Our Work – 2021
Our Work - 2021
Imported monkeypox case reported in Maryland
CDC is working closely with the Maryland Department of Health and other partners in response to a confirmed case of monkeypox in a Maryland resident who recently returned to the United States from Nigeria. CDC has been working with international health counterparts, the airline, and state and local health officials in the Washington, DC area to contact airline passengers and others who may have had contact with the patient. As of November 16, a total of 13 passengers, 6 community contacts, and 26 healthcare personnel were identified, but these numbers may change as the investigation continues. Because all travelers were required to wear masks on the US-bound flight and in the US airport, it’s believed the risk of spread of monkeypox to others through respiratory droplets on the plane is low. CDC laboratory scientists confirmed that the patient had monkeypox and that the infection matches the strain that is found in West Africa. The patient is currently in isolation in Maryland. Monkeypox re-emerged in Nigeria in 2017 and since then there have been eight exported cases in people, including the Maryland case and a case in Texas in July.
CDC testing of commercial lab vials rules out smallpox
CDC worked closely with other federal and state officials to find quick answers to an urgent situation. On November 18, CDC laboratory testing showed that recently discovered commercial vials labeled as “smallpox” contained vaccinia (the virus used in smallpox vaccine), and that they were negative for variola, the cause of smallpox. The vials marked “smallpox” were found by a laboratory worker on November 15 while cleaning out a freezer in a facility that performs vaccine research in Pennsylvania. The freezer was immediately secured, and staff followed standard protocols for notifying CDC. The vials were sent securely to CDC for testing on November 18. This effort reflected excellent collaboration between CDC centers and others.
Publicly available genomic data continue to grow: 1M SARS-CoV-2 genomes published in GenBank
In late October, the United States achieved a major milestone when it surpassed 1 million SARS-CoV-2 genome sequences in GenBank, the National Institute of Health’s genome repository of publicly available DNA sequences. These data come from the combined efforts of CDC, commercial, academic, and public health labs across the country, and the success represents how these organizations are working to accelerate the collection of real-time pathogen sequence data for surveillance and for the COVID-19 pandemic response.
CDC completes COVID-19 Genomic Epidemiology Toolkit

CDC recently posted the final modules for the COVID-19 Genomic Epidemiology Toolkit Module 2.7, “Wastewater-based epidemiology of SARS-CoV-2,” describes how the city of Burlington, Vermont used municipal wastewater surveillance as part of an integrated approach to mitigate the spread of COVID-19 among its residents. Staff from CDC, the Vermont Department of Health, and Burlington collaborated on this module. Other recently added modules include Module 2.6, “Detecting and prioritizing SARS-CoV-2 variants,” and Module 3.6, “Sequencing strategies for SARS-CoV-2.” CDC also redesigned the toolkit’s website to improve navigation and make individual modules easier to share.
New travel quiz helps you determine whether you can board a flight to the United States

This fall, CDC issued several orders affecting air travel to the United States from foreign countries, in accordance with the President’s Proclamation. The new travel policies went into effect on November 8. CDC also released a Travel Requirements Quiz to help people determine whether they are eligible to fly to the United States based on their citizenship and vaccination status. The quiz lets travelers know what requirements apply to them and what documentation they need to provide. To learn more about CDC’s new orders and travel requirements, visit the International Travel webpage.
CDC supports access to COVID-19 vaccines for long-term care settings
To prepare for a COVID-19 booster shot recommendation in long-term care (LTC) settings this fall, CDC’s LTC/Pharmacy team developed resources and provided technical assistance for LTC providers and jurisdictions to ensure that residents and staff continued to have access to COVID-19 vaccines — their primary series, additional doses, and booster shots. As part of this effort, the team launched a new webpage outlining options that LTC providers and jurisdictions have to coordinate vaccine access either onsite or in the local community. In addition, the team conducted direct outreach to staff at LTC settings and worked closely with federal partners, LTC organizations, and state and local immunization programs to identify settings that needed additional support.
Epidemiology & Laboratory Capacity (ELC) launches new reopening schools webpage

ELC recently launched its ELC Reopening Schools webpage, featuring an overview of the $10 billion ELC Reopening Schools supplement and information on each of the recipients’ school COVID-19 screening testing programs. The page also includes helpful resources for launching or maintaining school screening programs, such as CDC’s school testing toolkit, which has received over 70,000 visits this year. The webpage has already garnered attention after being included in a White House fact sheet highlighting tools to help schools get started with screening testing, an important facet of President Biden’s Path Out of the Pandemic Action Plan. This webpage is one of many tools ELC uses to provide ongoing technical assistance to recipients and promote their efforts to keep schools open safely.
Foodborne and animal contact disease outbreaks

CDC, with federal and state partners, is investigating several multistate foodborne and animal contact outbreaks, including the following:
- Listeria infections linked to packaged salads produced by Dole. As of December 22, there have been 16 illnesses, 12 hospitalizations, and two deaths from 13 states. See Dole’s recall notice for the full list of recalled salads.
- Listeria infections linked to packaged salads produced by Fresh Express. As of December 22, there have been 10 illnesses, 10 hospitalizations, and one death from eight states. On December 20, Fresh Express recalled several brands of packaged salad products.
- Salmonella infections linked to onions. As of November 16, there have been 892 illnesses and 183 hospitalizations from 38 states and Puerto Rico. Multiple companies have recalled onions. FDA’s website has a table with information about each company’s recall.
- E. coli infections linked to baby spinach. As of December 3, there have been 14 illnesses and four hospitalizations from nine states. Minnesota officials found E. coli O157:H7 in a package of leftover Josie’s Organics baby spinach collected from a sick person’s home. Investigators are working to determine if additional products may be contaminated.
CDC’s Arctic Investigations Program analyzes health disparities in respiratory infections
Lower respiratory tract infections (LRTI) cause excessive illnesses and deaths among American Indian and Alaska Native (AI/AN) people. Alaska has some of the highest LRTI infection and hospitalization rates in the country, and patients often travel from remote areas for long hospital stays. CDC’s Arctic Investigations Program (AIP) recently published an analysis of LRTI during 1998–2014 that describes how LRTI rates decreased 38% among non-AI/AN adults but increased 26% among AI/AN adults. These findings help quantify the health disparities affecting AI/AN communities. In Alaska, crowded households and a lack of piped water likely contributed to high LRTI rates. AIP continues to focus on health equity by partnering with tribal organizations and researching the effects of infectious diseases on Alaska Native communities.
CDC Helps Post More Genomic Sequencing Data : 1M SARS-CoV-2 genomes published in GenBank

On October 29, the United States achieved a major milestone when it surpassed one million , genome sequences of SARS-CoV-2, the virus that causes COVID-19, in GenBank, the National Institute of Health’s genome repository of publicly available DNA sequences. These data come from the combined efforts of CDC, commercial, academic, and public health labs across the country, and the success represents how these organizations are working to accelerate the collection of real-time pathogen sequence data for surveillance and for the COVID-19 pandemic response.
CDC completes COVID-19 Genomic Epidemiology Toolkit
CDC posted the final modules for the COVID-19 Genomic Epidemiology Toolkit in November. Module 2.7, “Wastewater-based epidemiology of SARS-CoV-2,” describes how the city of Burlington, Vermont used municipal wastewater surveillance as part of an integrated approach to mitigate the spread of COVID-19 among its residents. Staff from CDC, the Vermont Department of Health, and local departments collaborated on this module. Other recently added modules include Module 2.6, “Detecting and prioritizing SARS-CoV-2 variants,” and Module 3.6, “Sequencing strategies for SARS-CoV-2.” CDC also redesigned the toolkit’s website to improve navigation and make individual modules easier to share.
New travel quiz helps you determine whether you can board a flight to the United States

On October 25, CDC issued several orders affecting air travel to the United States from foreign countries, in accordance with the President’s Proclamation. The new travel policies went into effect on November 8. CDC also released a Travel Requirements Quiz to help people determine whether they are eligible to fly to the United States based on their citizenship and vaccination status. The quiz lets travelers know what requirements apply to them and what documentation they need to provide. To learn more about CDC’s new orders and travel requirements, visit the International Travel webpage. As of December 31, 2021, some travel restrictions that were previously in place have been lifted.
CDC supports access to COVID-19 vaccines for long-term care settings

In early September, CDC staff on the Vaccine Task Force prepared for a COVID-19 booster shot recommendation in long-term care (LTC) settings. The LTC/Pharmacy team developed resources and provided technical assistance for LTC providers and jurisdictions to ensure that LTC residents and staff continued to have access to COVID-19 vaccines to receive their primary series and booster shots. As part of this effort, a new webpage was launched outlining options that LTC providers and jurisdictions have to coordinate vaccine access either onsite or in the local community. In addition, the team conducted direct outreach to staff at LTC settings and worked closely with federal partners, LTC organizations, and state and local immunization programs to identify settings that needed additional support.
Epidemiology & Laboratory Capacity (ELC) launches new reopening schools webpage

ELC recently launched its ELC Reopening Schools webpage, featuring an overview of the $10 billion ELC Reopening Schools supplement and information on each of the recipients’ school COVID-19 screening testing programs. The page also includes helpful resources for launching or maintaining school screening programs, such as CDC’s school testing toolkit, which was visited more than 70,000 times in 2021 . The webpage garnered attention after being included in a White House fact sheet highlighting tools to help schools get started with screening testing, an important facet of President Biden’s Path Out of the Pandemic Action Plan. ELC continues to provide ongoing technical assistance to recipients and promote their efforts to keep schools open safely through this webpage.
CDC lab testing confirms cause of US melioidosis cases

CDC announced on October 26 that bacteria in an aromatherapy spray found in the home of a melioidosis patient in Georgia genetically matched the bacterial strains in the Georgia patient and three other patients in Kansas, Minnesota, and Texas who had become infected since March. CDC had announced earlier in October that the aromatherapy spray tested positive for Burkholderia pseudomallei¸ the type of bacteria that causes melioidosis, a rare and sometimes fatal disease. The finding reported on October 26 enabled CDC scientists to confirm that the spray or one of its ingredients caused the four melioidosis infections.
Foodborne and animal contact disease outbreaks

CDC, with federal and state partners, is investigating several multistate foodborne outbreaks, including the following:
- Salmonella infections linked to seafood. As of October 8, there have been 102 illnesses and 19 hospitalizations from 14 states. Northeast Seafood Products of Denver, Colorado, has issued a recall of fresh seafood products sold to restaurants and grocery stores in Colorado.
- Salmonella infections linked to salami sticks. As of November 12, there have been 31 illnesses and six hospitalizations from 10 states. Do not eat, sell, or serve Citterio brand premium Italian-style Salame sticks. California officials found Salmonella in these products.
- E. coli infections linked to baby spinach. As of November 15, there have been 10 illnesses and two hospitalizations from seven states. Josie’s Organics prepackaged baby spinach may be contaminated with E. coli and may be making people sick.
NCEZID releases new climate change and infectious disease report

Lyme disease, West Nile virus disease, and Valley fever are just some of the infectious diseases that are on the rise and spreading to new areas of the United States. Milder winters, warmer summers, and fewer days of frost make it easier for these and other infectious diseases to expand into new geographic areas and infect more people. To understand climate change’s impact, it’s important to look at some of the common ways these diseases spread—through water, fungi, mosquito and tick bites, and contact with animals. National Center for Emerging Zoonotic Infectious Diseases (NCEZID) scientists are at the forefront of CDC’s work, as part of a national effort, to protect the public’s health from the harmful effects of climate change. We recently released a report [PDF – 4 pages] that highlights some of that work and looks ahead to the important work yet to come.
Cross-border collaboration advances global health security in Africa

