Syphilis
Preface
The medical screening for syphilis among persons applying for adjustment of status for US permanent residence and others required to have a medical examination, hereafter referred to as applicants, is an essential component of the immigration process. Because syphilis may be challenging to diagnose and treat, these Technical Instructions provide civil surgeons with a method for recording findings from the immigration medical examination onto Form I-693, Report of Medical Examination and Vaccination Record, and provide additional guidance for the civil surgeon in classifying applicants.
The instructions in this document supersede all previous Technical Instructions, Updates to the Technical Instructions, memoranda and letters to civil surgeons. These Technical Instructions are to be followed for syphilis screening and treatment among all applicants and are effective as of September 1, 2021.
Visit the Technical Instructions for Civil Surgeons webpage for more information about the medical examination of applicants conducted in the United States.
Key Concepts
- All applicants aged 18 years to those aged less than 45 years must be tested for evidence of syphilis.
- Applicants aged less than 18 years or 45 years or greater must be tested if there is reason to suspect infection with syphilis.
- Nontreponemal and treponemal laboratory tests must be ordered by the civil surgeon at the time of the immigration medical examination and all tests for syphilis must be performed on the same blood sample. Testing performed elsewhere, or prior to the civil surgeon’s examination of the applicant, is not acceptable.
- Either the traditional or the reverse algorithm may be used for syphilis laboratory testing.
- External genital and rectal examinations for staging syphilis must only be performed AFTER laboratory confirmation of diagnosis and a chaperone must be present during these examinations.
Syphilis Screening
Syphilis is a sexually transmitted, systemic disease caused by the bacterium Treponema pallidum subspecies pallidum. The disease has often been called “the great imitator” because of its wide variety of signs and symptoms, with different stages having different clinical manifestations. There are three infectious stages (primary, secondary, and early latent disease) and two noninfectious stages (late latent and tertiary disease). Untreated syphilis can progress and lead to serious long-term sequelae and, rarely, death.
Medical History
Obtaining the applicant’s medical history must include inquiring about prior history and treatment for syphilis; a sexual partner with syphilis; or any history of painless sores on the genitals, anus, or mouth or a rash on the body, especially on the palms of the hands or soles of the feet.
Physical Examination
If the history or serologic tests are suggestive, a physical examination may be warranted that should include an evaluation for oral signs of syphilis, including mouth sores (chancres) or mucus patches, or rashes on the body, particularly on the palms of the hands or soles of the feet (a characteristic of syphilis infection that is unusual in other conditions). An external genital examination is not required and must not be performed unless the applicant has laboratory confirmation of diagnosis (see screening results below); these applicants should undergo an evaluation for external genital, anal, or perianal sores (chancres) or other lesions (i.e. condyloma lata) to help determine the stage of syphilis. A gown or sheet must be provided to allow privacy and a chaperone MUST be present, regardless of the applicant’s gender, if external genital and rectal exams are needed. The chaperone must be a staff member of the gender the patient feels most comfortable with and not a family member. However, family members may be present for the exam if the applicant requests. Sores consistent with syphilis are typically painless, indurated, clean-based genital, rectal or oral ulcers. Regional lymphadenopathy may be present in primary or secondary syphilis. Applicants with reactive serologic tests consistent with syphilis should be asked specifically about and evaluated for clinical symptoms or signs suggestive of neurosyphilis (e.g., cranial nerve dysfunction, meningitis, stroke, acute or chronic altered mental status, loss of vibration sense, and auditory or ophthalmic abnormalities). Neurosyphilis can occur at any stage of infection.
Histories or clinical signs suggesting syphilis in children must be properly evaluated with serologic testing. Physical examination findings of congenital syphilis in infants may include nonimmune hydrops, jaundice, hepatosplenomegaly, rhinitis, skin rash, and pseudoparalysis of an extremity. In older children, signs of untreated congenital infection may include interstitial keratitis (5-20 years of age), eighth cranial nerve deafness (10-40 years of age), Hutchinson teeth (peg-shaped, notched central incisors), anterior bowing of the shins, frontal bossing, mulberry molars, saddle nose, rhagades (linear scars around the mouth), and Clutton’s joints (symmetric, painless swelling of the knees).
