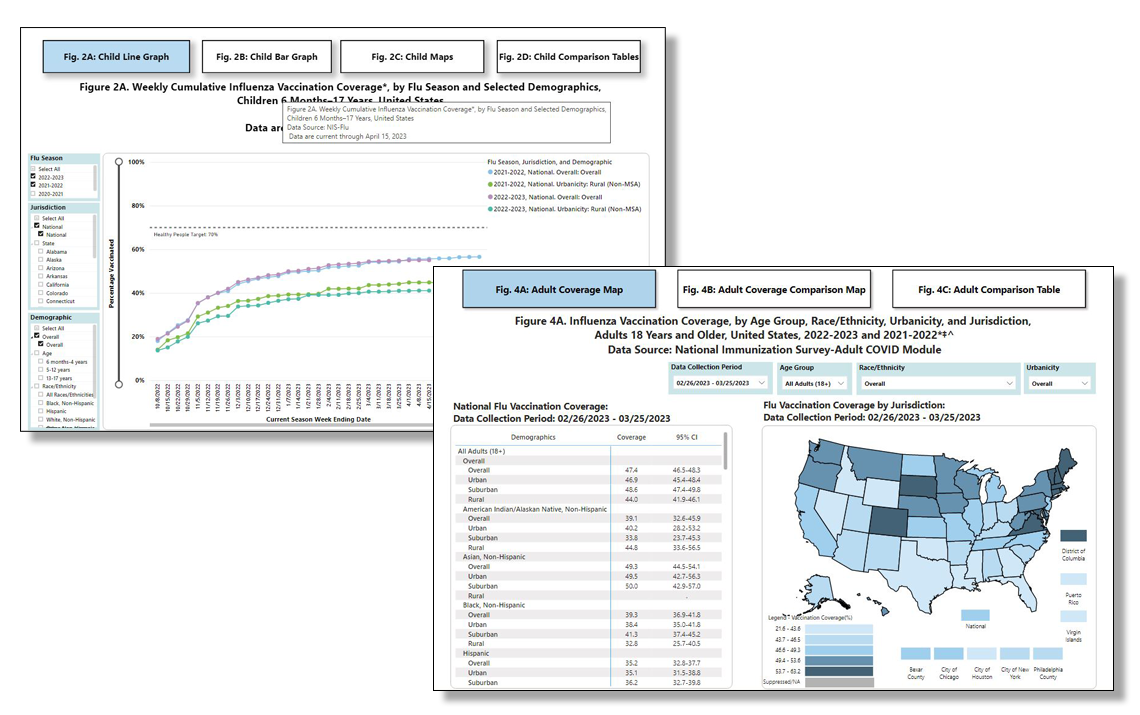
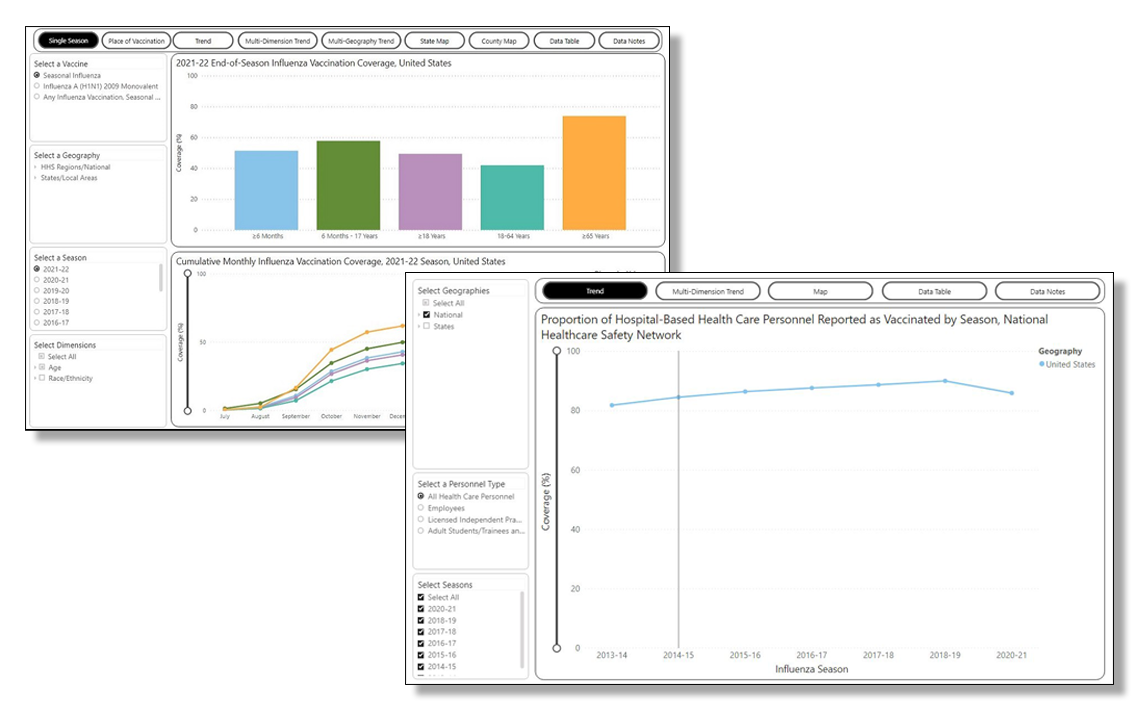
FluVaxView


Monitoring coverage for recommended vaccinations across the country helps CDC assess how well local areas, states, and the nation are protected from vaccine-preventable diseases. Find vaccination coverage data for all ages at:

A weekly influenza surveillance report prepared by the Influenza Division. All data are preliminary and may change as more reports are received.