In collaboration with the implementing partner Baylor-Uganda and the Democratic Republic of the Congo (DRC) and Uganda CDC country offices, CDC’s Global Border Health Team (GBHT) successfully brokered an in-person, binational, cross-border meeting between the DRC and the Uganda ministries of health. Participants in this meeting advanced cross-border information sharing commitments by finalizing a draft memorandum of understanding for collaboration. The meeting also featured a binational discussion on population movement patterns through and outside of identified points of entry between the countries using GBHT’s Population Connectivity Across Borders (PopCAB) toolkit. The meeting’s success advanced global health security in both countries by strengthening their partnership and heightening capacity for cross-border surveillance for communicable diseases and information sharing. The countries plan to rapidly leverage this momentum to address their responses to COVID-19, Ebola, plague, and meningitis.
NCEZID leads public health response for Operation Allies Welcome
About 124,000 people fleeing Afghanistan after the drawdown of US forces there have been airlifted out, and the US government is resettling evacuees in a humanitarian effort called Operation Allies Welcome (OAW), led by the Department of Homeland Security. CDC activated on September 9 under its Graduated Response Framework to head up OAW’s public health response, with the National Center for Emerging and Zoonotic Infectious Diseases (NCEZID) as the lead center. Staff from NCEZID and other CDC centers are supporting the response, which is focused on protecting the health of evacuees and taking precautions to prevent the spread of infectious diseases during resettlement. CDC staff have deployed to assist with public health operations at Department of Defense bases in the United States and Germany. These efforts include surveillance, infection control and prevention, quarantine and isolation, environmental health assessments, and vaccinations.
Pilot surveillance program focuses on identifying new SARS-CoV-2 variants entering the United States
CDC is collaborating with two industry partners on a pilot surveillance program to detect novel SARS-CoV-2 variants among incoming international travelers. In October, CDC launched the program at John F. Kennedy, San Francisco, Oakland, and Newark Liberty airports, focusing on travelers from India. The pilot features two types of testing: pooled PCR testing of passengers at the airport and take-home PCR tests for participating travelers to take within three to five days after their arrival and mail back. Specimens that test positive will be genetically sequenced. Both types of tests will enable detection of novel variants and help CDC understand more about variants spread through travel.
CDC issues new policies for safely resuming international travel
On October 25, the president issued a Proclamation on Advancing the Safe Resumption of Global Travel During the COVID-19 Pandemic, directing CDC to implement public health requirements for air travelers. CDC issued several orders with operational details, including a requirement for proof of vaccination and changes to COVID-19 testing requirements. These new travel policies became effective November 8, 2021. CDC also extended the Framework for Conditional Sailing Order (CSO) for cruise ship orders with minor modifications.
CDC also developed a Travel Requirements Quiz to help international travelers (US citizens and non-US citizens) learn what is required before traveling to the United States by air.
For more information, please visit the following pages:
- International Travel Landing Page
- International Travel: Information for US Citizens, US Nationals, US Lawful Permanent Residents, and Immigrants
- Non-US citizen, Non-US immigrants: Air Travel to the United States
- Travel Requirements Quiz
- Cruise Ship Travel During COVID-19
As of December 31, 2021, some travel restrictions that were previously in place have been lifted.
One Health Office investigates SARS-CoV-2 infection in otters at the Georgia Aquarium

The One Health Office (OHO) has been collaborating with state, tribal, local, and territorial partners to investigate outbreaks of COVID-19 infections in people and SARS-CoV-2 in animals since the beginning of the pandemic, helping us learn more about this emerging disease. One investigation close to home in Atlanta is highlighted in a new CDC Responder Story about how CDC collaborated in the spring with the Georgia Aquarium, the Georgia Department of Public Health, and other health partners to investigate infections of SARS-CoV-2, in Asian small-clawed otters.
CDC helped coordinate the genomic and diagnostic analysis of samples to support the epidemiologic investigation. This work contributes to our understanding of susceptibility to SARS-CoV-2 in animals and people. It also helps us understand whether mutations and human-to-animal transmission are possible. One Health investigations like this are crucial for controlling zoonotic diseases and preventing future outbreaks.
CDC estimates the impact of COVID-19 case investigation and contact tracing
Successful COVID-19 case investigation and contact tracing (CICT) have been challenged by limited resources and varying levels of public acceptance. Before the introduction of vaccines, health departments relied heavily on CICT and other nonpharmaceutical interventions (NPIs) like wearing masks to slow the spread of COVID-19. An analysis by CDC’s Health Economics and Modeling Unit of CICT’s use among 14 jurisdictions from June through October 2020 showed CICT programs notably reduced the transmission of COVID-19 in most locations and that the speed of contact notification had the greatest influence on program success. The researchers used the COVIDTracer Advanced modeling tool to estimate that CICT prevented 4%–97% expected cases and hospitalizations (after accounting for the effect of other NPIs). This variability suggests that there is an opportunity to improve CICT further. In a paper recently posted to a pre-print site (COVID-19 cases and hospitalizations averted by case investigation and contact tracing in the United States | medRxiv) , the same authors used a similar methodology to analyze CICT program data from 22 states and 1 territory, representing 42.5% of the US population (140 million people). They estimated that CICT programs prevented 1.11 million to 1.36 million cases and 27,231 to 33,527 hospitalizations over 60 days during the height of the pandemic (winter 2020-2021).
Identifying successful approaches to boils prevention in an Alaska Native village
In many villages in rural Alaska, running water is a luxury. Households lack indoor plumbing, leading residents to reuse water when washing hands, bathing, and doing laundry. As a result, people living in these villages are disproportionately affected by boils and other skin and soft tissue infections (SSTIs). CDC’s Arctic Investigations Program worked with tribal partners to investigate how to prevent boils in one village. The team promoted prevention practices and provided participating households with a kit of supplies to support these activities. They surveyed participants about the acceptability of the recommendations and found that most households did adopt new practices. The investigation highlighted that a household intervention that includes a combination of practices could be used to prevent SSTIs.
CDC staff discover evidence of Cache Valley virus transmission through blood transfusion
When a kidney transplant patient began showing symptoms of viral encephalitis about a month after transplant, CDC staff stepped up to verify the cause of infection and determine how the recipient was infected. They confirmed the cause as Cache Valley virus and worked through a list of 18 people, including the organ donor, other organ recipients, and blood donors, to identify the source of infection. Staff from health departments, donor organizations, and CDC’s Office of Blood, Organ, and Other Tissue Safety assisted with the investigation and determined that a blood donor had transmitted the virus. Previously, Cache Valley virus was only known to be transmitted through the bite of an infected mosquito. These findings increase our understanding of how the virus can be transmitted.
Foodborne and animal contact disease outbreaks

CDC, with federal and state partners, is investigating several multistate foodborne and animal contact outbreaks, including the following:
- Salmonella infections linked to backyard One in four sick people is a child younger than five years old. As of August 31, there are 863 illnesses, 209 hospitalizations, and two deaths from 47 states and the District of Columbia.
- Salmonella infections from an unknown food source. A specific food item has not yet been identified as the source of this fast-growing outbreak. As of September 30, there are 419 illnesses and 66 hospitalizations from 35 states.
- Salmonella infections linked to seafood. As of October 8, there have been 102 illnesses and 19 hospitalizations from 14 states.
Lyme disease clinician training modules launched
Recently, CDC released clinician training modules for Lyme disease. This four-part series will help frontline healthcare providers recognize, diagnose, and treat Lyme disease, an increasingly common tickborne illness. The free online curriculum serves as a valuable resource for primary care clinicians, public health professionals, pharmacists, and health educators who encounter patients with Lyme disease. Free continuing education credits are available. To register, go to CDC TRAIN. You can also view the modules here:
- Module 1: Introduction to Tickborne Diseases and Disease Prevention
- Module 2: Lyme Disease Clinical Overview
- Module 3: Lyme Disease Testing and Diagnosis
- Module 4: Lyme Disease Treatment and Management
CDC responds to multistate outbreak of Tuberculosis (TB) associated with a contaminated bone allograft material
On September 9, CDC published Notes from the Field: Tuberculosis Outbreak Linked to a Contaminated Bone Graft Product Used in Spinal Surgery—Delaware, March–June 2021 in Morbidity Mortality Weekly Report (MMWR). In May, a Delaware hospital notified the Delaware Division of Public Health (DPH) of a TB cluster among patients who had spinal surgery using a bone graft product. An investigation by DPH, CDC, and FDA found high rates of spinal and disseminated TB disease following implantation of the material. CDC recommended immediate treatment for TB disease for all patients and provided guidance to healthcare facilities to protect healthcare personnel and other patients who might have been exposed to M. tuberculosis during surgery and other patient care.
CDC staff co-author Candida auris article in Annals of Internal Medicine
In a recent article published in the Annals of Internal Medicine, CDC’s Mycotic Diseases Branch staff and partners describe how an aggressive regional public health approach to Candida auris found in an Orange County long-term acute care hospital was able to limit the spread of this deadly threat in 2019. Strong, coordinated public health action—including surveillance and admission screening of patients in at-risk facilities, antifungal susceptibility testing and whole genome sequencing by CDC’s AR Lab Network, and ongoing infection control assessments and improvements—can make a difference in the fight against C. auris.
CDC’s response to climate change

Climate change is expected to affect human health in many ways. Some existing threats will intensify, some new threats will emerge, and not everyone is equally at risk. CDC staff will play key roles in an agency-wide effort to meet those changing threats. Their work will help CDC address the public health implications of climate change strategically and develop a framework for action. Check out the CDC’s report [PDF – 4 pages] and read more about the CDC’s response to climate change.
CDC featured on EYE on Yellow Fever podcast

