How Influenza (Flu) Vaccines Are Made
For the United States there are three different flu vaccine production technologies approved by the U.S. Food and Drug Administration (FDA):
- egg-based flu vaccine,
- cell culture-based flu vaccine, and
- recombinant flu vaccine.
All commercially available flu vaccines in the United States are made by private sector manufacturers. Different manufacturers use different production technologies, but all flu vaccines meet FDA safety and effectiveness requirements. Different vaccines have different indications. See Different Types of Flu Vaccines for more information.
Egg-Based Flu Vaccines
The most common way that flu vaccines are made is using an egg-based manufacturing process
that has been used for more than 70 years. Egg-based vaccine manufacturing is used to make both inactivated (killed) vaccine (usually called the “flu shot”) and live attenuated (weakened virus) vaccine (usually called the “nasal spray flu vaccine”).
The egg-based production process begins with CDC or another laboratory partner in the WHO Global Influenza Surveillance and Response System providing private sector manufacturers with candidate vaccine viruses (CVVs) grown in chicken eggs per current FDA regulatory requirements. These CVVs are then injected into fertilized chicken eggs and incubated for several days to allow the viruses to replicate. The fluid containing virus is harvested from the eggs. For inactivated influenza vaccines (i.e., flu shots), the vaccine viruses are then inactivated (killed), and the virus antigen is purified. The manufacturing process continues with quality testing, packaging and distribution. For the nasal spray flu vaccine (i.e., the live attenuated influenza vaccine – LAIV), the starting CVVs are used to make live, but weakened viruses that are then used in vaccine production. FDA tests and approves all flu vaccines prior to release and shipment.
There are several different manufacturers that use egg-based production technology to make flu vaccines for use in the United States. This production method requires large numbers of chicken eggs to produce vaccine and may take longer than other production methods.
Cell Culture-Based Flu Vaccines
There also is a cell culture-based production process for flu vaccines that was approved by FDA in 2012. Originally, this production process also began with chicken egg-grown CVVs per FDA regulations. However, on August 31, 2016, FDA issued an approval for Seqirus, the sole FDA-approved cell culture-based flu vaccine manufacturer in the United States, to begin using cell culture-grown CVVs. Currently, cell culture-based manufacturing is used to make inactivated flu vaccines (e.g., flu shots), not LAIV (nasal spray flu vaccine).
The process of creating cell culture-based flu vaccines involves several steps. First, CDC or one of its laboratory partners, uses influenza viruses that have been grown in cultured mammalian cells to make CVVs, which are then provided to a vaccine manufacturer. Next, the vaccine manufacturer inoculates the CVVs into cultured mammalian cells (instead of into chicken eggs) and allows the CVVs to replicate (i.e., make copies) for a few days. Then, the virus-containing fluid is collected from the cells and the virus antigen is purified. The manufacturing process continues with purification, virus inactivation, and testing. Finally, FDA tests and approves the vaccines prior to release and shipment.
Cell culture-based flu vaccine production does not require chicken eggs because the vaccine viruses used to make vaccine are grown in mammalian cell cultures (no animals are harmed by this process). Cell culture-based technology also has the potential for a faster start-up of the flu vaccine manufacturing process.
While viruses used in previous seasons’ cell culture-based vaccine have been grown in cell cultures, prior to the 2019-2020 season, some of the viruses provided to the manufacturer had been first propagated in chicken eggs. Beginning with the 2021-2022 season, all four flu viruses used in the cell culture-based vaccine are grown in cell cultures only, making the vaccine completely egg-free.
For more information, see CDC’s Cell Culture-Based Flu Vaccines webpage.
Recombinant Flu Vaccines
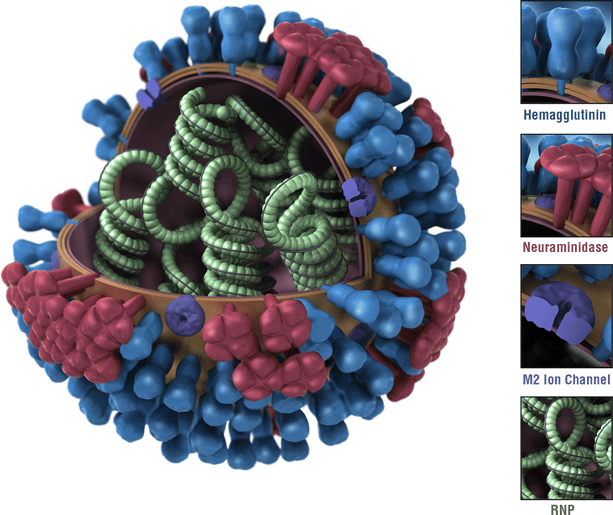
This is a picture of an influenza virus. The virus’ hemagglutinin (HA) surface proteins are depicted in blue. The HAs of an influenza virus are antigens. Antigens are features of the influenza virus that are recognized by the immune system and that trigger a protective immune response. Most flu vaccines are designed to trigger an immune response against the HAs of circulating influenza viruses.
A third production technology for flu vaccines was approved for use in the U.S. market in 2013 and involves using recombinant technology. Recombinant flu vaccines do not require the use of a candidate vaccine virus (CVV) for production. Instead, recombinant vaccines are created synthetically. To make a recombinant vaccine, flu scientists first obtain the virus’ gene that contains the genetic instructions for making the HA. HA is an antigen, which is a feature of a flu virus that triggers the human immune system to create antibodies that specifically target the virus. This HA gene is then combined with a baculovirus, a virus that infects invertebrates. This results in a “recombinant” baculovirus. The role of the baculovirus is to help deliver the genetic instructions for making flu HA antigen into a host cell. Once the recombinant virus enters a Food and Drug Administration (FDA) qualified host cell line, it instructs the cells to rapidly produce the HA antigen. This antigen is grown in bulk, collected, purified, and then packaged as recombinant flu vaccine. Prior to FDA approval and release of the vaccine lots to the public, these vaccines are tested for quality and potency by FDA.
This production method does not require an egg-grown vaccine virus and does not use chicken eggs at any stage of the production process. While there are other vaccines that use similar recombinant manufacturing processes, only one flu vaccine is FDA approved for use in the United States at this time. This production process is the fastest because it bypasses the need for CVVs adapted for growth in eggs or the development of cell culture-based vaccine viruses.
CDC and FDA monitor the safety of all vaccines licensed in the United States, including seasonal flu vaccines. More information about the safety of egg-based, cell culture-based, and recombinant flu vaccines, including adverse events, contraindications and precautions, screening, and safe vaccine administration is available at Seasonal Influenza Vaccine Safety: A Summary for Clinicians.
- The Evolution, and Revolution, of Flu Vaccines on the U.S. Food and Drug Administration website.
- Flu Vaccine Research on the National Institute of Allergy and Infectious Diseases Institute, NIH website.
- Influenza Vaccine Advances
