Presumptive Treatment and Screening for Strongyloidiasis, Infections Caused by Other Soil-Transmitted Helminths, and Schistosomiasis among Newly Arrived Refugees
- Background
- Summary of the Overseas Presumptive Treatment Information and the Impact on Post-arrival Management
- Resources and Summary Recommendations for Post-arrival Management of Strongyloidiasis, Other Soil-transmitted Helminths, and Schistosomiasis
- Persistent Eosinophilia in Refugee Populations
- Other Parasitic Infections Common in Refugees
- References
Background
Strongyloides parasites, other soil-transmitted helminths (STH), and Schistosoma species are some of the most common infections among refugees 1, 2. Among refugees resettled in North America, the prevalence of potentially pathogenic parasites ranges from 8% to 86% 1, 2. This broad range may be explained by differences in geographic origin, age, previous living and environmental conditions, diet, occupational history, and education level. Although frequently asymptomatic or subclinical, some infections may cause significant morbidity and mortality. Parasites that infect humans represent a complex and broad category of organisms. This clinical guidance provides detailed information regarding the most commonly encountered parasitic infections.
A summary table of current recommendations is included in Table 1. In addition, information on overseas pre-departure intervention programs can be accessed on the CDC Immigrant and Refugee Health website.
Strongyloides
Below is a brief summary of salient points about Strongyloides infection in refugees, especially in context of the presumptive treatment with ivermectin. Detailed information about Strongyloides for healthcare providers can be found at the CDC Parasitic Diseases website.
Background
- Ivermectin is the drug of choice for strongyloidiasis. CDC presumptive overseas ivermectin treatment was initiated in 2005.
Epidemiology
- Prevalence in serosurveys of refugee populations ranges from 25% to 46%, with a particularly high prevalence in Southeast Asian refugees 2 – 4.
Transmission
- The most common way of becoming infected with Strongyloides is by contacting soil that is contaminated with Strongyloides larvae. Therefore, activities that increase contact with the soil increase the risk of becoming infected, such as:
- walking with bare feet
- contact with human waste or sewage
- occupations that increase contact with contaminated soil such as farming and coal mining.
- Most infections are acquired through environmental exposure. Although the typical duration of infection is unknown, chronic, potentially life-long, infection occurs. Prevalence generally increases with age. The risk of acquisition (or transmission) after arrival in the US is negligible.
- A graphic describing Strongyloides lifecycle and transmission is available from the CDC Division of Parasitic Diseases and Malaria.
Clinical Information
- Infection is usually asymptomatic or has vague clinical symptoms.
- The most common clinical presentation in refugees is an elevated eosinophil count, although lack of eosinophilia does not exclude infection. Common clinical symptoms in refugees include abdominal complaints, nausea, diarrhea, constipation, dry cough, and skin manifestations (e.g., larva currens).
- Strongyloides autoinfection allows the parasite to persist for decades (e.g., lifelong) if not treated.
- Hyperinfection syndrome (disseminated strongyloidiasis) is associated with immunosuppression. Although most commonly associated with corticosteroid administration (independent of dose or duration used), hyperinfection syndrome may be caused by other immunosuppressive conditions or agents. Strongyloides hyperinfection syndrome is also associated with co-infection with human T-lymphotropic virus (HTLV-1). Hyperinfection may occur decades after last exposure and has mortality rates exceeding 50% 7 – 9.
- For unclear reasons, Strongyloides hyperinfection syndrome has rarely been reported in children.
- The natural history of infection is unknown and hence, when present, infection is considered lifelong unless treated 10.
Presumptive Treatment
- Most refugees receive overseas pre-departure treatment with ivermectin before departure, unless contraindicated (see Tables 1 and 2). A relative contraindication is confirmed or suspected concomitant infection with Loa loa, because persons with pre-existing Loa loa infection and a high microfilarial load are at risk of complications when treated with ivermectin. For this reason, refugees who have lived in areas endemic to Loa loa do not receive pre-departure presumptive treatment with ivermectin (see Loa loa section below). Additional information on treatment schedules for presumptive parasitic infections is available on the CDC Immigrant and Refugee Health website.