Outbreaks of yellow fever in urban centers could pose a global health risk. Listen to WHO’s latest podcast, featuring CDC’s own Dr. Erin Staples, to learn more about how yellow fever spreads and what can be done to stop it. https://www.who.int/initiatives/eye-strategy/podcast.
U.S. publishes over 1M SARS-CoV-2 sequences through monumental public health effort
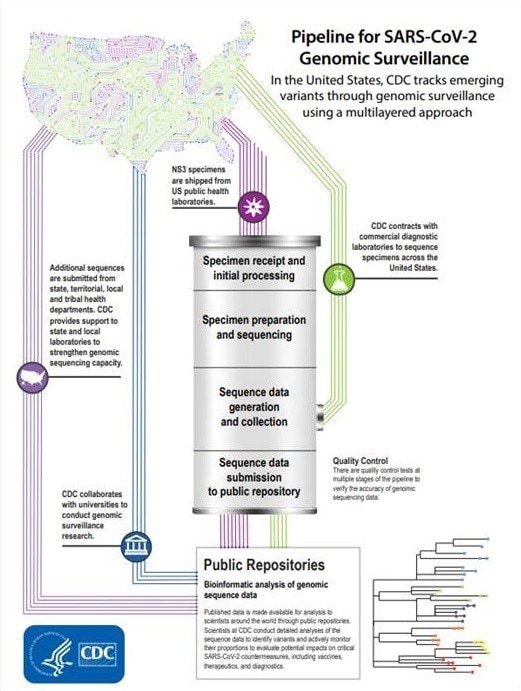
In September, the United States hit a major milestone in COVID-19 surveillance when it surpassed 1,000,000 U.S. SARS-CoV-2 genome sequences submitted to GISAID, a public data repository. By routinely acquiring sequences from a subset of COVID-19 cases, it is possible to monitor the spread of the virus across time and within populations. The combined efforts of academic, private, and public health laboratories across the nation have provided data that will improve surveillance and scientific inquiry now and for years to come. Learn more about CDC’s role in tracking variants.
CDC’s report on foodborne disease trends shows marked declines during the COVID-19 pandemic
Reported infections from pathogens commonly transmitted through food decreased markedly in the United States in 2020, according to a preliminary report from CDC’s Foodborne Diseases Active Surveillance Network (FoodNet). Overall incidence of infections caused by eight pathogens fell by 26%—the largest single-year change during 25 years of FoodNet surveillance. Reported incidence decreased for almost all these pathogens. Infections with Campylobacter and Salmonella remain the most commonly reported. Public health measures intended to slow the spread of SARS-CoV-2 likely contributed to this decline. Understanding the effects of the COVID-19 pandemic on people’s behaviors related to food could help us identify prevention measures and strategies for targeting particular pathogens and foods.
New data show an increase in healthcare-associated infections during the COVID-19 pandemic
The COVID-19 pandemic created a perfect storm for healthcare-associated infections (HAIs) and antibiotic resistant (AR) infections in healthcare settings. A new study from CDC is the first comprehensive look at the impact of COVID-19 on the incidence of HAIs that harm patients in U.S. hospitals. The findings show substantial increases nationally in HAIs and select AR infections in 2020 compared with 2019. For most of these infections, the increases seen in 2020 present a strong contrast to the success in reducing these infections prior to the pandemic.
CDC will continue to invest in healthcare infection prevention, training for all healthcare providers, and making certain that healthcare facilities and partners in state and local health departments have actionable data to ensure that patients who need healthcare during the pandemic and beyond are able to receive care safely.
CDC will invest $2.1 billion to protect patients and healthcare workers
In September, the Biden-Harris Administration announced a $2.1 billion investment to improve infection prevention and control (IPC) activities across the U.S. public health and healthcare sectors. This funding will allow the United States to strengthen and equip state, local, and territorial public health departments and other partner organizations with the resources needed to better fight infections, including COVID-19 and other known and emerging infectious diseases, in U.S. healthcare facilities.
Over the next three years, CDC will issue $1.25 billion of the total to 64 state, local, and territorial health departments to support this work. Part of the initial funding to jurisdictions this October will support new state-based nursing homes and long-term care strike teams to protect our most disproportionately affected population from COVID-19. The new funds will also be used to strengthen IPC capacities nationwide.
COVID-19 healthcare infection control guidance updated
In September, the COVID-19 Infection Prevention and Control (IPC) team updated its COVID-19 IPC guidance for healthcare personnel to further clarify, consolidate, and simplify existing healthcare IPC guidance. The updates streamline existing setting-specific guidance for staff in dental offices, assisted living facilities, EMS, and dialysis clinics and combine them into the main IPC guidance for healthcare settings. These updates will make it easier for healthcare personnel to find relevant guidance for the duration of the COVID-19 response. Core IPC measures like encouraging vaccination and testing; use of source control by healthcare personnel, patients, and visitors; and use of personal protective equipment by healthcare personnel caring for patients with suspected or COVID-19 infection continue to be emphasized to protect both patients and personnel in healthcare settings.
CDC releases new Technical Instructions for COVID-19
CDC’s Immigrant, Refugee, and Migrant Health Branch staff developed new COVID-19 Technical Instructions for civil surgeons and panel physicians. The new Technical Instructions incorporate COVID-19 vaccination for all people applying for lawful permanent resident status in the United States, people applying for immigrant or refugee status abroad, and non-immigrants who are required by the local U.S. embassy to have an overseas medical examination. The Technical Instructions are effective from October 1, 2021, until CDC determines that the instructions are no longer needed to prevent the importation and spread of COVID-19.
Wastewater surveillance and environmental metagenomics featured in Association of Public Health Laboratories (APHL) newsletter

Mia Mattioli prepares a sample of wastewater for PCR molecular testing.
CDC’s Mia Mattioli co-authored an article with National Center for Biotechnology Information (NCBI) scientist Ilene Mizrachi for the APHL summer issue of Lab Matters. The article features CDC and NCBI’s development of a metadata package for sequence data from wastewater surveillance to support the COVID-19 response. CDC launched the National Wastewater Surveillance System last September and has since partnered with over 40 state and local public health jurisdictions to monitor infection trends through wastewater. The standardized metadata submission will provide the infrastructure needed to connect wastewater genomic data around the globe and guide rapid public health decision-making about SARS-CoV-2 and other health concerns.
Foodborne disease outbreak investigations

CDC, with federal and state partners, is investigating several multistate foodborne and animal contact outbreaks, including the following:
- Salmonella infections linked to raw frozen breaded stuffed chicken products. As of August 11, there were 28 illnesses and 11 hospitalizations reported from eight states.
- Salmonella infections linked to frozen cooked shrimp. CDC identified three new illnesses related to this outbreak since the final update was posted on July 21. As of August 17, there were nine illnesses and three hospitalizations reported from four states.
- Salmonella infections linked to Italian-style meats. As of August 26, there were 36 illnesses and 12 hospitalizations reported from 17 states.
Melioidosis cluster expands to fourth state
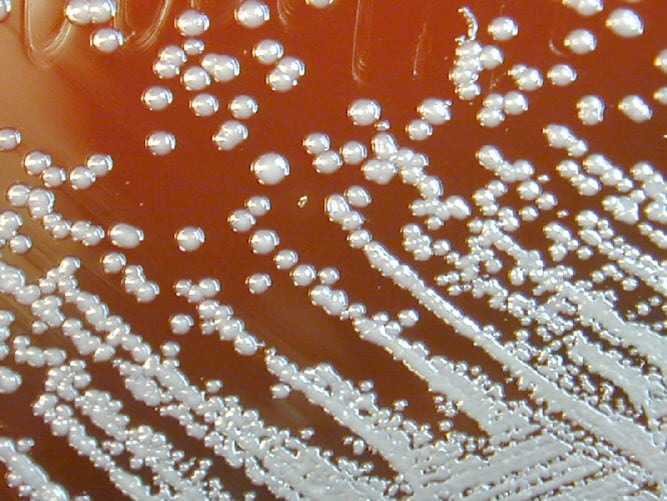
B. pseudomallei on a Petri dish culture plate. Image courtesy of Larry Stauffer, Oregon State Public Health Laboratory
Georgia’s Laboratory Response Network notified CDC in late July of a Burkholderia pseudomallei isolate from a deceased patient. Based on genomic analysis, this isolate from the Georgia patient closely matches the isolates from three cases previously identified in Kansas, Minnesota, and Texas over the past several months, indicating all four patients most likely share a common source of exposure. Melioidosis is typically found in tropical climates, especially Southeast Asia and northern Australia. Most previous cases reported in the United States have been in people who had recently traveled to a country where the disease is widespread, but the four people described here had no recent history of traveling outside of the United States. The investigation to find the source is ongoing.
CDC responds to multistate outbreak of TB associated with a contaminated bone allograft material
CDC is working with state and local health departments in 20 states and other partners to respond to tuberculosis (TB) cases in 115 patients who had spinal surgery using a single contaminated product lot of a bone allograft material. The manufacturer issued a voluntary nationwide recall of the single contaminated product lot linked to these patients on June 2. Through prompt action and collaboration among CDC, the US Food and Drug Administration, health departments, hospitals, and surgeons, all patients exposed to this product lot have been evaluated and have started treatment for TB disease. CDC is providing guidance and assistance to evaluate healthcare personnel and other patients exposed to TB during surgery and patient care activities. EIS officers and CDC staff provided in-person support to two states via Epi Aids; they described the initial cluster of patients and healthcare exposures, supported clinical management, and helped with contact tracing, notification, and testing for latent TB.
CDC and Wadsworth Center Laboratory pilot same-day whole genome sequencing for anthrax preparedness
If an intentional release of Bacillus anthracis happened, quick detection of antimicrobial resistance (AMR) would inform decisions about which antibiotics would be most effective in saving lives. CDC’s BioDefense Research and Development (BDRD) laboratory completed a pilot study of a method for rapid whole genome sequencing (WGS) of B. anthracis in collaboration with the Association of Public Health Laboratories and the New York State Department of Health Wadsworth Center, a member of the Laboratory Response Network (LRN). Laboratory scientists analyzed 11 strains of B. anthracis in a BSL-3 lab using sequencers and developed protocols, completing WGS and on-site analysis using a custom bioinformatics software. Laboratory scientists profiled plasmids, mutations, and genetic signatures associated with AMR in under five hours, demonstrating that trained LRN laboratory staff could rapidly sequence and analyze B. anthracis, strengthening public health emergency preparedness.
CDC and EID team up for a podcast episode
Emerging Infectious Diseases podcast host Sarah Gregory recently hosted an episode with CDC’s Cita Medalla, lead author of Increased Incidence of Antimicrobial-Resistant Nontyphoidal Salmonella Infections, United States, 2004–2016. You can listen to the episode on the Public Health Media Library.
Report on drug-resistant C. auris transmission in healthcare facilities

Illustration of Candida auris fungal organisms. Medical illustrator: Stephanie Rossow
MMWR recently published an article on transmission of Candida auris in healthcare facilities. While C. auris infections are more resistant to antifungal medications than similar fungal diseases, reports of resistance to all three classes of antifungal drugs (pan-resistant) have been rare in the United States. Among 101 cases in Washington, DC during January–April 2021, three pan-resistant cases were identified in residents at a long-term care facility. Among 22 cases in Texas during January–April 2021, two were pan-resistant and five were resistant to echinocandins and fluconazole. Each cluster involved common health care encounters and no known previous echinocandin exposure, suggesting transmission of pan- and echinocandin-resistant strains for the first time in the United States. Early identification through screening and testing, rigorous infection control, and coordinated communication are essential to help prevent the spread of C. auris, particularly pan-resistant and echinocandin-resistant strains. Antifungal susceptibility testing is critical to identify resistance and prevent spread.
Project Firstline launches refreshed website, new materials

Continuing its efforts to provide easily accessible infection control content and training materials to frontline healthcare workers, CDC’s Project Firstline launched a refreshed website with exciting new features. The website now makes it easier to find infection control information on a variety of topics and learn what Project Firstline’s partner organizations are doing. New features and webpages include the following:
- Redesigned Facilitator Toolkit page
- Enhanced Partnerships page
- New Project Firstline Community College Collaborative page
- New Multimedia Resources page
- Details on how to earn continuing education (CE)
Project Firstline also released a new suite of materials to help healthcare workers learn how to use multi-dose vials safely when giving injections, like the current COVID-19 vaccines. Resources include an explainer video, printable checklist [PDF – 1 page], and a training session plan and presentation. Stay tuned for more new materials coming soon on the Project Firstline website.
CDC launches school testing communication toolkit

