Our Work: 2022
Our Work - 2022
Monkeypox case counts grow in US and globally
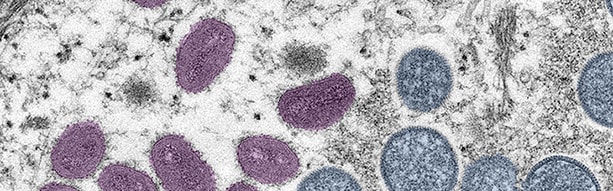
Colonized electron microscope image depicting a monkeypox virion.
As of July 28, 46 states, Washington DC, and Puerto Rico had reported 4,907 suspected or confirmed cases of monkeypox. The global case count is growing as well, with more than 21,000 confirmed or suspected cases reported worldwide. CDC is intensifying its efforts to raise awareness among clinicians and the public about the need for early recognition and diagnosis to prevent further spread. CDC is also working with its partners to expand access to vaccinations and to laboratory testing. Find the latest US and global case counts on CDC’s U.S. Monkeypox Outbreak 2022: Situation Summary webpage.
LRN preparedness pays off in monkeypox response

In 2005, NCEZID scientists made sure that the Laboratory Response Network (LRN) had an FDA-cleared laboratory test that could detect non-variola orthopoxviruses, the family of viruses that includes smallpox and monkeypox. Those steps to strengthen preparedness 17 years ago meant that LRN laboratories across the country were ready for this outbreak with a test that gives rapid results to guide patient care and help manage the spread of monkeypox. To meet increased testing demand, NCEZID’s scientists worked with FDA to allow five large commercial laboratories to use the test, increasing testing capacity by up to 10,000 specimens per week, per laboratory. The actions by NCEZID’s scientists have made it easier for clinicians to diagnose patients with monkeypox, which enables isolation and treatment of patients, administration of vaccines, and other public health actions to prevent the spread of disease.
Salmonella infections linked to backyard poultry

Backyard poultry, such as chickens and ducks, can carry Salmonella germs even if they look healthy and clean. These germs can easily spread to anything in the areas where the poultry live and roam. You can get sick from touching your backyard poultry or anything in their environment and then touching your mouth or food and swallowing Salmonella germs.
CDC is working with federal and state partners to investigate Salmonella outbreaks linked to backyard poultry. As of July 13, there have been 572 illnesses, 2 deaths, and 92 hospitalizations from 48 states. Learn more about the current outbreaks and what you can do to protect yourself and others whether you own backyard poultry or work in a hatchery or store selling poultry from hatcheries: Salmonella Outbreaks Linked to Backyard Poultry.
One Health approach identifies priority zoonotic diseases in Thailand, Southeast Asia

Staff from CDC’s One Health Office recently held a One Health Zoonotic Disease Prioritization workshop in Bangkok, Thailand, where they collaborated with the Thailand Coordinating Unit for One Health and the Food and Agriculture Organization of the United Nations to identify zoonotic diseases to prioritize for action. Thailand’s priority zoonotic disease list now includes zoonotic avian influenza, emerging coronaviruses, Nipah virus infection, Ebola, and rabies. CDC staff also created action plans to address the newly prioritized zoonotic diseases using a One Health approach. While in Thailand, CDC staff also provided technical assistance on SARS-CoV-2 zoonotic surveillance activities being conducted through CARES Act funding and developed and strengthened One Health partnerships in Thailand and the Southeast Asia region for future work. Pictured above: CDC One Health Office staff meet with staff from Thailand’s Department of Livestock Development and CDC Thailand.
Webpage focuses on challenging bacteria strains that threaten food safety

On the new Reoccurring, Emerging, and Persisting (REP) Enteric Bacterial Strains webpage, you will find helpful information on REP strains of bacteria, which can cause foodborne outbreaks over a period of years, are often linked to multiple sources, and are not stopped by a single intervention. Visit the webpage to learn more about REP strains and how CDC works to better understand their spread.
BEAM Dashboard allows users to visualize data needed to prevent enteric diseases
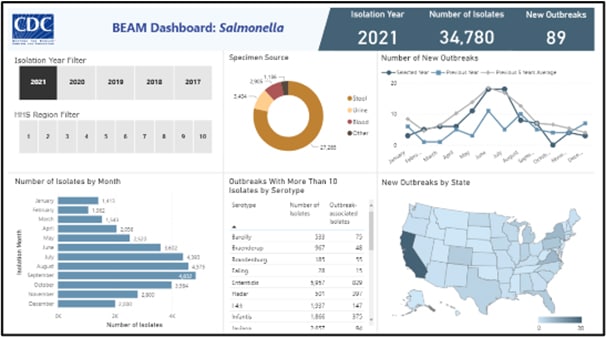
CDC staff have developed an interactive tool that allows you to access and visualize data from the System for Enteric Disease Response, Investigation, and Coordination. The Bacteria, Enterics, Amoeba, and Mycotics (BEAM) Dashboard will inform work to help prevent illnesses associated with food, water, the environment, and animal contact.
The first version of BEAM focuses on data for Salmonella bacteria isolated from human samples (stool or blood). The dashboard will eventually include additional pathogens, antimicrobial resistance data, and epidemiologic data from outbreak investigations. CDC plans to update the dashboard quarterly, with an aim to provide near real-time data in future versions. BEAM will provide timely pathogen, outbreak, and serotype-specific information to industry partners such as poultry producers, public health partners, and the public. Get more information in the BEAM Dashboard Frequently Asked Questions.
MMWR: Soft cheeses like queso fresco can still be contaminated after pasteurization

A recent MMWR article highlights the risk for pregnant people, older adults, and people with weakened immune systems of getting a serious Listeria infection from queso fresco, even if it’s made from pasteurized milk. CDC and federal, state, and local partners began investigating a cluster of Listeria monocytogenes infections in 2021.
In total, 13 people got sick, including one person who died. Four people got sick during pregnancy, resulting in two pregnancy losses and one premature birth. Investigators were able to identify a brand of queso fresco made with pasteurized milk as the likely source of infections. Although pasteurizing milk kills Listeria, this outbreak investigation was able to show that soft cheeses like queso fresco can still become contaminated after pasteurization.
CDC updates guidance for before, during, and after travel