- Most refugee children weighing more than 15 kilograms receive presumptive treatment with ivermectin before resettlement, unless contraindicated (see Tables 1 and 2). Children weighing 15 kg or less have a lower likelihood of infection and therefore do not receive presumptive treatment.
Post-arrival Management
- Asymptomatic refugees who did not receive overseas presumptive ivermectin treatment may be presumptively treated at arrival, or screened (“test and treat”) if contraindications to presumptive treatment exist or ivermectin is unavailable.
- A “test and treat” approach for strongyloidiasis should include a Strongyloides IgG serology. Stool ova and parasite testing, similar to STH testing, may also be done in conjunction. Notes on screening:
- Stool ova and parasite testing lacks sensitivity for Strongyloides infection and should not be used to rule out infection.
- Other testing modalities are sometimes used but are not widely available, and data on screening are limited (e.g., agar technique, polymerase chain reaction).
- Strongyloides IgG generally declines with resolution of infection and declines following treatment. The sensitivity and specificity vary according to the test being used, and false negative test results do occur, particularly in high-risk individuals.
- Refugees who have lived in a Loa loa-endemic country (see below) should be tested for the presence of Loa loa microfilaremia BEFORE being treated with ivermectin.
- In patients with a high microfilarial Loa loa load, treatment with ivermectin has been associated with fatal encephalopathy. Detailed information on Loa loa can be found on the CDC Parasitic Diseases website.
- Infection should be considered in children less than 15 kilograms with signs or symptoms of infection (e.g., elevated absolute eosinophil count).
- Mothers infected with Strongyloides are known to pass IgG to their infants.
- The duration of infant Strongyloides IgG positivity from maternal-fetal transmission of immunoglobulins is unknown.
- Treatment is not 100% effective, and continuation or recurrence of signs and symptoms (e.g., ongoing eosinophilia) should prompt further investigation and may necessitate repeat treatment.
See additional detailed information on Strongyloides.
Loa loa
Background
- Loa loa is one of several filarial infections that may affect refugees. These infections, including Loa loa, are not discussed in detail in this document. Further information is available at the CDC Parasitic Diseases website.
- With respect to this document, Loa loa is discussed in relation to treatment considerations for strongyloidiasis with ivermectin.
- Refugees are not presumptively treated for Loa loa before departure.
- Refugees from areas with Loa loa are not treated with ivermectin for strongyloidiasis.
Epidemiology
- Loa loa is typically found in West and Central Africa.
- The following countries are endemic for Loa loa, and refugees who have lived in any of the affected areas or in these countries do not receive presumptive ivermectin before departure:
- Angola
- Cameroon
- Central African Republic
- Chad
- Congo
- Ethiopia*
- Equatorial Guinea
- Gabon
- Nigeria
- South Sudan
- Democratic Republic of Congo
*Several areas of Ethiopia currently have endemic Loa loa. However, resettling refugees are not from these areas. Ethiopian refugees are receiving presumptive treatment with ivermectin for Strongyloides infection.
- A graphic describing Loa loa lifecycle and transmission is available from the CDC Division of Parasitic Diseases and Malaria.
Post-arrival Management for Strongyloides in those at risk for Loa loa infection
- Refugees who are from or who have lived in an area or country endemic for Loa loa are not treated presumptively for Strongyloides with ivermectin until high microfilarial load from Loa loa has been ruled out.
- For refugees who are at significant risk of Strongyloides, have potential past exposure to Loa loa, and have not received pre-departure (or previous) treatment with ivermectin:
- A single thin and thick blood smear between 10 a.m. and 2 p.m. can be performed (Inform the laboratory there is concern of Loa loa infection).
- If the thin and thick smear is negative: High-burden Loa loa parasitemia has been ruled out, and ivermectin may be offered.