Laboratory Testing
CDC allows either the traditional or the reverse algorithm to be used by civil surgeons when screening applicants for syphilis.
- The traditional algorithm starts with a nontreponemal test with positive (or reactive) samples being reflexed to a treponemal test.
- The reverse algorithm starts with a treponemal test with positive (or reactive) samples being reflexed to a nontreponemal test. Discordant samples are resolved with a second treponemal test, preferably one based on different antigens than the original treponemal test.
Laboratories should consider their patient population and syphilis risk when considering the traditional or the reverse screening algorithm. More information is provided here [PDF – 40 pages]. The reverse screening algorithm can be more convenient for laboratories, but its clinical interpretation is problematic, as this testing algorithm can identify persons previously treated for syphilis, those with untreated or incompletely treated syphilis, and persons with false-positive results that can occur with a low likelihood of infection. Therefore, the reverse screening algorithm is not recommended in high prevalence settings or patient populations. All tests used by civil surgeons must be conducted by appropriately trained technologists following standard operating procedures in a qualified laboratory that is enrolled in an external quality assurance program (e.g., proficiency testing) program.
Regardless of the algorithm used, all nontreponemal and treponemal tests must be performed on the same blood sample. A nontreponemal test (i.e., Venereal Disease Research Laboratory [VDRL] or Rapid Plasma Reagin [RPR]) with positive or reactive results must have a quantitative titer result reported (e.g., 1:4) and documented on the Form I-693 at Part 8, Civil Surgeon Worksheet, Section 1B. Communicable Diseases, Syphilis. The following tests are permitted for treponemal testing: T. pallidum passive particle agglutination (TP-PA) assay, Treponema pallidum haemagglutination (TPHA) test, enzyme immunoassays (EIAs), chemiluminescence immunoassays (CIAs), fluorescent treponemal antibody absorbed (FTA-ABS) tests, immunoblots (use products of this test that are approved by the US Food and Drug Administration [FDA]), or rapid treponemal assays (use products of this test that are FDA-approved).
Syphilis testing must be performed at the time of the immigration medical examination. Civil surgeon sites that are independent of US health departments must not refer applicants to a health department for syphilis testing; all nontreponemal and treponemal tests must be ordered by the civil surgeon and must be performed independently of a health department. Tests performed elsewhere, or prior to the civil surgeon’s examination of the applicant, are not acceptable.
Children with history of physical stigmata suggesting syphilis infection must have serologic testing for syphilis. Older infants (aged ≥1 month) and children who are identified as having reactive serologic tests for syphilis should have maternal serology and records reviewed to assess whether they have congenital or acquired syphilis and whether they have been previously treated. Any child at risk for untreated congenital syphilis should be treated, and recommended to have a full evaluation, which may include CSF analysis, complete blood count and differential, and long-bone radiographs, depending upon previous documented evaluation. Dark field microscopic examination of suspicious lesions or body fluids (e.g., nasal discharge) also should be performed. Civil surgeons should refer to CDC’s Sexually Transmitted Infections (STI) Treatment Guidelines for specific guidance on testing in children.
All applicants diagnosed with syphilis should be advised to be tested for other STIs, including chlamydia, gonorrhea, and HIV. The consent for HIV testing should include the following:
- Applicants understand they do not have to be tested for HIV.
- Applicants understand that if they would like to be tested for HIV, they do not have to be tested for HIV by a civil surgeon.
- Applicants understand that civil surgeons must include the test results on the paperwork they complete.
If the applicant consents, civil surgeons should perform HIV testing.
- Applicants with a positive (or reactive) nontreponemal test (i.e., RPR or VDRL) and a negative or (nonreactive) treponemal-specific test are No Class for syphilis.
- Applicants with a positive (or reactive) nontreponemal test (i.e., RPR or VDRL) and a positive (or reactive) treponemal test are Class A for syphilis and will remain Class A until treated.
- If the reverse algorithm is used, applicants with a positive (or reactive) treponemal test and a negative (or nonreactive) nontreponemal test must undergo a second treponemal test, preferably one based on different antigens than the original treponemal test.