In May, CDC updated guidance on wearing masks during travel and in public transportation settings and updated recommendations on what to do before and after travel
- When traveling and in public transportation settings: Everyone aged 2 years or older—including passengers and workers— should properly wear a well-fitting mask or respirator in indoor areas of public transportation (such as airplanes, trains, buses, ferries) and transportation hubs (such as airports, stations, and sea-ports), especially in locations that are crowded or poorly ventilated such as airport jetways.
- This guidance is based on currently available domestic and global data, current trends in COVID-19 community levels within the United States, and projections of COVID-19 trends in the coming months. Continue to keep yourself and others protected from COVID-19 by staying up to date with COVID-19 vaccines, avoiding crowds when possible, washing your hands, and properly wearing a well-fitting mask or respirator.
- Before travel: Regardless of your destination (domestic or inter-national) and vaccination status, consider getting tested for current infection with a viral test as close to the time of your departure as possible (no more than 3 days).
- After domestic travel: Regardless of your vaccination status, get tested if your travel involved situations with a greater risk of expo-sure, such as being in crowded places while you were not wearing a well-fitting mask or respirator, and follow additional guidance if you know you were exposed to a person with COVID-19. Monitor yourself for COVID-19 symptoms, and isolate yourself and get tested if you develop symptoms.
To learn more, visit CDC’s Wearing Masks in Travel and Public Transportation Settings, Domestic Travel during COVID-19 and International Travel webpages.
Removal of COVID-19 testing requirement to board flight to the US

The Order requiring people to show a negative COVID-19 test result or documentation of recovery from COVID-19 before boarding a flight to the United States has been removed. Effective June 12, 2022, air passengers do not need to get tested and show the negative COVID-19 test result or documentation of recovery from COVID-19 prior to boarding a flight to the United States. For more details, visit CDC’s Rescission: Requirement for Negative Pre-Departure COVID-19 Test Result or Documentation of Recovery from COVID-19 for all Airline or Other Aircraft Passengers Arriving into the United States from Any Foreign Country webpage.
New tool informs travelers of requirements to board flight to the US

Travel Assessment is a new tool from CDC that helps travelers learn the latest requirements to board a flight to the United States during the pandemic. This tool gives travelers a customized response based on their circumstances, and it improves the user experience for all travelers. Visit the COVID-19 Travel website to use the Travel Assessment tool.
Improvements needed in outpatient antibiotic prescribing for older adults

In a recent JAMA research letter, CDC authors showed that during the first year of the COVID-19 pandemic (April 2020–April 2021), 30% of outpatient visits for COVID-19 among Medicare beneficiaries were associated with an antibiotic prescription. Azithromycin was the most frequently prescribed antibiotic (50.7%), followed by doxycycline (13.0%), amoxicillin (9.4%), and levofloxacin (6.7%). Prescribing rates were highest for emergency department (33.9%) and telehealth encounters (28.4%).
Non-Hispanic white beneficiaries were prescribed antibiotics at a higher rate than African American or Black beneficiaries. Antibiotics, including azithromycin, are not effective against COVID-19 and should not be prescribed to most outpatients with viral infections. Findings from this study reinforce the importance of improving appropriate antibiotic prescribing in outpatient settings and avoiding unnecessary antibiotic use for viral infections, including COVID-19, in older adult populations.
CDC and Partners Collaborate on Monkeypox Response

In May, CDC activated a response for the rapidly evolving outbreak of monkeypox virus infections in the United States and other countries. Testing by NCEZID’s Poxvirus and Rabies Branch on May 18 confirmed monkeypox (West African clade) in a Massachusetts resident with rashes characteristic of monkeypox who had recently traveled internationally. Since then, the Laboratory Response Network has identified additional Orthopoxvirus-positive cases in multiple states. CDC officials are also tracking several clusters of monkeypox cases recently identified in countries that do not typically report monkeypox.
Although the majority of cases in the current outbreak are among gay, bisexual, or other men who have sex with men anyone can get monkeypox. Monkeypox spreads person to person through contact with infectious rashes, close face-to-face contact, or items that previously touched the infectious rash or body fluids.
CDC issued a HAN advisory on May 20 urging US healthcare providers to be alert for patients who have a rash consistent with monkeypox, regardless of whether they have recently travelled to a country where monkeypox has been reported or have other risk factors for monkeypox. CDC updated the case definition for monkeypox in a June 14 HAN update. Learn the latest about this outbreak on CDC’s Monkeypox | Poxvirus web page.
Salmonella outbreak linked to peanut butter

CDC issued a Food Safety Alert on May 21 warning consumers about Salmonella infections linked to Jif brand peanut butter, sold at stores nationwide. NCEZID scientists are working with public health and regulatory officials in several states and the US Food and Drug Administration to investigate the multistate outbreak of Salmonella Senftenberg infections. As of June 1, a total of 16 people infected with the outbreak strain had been identified in 12 states. On May 20, J.M. Smucker Company announced a voluntary recall of multiple Jif brand peanut butter types.
People can identify recalled products by their lot code. If the first four numbers are 1274 through 2140 and the next three numbers are 425, the product has been recalled. For example, the lot code 1352425 has been recalled (see photo). Additional companies have recalled food items made with Jif brand peanut butter, such as snack trays, chocolates, or ice cream. The CDC alert recommends that the recalled peanut butter products should not be eaten, sold, or served. Also, people should not feed recalled food to pets or other animals.
New report shows concerning resistance in enteric bacteria

The National Antimicrobial Resistance Monitoring System (NARMS) regularly collects and tests bacteria from people, retail meats, and food animals to examine trends in resistance of enteric pathogens to important antimicrobial drugs. Each year, NARMS—a partnership between FDA, CDC, and USDA—releases a summary of its latest findings.
The 2019 NARMS Update: Integrated Report Summary Interactive Version shows a similar level of antimicrobial resistance in 2019 as in 2018 for enteric bacteria from humans, but the report identifies some concerning trends. For example, resistance to antibiotics commonly used to treat intestinal infections increased for some serotypes of Salmonella. Read the report summary and explore the interactive version for details on specific bacteria, drugs, and trends.
Free continuing education on ehrlichiosis and anaplasmosis
Ehrlichiosis and anaplasmosis are emerging tickborne diseases. Signs and symptoms of these diseases can be difficult to differentiate from other diseases, but early recognition and treatment are key to preventing severe and fatal outcomes.
NCEZID’s Rickettsial Zoonoses Branch has created a new module to help clinicians diagnose and treat these two diseases. The module includes knowledge checks and a case-based scenario to help providers apply what they have learned. Free continuing education credit is available for physicians, nurse practitioners, pharmacists, veterinarians, nurses, epidemiologists, public health professionals, educators, and health communicators.