- If the thin and thick smear is positive:Consult CDC for further guidance regarding evaluation and treatment options for Loa loa.
- NOTE: A negative thin and thick smear does not rule out Loa loa infection; it only rules out a high microfilarial load.
- Alternatively, these refugees may be offered a Strongyloides serologic test. If negative for Strongyloides, no ivermectin is needed. If positive, a thin and thick blood smear between 10 a.m. and 2 p.m. for Loa loa microfilaria (as above) should be performed before treatment with ivermectin.
- A single thin and thick blood smear between 10 a.m. and 2 p.m. can be performed (Inform the laboratory there is concern of Loa loa infection).
See additional information on Loa loa, including treatment.
Soil-transmitted Helminth Infections
Background
- CDC pre-departure albendazole treatment was initiated in Africa and Asia in 1999, and in the Middle East in 2008.
Epidemiology
- Soil-transmitted helminth infections, including Ancylostoma duodenale and Necator americanus (hookworm), Ascaris lumbricoides (ascariasis), and Trichuris trichiura (whipworm) are often found in areas with warm and moist climates where sanitation and hygiene are poor, including in temperate zones during warmer months.
Transmission
- Ascaris and Trichuris infections occur through indirect exposure. These infections require exposure to eggs that have embryonated in the environment.
- Hookworm infections require direct contact of skin with contaminated soil, and penetration of the larval worm through skin. One kind of hookworm can also be transmitted through the ingestion of larvae.
Clinical Information
- Infections are most frequently asymptomatic or subclinical.
- Ascariasis, when symptomatic, may be associated with abdominal pain, cough (Löffler syndrome), and if high-burden, intestinal obstruction, appendicitis, or cholecystitis.
- Trichuriasis (whipworm)may cause bloody stools, anemia, and rarely, rectal prolapse.
- Hookworm may cause anemia, which is of particular concern in young children and pregnant people. The most common signs and symptoms include those associated with anemia (e.g., weakness, pallor), as well as abdominal pain, diarrhea, and eosinophilia. Chronic infection with Ascaris, Trichuris, or hookworm is associated with poor growth in children.
Presumptive Treatment
- Most refugees receive overseas presumptive treatment with albendazole, unless contraindicated (see Tables 1 and 2). A single dose of albendazole is highly effective against ascariasis, and moderately effective against hookworm and trichuriasis. Additional information on treatment schedules for presumptive parasitic infections is available on the CDC Immigrant and Refugee Health website.
Post-arrival Management
- Refugees who have received pre-departure treatment do not require further screening or testing for soil-transmitted helminths unless they have symptoms, or have signs of infection such as persistent eosinophilia (Table 3).
- Asymptomatic refugees who did not receive overseas presumptive treatment may be presumptively treated upon arrival with a single dose of albendazole. Asymptomatic refugees can also be screened (“test and treat”) if contraindications to presumptive treatment exist or albendazole is unavailable.
- “Test and treat” approach for STHs is generally done with two or more separate stool ova and parasite tests done by concentration technique; samples must be collected 12 to 24 hours apart because shedding may be intermittent.
- Young infants ( less than 6 months) without history of direct skin contact with soil or fecal-oral contact are at low risk of infection. Therefore, the value of routine screening in infants is unknown. Testing should be performed if the infant has symptoms or signs of infection (including elevated eosinophil count).
See additional information on soil-transmitted helminths.
Schistosoma Species
Background
- Praziquantel is the drug of choice for treating schistosome infections. CDC presumptive praziquantel treatment was initiated in 2010 for sub-Saharan African (SSA) refugees.
Epidemiology
- All SSA countries (except Lesotho [PDF – 6 pages]) are considered endemic for schistosomiasis. The majority of infections are caused by Schistosoma mansoni or S. haematobium.
- Documented seroprevalence in some groups of SSA refugees has exceeded 70% (such as Somali Bantu) 3.
Transmission
- Infection requires direct skin contact with schistosoma-contaminated fresh water.