This social media message is part of the school testing communication effort
CDC launched a COVID-19 School Testing Toolkit on August 25 to support schools that are implementing COVID-19 testing programs this fall. Many students are returning to in-person learning, and some schools are offering free, routine COVID-19 testing for students and staff. This type of regular testing—in addition to COVID-19 vaccination, physical distancing, and mask wearing—helps protect students, staff members, family members, and others who are not vaccinated against COVID-19 or are otherwise at risk for getting seriously sick from COVID-19. These regular testing programs can serve as an early warning system to help prevent an outbreak before it starts. The toolkit, posted to CDC’s COVID-19 website, provides materials that jurisdictions, schools, and health departments can customize and use to encourage participation in school COVID-19 testing programs to help keep schools open while students take part in activities they love. The communication effort includes posters, flyers, customizable letters, FAQs, social media ads, and outreach to partners.
CDC and the National Resource Center for Refugees, Immigrants, and Migrants support vaccination efforts
CDC’s partnership with the National Resource Center for Refugees, Immigrants, and Migrants (NRC-RIM), funded by CDC/HHS, supports health departments and community organizations working with RIM populations, and has a breadth of communication resources in over 40 languages about COVID-19 vaccines, case investigation/contact tracing, and community engagement. NRC-RIM works to encourage vaccination, dispel myths, and provide culturally and linguistically appropriate resources such as
- Vaccine Central—a website that supports vaccine planning, checklists to follow, vaccine knowledge, and confidence among RIM communities
- Get the Facts—a campaign with facts about COVID-19 vaccines in fully customizable and translated social media, fact sheets, and audio/video public service announcements
- Vaccination Is—a campaign, with a community-led, hyperlocal communications strategy developed by, and for members of, narrowly defined local groups to align with their communities’ unique priorities and values, and the expertise of local leaders
Genomic Epidemiology Toolkit developers partner with Africa CDC’s Institute of Pathogen Genomics
This summer, CDC participated in a live Q&A session for a next-generation training event on SARS-CoV-2, the virus that causes COVID-19. The event was held by Africa CDC’s Next Generation Sequencing Academy for the Pathogen Genomics Initiative. NGS Academy Training Coordinator Kirsty Lee Garson incorporated the four introductory modules from CDC’s COVID-19 Genomic Epidemiology Toolkit into a virtual course for personnel involved with SARS-CoV-2 surveillance at African institutions.
The training materials “laid the foundation for the course,” said Kirsty. “It was a privilege to partner with [the CDC] on this training initiative.”
An important part of CDC’s response to the COVID-19 pandemic is the financial and technical assistance provided to public health partners. Here are two recent examples of support awarded to state, local, and territorial health departments through NCEZID’s Epidemiologic and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases Cooperative Agreement (ELC).
- In July, CDC awarded $33 million to ELC recipients in support of the National Wastewater Surveillance System (NWSS), an initiative launched in 2020 to test wastewater (sewage) for evidence of the virus that causes COVID-19 and other emerging pathogens (microorganisms that can cause disease). This project is managed by CDC in collaboration with state and local health departments and national public health organizations. Once fully operational, NWSS will be able to identify signals of possible outbreaks sooner than traditional clinical testing and reporting.
- In partnership with the Department of Justice’s Office of Justice Programs and the U.S. Department of Health and Human Services (HHS), CDC is distributing $700 million to 64 ELC recipients to detect and help prevent the spread of COVID-19 in confinement facilities, including prisons, jails, and juvenile confinement facilities. Among other things, these funds will allow facilities to launch COVID-19 diagnostic and screening programs for people who reside in these facilities, as well as staff and visitors.
SPHERES Initiative funding

CDC issued 22 awards as part of our Office of Advanced Molecular Detection’s (OAMD) SARS-CoV-2 Sequencing for Public Health Emergency Response, Epidemiology, and Surveillance (SPHERES) Initiative. These awards, along with the seven awarded in 2020, fund innovative research and collaborative projects to fill knowledge gaps and expand innovative responses to the COVID-19 pandemic. Some of the projects will:
- integrate viral genomic data with patient data and other relevant data to better understand patient risk factors and clinical outcomes;
- compare strains of the virus that cause COVID-19 between geographic regions; and
- develop or enhance public bioinformatics software, tools, and databases to improve the availability, use, and impact of genomic data in the public domain for the virus that causes COVID-19.
- These awards are meant to help combine the efforts of academic and research institutions with those of public health agencies. They will substantially add to the availability of timely genomic surveillance data and maximize the usefulness of those data for public health.
Clinical Infectious Diseases journal publishes a supplement on the public health response to COVID-19

Clinical Infectious Diseases’ recent supplement, published online on July 15, looks back at the public health response to COVID-19 and what can be learned and built upon during the next phase of this pandemic and for future pandemics. “A Snapshot of Work by the COVID-19 Public Health Response” touches on five major themes: clinical outcomes of long-term COVID-19 infection, COVID-19 testing, COVID-19’s interaction with other infectious diseases, how COVID-19 disproportionately affects persons, and COVID-19 transmission. The supplement’s cover features a photo of CDC medical officer Kiran Mayi Perkins.
Foodborne disease outbreak investigations

CDC, with federal and state partners, is investigating several multistate foodborne and animal contact outbreaks, including the following:
- Listeria infections linked to fully cooked chicken. As of July 14, there were three illnesses and one death from two states. Tyson Foods Inc. recalled over 8 million pounds of ready-to-eat chicken products. CDC advises people not to eat, sell, or serve recalled products. You can find the lists of recalled products on USDA’s and FDA’s
- Salmonella infections linked to BrightFarms packaged salad greens. As of July 28, there were 11 illnesses and no deaths from three states. BrightFarms recalled salad greens produced in Rochelle, IL.
- Salmonella infections linked to raw frozen breaded stuffed chicken products. As of August 11, there were 28 illnesses eight states. All products have the establishment number “P-2375” inside the U.S. Department of Agriculture’s mark of inspection. See recall notice for more details.
- Salmonella infections linked to cooked frozen shrimp. CDC identified three new illnesses in this outbreak since the final update was posted on July 21, 2021. At least one of these people ate frozen cooked shrimp supplied by Avanti Frozen Foods that was not part of the initial recall on June 25, 2021. On August 13, 2021, Avanti expanded their recall to include additional brands and expiration dates of frozen cooked shrimp products.
- E. coli infections linked to cake mix. As of July 28, there were 16 illnesses, seven hospitalizations, and no deaths from 12 states. Seventy-five percent of sick people are children younger than 18.
CDC responds to a case of rabies in an imported dog

In June, CDC was notified about a dog in Pennsylvania that tested positive for rabies. The dog was among a group of 33 dogs and one cat imported into the United States from Azerbaijan by an animal rescue group. CDC coordinated a response to this case with multiple state health departments, the US Department of Agriculture, and US Customs and Border Protection. Public health officials identified at least 19 people who have been exposed to the rabid dog. CDC testing indicated that the rabid dog was infected in Azerbaijan before it arrived in the United States. This rabies case highlights the critical importance of CDC’s recent temporary suspension of the importation of dogs into the United States from countries with a high risk of dog rabies as well as the potential cost to taxpayers. With every imported dog rabies case, the cost for the investigation is half a million dollars. The suspension went into effect on July 14.
Monkeypox case investigation update
CDC led the investigation of monkeypox in a US resident who traveled from Nigeria to the United States in mid-July. The coordinated response involved more than 70 CDC staff who identified contacts, performed laboratory testing, developed guidance to categorize exposed persons into risk categories, and oversaw the first ever use of TPOXX, an antiviral medical countermeasure stockpiled by the Strategic National Stockpile among many other tasks. Currently, over 150 contacts have completed their monitoring period without any incident (i.e., airline passengers who were on the flight with the patient, laboratorians from an out-of-state lab that processed clinical samples, and family members). Some healthcare workers who cared for the patient continue to be monitored but are low risk for acquiring infection because appropriate personal protective equipment was worn. Tools developed and posted to the CDC website will be used if additional cases are imported from Nigeria.
Melioidosis cluster expands to 4th state
In late July, Georgia’s Laboratory Response Network notified DHCPP’s Bacterial Special Pathogens Branch of a Burkholderia pseudomallei isolate from a deceased patient. Based on genomic analysis, this case in Georgia closely matches the three cases previously identified in Kansas, Minnesota, and Texas in the past several months, indicating they most likely share a common source of exposure. Melioidosis is typically found in tropical climates, especially Southeast Asia and northern Australia. Most previous cases reported in the United States have been in people who had recently traveled to a country where the disease is widespread. However, the four people recently diagnosed with melioidosis had no recent history of traveling outside of the U.S. The investigation to find the source is ongoing.
CDC publishes updated guidelines for treatment and prophylaxis of plague

Plague has killed millions worldwide and still causes disease in the United States and around the world. A bioweapon attack with Yersinia pestis, the bacterium that causes plague, could potentially infect thousands, requiring rapid and informed decision-making by clinicians and public health agencies to prevent deaths. Fortunately, plague is treatable with antibiotics. After a comprehensive review process, CDC staff developed new plague treatment and prophylaxis recommendations for clinicians and public health officials. These guidelines include several important updates and expanded antibiotic treatment options. The recommendations can inform the treatment of patients infected with plague by naturally occurring means (most commonly by the bite of an infected rodent flea) or in the aftermath of a bioterrorist attack.
Laboratory-associated Zika virus infections explored

Laboratory-associated Zika virus.
When the Zika virus began spreading through the Americas in 2015, the risk for being exposed to the virus increased for laboratory workers and biomedical researchers. Four cases of laboratory-associated Zika virus disease in the United States reported during 2016–2019 are described in a recent article in NCEID’s journal Emerging Infectious Diseases. Two of these cases were associated with needlestick injuries. For the other two cases, the route of transmission was undetermined. In laboratories where work with Zika virus is performed, good laboratory biosafety practices must be practiced to reduce the risk for infection among laboratory personnel.
In this podcast, author Susan Hills talks about these four U.S. cases and notes that decades ago, one of the earliest reports of human Zika virus infection could have been acquired in a laboratory. That case occurred in 1963 when a researcher was working in a laboratory in Uganda with Zika virus strains that he had isolated from mosquitoes.
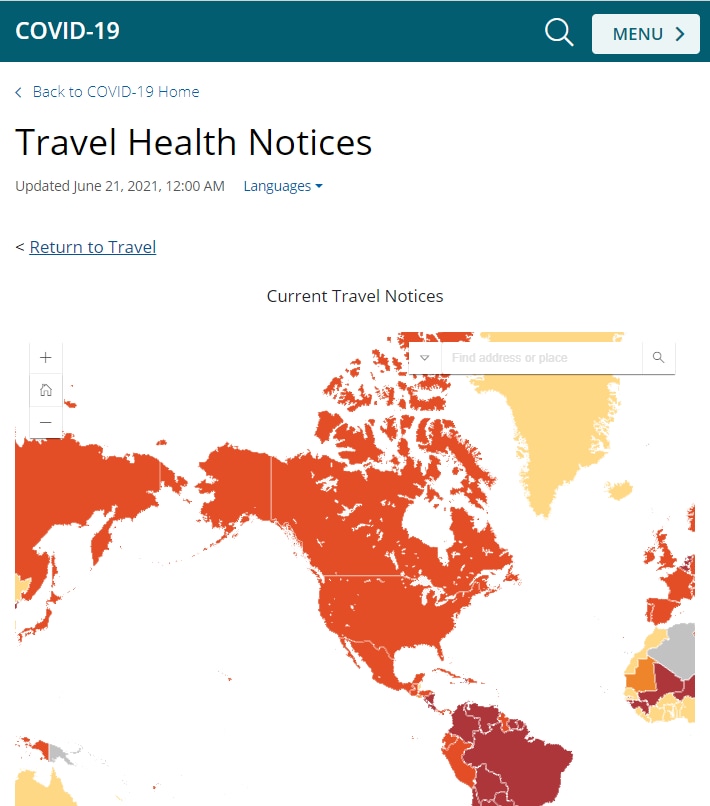
To reflect the current state of the COVID-19 pandemic, NCEZID updated the COVID-19 Travel Health Notices (THN) to better differentiate countries with severe outbreak situations from countries with sustained, but controlled, COVID-19 spread. THNs give specific travel advice for vaccinated and unvaccinated people according to the THN level. Every Monday, THNs are updated to ensure levels reflect the current global situation and are aligned with guidance for international travel.

On June 5, 1981, CDC published a description of the first known cases of what would come to be known as AIDS. The pandemic disease has since infected 75 million people globally, killing 32 million. To mark the 40th anniversary of the discovery of HIV/AIDS, three veteran CDC leaders in the response to the pandemic offer their reflections in a June 2021 article in NCEZID’s journal Emerging Infectious Diseases. The authors examine efforts of the past 10 years with an emphasis on biomedical solutions to treat HIV/AIDS and slow its spread. They also describe other strengths of the response to HIV/AIDS from funding to coordination and tragic challenges like discrimination and exclusion. The response to HIV/AIDS has been useful in gauging successes and challenges in pandemic responses that have followed. The authors remind us that, despite medical advances that are saving millions of lives, HIV/AIDS is far from over, and the global rate of new infections is high.
In this podcast, author Dr. Kevin De Cock relates the fight against HIV/AIDS to other disease responses, including to COVID-19.
On June 19, the Republic of Guinea (Guinea) Ministry of Health and the World Health Organization declared the end of the Ebola outbreak in Guinea. A total of 23 cases and 12 deaths were reported in the outbreak that was announced on February 14, 2021. A 90-day enhanced surveillance period began at the end of the outbreak to ensure that any new case is quickly detected and responded to. In the Democratic Republic of the Congo, the post-outbreak 90-day enhanced surveillance period, which began on May 3, 2021, is set to end in early August if there are no new cases.