National Healthcare Safety Network Dialysis Data Now Available on COVID Data Tracker
In March, the Dialysis COVID-19 Data Dashboard was added to CDC’s COVID Data Tracker. This new dashboard displays data on COVID-19 cases and deaths among patients and staff of dialysis facilities at the national or state level. This information helps CDC assess the effect of COVID-19 on patients and staff in more than 7,500 dialysis facilities in the United States.
These summaries use data that dialysis facilities submit to the National Healthcare Safety Network COVID-19 Module. The dashboard is available via the Health Care Settings data grouping on the COVID Data Tracker and is updated weekly.
Personalized Quarantine and Isolation Recommendations at the Touch of a Finger
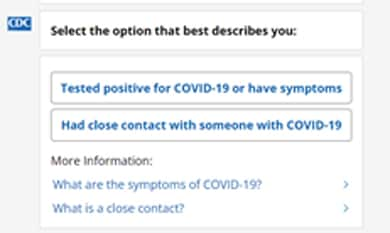
CDC published the Quarantine and Isolation (Q and I) Calculator, an online tool that helps people determine how long they need to isolate, quarantine, or take other steps to prevent spreading COVID-19. The calculator, featured on Good Morning America, is a quick and easy tool for people who have COVID-19 or have been in close contact with someone who has COVID-19. Users answer questions about their close contact exposure or symptom onset and get personalized recommendations and information quickly. As home self-testing becomes more common, the Q and I Calculator helps people make informed decisions about quarantine and isolation for themselves and their loved ones. The calculator is available in both English and Spanish.
CDC discovers a new type of HPV
Scientists at CDC’s Chronic Viral Diseases Branch (CVBD) used their own laboratory-developed method of targeted whole genome sequencing (eWGS) to identify a new type of human papillomavirus (HPV). The sequenced samples from eWGS are entered into a database, which finds matches to existing types. Recently, the results revealed three unidentified types of HPV. One of them was confirmed last month as a new type (called HPV229) by the International HPV Reference Center. CDC has for years worked in HPV research and analysis, but this is the first time they have identified a new HPV type. Of the currently known 200 HPV types (out of 229), about 40 cause genital infection and 14 are highly associated with causing cancer. The discovery of new HPV types is a step toward identifying potential associations between novel HPV types and additional diseases.
CDC and LRN laboratories join forces to pilot detection of antibiotic-resistant B. anthracis
During an anthrax emergency, rapid laboratory detection and quick action would be key to an effective public health response. To strengthen CDC’s capacity to act quickly if such an emergency were to occur, the Laboratory Preparedness and Response Branch (LPRB) kicked off a new pilot study of real-time whole genome sequencing testing that could detect genetic evidence if Bacillus anthracis bacteria is ever bioengineered to be resistant to antibiotics. The study, a collaboration with the Association of Public Health Laboratories, is being conducted in three state Laboratory Response Network (LRN) laboratories. The findings from this study will help CDC improve DNA sequencing-based laboratory methods currently under development and lead to more rapid laboratory results during a public health emergency.
CDC addresses health disparities related to antibiotic resistance

CDC recently released a new Health Equity and Antibiotic Resistance fact sheet [PDF – 2 pages] highlighting health disparities related to specific antibiotic-resistant pathogens. Antibiotic resistance contributes to health disparities across age, race, and gender in settings spanning the One Health spectrum. The fact sheet describes examples of known health disparities linked to many antibiotic-resistant threats.
CDC is taking concrete steps to reduce these disparities through its agency-wide CORE Initiative goals and across programs, including:
- Project Firstline, which addresses training gaps for healthcare workers from diverse educational and training backgrounds
- Antibiotic resistance laboratory networks, which analyze patient demographic data alongside laboratory test results to provide a comprehensive picture of antibiotic resistance in certain populations
- The National Healthcare Safety Network, which provides demographic data on patients for future analysis and drives improvement in healthcare quality
Advancing One Health in Colombia

stand with National Institute of Health staff. Grace is in the front row, second from left; Kate, in the front row, is the first person on the right. Italo is in the back row, second from right.
CDC’s One Health Office (OHO) global health security. supporting Colombia’s National Institute of Health (INS) in its implementation of One Health activities to address outcomes from Colombia’s One Health Zoonotic Disease Prioritization (OHZDP) workshop in 2019. The workshop brought together representatives from human, animal, and environmental health sectors to identify zoonotic diseases that are of greatest concern in Colombia.
CDC staff recently traveled to Colombia to provide technical assistance to INS colleagues and discuss plans for One Health efforts in the country. During this visit, CDC staff helped INS prepare to conduct subnational OHZDP workshops in four regions across Colombia, which have different cultural and environmental characteristics. The subnational workshops will give representatives from human, animal, and environmental health sectors the opportunity to identify subnational zoonotic diseases that are high priorities (to complement the existing national list of priority zoonotic diseases) and develop next steps to address these priority diseases. In addition, INS began working with CDC’s Poxvirus and Rabies Branch to identify gaps and opportunities to collaborate on rabies prevention and control because rabies was identified as a priority zoonotic disease This partnership is a great example of a One Health collaboration that will help advance and strengthen global health security.
CDC launches webinar on dengue vaccine

CDC launched the “Dengvaxia: What Healthcare Professionals Need to Know” webinar about the newly approved dengue vaccine. The dengue vaccine was approved and recommended for use in children 9–16 years old who have laboratory-confirmed evidence of a previous dengue virus infection and who live in areas of the United States where dengue is common (this includes US territories of American Samoa, Puerto Rico, and the US Virgin Islands, and the freely associated states, including the Federated States of Micronesia, the Republic of Marshall Islands, and the Republic of Palau). This 30-minute learning activity provides an overview of the vaccine’s eligibility criteria; pre-vaccination screening recommendations; and vaccine storage, handling, and administration.
Professionals will be able to get free continuing education credits after completing the webinar, which you can find online.
Genomic Surveillance System Enables Early Detection of Variants among Arriving International Travelers

By testing nasal and saliva samples from international air passengers arriving at four United States airports, CDC’s genomic surveillance program for SARS-CoV-2, the virus that causes COVID-19, identified importations of Omicron variants days before they were reported elsewhere in the United States and North America— 7 days earlier for variant BA.2 and 43 days earlier for variant BA.3. This voluntary surveillance program is a partnership with XpresCheck, Ginkgo Bioworks, and CDC.
The program began in September 2021 and operates out of testing booths at airports in New York, New Jersey, Miami, San Francisco, and Atlanta. Staff members recruit international air passengers to voluntarily participate. Participants provide saliva samples at airports when they arrive and 3 to 5 days later by using at-home saliva sampling kits. Samples undergo RT-PCR testing, and those that test positive are sequenced to determine the strain of the virus This program serves as an early warning system to detect future COVID-19 outbreaks introduced by international travelers.
Identifying CDC Partnerships with Faith-Based Organizations to Promote COVID-19 Vaccination