- The larval parasite enters by penetrating the skin.
- Young infants without direct skin exposure to contaminated fresh water are at low risk of infection. However, babies bathed in basins with contaminated fresh water are at risk of infection.
- Graphics describing lifecycle and transmission of schistosoma species are available from the CDC Division of Parasitic Diseases and Malaria.
Clinical Information
- Infections are frequently asymptomatic or subclinical.
- Infection may persist for more than 50 years, causing ongoing damage. Clinical cases have been documented years after migration from an endemic area.
- Infection may cause many clinical manifestations.
- The most common sign of infection is asymptomatic eosinophilia.
- Severe manifestations of chronic S. mansoni infections include bowel wall pathology, liver cirrhosis, and portal hypertension.
- Severe manifestations of chronic S. haematobium infections include dysuria, hematuria, urinary tract obstruction, renal failure, and bladder cancer.
- While less common, acute myelitis can result from ectopic deposition of schistosome eggs in the spinal cord. This is a severe complication that occurs when the infection involves the central nervous system.
Presumptive Treatment
- Most SSA refugees receive overseas pre-departure treatment with praziquantel, unless contraindicated (see Tables 1 and 2). Additional information on treatment schedules for presumptive parasitic infections is available on the CDC Immigrant and Refugee Health website.
Post-arrival Management
- Asymptomatic SSA refugees who did not receive overseas presumptive praziquantel treatment may be presumptively treated after arrival, or screened (“test and treat”) if contraindications to presumptive treatment exist or if praziquantel is unavailable or inaccessible (see Tables 1 and 2).
- A “test and treat” approach for schistosomiasis should include a schistosoma IgG serology. Testing of stool and urine for eggs and urine analysis for red blood cells, may also be done in conjunction. However, because of lack of sensitivity, these tests should not be used alone to rule out infection 5, 6, 11, 12. Notes on screening:
- Previous treatment does not decrease schistosoma IgG levels, so persistently positive schistosome antigen-specific IgG results do not necessarily indicate current infection and should not be used to monitor treatment success 3.
- Mothers with schistosomiasis are known to pass IgG to their infants. The duration of infant schistosoma IgG positivity from maternal-fetal transmission of immunoglobulins is unknown. Positive IgG results in infants should be interpreted in consultation with CDC experts. Unless an SSA infant has direct skin-to-fresh water contact, infection with Schistosoma species is relatively unlikely and testing is not recommended.
- Treatment is not 100% effective, and continuation of symptoms or signs of infection (e.g., ongoing eosinophilia) should prompt further investigation; repeat treatment may be necessary.
See additional information regarding schistosomiasis.
Giardia
Background
- Refugees are not presumptively treated for Giardia before departure.
Epidemiology
- Giardia is found worldwide, including in every region of the United States.
- Giardia is the most prevalent pathogenic intestinal parasite among newly arrived refugees. There have been no reported outbreaks of giardial infection linked to refugees 13, 14.
Transmission
- Infection requires ingestion of the Giardia parasite, through either drinking/swallowing contaminated water or eating uncooked food that contains Giardia organisms.
- Anything that comes into contact with feces from infected humans or animals can become contaminated with the Giardia parasite. Infection can also be caused by ingesting Giardia resulting from contact with surfaces (bathroom handles, changing tables, diaper pails, or toys) contaminated with feces from an infected person or animal.
- Person-to-person transmission can also occur through fecal-oral exposure.
- Transmission is not possible through contact with blood or other body fluids.
- A graphic describing Giardia lifecycle and transmission is available from the CDC Division of Parasitic Diseases and Malaria.
Clinical Information
- The majority of Giardia infections are asymptomatic, and it is unknown whether treatment of asymptomatic infection is of any benefit to the infected host.
Post-arrival Management
- Testing is recommended for those with symptoms.
- Symptoms of chronic infection may include diarrhea, anorexia, poor weight gain/failure to thrive/malnutrition, abdominal distension, and/or flatulence. Giardiasis is not associated with eosinophilia.