- If the repeat treponemal test is negative (or nonreactive), applicants are No Class for syphilis
- If the repeat treponemal test is positive (or reactive) and there is unknown or no history of prior syphilis treatment, applicants are Class A for syphilis and will remain Class A until treated.
- If the repeat treponemal test is positive (or reactive) and there is documented history of prior syphilis treatment and no concern for re-exposure, applicants are No Class for syphilis.
- After completing treatment, they are classified as Class B.
- The evaluation is complete when the required aspects of the medical examination have been completed and the applicant can be assigned a syphilis classification.
It is important that syphilis be correctly diagnosed. Correct diagnosis of syphilis will ensure that affected applicants receive appropriate treatment minimizing long-term sequelae and reducing further spread of the disease. False-positive nontreponemal test results can be associated with multiple medical conditions and factors unrelated to syphilis, including other infections (e.g., HIV, tuberculosis, hepatitis, malaria), autoimmune conditions (e.g., systemic lupus, rheumatoid arthritis), vaccinations (e.g., smallpox, MMR), injection drug use, pregnancy, and older age. Therefore, persons with a positive (or reactive) nontreponemal test must always receive a treponemal test to confirm the syphilis diagnosis (traditional algorithm). Treponemal tests might be positive (or reactive) with serum from patients with other infectious and noninfectious diseases, conditions, Yaws (T. pallidum subspecies pertenue), Pinta (T. carateum), Bejel (T. pallidum subspecies endemicum) or other treponemal diseases.
Civil surgeons must refer to CDC’s STI Treatment Guidelines for more details regarding interpretation of nontreponemal test titers for applicants with a positive (or reactive) treponemal test and documented history of prior syphilis treatment. Only nontreponemal test titers from the same assay type can be compared directly. Official documentation of treatment history and previous laboratory results should be included with the applicant’s exam packet.
Figure 1. Syphilis Screening using the Traditional Algorithm for All Applicants Aged 18 Years to Those Aged less than 45 Years
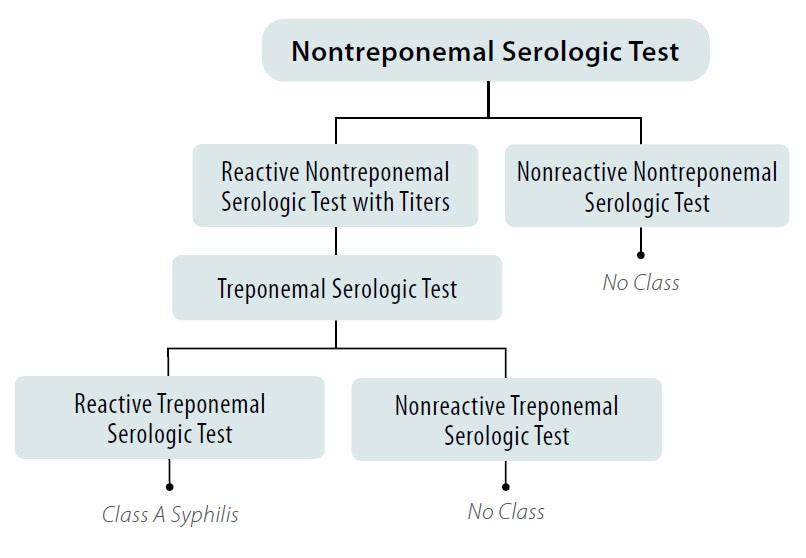
*And Any Applicant Aged less than 18 Years or 45 Years or greater Suspected of having Syphilis Infection
Table 1. Nontreponemal and treponemal test results and syphilis classifications using the traditional algorithm.