During the COVID-19 pandemic, state health departments engaged faith-based organizations to support response activities. To understand the scope of activities, CDC, along with staff from HHS, and the Association of State and Territorial Health Officials (ASTHO), surveyed ASTHO member jurisdictions and recently published their results. Almost all respondents (92%) reported partnerships to promote vaccination, and all viewed the partnerships as valuable for reaching racial and ethnic minority groups. Many jurisdictions attempted to be inclusive, working with churches, mosques, synagogues, and temples. Barriers to engagement included lack of established relationships, resource limitations, and distrust of government. These findings demonstrate a promising approach to reaching diverse communities that may be useful for future preparedness and response efforts.
CDC and Tribal Partner Assess Antigen Test Positivity after COVID-19 Isolation in Southwest Alaska
The arrival of the Omicron variant ushered in a new phase to the COVID-19 pandemic. With this phase came additional questions about the how antigen tests could guide recommendations for isolating people with confirmed or suspected cases of COVID-19. CDC’s Arctic Investigations Program, in collaboration with the Yukon-Kuskokwim Health Corporation, published a Morbidity and Mortality Weekly Report (MMWR) article on antigen test positivity after COVID-19 isolation in Alaska’s Yukon-Kuskokwim Delta Region. Their investigation found that most people tested still had a positive antigen test result 5 to 9 days after they first had symptoms or after their initial positive test. Their findings support the recommendation that people who tested positive for COVID-19 should follow CDC’s quarantine and isolation guidance following infection, including wearing a well-fitted mask and avoiding being around people who are at higher risk for severe COVID-19 outcomes.. The study demonstrates that antigen tests might be a useful tool to guide recommendations for isolation after SARS-CoV-2 infection.
CDC Supports One Health Investigations of SARS-CoV-2 in Big Cats

CDC’s One Health Office (OHO) experts helped investigate SARS-CoV-2–associated deaths in seven big cats (five snow leopards and two lions) from five zoos across the United States. OHO, USDA, and state partners collaborated with zoo veterinarians, veterinary pathologists, and CDC’s Infectious Diseases Pathology Branch (IDPB) to help determine the role of SARS-CoV-2 in the animals’ deaths. IDPB detected SARS-CoV-2 in respiratory tissues of the six big cats they evaluated. Five big cats also had severe pneumonia attributed to bacterial and fungal coinfections. Pseudomonas spp. and Aspergillus spp. were identified; two animals had systemic aspergillosis. SARS-CoV-2 likely caused initial damage to their respiratory tracts, which predisposed the animals to fatal bacterial and fungal coinfections. These cases highlight the importance of collaborative One Health investigations to understand transmission dynamics between animals and people with SARS-CoV-2 and underscore the similarity in outcomes between animals and people with SARS-Cov-2 and fatal secondary infections. The SARS-COV-2 antibody assay is now used as part of CDC’s ongoing One Health investigations.
CDC finds new hope for an old disease
A new study comparing treatment outcomes for children with Kawasaki disease (KD) has yielded results that could substantially improve clinical management. Using data from KD surveys conducted in Japan, CDC and Japanese collaborators evaluated the effectiveness of two different steroid regimens by comparing KD patients receiving a single high-dose pulse of steroids to those receiving multiple steroid doses over a longer period. The differences observed between patient outcomes following the different treatments were striking. For patients receiving pulse steroid treatment, treatment failed to reduce fever in about 41% of severe KD infections. However, multiple doses over a longer period cut the rate of treatment failure in half—to 21%. These patients also had a 34% lower likelihood of developing coronary artery damage.
CDC authors identify higher-volume antibiotic outpatient prescribers by using Medicare Part D data

A new Morbidity and Mortality Weekly Report (MMWR) article from CDC shows that in 2019, 41% of all Medicare Part D antibiotic prescriptions were prescribed by 10% of prescribers. This finding indicates that a small percentage of healthcare providers were responsible for prescribing many antibiotic prescriptions. Prioritizing higher-volume prescribers (the top 10% of antibiotic prescribers by volume) for outreach and interventions could result in larger improvements in antibiotic prescribing. Public health and healthcare organizations can use publicly available Medicare Part D prescription data to optimize antibiotic prescribing, limit the development of antibiotic resistance, and improve patient outcomes, according to the report.
Tick-borne encephalitis vaccine recommended for some travelers
On February 23, the Advisory Committee on Immunization Practices (ACIP) approved recommendations for use of a newly licensed vaccine against tick-borne encephalitis (TBE) among US travelers. TBE is a rare but potentially severe disease; TBE virus is found in parts of western and northern Europe through northern and eastern Asia. Risk for TBE is low for most travelers. However, vaccination is recommended for those planning to visit TBE-endemic areas if they will have extensive exposure to ticks. In addition, vaccination may be considered for travelers without extensive tick exposure if they might engage in outdoor activities where ticks are likely to be found. In those cases, the decision to vaccinate is based on an assessment of their planned activities and itinerary, risk factors for a poorer medical outcome, and personal perception and tolerance of risk. Recommendations were also approved for laboratory workers with a potential for exposure to TBE virus. For more information about the TBE vaccine, check out the March ZOHU Call webinar.
“We Were There” to honor Sherif Zaki

CDC’s “We Were There” lecture features a very special presentation focusing on the work and life of the late Sherif R. Zaki, MD, PhD, who started CDC’s Infectious Disease Pathology Branch and for many years managed this unique team of experts. The theme for the webinar is “Honoring Sherif Zaki—the Science and the Man.” The webinar will be held Tuesday, April 26, from 1:30 to 3:00 pm.
CDC Director Rima Khabbaz to retire March 31, 2022

CDC’s National Center for Emerging and Zoonotic Infectious Diseases (NCEZID) Director Rima Khabbaz, MD, retired March 31, after nearly 4 decades of service at CDC. She had been the director of NCEZID since 2017. Under her guidance, NCEZID confronted disease threats that included antibiotic resistance, Ebola, Zika, COVID-19, and many more. As director, Dr. Khabbaz also supported the One Health initiative and One Health capacity building worldwide and furthered CDC’s efforts to prevent and control a multitude of emerging and zoonotic infectious diseases.
CDC’s Arjun Srinivasan in the news