- Stool ova and parasite testing has low sensitivity. Stool antigen is the preferred test for identifying Giardia infections.
- Screening asymptomatic children younger than 5 years of age for Giardia stool antigen and treating those who test positive may be beneficial and is done by some experts in refugee children, although data supporting this approach are lacking.
See additional information on Giardia, including treatment.
Table 1
Recommended Medication Regimen for Presumptive Treatment or Treatment of Identified Parasitic Infections in Adults, Pregnant People, and Childrenx
| Adults | |||
|---|---|---|---|
| Refugee Population | Treatment Regimens by Pathogen | ||
| Albendazole for Soil-transmitted Helminths | Ivermectin for Strongyloidiasis | Praziquantel for Schistosomiasis | |
| Asia, Middle East, North Africa, Latin America, and Caribbean | 400 mg orally as a single dose | 200 μg/kg orally as a single dose | Not recommended |
| Sub-Saharan Africa (non Loa loa-endemic) | 400 mg orally as a single dose | 200 μg/kg orally as a single dose | 40 mg/kg‡ orally as a single dose |
| Sub-Saharan Africa (Loa loa– endemic) | 400 mg orally as a single dose | 200 μg/kg orally as a single dose if no Loa loa infection | 40 mg/kg‡ orally as a single dose |
| Pregnant People§ | |||
|---|---|---|---|
| Refugee Population | Treatment Regimens by Pathogen | ||
| Albendazole for Soil-transmitted Helminths | Ivermectin for Strongyloidiasis | Praziquantel for Schistosomiasis | |
| Asia, Middle East, North Africa, Latin America, and Caribbean | Not recommended for presumptive treatment. Relative contraindication for diagnosed infection | Not recommended for presumptive treatment. Relative contraindication for diagnosed infection | Not recommended |
| Sub-Saharan Africa | Not recommended for presumptive treatment. Relative contraindication for diagnosed infection | Not recommended for presumptive treatment. Relative contraindication for diagnosed infection | 40 mg/kg‡ orally as a single dose |
| Children‖ | |||
|---|---|---|---|
| Refugee Population | Treatment Regimens by Pathogen | ||
| Albendazole for Soil-transmitted Helminths | Ivermectin or High-dose Albendazole for Strongyloidiasis | Praziquantel for Schistosomiasis | |
| Asia, Middle East, North Africa, Latin America, and Caribbean | Presumptive therapy is not recommended for any infant less than 12 months of age.
12–23 months of age: 200 mg orally as a single dose >2 years: 400 mg orally as a single dose |
200 μg/kg orally as a single dose
Should not be used presumptively if child is <15 kg or from Loa loa-endemic country |
Not recommended |
| Sub-Saharan Africa | Presumptive therapy is not recommended for any infant less than 12 months of age.
12–23 months of age: 200 mg orally as a single dose >2 years: 400 mg orally as a single dose |
200 μg/kg orally as a single dose
Should not be used presumptively if ≤15 kg or from Loa loa-endemic country |
Presumptive treatment is not recommended for children < 4 years of age.
≥4 years: 40 mg/kg‡ orally as a single dose |
xInformation on overseas pre-departure intervention programs is available on the CDC Immigrant and Refugee Health website.
‡ May be divided and given in two doses for better tolerance.
§ The risk of strongyloidiasis during pregnancy needs to be balanced with the risk of treatment. While the infection itself does not pose a particular pregnancy risk, if a pregnant person with strongyloidiasis receives immunosuppressive drugs (e.g., betamethasone for preterm labor), she is at risk of hyperinfection syndrome and death.
‖If Strongyloides, other STH, or schistosomiasis infection is identified in children below age or weight limitations, treatment should still be offered.