Starting July 14, 2021, new rules were enacted to prohibit dogs entering the United States from countries at high risk for dog rabies. CDC has published a Federal Register Notice [PDF – 43 pages] announcing the temporary suspension. There is an exception: dogs with a CDC Dog Import Permit issued in advance of the dog’s arrival may enter. “Then and Now” changes are outlined and explained in FAQs. The suspension protects dogs and people from a reintroduction of dog rabies into the United States. About 98% of human victims of rabies around the world get this fatal disease from dogs. In June, CDC was informed that a dog confirmed to have rabies was brought into the United States. This shows how timely and necessary the new suspension is.
NCEZID is investigating several multistate foodborne and animal-borne outbreaks, including the following:
- Salmonella infections linked to raw frozen breaded stuffed chicken. As of June 2, six states have reported 17 cases with eight hospitalizations and no deaths.
- Salmonella infections linked to backyard poultry. As of June 17, a total of 474 people in 46 states have been reported as infected with one of the outbreak strains. Illnesses started on dates ranging from December 15, 2020, to June 4, 2021.
- Salmonella infections linked to frozen cooked shrimp. As of June 25, two states have reported 6 cases with two people hospitalized and no deaths. A Food Safety Alert was posted June 25, and the product has been recalled.
What impact does the work of a public health laboratory (PHL) have on detecting and responding to a Salmonella outbreak? To help put the impact in numbers, NCEZID partnered with the Association of Public Health Laboratories to create the PHL Impact tool. It uses data from the lab’s jurisdiction to show a laboratory’s contribution to testing for Salmonella. The tool can also be used to estimate how many people a public health response has protected from infection and hospitalization and how much in medical costs a public health response saved.

Illustration of Candida auris fungi. Medical illustrator: Stephanie Rossow
NCEZID has been investigating two clusters of infections with the fungus Candia auris that were reported between January and April 2021 in Texas and Washington, DC. Of 126 cases identified, five were confirmed to be caused by C. auris resistant to all major antifungal drugs. All infections in both states were identified in healthcare settings. Although infections with strongly drug-resistant C. auris have been identified previously in the United States, these clusters of infections are the first in the country suspected of spreading from person to person. So far, the infections in both states do not appear to be connected.

More healthcare professionals will begin their careers with the knowledge they need to protect themselves and patients from infectious diseases thanks to a new NCEZID collaboration. Project Firstline, CDC’s infection control training collaborative, is now partnering with the American Hospital Association and the League for Innovation in the Community College to integrate enhanced infection control coursework into community colleges that train healthcare professionals. The program launched this summer with more than a dozen participating community colleges. Faculty teams across the collaboration will help tailor coursework for nursing and allied health students.

Experts in antibiotic resistance (sometimes called AR) continually learn from one another. To help them do so, NCEZID’s AR Coordination and Strategy Unit started the Global AMR Exchange webinar series as a way to share knowledge and information with partners around the world. The first webinar, available online, featured experts from CDC and international organizations to discuss the latest research and lessons learned, while identifying solutions to help fight antibiotic resistance after COVID-19 with a One Health approach. Antibiotic-resistant germs—bacteria and fungi that resist drug treatment—cause over 2.8 million infections in people each year in the United States. More than 35,000 of these infected people die.
You can now register for the second webinar, featuring experts discussing antibiotic resistance and water.

More than 450,000 people from around the country attended the Sturgis Motorcycle Rally in South Dakota on August 7–17, 2020. Public health officials already knew then that a gathering that large could cause significant spread of COVID-19. A recent report by NCEZID staff and members of CDC’s COVID-19 response team traced 649 COVID-19 cases, 17 hospitalizations, and one death in various parts of the country back to the motorcycle rally. People who got COVID-19 included rally attendees, their close contacts, and contacts of contacts. The actual numbers of infections that came from the rally were likely higher than those in the study, but the data in the study alone show that the rally was a superspreading event. The study is a reminder of how important it is to communicate with the public about COVID-19, to take steps like wearing a mask and staying at least 6 feet apart if not fully vaccinated, and to encourage people to get tested during and after large events. Although COVID-19 vaccines were not available at the time of the rally, the report also serves as a reminder that people should be vaccinated before attending large gatherings to keep from getting and spreading COVID-19.

Handwashing station in Sierra Leone. Stations like these help prevent Ebola, which spreads through contact with infected body fluids. Photo credit: Caitlin Cossaboom
On May 3, the Democratic Republic of the Congo (DRC) Ministry of Health and the World Health Organization declared DRC’s Ebola outbreak over. For the outbreak to be declared over, 42 days had to pass with no new cases since the last survivor tested negative and was discharged from the Ebola treatment unit. During the outbreak, 12 people became sick with Ebola, six of whom died. A 90-day enhanced surveillance period started at the end of the outbreak to quickly detect and respond to any new cases. In Guinea, the countdown to the end of their Ebola outbreak started on May 8. As of June 6, 23 people have become sick, 12 of whom have died. NCEZID staff continue to support the preparedness, response, and post-outbreak efforts in affected and border countries.

Outbound Travel Health Alert Notice in English and Spanish on the front (French and Creole on the back).
The spread of rabies in dogs is increasing in Haiti, and NCEZID staff have created materials to alert travelers to the danger. The materials include an online Travel Health Notice, an alert card for airline gate agents to distribute to passengers traveling to Haiti, and a script for airline crew to read aloud to passengers on direct flights between Miami/Fort Lauderdale and Haiti. The information tells travelers to avoid any contact with dogs and cats in Haiti and to seek medical care right away if they are bitten, licked, or scratched. The materials also advise travelers to seek medical care outside of Haiti because human rabies vaccine is in limited supply in that country. Since April, three residents of Haiti have died from rabies, highlighting the need for these prevention materials. Only national and routine dog vaccination campaigns will eliminate rabies from Haiti, and it is unclear when those campaigns – halted due to COVID-19 – will resume.
Infections with monkeypox, a disease similar to smallpox but with milder symptoms, have nearly doubled in the Democratic Republic of the Congo (DRC) since the early 1980s. A report co-authored by NCEZID staff on the spread of monkeypox in Tshuapa Province, DRC, was recently published in the Journal of Infectious Diseases. In the report, more men than women had monkeypox. The single largest group of people who became infected were males 10-19 years old. This group also reported the most contact with animals likely to carry monkeypox, such as small rodents. Women aged 20-29 had the second largest rate of monkeypox infections. Women in that age group had frequent contact with other people who had common monkeypox symptoms. Rates of monkeypox were lower for people who had received a smallpox vaccine prior to the disease’s eradication. This suggests that the vaccine, which these people received prior to its discontinuation in 1980, provides some long-term protection against monkeypox. Monkeypox is an understudied disease and can be deadly for as many as 10% of people who fall sick with it.
CDC has published the first comprehensive guidelines for healthcare professionals to treat botulism. Current and former NCEZID staff spent years developing the guidelines using extensive expert input and scientific reports spanning 100 years. The guidelines provide
- Best practices for diagnosing, treating, and monitoring people with botulism
- Special considerations for infants, children, and pregnant or breastfeeding patients
- Suggestions for supportive care, including helping patients and family members cope with the disease
Healthcare professionals can use the guidelines if they’re treating one patient or many patients. The guidelines would be especially useful during an outbreak in which resources such as ventilators, medical staff, and botulism antitoxin might be in short supply.
May was Lyme Disease Awareness Month, and NCEZID developed communication materials urging people to “get the tips” to “stop the ticks.” Staff worked with a marketing agency to develop entertaining cartoon tick bite prevention videos and images for social media, as well as a full-page tick bite prevention insert in the May 16 edition of the Washington Post. See the content (and like it) by following @CDC_NCEZID and @CDC on Twitter.
CDC continues support to recipients through ELC Reopening Schools awards
CDC’s Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases (ELC) program continues to distribute funds and provide technical support to recipients as they respond to COVID-19. On April 9, ELC awarded $10 billion to 64 recipients through the American Rescue Plan. ELC Reopening Schools awards support COVID-19 screening testing programs to help K–12 (public and private) schools to reopen or stay open safely. ELC recently hosted a webinar for 600+ attendees, where they were engaged by CDC, White House Task Force, Health and Human Services (HHS), Rockefeller Foundation, and school subject matter experts from the field. This webinar provided technical assistance and actionable information on using ELC funds to launch or expand school screening testing programs.
Ebola outbreaks in Democratic Republic of the Congo and Guinea
After reaching 42 days with no new cases since the last survivor tested negative and was discharged from the Ebola treatment unit, the Ministry of Health in the Democratic Republic of the Congo and WHO declared the outbreak in North Kivu Province over on May 3.
Unfortunately, the countdown to the end of the outbreak in Guinea ended soon after it began. In late March, a cluster of deaths, a woman and her two adult daughters, was reported in a village in Soulouta, a sub-prefecture of N’Zérékoré Prefecture. They were determined to be probable cases of Ebola after the son of one of the daughters tested positive for the virus on April 1. Another case was confirmed on April 3. With the new confirmed and probable cases, the Ebola outbreak in Guinea stands at 23 cases and 12 deaths as of May 6, 2021.
Foodborne and animal contact disease outbreaks

CDC, with federal and state partners, is investigating several multistate foodborne and animal contact outbreaks, including the following:
- Salmonella infections linked to wild songbirds. As of April 1, eight states had reported 19 cases with eight hospitalizations and no deaths. CDC and public health officials in several states are collecting different types of data to investigate a multistate outbreak of Salmonella Typhimurium infections in people. Data show that contact with wild songbirds and bird feeders is likely making people sick in this outbreak.
- Salmonella infections linked to ground turkey. As of April 12, 12 states had reported 28 cases with two hospitalizations and no deaths. The ground turkey products have the establishment number “P-244” inside the USDA’s mark of inspection. The ground turkey products were made during December 18–29, 2020 and sold nationwide. These products are no longer available in stores but could still be in your freezer. Investigators are working to determine if additional turkey products are linked to illnesses.
- Salmonella infections linked to Jule’s Cashew Brie. As of April 23, three states had reported five cases with two hospitalizations and no deaths. CDC, public health and regulatory officials in several states, and the FDA are collecting different types of datato investigate a multistate outbreak of Salmonella Duisburg infections. The recalled food can be found can be found here. Do not eat any recalled products.
CDC and partners strengthen surveillance for cholera in Haiti
Haiti’s Ministry of Public Health and Population (MSPP) and CDC are collaborating with the Pan American Health Organization, the CDC Foundation, CDC Haiti, and the Global Task Force on Cholera Control to strengthen Haiti’s surveillance system for cholera and end the disease’s transmission in that country. Cholera first appeared in Haiti in October 2010; since then, the country has reported more than 820,000 cases of the disease, and nearly 10,000 people have died. Cholera cases in Haiti have dropped due to ongoing efforts by MSPP and partners; the last confirmed case was reported in February 2019. With funding from the Bill and Melinda Gates Foundation, partners are working to ensure the timely detection, investigation, and confirmation of any new, suspected cholera cases and hope to help Haiti reach the 3-year mark without any confirmed cholera cases in early 2022.
A mobile application simplifies data collection in the field