CBS News chief medical correspondent Jon LaPook interviewed CDC’s Dr. Arjun Srinivasan for a segment answering common questions about COVID-19. Dr. Srinivasan discussed CDC’s latest mask guidance, the future of COVID testing, and what a “new normal” might look like. The interview was featured on 60 Minutes Overtime on Sunday, March 6.
CDC’s Project Firstline releases new infection control resources for healthcare workers

CDC’s Project Firstline launched a new suite of innovative infection control educational resources designed to help healthcare workers think critically about infection control. The new resources provide foundational knowledge about where germs live in health care and how they spread to ensure that all healthcare workers know when and how to take action to protect patients and themselves from infections.
New resources were developed with and for healthcare workers to meet their unique needs and the time constraints of their busy schedules. The resources are available in a variety of formats, including
- Educational webpages that provide foundational information on infection control.
- Training toolkits for those looking to host their own infection control training.
- Interactive resources for testing your infection control knowledge.
- Videos and social media that can be easily accessed on the go.
- Posters and infographics to keep infection control front and center throughout healthcare facilities.
Check out the newly updated Antibiotic Resistance website
The Antibiotic Resistance Coordination & Strategy Unit is excited to share its updated website, refreshed to better engage audiences and share information about antibiotic resistance (AR) in the United States and around the world. The reorganization is intended to help all audiences quickly access CDC’s latest AR information resources.
Discover new features and updated content:
- Share print and digital educational resources.
- Read about the five areas where resistance emerges and spreads.
- Learn about national infection and death estimates and how CDC tracks and monitors antibiotic resistance threats in the United States.
- Find out how CDC is combating antibiotic resistance, plus ways you can fight AR.
NORS outbreak data available through 2020
National Outbreak Reporting System (NORS) outbreak data are now available through 2020 for all modes of transmission in NORS Dashboard. With the Dashboard’s interactive maps and charts, you can learn about foodborne, waterborne, and other enteric disease outbreaks and gain valuable insight about the pathogens, foods, water, states, and settings linked to them.
Food safety microsite now available in Spanish and Haitian Creole

CDC developed a food safety microsite, a collection of key food safety webpages that can be embedded in other websites. It’s now available in English, Spanish, and Haitian Creole. The microsite allows partners to add CDC information to their websites, and the content updates automatically when CDC makes changes.
Emerging Infectious Diseases podcasts spotlight Salmonella illnesses, West Nile virus transmission
The winter holidays have come and gone, but CDC continues to share insights from research on foodborne illnesses that happen around the holidays. An episode from the Emerging Infectious Diseases (EID) journal’s podcast dives into a recent study that analyzed 20 years of data on Salmonella infections that occurred soon after the Thanksgiving holiday. Podcast host Sarah Gregory and CDC study author Farrell Tobolowsky discuss Salmonella illnesses and outbreaks, how the authors linked illnesses to the Thanksgiving holiday, measures that can make turkey safer, and more.
Another recent EID podcast episode covers transmission of West Nile virus through solid organ transplantation. CDC Epidemic Intelligence Service Officer Raymond Soto and Sarah Gregory discuss this issue, and you can learn more about Soto and his coauthors’ investigation in EID’s synopsis. Find these episodes and many others from EID’s podcast in CDC’s Public Health Media Library.
Wastewater data now on COVID Data Tracker
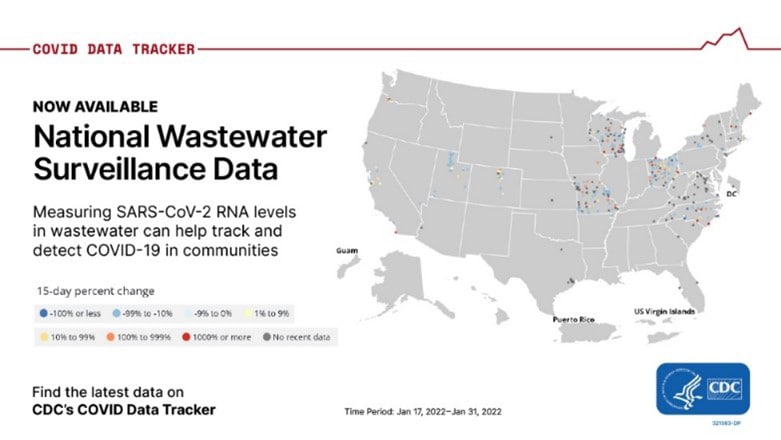
CDC’s COVID Data Tracker now shows wastewater surveillance data from more than 400 testing sites around the country. Surveillance data are available for download. As many as 80% of people who get COVID-19 shed viral RNA in their feces, making wastewater testing data important for enhancing COVID-19 surveillance. Currently, CDC is supporting 37 states, four cities, and two territories to help develop wastewater surveillance systems that can provide an early warning of COVID-19’s spread in their communities.
COVID-19 vaccine boosters remain safe

People 18 years and older who received the same mRNA vaccine brand for all their vaccinations experienced fewer adverse reactions following the booster dose than they did after their second dose, according to a new CDC report. COVID-19 vaccination data for the report came from two CDC vaccine safety monitoring systems, v-safe and the Vaccine Adverse Event Reporting System (VAERS). More than 90% of side effects reported to VAERS were not considered serious, with headache, fever, and muscle pain among the most common reactions. V-safe data showed that people rarely received medical care after a booster dose. Read the study in MMWR: Safety Monitoring of COVID-19 Vaccine Booster Doses Among Adults — United States, September 22, 2021–February 6, 2022.
5 million COVID-19 rapid antigen tests monthly for K–12 schools