Table 2
Contraindications to Presumptive Treatment with Albendazole, Ivermectin, and Praziquantel
*Cysticercosis (e.g., subcutaneous nodules) is a parasitic tissue infection caused by Taenia solium. Taenia brain cysts (neurocysticercosis) may cause seizures or other neurologic disorders. Presumptive treatment with albendazole and/or praziquantel is contraindicated in these cases, and expert consultation is strongly recommended.
**Treatment of identified strongyloidiasis is contraindicated in patients with high-burden Loa loa parasitemia (see Loa loa endemic country list, above). In these cases, ivermectin may precipitate encephalopathy.
Summary of the Overseas Presumptive Treatment Information and the Impact on Post-arrival Management
The majority of resettling populations receive pre-departure albendazole, making stool testing or treatment for STH unnecessary for most refugees.
Most refugees from SSA have been presumptively treated for schistosomiasis with praziquantel; therefore, domestic testing and treatment for schistosomiasis are not necessary for most refugees from SSA.
Nearly all refugees (regardless of country of origin) are at risk for Strongyloides and should be tested and treated with ivermectin, if not previously pretreated. However, refugees from areas of SSA endemic for Loa loa deserve special attention. These refugees should have serologic testing for Strongyloides parasites and if positive, high-burden Loa loa parasitemia should be ruled out before treatment is initiated. Ivermectin given in the setting of high-burden Loa loa infection may result in encephalopathy. Screening for high-burden Loa loa parasitemia may be achieved by collecting a daytime (10 a.m.–2 p.m.) thin and thick blood smear for Loa loa. If the thin and thick blood smear is negative, it is safe to treat the Strongyloides infection with ivermectin. If the daytime thin and thick blood smear is positive, contact CDC for further guidance regarding workup and management. Additional information on loiasis is available on the CDC Parasitic Diseases website.
Detailed information regarding overseas presumptive treatment, including regions and countries where presumptive treatment has been implemented, can be found on the CDC Immigrant and Refugee Health website. Pre-departure presumptive treatment for strongyloidiasis will likely be expanded to other refugee populations in the future. It is important to note that certain populations may receive incomplete or no pre-departure presumptive treatment. Specific individuals may not be eligible for certain pre-departure medications because of pregnancy, breastfeeding, young age, or other contraindications, such as known hypersensitivity, allergies, history of seizures, or known neurocysticercosis (see Table 2). Thus, post-arrival guidance is designed to be flexible and are contingent upon whether the pre-departure presumptive treatment was received. Ideally, clinicians will have documentation of presumptive therapy received by each refugee at the time of post-arrival screening.
Finding Information on Presumptive Treatment Received
There have been substantial challenges in providing the records of presumptive parasite treatment for individual refugees to US clinicians at the time of their post-arrival medical examination. Therefore, individual refugee presumptive treatment records are made available in electronic and hard copy formats, and general information is provided on the CDC Immigrant and Refugee Health website.
- International Organization for Migration (IOM) Bag: Records of presumptive pre-departure treatment received by the refugee are usually available in the IOM bag, also known as the “blue and white bag,” carried by the refugee. The refugee should be directed to bring the IOM bag to the clinic at the time of the screening appointment. The provider may also check with the voluntary resettlement agency coordinating the refugee’s care, as it may have a copy of these records.
- Electronic Disease Notification System (EDN): Records will be made available to state refugee health coordinators and some clinics through EDN. Clinicians may request this information from their state’s refugee health program.
- CDC Website: Information regarding overseas presumptive treatments, including specifics on refugee populations receiving presumptive treatments, and countries where pre-departure presumptive treatment has been implemented, is available on the CDC Immigrant and Refugee Health website.
Resources and Summary Recommendations for Post-arrival Management of Strongyloidiasis, Other Soil-transmitted Helminths, and Schistosomiasis
The following sections recommend approaches for evaluating refugees during the post-arrival domestic medical visit based on whether the refugee had documented or presumed pre-departure treatment. For the purpose of post-arrival management of parasites, refugees may be divided into two groups—incomplete presumptive treatment and complete presumptive treatment.