A COPA staff member uses HTrack on a mobile device to record data during a home visit in Ponce, Puerto Rico.
Mobile technology allows community health workers to capture and store data as they go from house to house for community-based research studies. However, until now, no single mobile data application has been able to provide everything community health workers need to conduct a home visit, and they end up using multiple devices and apps.
Data collected during home visits are crucial to the success of the CDC-led Communities to Prevent Arboviruses (COPA) project in Ponce, Puerto Rico. COPA project staff visit 3,800 participants annually to determine how many got infected with a virus spread by mosquitoes and to assess disease outcomes. For this big task, CDC’s Dengue Branch Data Team created HTrack (Household Tracking), a mobile application that provides COPA staff with integrated components needed for a successful home visit — for example, pre-loaded maps that allow offline navigation and pre-populated data collection forms that eliminate the need for data reentry after each visit. HTrack can also securely link to other popular data collection platforms used by community health workers. Read more on our monthly spotlights webpage.
CDC adds population survey data to FoodNet Fast
CDC’s Foodborne Diseases Active Surveillance Network (FoodNet) periodically surveys people in its surveillance area to learn more about foods and behaviors linked to diarrheal illness. Data from the latest survey are now available in FoodNet Fast, which allows you to search FoodNet data and see results displayed on interactive graphs and charts. One new search tool, the Population Survey Tool, allows you to see people’s responses to questions about food consumption, contact with animals, dietary practices, food handling practices, and raw milk. The tool also lets you see how responses vary for different groups of people, with the option to filter results based on age group, gender, race and ethnicity, geographic location, and season. Try out the FoodNet Fast Population Survey Tool for yourself!
New antibiotic resistance data and features on NARMS Now
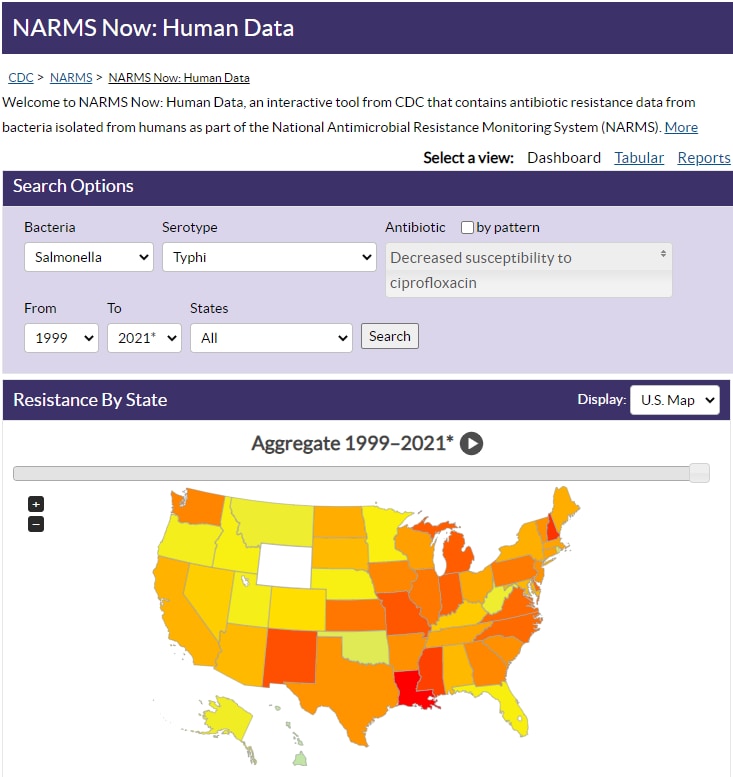
A suite of upgrades to NARMS Now: Human Data makes it even easier and faster to access CDC’s antibiotic resistance data for five enteric (intestinal) bacteria commonly transmitted through food: Campylobacter, E. coli O157, Salmonella, Shigella, and Vibrio (species other than V. cholerae). Data in this interactive, online database come from the National Antimicrobial Resistance Monitoring System (NARMS). The latest version of NARMS Now includes new search, report, and export features—and more data and categories for you to explore.
CDC deploys BioFire Film Array and Warrior Panel to Laboratory Response Network
In more technology news, CDC recently distributed the BioFire FilmArray 2.0 instrument and Warrior Panel, which are used to test for biothreat agents, to 16 high-priority Laboratory Response Network (LRN) facilities. These LRN laboratories will be able to test clinical specimens for anthrax, tularemia, plague, Q fever, and hemorrhagic fevers caused by Ebola and Marburg viruses. CDC staff have also worked to support Clinical Laboratory Improvement Amendments (CLIA) requirements for assay verification, including providing materials that will test positive for these biothreat agents. CDC worked with an external vendor to create nucleic acids to be used for CLIA verification, evaluated performance, and distributed them to LRN laboratories.
CDC releases new resources to improve providers’ understanding of rickettsial disease testing options
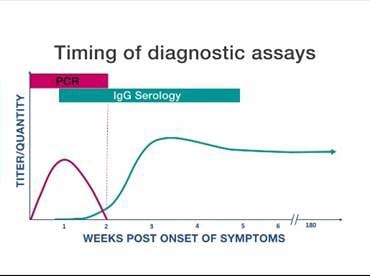
Graph shows timing of specimen collection for PCR and serologic tests
CDC released a new video, “Rickettsial Disease Diagnostic Testing and Interpretation,” to help healthcare providers better understand rickettsial disease testing options. The 10-minute video provides a short overview of the different diagnostic assays, the best times to use each option, and an outline of the most appropriate samples to collect. A companion quick reference guide [PDF – 1 page] directly compares the two most commonly available diagnostic methods: polymerase chain reaction (PCR) and the indirect immunofluorescence antibody (IFA) assay. The video and guide provide quick and easy references that healthcare providers can use when selecting diagnostic tests for their patients.
CDC and partners advance treatment recommendations for antibiotic-resistant Shigella
This spring, the Clinical and Laboratory Standards Institute’s (CLSI) Antimicrobial Susceptibility Testing Subcommittee published new recommendations in CLSI’s M100: Performance Standards for Antimicrobial Susceptibility Testing that include the first “breakpoints”—numerical definitions of Shigella’s susceptibility to azithromycin. With the tools to determine whether a Shigella infection will respond to azithromycin, clinicians can now get patients on the right treatment faster, improving the patients’ care and outcomes. The new recommendations follow many months of hard work and careful deliberations. At an August 2020 meeting of the CLSI Breakpoint Working Group, staff from CDC’s National Antimicrobial Resistance Monitoring System (NARMS), the California Department of Health, and University of Virginia presented clinical and laboratory findings that helped CLSI propose the first breakpoints. Read more on NARMS’ webpage.
New publications on antibiotic prescribing
CDC published a new study estimating that more than half of antibiotics prescribed in hospitals in 2015 for selected prescribing events were not consistent with recommended prescribing practices. This analysis is one of the largest to assess the appropriateness of antibiotic prescribing and establishes baseline estimates and a method for future analyses to measure the impact of antibiotic stewardship programs on prescribing antibiotics in hospitals.
CDC also published two new papers describing trends in antibiotic prescribing during the COVID-19 pandemic, showing a substantial decrease of antibiotic use in outpatient settings and spikes in the use of specific antibiotics intended to treat COVID-19 among nursing home residents.
Project Firstline releases infection control training toolkit for facilitators

Project Firstline is making it easier than ever for facilitators to lead virtual infection control trainings for public health and frontline healthcare workers, even if they are not infection control experts.
The Project Firstline Facilitator Toolkit is now available online and includes infection control training resources that work for a variety of learning styles and fit into busy schedules. The toolkit is divided into session plans and slide decks for 10-, 20-, and 60-minute presentations on foundational infection control topics that facilitators can mix and match to meet the needs of their group. Initial topics include
- The Concept of Infection Control
- The Basic Science of Viruses
- How Respiratory Droplets Spread COVID-19
- How Viruses Spread from Surfaces to People
- How COVID-19 Spreads: A Review
Project Firstline will continue to expand the infection control topics and facilitator resources in the toolkit. To stay in the loop as new resources are made available, follow Project Firstline on Facebook or Twitter or sign up for the newsletter.

CDC-developed, award-winning AGOs (autocidal gravid ovitraps) capture mosquitoes in neighborhoods around Puerto Rico
CDC researchers in San Juan, Puerto Rico invented, field-tested, and improved a mosquito trap called the autocidal gravid ovitrap (AGO), which uses no chemicals. CDC’s Technology Transfer Office, which transfers CDC inventions to partners for public health benefit, and the CDC team at the National Institutes of Health’s National Institute of Allergy and Infectious Diseases Technology Transfer and Intellectual Property Office oversaw the licensing and patenting process. Once a patent license agreement was in place, AP&G Company began selling AGOs. The Federal Laboratory Consortium for Technology Transfer (FLC) recognized the work with the 2021 Excellence in Technology Transfer award. This award recognizes employees of FLC member laboratories and non-laboratory staff who have accomplished outstanding work in the process of transferring federally developed technology.
Emerging Infectious Diseases continues to see growing success with its weekly podcasts, which garner millions of hits each year. EID’s podcasts were developed to inform the general public about the latest scientific research on emerging infectious diseases, and the journal’s podcast library has amassed almost 350 titles that highlight a wide variety of subjects, including antimicrobial resistance, influenza, Lyme disease, E. coli, Ebola, and the COVID-19 pandemic. You can access EID’s podcasts through EID’s website, CDC’s Public Media Library, or wherever you listen to your podcasts.
President, Vice President visit with COVID-19 responders

President Joseph Biden and Vice President Kamala Harris listen as Incident Manager Henry Walke provides an update about CDC’s COVID-19 response.
CDC Director Dr. Rochelle Walensky warmly welcomed President Biden and Vice President Harris to the agency’s Emergency Operations Center on March 19 to brief them on CDC’s COVID-19 response activities. President Biden thanked CDC’s workforce “not only for your intellectual skills but your heart, your determination.” Vice President Harris added that CDC is making decisions “based on science, based on hard work, and based on commitment to public health.” Dr. Walensky thanked President Biden for his leadership, which she said has reinvigorated the whole-of-government response and CDC. Dr. Henry Walke, who oversees CDC’s COVID-19 response, told the President and Vice President that thousands of CDC responders have worked tirelessly and sacrificed much throughout the response.
Congressional staff learn about tracking COVID-19 variants

A CDC scientist briefed congressional staffers on the usefulness of genomic sequencing to track the virus that causes COVID-19. In a webinar, NCEZID’s Greg Armstrong told participants about the data coming to CDC from public and private partnerships sequencing thousands of virus samples every week from across the nation. Dr. Armstrong, who leads CDC’s Office of Advanced Molecular Detection, also spoke on particularly contagious variants of the virus. He stressed the need to increase the use of genomic sequencing in the nation’s public health departments and elsewhere to prepare for future infectious disease threats. The public webinar was hosted by the American Society for Microbiology and opened by US Senator Tammy Baldwin (D-WI). US Representative Ami Bera (D-CA) also delivered remarks.
Nurses contribute to Alaska’s COVID-19 vaccine success

Alaska recently made headlines as having the highest rate of COVID-19 vaccination per capita in the United States. Public health nurses at NCEZID’s Arctic Investigations Program (AIP) in Anchorage have contributed to that success by helping with vaccinations. Three nurses have volunteered at more than 14 sites, which administered nearly 13,000 shots by the end of March. State and local public health officials established vaccination sites in cooperation with Alaska Respond, a volunteer healthcare corps that is part of the state health department and which responds to public health emergencies. Sites were established in Anchorage and the Matanuska Valley, including at the Alaska Native Medical Center. AIP works to prevent the spread of infectious disease in the region and has played an important role in efforts to vaccinate people against COVID-19.
Order to collect contact information from travelers from DRC and Guinea

As a precaution due to recent Ebola outbreaks in Africa, CDC has issued an order requiring airlines and other aircraft operators to collect contact information from passengers who were in Guinea or the Democratic Republic of the Congo (DRC) within 21 days before arriving in the United States. The information is transmitted electronically to CDC, which provides it to state and local health departments. They are then able to monitor travelers and ensure that those who develop Ebola symptoms are quickly isolated and receive medical evaluation and care. Additionally, CDC is providing a Travelers’ Health Alert Notice (or T-HAN; image at left) to travelers arriving in the United States from DRC and Guinea, directing them to a webpage with information in English and French on how to monitor themselves for symptoms and what to do if they have symptoms.
Ensuring access to Ebola vaccine in the United States
People in the United States who work to help stop Ebola outbreaks abroad or who are involved in the detection, prevention, and/or treatment of Ebola can now get a vaccine that protects them from infection. While an Ebola vaccine has been FDA approved, this licensed vaccine is not yet available in the United States. Therefore, a CDC-sponsored Investigational New Drug (IND) program has been in place since October 2020 to allow people who are at risk of occupational exposure to Ebola to get a vaccine in the interim. NCEZID scientists collaborated to get the FDA’s permission on the IND program, and recently, they expanded the IND protocol to include a booster dose, since it is not yet known how long protection from the initial dose of vaccine lasts.
Was it or wasn’t it a multistate outbreak of intestinal disease?