CDC, in partnership with the Office of the Assistant Secretary for Preparedness and Response, is providing free COVID-19 rapid antigen tests to K-12 schools. The tests are point-of-care: they give the user rapid results and don’t have to be sent to a lab for processing. Requests to receive tests are prioritized for school districts that have:
- An immediate need for testing support
- A high social-vulnerability index score
- The ability to rapidly implement screening tests or test-to-stay programs
The 64 jurisdictions funded through the Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases (ELC) cooperative agreement are coordinating the allocation of the rapid tests to the school testing programs they support. In the program’s first seven weeks, CDC received requests from 50 jurisdictions and allocated 5.3 million tests to more than 1,000 school districts. These tests are supplementing the over 30 million tests performed in schools across the country. This initiative compliments the ELC Reopening Schools program.
Record funding strengthens health departments’ response to COVID-19
NCEZID has distributed a record $42.6 billion to help health departments modernize data reporting and lab equipment and support testing to fight the spread of COVID-19. The sum, which was allocated through the Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases (ELC) cooperative agreement, has supported over 51,000 employees across the country and funded innovations to fight COVID-19, including:
- Pairing whole genome sequencing with wastewater surveillance to identify COVID-19 variants
- Transforming siloed data systems into more unified public health information infrastructure
- Modernizing diagnostic data reporting
- Supporting testing and results reporting for over a billion tests, including 37.6 million COVID-19 tests at schools to maintain safe, in-person learning
- Equipping labs to speed up processing of COVID-19 tests
These investments in laboratories, epidemiology, health information systems, and the public health workforce have been crucial to saving more lives in the COVID-19 pandemic and can help the country prevent future disease threats.
Africa Field Program supports public health in Kenya and East Africa
Public health officials in Kenya and neighboring countries collaborated with NCEZID’s Africa Field Program to develop ideas to tackle COVID-19 and other infectious diseases. Several projects improved border health coordination between Kenya and its neighbors and improved Kenya’s overall public health capacity. Projects included establishing a hybrid (virtual and in-person) meeting between Kenya and Uganda and supporting the development of an app called Jitenge, which enables users traveling to and from Kenya to report COVID-19 symptoms to Kenya’s Ministry of Health.
COVID-19 Program for Cruise Ships
CDC is working closely with the cruise industry to implement the COVID-19 Program for Cruise Ships, a set of recommendations and guidance for cruise ships operating in US waters. Participation is voluntary in the program, which replaced the mandatory Conditional Sailing Order that expired on January 15. Cruise ships choosing to participate in the new COVID-19 Program for Cruise Ships receive a color status designation to indicate:
- The level of COVID-19 cases reported onboard
- Whether an investigation is needed
- Additional public health measures the ship is taking
- Whether a ship has opted out of the program
Foreign-flagged cruise ships choosing not to participate in the new program receive a gray color designation. Gray designated cruise ships may have their own COVID-19 health and safety protocols, which CDC has not reviewed or confirmed. US-flagged cruise ships may choose to follow CDC’s COVID-19 Program for Cruise Ships at the cruise ship operator’s discretion. CDC will continue working closely with the cruise ship industry and monitoring COVID-19 preventive measures and cases onboard ships through inspections and daily data collection. CDC’s recommendations are aimed at protecting people onboard and on land.
NCEZID Accomplishments 2021 report out now

The NCEZID Accomplishments 2021 report is available online. It summarizes our staff’s and partners’ achievements in 2021 in fighting the spread of COVID-19 and protecting people from many other domestic and global infectious disease threats. The report is a reminder of how much NCEZID and its colleagues and partners do to reduce illness and death from emerging and zoonotic infectious diseases.
NCEZID deactivates public health response in resettlement of Afghan evacuees
On February 18, NCEZID deactivated the public health response that was part of Operation Allies Welcome (OAW), the US government mission to safely evacuate people from Afghanistan and resettle them in the United States. NCEZID led OAW’s public health efforts, which included medical screenings and the vaccination of more than 80,000 Afghans at designated military bases, where evacuees lived before resettling in US communities. Nearly 285 staff from across CDC participated in the response, including more than 140 who deployed to the field. Responders helped prevent measles, chickenpox, and other diseases detected among the evacuees from spreading in this country and helped protect evacuees from the spread of COVID-19 on military bases. NCEZID is continuing some response measures beyond the deactivation.
NCEZID celebrates Black History Month and girls and women in science

NCEZID celebrated Black History Month in February, including on social media, where the center honored staff who have made a substantial impact in their fields and on public health. Achievements by those honored touch many roles at NCEZID—from public health analyst and quarantine officer to associate director. These colleagues’ contributions make CDC stronger—from mentoring and improving infection prevention and control to managing and leading strategic development.
On February 11, International Day of Women and Girls in Science, NCEZID’s Office of Advanced Molecular Detection (AMD) promoted the participation of women and girls in STEM fields with a post on Twitter that featured members of the AMD team and highlighted the role of women in public health.
Improving prevention measures against Chapare virus and hantavirus in Bolivia
Arenavirus / CDC
NCEZID staff recently visited Bolivia to collaborate with health officials to use new diagnostic assays and improve surveillance and response against Chapare virus and hantavirus. Both viruses cause viral hemorrhagic fevers. The work was done at Bolivia’s National Center of Tropical Diseases and was in response to requests for NCEZID support from the Pan American Health Organization and the Bolivian Ministry of Health following a cluster of Chapare virus cases in Bolivia in 2019.
Superbug Candida auris identified in four new states

In December and January, four more states—Oregon, Louisiana, Tennessee, and Wisconsin—reported their first cases of infection with Candida auris, a highly drug-resistant fungus that primarily spreads in healthcare facilities. Most initial cases were detected in acute care hospitals, but additional cases were identified in nearby long-term care facilities. Only two cases have been linked to travel, a finding that suggests C. auris may be spreading locally in these states. NCEZID staff are providing expertise to health departments and facilities in these states, which have issued public health alerts and supported infection control efforts and screening of patients at high risk for C. auris. Specimens will be tested for their genetic profiles and potential antifungal resistance, a hallmark of C. auris infection. With these new cases, 32 states have now identified at least one case of C. auris.
Use doubles of MicrobeNet, which provides access to CDC’s pathogen library

Use of NCEZID’s MicrobeNet more than doubled during 2021. This online tool gives users access to CDC’s free virtual pathogen library, which helps medical and laboratory personnel anywhere in the world identify bacterial and fungal pathogens. They can submit their genomic sequences or test results, and MicrobeNet returns the closest identified match within minutes. In 2021, MicrobeNet received 151,101 submissions, an increase of 104% over 2020. The biggest jump in usage during 2021 came from users in China, who made more than 50% of all submissions to the online platform.
MicrobeNet staff are expecting continued increases in submissions this year due to two expansion projects—an international collaboration to build more databases and the development of a module for whole genome sequence (WGS) identification. The latter will allow users to submit raw WGS files for identification of more than 6,500 pathogens. To handle the anticipated increase in demand, MicrobeNet will be migrating to the cloud.
Interactive maps show investments in Advanced Molecular Detection

The Office of Advanced Molecular Detection (OAMD) has launched interactive investment maps displaying OAMD’s contributions to build and integrate laboratory, bioinformatics, and epidemiology technologies across CDC and nationwide.
The map shows AMD’s support for state and local jurisdictions. The office helps build genomic sequencing capacity and provides workforce training to prepare states, communities, and the nation for future disease outbreaks.
Map showcases innovations against antibiotic resistance