Incomplete Presumptive Treatment
- Refugees who did not receive any presumptive treatment before departure for the United States due to contraindication, as well as those from countries where pre-departure presumptive treatment has yet to be implemented.
- Refugees who received partial presumptive treatments, such as those excluded due to contraindications to presumptive treatment with albendazole, praziquantel, or ivermectin (see Table 2).
Complete Presumptive Treatment
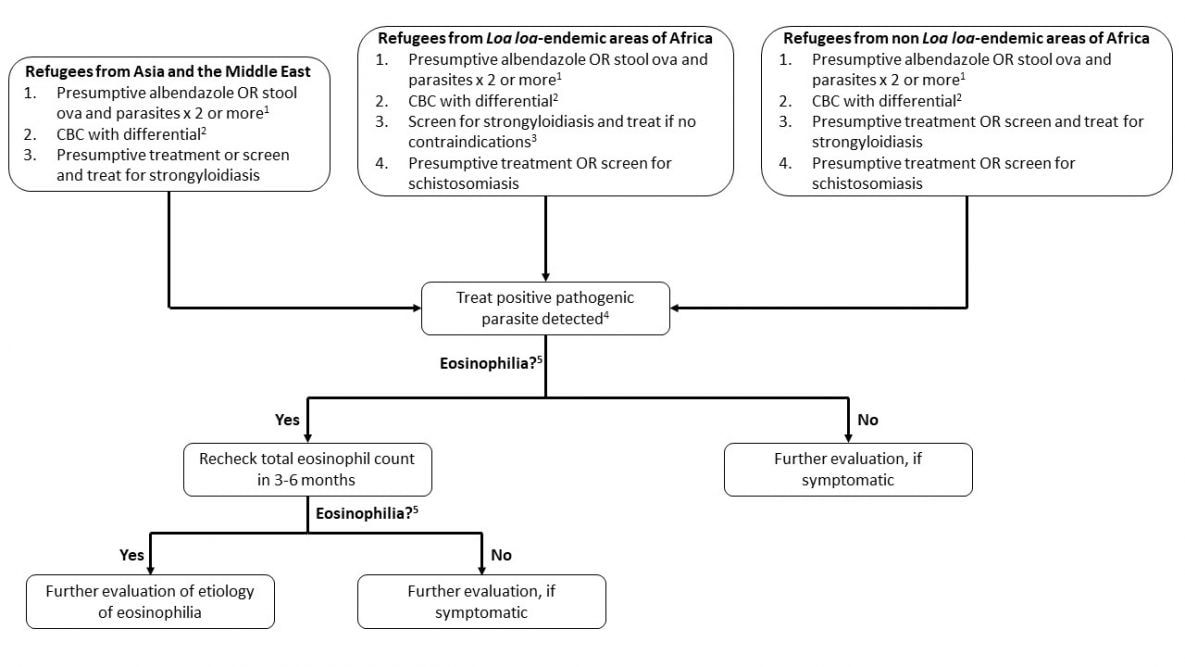
Refugees in this category received all recommended overseas presumptive treatment for parasites before departure. These individuals require no further testing for these infections unless they have persistent eosinophilia or other signs or symptoms of infection 3 to 6 months after initial treatment (Figure 1). Persistent eosinophilia may indicate treatment failure or unidentified parasitic infection (Table 3), and follow-up is recommended (Figure 1). In these cases, please consult a tropical medicine specialist.
Figure 1
Management of Asymptomatic Refugees for Parasitic Infection if They Received No or Incomplete Pre-departure Treatment and Initial Approach to Persistent Eosinophilia
- Sensitivity varies according to parasite (e.g., very sensitive for Ascaris but may need more than six specimens to reliably exclude Strongyloides), and a minimum of two specimens is suggested. Many pathogens and nonpathogenic organisms may be detected on stool ova and parasite testing (Table 4).
- CBC: Complete blood count and differential (not recommended as screening test for parasitic infection but routinely obtained on screening for newly arrived refugees)
- See text for discussion of screening for Loa loa.