Was or wasn’t it a multistate outbreak? The first-ever issue of a new annual NCEZID report gives answers that may surprise you about multistate outbreaks of Listeria, Salmonella, and E. coli., three common causes of intestinal disease. In 2016, investigations coordinated by CDC determined that 50 out of 118 possible outbreaks actually met the criteria. Contact with animals was found to be the cause of nearly twice as many of these multistate outbreaks as contaminated food. The report reveals new concerns about foods that carry these diseases, like flour and frozen vegetables, and persistent multistate outbreaks linked to contact with backyard poultry and small turtles. CDC is working on similar reports that cover the years 2017-2019. You can find highlights from the 2016 report online, read the related MMWR article, and see the data visualized in charts in the PDF version [PDF – 56 pages].
Six antibiotic-resistant threats that cost the United States $4.6 billion each year

Six multidrug-resistant pathogens generate national healthcare costs of more than $4.6 billion annually, according to a new study. CDC published the data in the journal Clinical Infectious Diseases in partnership with the University of Utah. This study is one of the largest of its kind to provide national cost estimates for antibiotic-resistant infections. Of note, it highlights a major theme from our 2019 AR Threats Report [PDF – 150 pages]: Resistant infections that occur in communities – as opposed to healthcare settings – account for more costs than hospital-associated infections. CDC funds research like this to help public health experts and leaders better plan for prevention and response activities.
Estimating the annual burden of Lyme disease

The western black-legged tick (above) can infect people with bacteria that cause Lyme disease. Photo credit: James Gathany
One of the most common questions about Lyme disease is, “How many people are diagnosed and treated each year?” Although 30,000–40,000 cases of Lyme disease are reported to CDC each year, the actual number of people diagnosed and treated for Lyme disease is much higher. In the February issue of Emerging Infectious Diseases, researchers assessed whether commercial insurance claims could provide reliable data on diagnoses of Lyme disease. They then used those data to estimate that an average of 476,000 people were diagnosed with and treated for Lyme disease annually in the United States during 2010-2018. The authors noted that the estimate likely includes some overdiagnosis. But the fact that Lyme disease is diagnosed so often underscores the need for effective tick bite prevention methods.
Prestigious infectious disease journal achieves 25 years of success

As COVID-19 emerged and spread around the globe last year, the CDC journal Emerging Infectious Diseases had its 25th anniversary with little fanfare. However, the number of submissions surged to nearly 5,000 articles in 2020, up by more than 3,000 articles than in 2019. EID, founded in 1995, published more articles, figures, tables, and pages, and more pages of appendices during the pandemic than ever before. EID articles also had a broad news and social media reach, as documented by Altmetric, a service that collects data on digitally accessible scientific publications worldwide. Three EID articles are ranked among Altmetric’s top 50, eight among its top 250, and 35 among its top 5,000―out of nearly 17 million tracked scholarly articles.
Haiti faces rabies challenges due to vaccination campaign delays

Puppies play in a basket in Haiti
The COVID-19 pandemic threatens to hinder some of the progress made toward the global goal of achieving zero human deaths from rabies caused by dog bites. The World Health Organization and its partners in the United Against Rabies collaboration want to reach that goal by 2030. Since 2013, with CDC’s support, Haiti has dramatically reduced cases of rabies in dogs. But recently the country has had to delay mass dog rabies vaccination campaigns, in part because rabies response partners needed to divert resources to respond to COVID-19. An eight-fold increase in rabid dogs was recorded for the last half of 2020 in Haiti’s capital, Port-au-Prince. Additionally, Haiti’s entire stockpile of post-exposure rabies vaccine expired last September, prompting CDC to issue a travel alert warning travelers of the risk. Despite positive steps that have been made toward eliminating rabies, only national and routine dog vaccination campaigns will eliminate rabies from Haiti, and it is unclear when those campaigns will resume.
Diversity and Inclusion Council Roundtable

To promote diversity and inclusion at NCEZID, the center holds monthly internal roundtable discussions, which have been virtual during the COVID-19 pandemic. The theme of the February roundtable was “We Shall Overcome: Building an Equitable, Safe, and Fair Workplace.” It explored how vocational barriers such as sexism, racism, and other prejudices in the workplace can limit staff opportunities for growth and productivity. NCEZID’s Diversity and Inclusion Council used a combination of informative videos, NCEZID survey results, and dialogue with staff to define inequity and fairness problems in the workplace and identify options for moving forward.
Interstellar chat with NASA astronauts

Astronauts Kate Rubins and Shannon Walker (on screen) talk with Division of High Consequence Pathogens and Pathology Director Dr. Inger Damon (in studio)
As COVID-19 wears on, it has become normal to video chat with people. But it’s still a little odd to think that you can have a video chat with people in outer space almost as easily as with a coworker across town. NCEZID’s Dr. Inger Damon hosted a live video chat with NASA astronauts Shannon Walker and Kate Rubins aboard the International Space Station on February 10, 2021. Dr. Damon asked them about their experiments in space and how they were dealing with the COVID-19 pandemic. The astronauts are tackling projects that include DNA sequencing and cataloging of microbes they found on the space station. They even gave a collective shout-out to a rare pathogen that NCEZID is known for studying – Elizabethkingia – that was also found on the space station Mir.
Over $30 billion to help health departments fight COVID-19
As of January, NCEZID has awarded over $30 billion in COVID-19 funding to 64 public health departments across the country. This includes a recent distribution through the Coronavirus Response and Relief Supplemental Appropriations (CARES) Act [PDF – 13 pages] of $19.11 billion to expand COVID-19 testing and practices to track the spread of the disease. In December 2020, $87 million went to special projects like preparing public health laboratories to detect molecular characteristics of the virus that causes COVID-19. These efforts will strengthen public health departments beyond the current crisis, readying them for future threats. These awards were made through a long-standing funding and cooperation agreement with health departments, the Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases (ELC) program, which boosts the departments’ ability to prepare for and respond to infectious diseases.
COVID-19 funding strengthens laboratories’ ability to respond
In the marathon response to COVID-19, state and local public health laboratories overcame many obstacles despite chronic underfunding that left little room to advance lab capabilities. NCEZID recently strengthened public health labs by awarding COVID-19 funding to the Laboratory Response Network (LRN). LRN is a network of laboratories ready to respond to biological and chemical threats, and it includes state and local public health laboratories. LRN partners will use these funds to invest in new technologies such as next generation whole genome sequencing and to expand their capabilities to detect infectious diseases. The funds also will help proactively get necessary equipment and supplies in place in LRN member laboratories and enable the shipping of lab samples to other laboratories for deeper examination. And more labs will be able to connect to electronic laboratory reporting systems that enable them to report results faster and more efficiently.
CDC issues mask order for travelers

On January 29, CDC issued an order requiring people to wear masks on public transportation and at transportation hubs in the United States. CDC staff have worked around the clock to apply this and other provisions of President Biden’s Executive Order on Promoting COVID-19 Safety in Domestic and International Travel, issued January 21. Prior to this, on January 12, CDC issued an order requiring a negative COVID-19 test result or documentation of recovery from COVID-19 for all air passengers coming to the United States from a foreign country.
Project Firstline launches video series for healthcare workers

In February, Project Firstline continued its Facebook video series for frontline healthcare workers called Inside Infection Control. The videos have been viewed over 70,000 times on YouTube. February’s videos answered questions that frontline healthcare workers may have during the current stage of the pandemic. New episodes include content on personal protective equipment (PPE) worn for respiratory protection and were produced in collaboration with the National Institute for Occupational Safety and Health. Project Firstline also published videos about why cleaning, disinfection, and ventilation matter for infection control. It dedicated one episode to discussing what new strains of COVID-19 mean for infection control. New Inside Infection Control videos post every Tuesday and Thursday on Project Firstline’s Facebook page.
Looking back on 2020’s fight against emerging infectious diseases

Last year was a year like no other. In 2020, nearly 2,000 of NCEZID’s staff supported CDC’s COVID-19 emergency response, while other staff stretched to meet ongoing emerging disease threats and challenges. The year’s work is summed up in the recently released Protecting Health in 2020: NCEZID Progress Report [PDF – 16 pages]. The report highlights our involvement in new discoveries, unprecedented support for states during the COVID-19 pandemic, accelerated data use, and advances that are saving lives and strengthening preparedness across all our efforts against emerging infectious diseases.
Ebola vaccine recommendations

In December 2019, the Food and Drug Administration approved the first vaccine to prevent Ebola infections caused by the most deadly species of the virus, Zaire ebolavirus. CDC’s Advisory Committee on Immunization Practices developed recommendations for the use of the vaccine, which is called Ervebo®, in the United States. NCEZID staff played a leading role in these recommendations, which were published in the January 8 Morbidity and Mortality Weekly Report. The report outlined three groups of adults considered at highest risk for occupational exposure to Ebola – responders to outbreaks, healthcare professionals at Ebola treatment centers, and staff at laboratory facilities that require the highest level of safety precautions.
New cases of Ebola confirmed in DRC and Guinea
As we were nearing the anticipated deactivation of CDC’s Ebola response, the Democratic Republic of the Congo’s (DRC) Ministry of Health announced on February 7 that a case of Ebola had been confirmed in North Kivu Province. As of February 24, eight cases had been confirmed in DRC. Sequencing of samples suggests that those cases are linked to cases in the area during the 2018-2020 outbreak and were likely caused by a persistent infection in a survivor who either relapsed or sexually transmitted the virus. Then, on February 14, an Ebola outbreak was confirmed by Guinea’s Ministry of Health. The outbreak is in an area affected by the 2014-2016 West Africa Ebola outbreak, which was the largest Ebola outbreak in history. Sequencing of samples will be conducted to determine if the cases are due to a new introduction of the virus or are linked to the 2014-2016 outbreak. As of February 24, eight cases had also been recorded in Guinea. CDC is supporting response and preparedness efforts in DRC, Guinea, and surrounding countries.
New botulism treatment resources for healthcare professionals

A new video demonstrates to healthcare professionals how to prepare a treatment for botulism and give it to a patient. The video, titled Preparing and Administrating Heptavalent Antitoxin for the Treatment of Botulism, also answers questions that healthcare professionals often ask during clinical consultations with CDC. In addition, public health officials and first responders may turn to the video for guidance if a large botulism outbreak occurs.
NCEZID also posted a new page on CDC’s botulism website that guides healthcare providers on what to do if a baby has signs of infant botulism. The webpage, “Infant Botulism: Information for Clinicians,” includes detailed information on treatment, diagnostic testing, reporting, and more.
Cheese and turtles are focus of disease outbreak investigations