In February, CDC released an updated Antibiotic Resistance Investment Map, highlighting CDC’s antibiotic resistance funding to global and domestic partners—from academic centers to local public health departments.
Highlights of this year’s map include:
- An interactive map of CDC’s antibiotic resistance funding that supports state and local activities
- A global investment fact sheet featuring CDC’s antibiotic resistance partnership work in more than 50 countries
- A fact sheet showing how CDC’s COVID-19 efforts have also worked to address antibiotic resistance
Animated video explains how CDC investigates foodborne outbreaks

PulseNet is a vast network of laboratories across the United States that monitor outbreaks of illnesses transmitted through food, water, and the environment. To show how PulseNet works—in plain language—NCEZID recently released an animated video of an epidemiologist interviewing a patient who just recovered from a Salmonella infection.
The patient learns how PulseNet investigators detect outbreaks and how they use whole genome sequencing to nail down the outbreak’s possible source, like a certain food. The video explains how investigators use PulseNet to identify outbreaks and find possible gaps in food safety systems so they can quickly alert the public to reduce foodborne illnesses.
New population survey data about diarrheal illness on FoodNet Fast

What do people eat, drink, or do before getting a diarrheal illness? To find out, NCEZID’s Foodborne Diseases Active Surveillance Network (FoodNet) periodically surveys people in its surveillance area, which includes 15% of the US population. FoodNet has added data from its most recent population survey (2018–2019) to its online toolbox, FoodNet Fast. FoodNet Fast’s Population Survey Tool displays responses to questions, including about food, water, contact with animals, and food safety behaviors. You can also compare responses for different groups of people by filtering results by age group, sex, race and ethnicity, geographic location, and season.
CDC health equity strategy during COVID-19

CDC’s strategies to address COVID-19 inequities affecting racial and ethnic minority groups are described in Promoting health equity during the COVID-19 pandemic, United States [PDF – 3 pages], published December 29, 2021, in the Bulletin of the World Health Organization:
- The first strategy is to expand the evidence base: detailed race and ethnicity data can help guide ongoing emergency response efforts.
- The second strategy is to expand programs and practices to reach populations at increased risk for COVID-19—populations that can also be disproportionately affected by unintended economic consequences from COVID-19 mitigations.
- The third strategy focuses on supporting essential and frontline workers, in which racial and ethnic minority groups are overrepresented and who face higher risk of SARS-CoV-2 exposure.
- The fourth strategy aims to build a diverse and inclusive public health workforce that is equipped to address the needs of an increasingly diverse population.
The paper describes how CDC is applying each of these strategies, the agency’s other efforts to promote health equity, and lessons learned. The authors conclude that the continued financing and implementation of CDC’s measures, along with community and partner engagement and national policies, can ensure that health equity considerations are built into all COVID-19 programs and future emergency responses.
COVID-19 vaccine safety in children 5 to 11 years old

A new MMWR article that reviewed early safety data for children 5–11 years old who received the Pfizer-BioNTech COVID-19 vaccine has shown that reports of serious reactions, including myocarditis, have been rare. If serious reactions occurred, it was usually the day following vaccination. Most reported reactions to the vaccine were mild and brief and included pain in the arm where the shot was given, tiredness, and headache. Reactions ranged from mild (didn’t interfere with normal daily activities) to moderate (interfered somewhat with normal daily activities). CDC and FDA continue to monitor vaccine safety, communicate findings, and use vaccine safety data to help shape COVID-19 vaccination recommendations.
Nursing home booster data now available on CDC’s COVID Data Tracker
In December 2021, CDC updated the COVID Data Tracker Nursing Home COVID-19 Vaccination Data Dashboard to include weekly data on nursing home residents and staff who received booster doses of a COVID-19 vaccine. The data, reported to the National Healthcare Safety Network (NHSN), provide a snapshot of booster vaccine uptake among residents of the nation’s approximately 15,400 nursing homes certified by the Centers for Medicare & Medicaid. The vaccine data also include reports of residents who received an additional primary dose (a vaccine dose given not long after the initial vaccination to people who could not generate adequate immunity to COVID-19 through that initial vaccination). In addition, the updated dashboard displays the vaccination status of nursing home residents who became infected with COVID-19. The data can be used to support efforts to increase the number of people receiving boosters among the more than 1.1 million residents and 1.8 million staff at US nursing homes.
COVID-19 tests distributed to arriving international travelers at select US airports

Test kits distributed at Detroit Airport. Photo credit: Amanda Bierkamp, NCEZID
On December 13, 2021, the White House launched a month-long initiative encouraging international travelers to self-test for COVID-19 three to five days after arriving in the United States. To support this initiative, NCEZID staff members, with the help of colleagues from around CDC, distributed free self-test kits at eight airports: Chicago, Dallas, Detroit, Honolulu, Miami, Minneapolis, Philadelphia, and San Juan. The team offered over 400,000 test kits to travelers, regardless of their country of origin or departure, through January 15.
DRC and WHO declare the end of DRC’s 13th Ebola outbreak

On December 16, 2021, the Democratic Republic of the Congo’s (DRC) Ministry of Health and the WHO declared the end to the Ebola outbreak that began in eastern DRC on October 8. The outbreak—DRC’s thirteenth and its sixth since 2017—concluded with 11 cases (eight confirmed, three probable) and six deaths.
CDC provided remote technical assistance for surveillance, transmission chain tracking, laboratory efforts, infection and prevention control, vaccination efforts, social and behavioral science, and communications. CDC continues to provide support during the ongoing 90-day enhanced surveillance period. NCEZID is working with the CDC country office and DRC’s Ministry of Health to support the use of rapid diagnostic tests (RDTs) for cadaver surveillance. This involves collecting oral swabs from people who died, with their families’ approval, in communities where Ebola outbreaks recently occurred. When the result is positive, staff can take measures to prevent the additional spread of Ebola. RDTs have helped public health staff quickly identify and curb any flare-ups of an Ebola outbreak.
DRC’s last three Ebola outbreaks appear to be linked to either sexual transmission or relapse from survivors, highlighting the need to continue monitoring for Ebola and supporting programs for Ebola survivors after an outbreak has ended.
Dengue vaccine available

On December 16, 2021, NCEZID published an MMWR article summarizing the Advisory Committee on Immunization Practices recommendations for the first dengue vaccine approved by the US Food and Drug Administration. The vaccine is recommended for use in children 9–16 years old who have previously had a dengue infection and who live in areas where dengue is endemic. The vaccination campaign will begin in Puerto Rico in 2022, where 95% of US dengue cases occur. CDC is collaborating closely with the Puerto Rico Department of Health to assist with the rollout of this vaccine. Learn more about the dengue vaccine on our website.
Raising awareness about rabies