- See DPDx Laboratory Identification of Parasites of Public Health Concern for more information and diagnostic and management assistance.
- Eosinophilia = eosinophil count of > 450 per microliter (μL)
Persistent Eosinophilia in Refugee Populations
If the initial eosinophil count is normal, repeat testing for eosinophilia in an asymptomatic person is not indicated. If the initial absolute eosinophil count is greater than 450 cells/μL, clinical decision making should be guided by the history of presumptive treatment. Although many parasites are associated with eosinophilia, others are not. In addition, eosinophilia may also be associated with noninfectious disorders, and the broader differential diagnosis should be kept in mind (Table 3). Persistent eosinophilia in a refugee is most likely due to parasitic infection, and the most commonly identified etiologies are failed treatments of strongyloidiasis, schistosomiasis, trichuriasis, and other commonly encountered parasites such as tapeworm (e.g., Hymenolepis nana, Taenia species [spp]). Persistent eosinophilia frequently requires expert consultation.
Table 3
Parasitic and Non-parasitic Causes of Eosinophilia
| Parasites that cause eosinophilia commonly found in stool examination | Other parasitic infections associated with eosinophilia | Nonparasitic causes of eosinophilia | Parasites commonly found in the stool NOT typically associated with eosinophilia |
|---|---|---|---|
| Ascaris lumbricoides
Hookworm (Ancylostoma spp, Necator spp) Trichuris trichiura Strongyloides stercoralis* Tapeworm (Taenia solium, T. saginata) Schistosoma (most commonly S. mansoni*, S. haematobium*, S. japonicum*) Other flukes (Paragonimus spp*, Opisthorchis spp*, Fasciola spp*) |
Angiostrongylus
Anisakis Capillaria spp Echinococcus spp Filariasis (Wuchereria bancrofti, Brugia spp, Mansonella spp, Onchocerca volvulus, Dracunculus medinensis, Loa loa) |
Asthma
Atopy Drug allergy Eosinophilic leukemia Hodgkin’s lymphoma Hypereosinophilic syndrome Pemphigoid Pemphigus Polyarteritis nodosa |
Entamoeba spp (histolytica, dispar, other)
Cryptosporidium spp Giardia intestinalis (also known as G. lamblia or G. duodenalis) |
* These parasites commonly cause eosinophilia, but stool examination alone is insufficient for diagnosis. Special testing is frequently needed.
Other Parasitic Infections Common in Refugees
Many parasitic infections in refugees may be detected when stool exams are performed (Table 4). Some (pathogenic) require treatment, while others do not.
Table 4
Common Parasites Detected on Stool Examination
| Pathogenic | Controversial | Nonpathogenic | ||||
|---|---|---|---|---|---|---|
| Nematodes | Trematodes | Cestodes | Protozoa | Protozoa | Other | Protozoa |
| Ascaris lumbricoides; Hookworm (Necator americanus and Ancylostoma duodenale);
Trichuris trichiura; Strongyloides stercoralis |
Schistosoma (S. mansoni, S. haematobium, S. japonicum);
Other flukes (Opisthorchis spp) |
Taenia solium; Taenia saginata;
Hymenolepis nana |
Entamoeba histolytica* Giardia intestinalis (G. lamblia or G. duodenalis) | Dientamoeba fragilis; Entamoeba polecki | Blastocystis hominis | Entamoeba dispar*; Entamoeba moshkovskii*;
Entamoeba coli; Entamoeba hartmanii; Endolimax nana; Iodamoeba butschlii; Chilomastix mesnili |
*The cysts of E. histolytica, E. dispar, and E. moshkovskii are morphologically identical by stool microscopy. When cysts are detected, stool antigen testing is recommended to distinguish the potentially pathogenic E. histolytica from the more common, nonpathogenic species.
Further information on these parasitic infections and assistance with diagnosis and treatment may be provided by the Centers for Disease Control and Prevention’s Division of Parasitic Diseases.