use ventilator data from the 2 calendar days prior to transfer to determine whether there is a PedVAE early in the course of hospitalization in your receiving facility that would be attributed back to the transferring facility.
Home ventilators
The first step in determining whether such patients should be included in PedVAE surveillance is to decide whether the patient is on invasive mechanical ventilation, as defined by the NHSN. The NHSN definition of a ventilator is, “Any device used to support, assist, or control respiration (inclusive of the weaning period) through the application of positive pressure to the airway when delivered via an artificial airway, specifically an oral/nasal endotracheal or tracheostomy tube.” Note: Ventilation and lung expansion devices that deliver positive pressure to the airway (for example, CPAP, BiPAP, Bi-level, IPPB, and PEEP) via non-invasive means (for example, nasal prongs, nasal mask, full face mask, total mask, etc.) are not considered ventilators unless positive pressure is delivered via an artificial airway (oral/nasal endotracheal or tracheostomy tube).
Based on this definition, patients on home mechanical ventilators or patients supported by devices typically considered non-invasive ventilator devices should be included in PedVAE surveillance if the ventilator support is administered via an endotracheal or tracheostomy tube, even if the support is administered only for portions of each day (for example overnight). Patients receiving non-invasive ventilation (such as BiPAP via a face mask or nasal mask) should not be included in PedVAE surveillance.
The second step in determining whether such patients can be included in PedVAE surveillance is to determine whether the FiO2 can be set at a specific level on the home mechanical ventilator or other ventilator device or if a MAP reading can be provided. If neither FiO2 or MAP can be provided, a patient could not be included in PedVAE surveillance because it would not be feasible to assess changes in the level of FiO2 settings or MAP readings. If the FiO2 can be set or MAP readings can be provided and either are monitored and recorded, then these patients should be included in PedVAE surveillance.
If the patient is switched from a home mechanical ventilator or other device to a critical care unit mechanical ventilator, then they can be included in PedVAE surveillance at that time (taking into account that a baseline period of stability or improvement will need to be established on the critical care mechanical ventilator).
NOTE: Patients on home ventilators in locations where in-plan PedVAE surveillance is being performed will be included in the ventilator day denominator count regardless of their eligibility for PedVAE surveillance.
Location of mechanical ventilation
This field should reflect the location where the patient was intubated. For example, if the patient was intubated by first responder personnel in the field prior to arrival in the facility where mechanical ventilation was eventually initiated, the location chosen should be Mobile Emergency Services/EMS. If the patient was intubated at another facility, the location chosen should be “Location outside facility.” You will want to map these locations into your facility’s Locations list as appropriate.
Date of mechanical ventilation initiation
When a patient is admitted to a facility on a mechanical ventilator the date of mechanical ventilation initiation should reflect the actual date of mechanical ventilation initiation, not the date of admission to the facility. If necessary, an estimate of the actual date of mechanical ventilation initiation can be used. In the situation where a patient’s mechanical ventilation initiation is begun prior to admission to a facility, only in circumstances where the actual date or an estimate of the actual date cannot be determined should the date of mechanical ventilation initiation default to the date of admission to the facility.