Photo credit: CDC
On January 6, NCEZID’s Rabies Team published an MMWR—Notes from the Field article about an uptick in human rabies cases in the United States in 2021.
Four of the five rabies cases in humans during 2021 were due to bat exposures, signaling a fresh need to warn people of the potential dangers of having contact with bats. The article also highlighted a possible lack of awareness about lifesaving postexposure prophylaxis (PEP) for rabies—or rabies shots—following contact with a rabid or possibly rabid animal.
The article underscored that people who have contact with a bat should seek immediate medical evaluation and potentially PEP. They should also try to have the bat tested for rabies with help from local health authorities.
Two new global CDC networks will combat antibiotic resistance
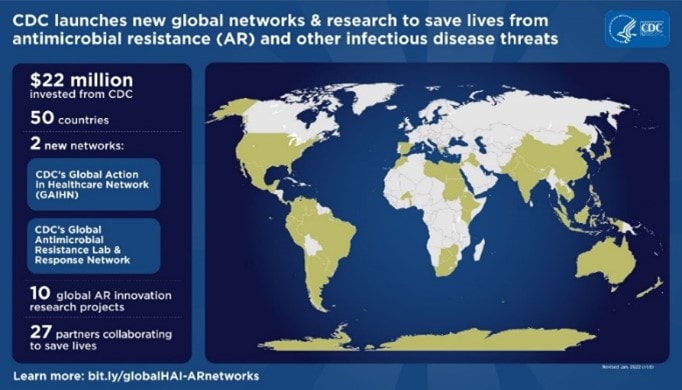
In December 2021, CDC announced it had awarded $22 million to 27 organizations, spanning more than 50 countries around the world. They will combat antibiotic resistance (AR) and other infectious disease threats through two new networks: the Global AR Laboratory and Response Network and the Global Action in Healthcare Network (GAIHN).
The Global AR Lab & Response Network will improve the detection of new and existing AR threats, identify risk factors that drive the emergence and spread of AR across the world, and strengthen efforts to respond on the ground to emerging threats. In collaboration with the Global AR Lab & Response Network, GAIHN will identify AR-related threats in healthcare settings and strengthen our partners’ efforts to prevent, detect, and respond to other infectious disease threats in healthcare settings.
CDC is also investing in short-term global AR innovation research to find public health solutions that save lives and transform the global response to AR across One Health.
Webinar on antibiotic resistance and animals

Nearly 700 people from 69 countries tuned in for NCEZID’s recent AMR Exchange webinar on the use of antibiotics in pets and livestock.
The panel was moderated by a CDC expert, and panelists included animal health experts from The Ohio State University, Iowa State University, and EpiX Analytics.
Discussions included:
- Implementing antibiotic stewardship programs and their effectiveness in animals using a One Health approach
- How antibiotic resistance impacts animal health
- How engaging pet owners and livestock producers can improve antibiotic stewardship and prescribing practices
If you missed the webinar, you can still watch the recording.
EID podcast spotlights Zika in Puerto Rico and antibiotic resistance in medical tourism
In a recent episode of the Emerging Infectious Diseases podcast, host Sarah Gregory spoke with NCEZID’s Dengue Surveillance and Research Laboratory Team Lead Jorge Muñoz-Jordan. They discussed the origin, spread, and evolution of the Zika virus in Puerto Rico from 2016 through 2017. In another recent episode, Sarah spoke with CDC medical officer Cal Ham about drug-resistant infections linked to medical tourism from the United States to Mexico.
You can find these episodes and many others in the Public Health Media Library.
Was It Something I Ate? Series features Harlee’s story about eating raw cake batter

Harlee, 11, was hospitalized with an E. coli infection after eating raw cake mix batter.
Eleven-year-old Harlee began having sharp stomach pains on a Saturday evening in July 2021, a week after tasting raw batter for a cake she had baked. Harlee’s story is the latest addition to the NCEZID series, Was It Something I Ate?, that includes accounts about people who developed serious infections from food or contact with an animal. Harlee was hospitalized with an E. coli infection, and an investigation found that her illness was part of a multistate outbreak linked to cake mix. You can read Harlee’s story on our Food Safety webpage.
2021 overview of Salmonella outbreaks linked to backyard poultry

In 2021, NCEZID staff collaborated with public health officials on 12 outbreak investigations of Salmonella infections linked to backyard poultry. Six strains of Salmonella were identified, and 1,135 people infected with outbreak strains were reported from 48 states, the District of Columbia, and Puerto Rico. Public health officials from several states found two of the outbreak strains in backyard poultry and backyard poultry environments of people who became infected. Of 293 ill people who reported having contact with backyard poultry and provided more information, 212 (72%) reported buying backyard poultry. CDC has conducted focus groups with backyard flock owners to assess their knowledge, attitudes, and behaviors around safe handling of backyard poultry and to improve how CDC communicates about preventing disease spread via poultry. CDC works with hatcheries and stores that sell poultry to reduce the spread of Salmonella at hatcheries and to educate new poultry owners on how to avoid getting and spreading Salmonella.
Campylobacter infections linked to water

Large-volume ultrafiltration to sample water from a municipal water tower. Photo credit: Mia Mattioli, NCEZID
In support of Nebraska’s Division of Public Health and its Department of Environment and Energy, NCEZID completed an environmental investigation of an untreated municipal drinking water system associated with a large outbreak of Campylobacter infections. Evidence of bird feces was found in a drinking water sample that contained Campylobacter DNA together with other bacteria associated with fecal matter. These findings suggested that bird droppings had contaminated the town’s water system, leading to the outbreak. In light of these results, the state water authority is requiring thorough inspection and repairs of the municipal water tower. Bird intrusion into water towers has previously been linked to drinking water associated with Campylobacter outbreaks.
Investigating outbreaks linked to food or animal contact

NCEZID is working with federal and state partners to investigate several multistate foodborne and animal contact outbreaks, including the following:
- E. coli infections linked to packaged salads. As of January 6, there were 10 illnesses and four hospitalizations from four states.
- Listeria infections linked to packaged salads produced by Dole. As of February 1, there have been 17 illnesses, 13 hospitalizations, and two deaths from 13 states. See Dole’s recall notice for the full list of recalled salads.
- Listeria infections linked to packaged salads produced by Fresh Express. As of December 22, from eight states, there have been 10 illnesses, 10 hospitalizations, and one death. On December 20, 2021, Fresh Express recalled several brands of packaged salad products.
- Salmonella infections linked to pet bearded dragons. As of January 11, there have been 44 illnesses and 15 hospitalizations from 25 states.