NCEZID and its federal and state partners are investigating several outbreaks of foodborne diseases and infections spread through animal contact:
- Listeria infections linked to queso fresco made by El Abuelito Cheese Inc. As of March 1, four states reported 11 ill people with ten hospitalizations and one death. Connecticut officials found the outbreak strain of Listeria in samples of El Abuelito brand queso fresco cheese. El Abuelito Cheese Inc. recalled all cheeses, sold under multiple brands, made in the same facility as the contaminated queso fresco cheese.
- Salmonella infections linked to contact with small pet turtles. As of February 20, seven states reported 22 ill people with eight hospitalizations and one death. Most people sick in this outbreak are children. The U.S. Food and Drug Administration bans the sale and distribution of the turtles involved. They can sometimes be found illegally at stores, flea markets, and roadside stands.
Tick bite data tracker displays emergency department visits
Your chances of being bitten by a tick and getting a tickborne disease rise in the spring and stay higher through the fall. In January, NCEZID launched a new tick bite data tracker. The dashboard displays data on the number of people visiting emergency departments with symptoms of or concerns about tick bites—before a diagnosis of a tickborne disease is confirmed. Updated weekly, these data give public health officials a rapid indicator of when cases of tickborne illness might be rising in different parts of the country. It can also help public health officials and healthcare providers target outreach on tick bite prevention and risk to the public.
New map shows CDC investments to combat antibiotic resistance
NCEZID recently updated its Antibiotic Resistance (AR) Investment Map, showing CDC’s funding in 2020 to its partners and continued collaborative efforts to combat AR. The site includes several of CDC’s COVID-19 response efforts that also slow the spread of AR in the United States, including in healthcare settings. The map showcases how CDC’s AR investments are working in every US state, some large cities, and in more than 15 other countries.
Rare fungal infections investigated at a medical center in Arkansas
In February, a CDC team deployed to Little Rock, Arkansas, to help state public health colleagues investigate fungal infections called mucormycosis occurring in a hospital. The source of the infections is unknown. CDC has provided guidance and expertise remotely to the Arkansas Department of Health (ADH) since February 2020, but after a new patient with a compromised immune system came down with a mucormycosis skin infection, ADH requested in-person help from CDC. The CDC team included NCEZID staff and a colleague from the Center for Surveillance, Epidemiology, and Laboratory Services (CSELS).
Toolkit helps epidemiologists better use genomic data to track COVID-19 variants

Epidemiologists tracking variants of the virus that causes COVID-19 in states and localities have a new tool to help them stay on top of the latest scientific data. CDC’s Office of Advanced Molecular Detection recently released a COVID-19 Genomic Epidemiology Toolkit to teach epidemiologists how to optimize the use of whole genome data in their investigations. In infectious disease, molecular detection technology identifies important clues in germs. Identifying their genetic blueprints, or whole genomes, is among the most advanced available forms of molecular detection. The new toolkit consists of video training modules, audio transcripts, case studies, and more. The toolkit includes introductions to genomic epidemiology and concentrates on the genome of the virus that causes COVID-19. Sign up to receive email updates when new modules are added to the COVID-19 Genomic Epidemiology Toolkit.
Testing order for air travelers flying to the US from other countries
CDC has issued an Order requiring a negative COVID-19 test or documentation of recovery for all air passengers 2 years of age or older before boarding a flight to the United States from another country. The order took effect on January 26 and includes US citizens and legal permanent residents. Testing before and after travel is a critical step to slow the introduction and spread of COVID-19 in the United States. As variants of the virus that causes COVID-19 continue to emerge around the world, there is growing evidence that some of them are more contagious. With COVID-19 surging in the United States, the testing requirement for air passengers will help slow the spread of the virus as more people in the United States get vaccinated.
Serving as a COVID-19 vaccine hub for Alaska
When Alaska needed help distributing a vaccine to prevent COVID-19, it turned to its partners at CDC. The Alaska Department of Health and Social Services (DHSS) asked NCEZID’s Arctic Investigations Program (AIP) for assistance in receiving and redistributing the vaccine developed by Pfizer-BioNtech. AIP manages several ultracold freezers, the same type needed to store the vaccine, and agreed to serve as one of Alaska’s three vaccine hubs. AIP developed a database to track vaccine shipments, completed vaccine order redirects from Pfizer, and established a process to redistribute the vaccine to communities across the state. AIP helped distribute more than 23,000 doses of the vaccine to sites across the state, including to remote villages and smaller communities. AIP has since transitioned its vaccine hub responsibilities to the state.
Yellow fever vaccination campaigns continue safely during COVID-19 pandemic
Yellow fever, a disease transmitted by mosquitoes, kills an estimated 30,000 people annually. A WHO strategy aims to drive deaths down to zero by 2026, but COVID-19 has threatened prevention campaigns in countries at risk for yellow fever. For example, a vaccination campaign targeting 5.6 million people in Ghana was planned for November 2020, but personal protective equipment (PPE) and infection prevention and control (IPC) supplies were too low to adequately prevent the spread of COVID-19. Also, officials planned to use “yellow cards” to note who was vaccinated, but the country did not have supplies needed to create them. To purchase all necessary PPE, IPC, and yellow cards, CDC, in particular its Arboviral Diseases Branch, a close partner with WHO on insect-borne diseases, raised over $7 million internally, including from CDC Foundation. The vaccination campaign in Ghana was successfully carried out at the end of November 2020. The funding will also allow yellow fever vaccination campaigns to move forward in Nigeria and the Democratic Republic of the Congo.
Project Firstline launches video series for healthcare workers

In December, Project Firstline launched a Facebook video series for frontline healthcare workers called Inside Infection Control. The videos range from three to five minutes and are designed to answer questions received from frontline healthcare workers. December’s episodes describe important infection control concepts and serve as a foundation for understanding infection control in healthcare. New Inside Infection Control videos are posted every Tuesday and Thursday. January videos focused on infection control actions that are important for healthcare workers to apply during this phase of the COVID-19 pandemic. To stay up to date on new video and infection control content from Project Firstline, follow along on Facebook and Twitter.
Report on the “food” in “foodborne” illness is packed with data
Illustration of Salmonella Typhi bacteria. Medical illustrator: Alissa Eckert
A report [PDF – 15 pages] with NCEZID co-authors examines which foods are most commonly responsible for illnesses caused by the bacteria Salmonella, Campylobacter, E. coli O157, and Listeria. The report uses data collected over 21 years to mathematically estimate the major food sources of these illnesses during 2018. Combined with other analyses, this report [PDF – 15 pages] could help shape measures to keep more people safe from foodborne illnesses. It was produced by the Interagency Food Safety Analytics Collaboration, which includes CDC, the US Food and Drug Administration, and the Food Safety and Inspection Service of the United States Department of Agriculture. The methods used in this and prior annual reports are detailed in an article in the journal Emerging Infectious Diseases.
Monitoring drug-resistant bacteria that can come from food or live animals
Some infectious bacteria that people catch from contaminated food or contact with animals are resistant to antibiotics. A new report, the 2018 NARMS Integrated Summary, summarizes data on the presence of antibiotic-resistant bacteria in raw retail meat, food-producing animals at slaughter, and infected people. The report also includes data on antibiotic-resistant bacteria from dogs collected through a pilot (new) surveillance system. The National Antimicrobial Resistance Monitoring System (NARMS), a collaboration between the Food and Drug Administration, the U.S. Department of Agriculture, and CDC, issued the 2018 report. It adds data on more antimicrobial drugs and expands the number of retail meat collection sites since the last report. The new report can also be viewed in an interactive format with convenient data charts.
Reducing the spread of infections in healthcare settings
The United States has continued to drive down several kinds of infections that spread in healthcare settings. But there is still room for improvement. Both aspects are highlighted in a recent CDC report, the 2019 National and State Healthcare-Associated Infections (HAI) Progress Report. The report’s findings show that HAI prevention is possible, which is good news for patient safety. Notably, there has been a decrease of about 18% in hospital-onset infections caused by Clostridioides difficile (C. difficile) in acute care hospitals. C. difficile can cause the colon to inflame and can be deadly. The 2019 Progress Report includes data from the National Healthcare Safety Network. The report’s data are also available in CDC’s Antibiotic Resistance & Patient Safety Portal. Continued collaboration with public health departments, healthcare professionals, and other partners is critical to sustain progress in eliminating HAIs and to ensure patient safety.
A call to end deaths from an HIV-related fungal meningitis by 2030
About 20 percent of people who die in connection with HIV/AIDS globally succumb to a fungal infection called cryptococcal meningitis. CDC researchers have co-authored an article in the medical journal The Lancet Infectious Diseases arguing for a global strategy to end these deaths by 2030. The article appeared in late November just before World AIDS Day on December 1, 2020. It highlights major gaps in access to diagnostics and treatment that need to be addressed for this goal to be achieved. CDC is supporting a global effort against the AIDS-related disease at a time when the strategic framework to fight HIV/AIDS is being updated.
Modeling a path to fewer rabies deaths around the world

A little girl waits in line to get her puppy vaccinated against rabies at a clinic in Haiti. Photo credit: Kelly Crowdis
About 59,000 people around the world die every year needlessly from rabies infections passed on by dogs. To combat the problem, NCEZID has produced RabiesEcon, an analytic tool that estimates the impacts of policy choices to reduce the spread of rabies. CDC has collaborated with stakeholders in Zambia, Mexico, Guatemala, Vietnam, and Haiti to model rabies cases and the human deaths that can be averted by expanding or continuing rabies vaccination programs for dogs. RabiesEcon models helped strongly persuade policymakers in Mexico to continue funding for the country’s successful 20-year rabies vaccination program for dogs.
City water evaluated following a death from brain-eating amoeba

DFWED's Mia Mattioli and Travis Brown collect water from a water tower in Lake Jackson. Photo credit: Jenna Kieser
CDC staff recently conducted water evaluations in the city of Lake Jackson, Texas, after disinfection of its drinking water pipes. In September, CDC had detected Naegleria fowleri, commonly called brain-eating amoeba, within the city’s water system after a child was exposed at a city splash pad. The child later died. The city treated its water distribution network for 60 days with a high dose of chlorine. Afterward, CDC staff took large water samples throughout the city from water towers, fire hydrants, and monitoring points which, before treatment, had shown low levels of chlorine. CDC detected no evidence of N. fowleri in drinking water samples after the treatment. Water testing results, along with the positive data about the disinfection levels, are helping restore the public’s and local authorities’ confidence in their drinking water.
Studying mosquitoes that transmit diseases in time to take action

Staff from CDC, US Virgin Islands Department of Health, and the Puerto Rico Vector Control Unit collect mosquito larvae and pupae from a cistern.
Outbreaks of Zika and other mosquito-borne diseases have highlighted the need to control a particular species of mosquito called Aedes aegypti. This requires a complex, layered approach that includes identifying places where mosquitoes lay eggs and detecting mosquitoes that are resistant to insecticide early enough to take effective action. A CDC team traveled to the US Virgin Islands in November to contribute knowledge and skills through an agreement that facilitates public health support in areas affected by hurricanes. On the island of St. Croix, the team worked with staff from the Virgin Islands Department of Health (VIDOH) and the Puerto Rico Vector Control Unit to collect mosquito larvae and pupae living in water-collecting containers like rain barrels or basins. In St. Thomas, the CDC team helped VIDOH colleagues collect Ae. aegypti eggs that will be tested for insecticide resistance.
On the trail of pneumonia caused by bacteria that make an anthrax toxin
A CDC disease detective and a colleague from the National Institute of Occupational Safety and Health traveled to Houston to help the Texas Department of Health investigate the source of an unusual pneumonia infection. It was caused by a rare strain of Bacillus bacteria that produces a toxin normally made by anthrax. (The infecting bacteria were not anthrax.) The team wanted to know where the patient, a welder, contracted the bacteria. This was the second time in six months that a welder came down with pneumonia caused by Bacillus bacteria containing DNA to make the anthrax toxin. It was the seventh known such case in total in the Southern United States.