| Treponemal Test Results | ||
|---|---|---|
| Nontreponemal Test Results | Reactive | Non-Reactive |
| Reactive > 1:8 |
|
|
| Reactive < 1:8 |
|
|
| Non-reactive |
|
|
Figure 2. Syphilis Screening using the Reverse Algorithm for All Applicants Aged 18 Years to Those Aged less than 45 Years
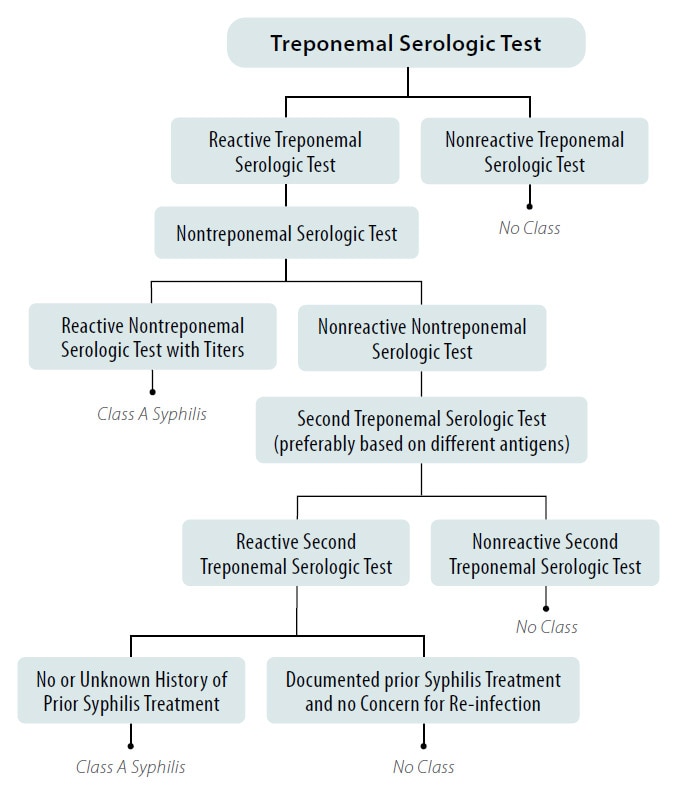
*And Any Applicant Aged less than 18 Years or 45 Years or greater Suspected of having Syphilis Infection
Table 2. Nontreponemal and treponemal test results and syphilis classifications using the reverse algorithm.
| Treponemal Test Results | Nontreponemal Test Results | ||
|---|---|---|---|
| Reactive ≥ 1:8 | Reactive < 1:8 | Nonreactive | |
| Reactive |
|
|
|
| Nonreactive |
|
||
Syphilis Treatment
Civil surgeons must treat syphilis according to CDC’s Sexually Transmitted Infections (STI) Treatment Guidelines, which are periodically updated.
- Details of treatment must be provided in Part 7, Civil Surgeon Worksheet, Section 1.B., Communicable Diseases, and Syphilis of Form I-693 [PDF – 14 pages].
- Benzathine penicillin G (BPG) is the preferred treatment regimen for syphilis. Although alternative regimens are known to be effective, applicants should be informed that BPG is the first-line recommendations in the Treatment Guidelines.
- No proven alternatives to injectable penicillin regimens are available for treating neurosyphilis, congenital syphilis, or syphilis in pregnant women. Therefore, infected applicants with a history of penicillin allergy who have neurosyphilis or congenital syphilis, or are pregnant advised to be desensitized and treated with penicillin if appropriate facilities are available.
Treatment – Post Evaluation
- Adult applicants treated for syphilis should be informed by civil surgeons that they will need follow-up care for clinical and serologic re-evaluation in 6 months (3 months if HIV positive and treated for primary or secondary syphilis).
- Children should receive follow-up care 2-3 months after treatment by the treating physician; this follow-up does not have to occur before finalizing the medical examination or affect the examination validity period.
- A provision allows applicants undergoing treatment for syphilis to apply for a Class A waiver.
- Waivers will become unnecessary after completion of treatment, as the applicant will be classified as Class B for syphilis.
A provision allows applicants with a Class A physical disorder to petition for a Class A waiver. The Application for Waiver of Grounds of Inadmissibility Form (I-601) must be completed. These waivers are submitted to the Department of Homeland Security (DHS), US Citizenship and Immigration Services (USCIS) on an individual basis. DGMQ also reviews the waivers and supporting medical examination to provide an opinion regarding the case to the requesting entity (Department of State or DHS, USCIS). DGMQ’s review of the waiver and supporting medical examination documentation is to ensure that the applicant has been classified properly and that an appropriate US healthcare provider is identified for the applicant. DHS, USCIS has the final authority to adjudicate the waiver request.
Documentation
The Form I-693 [PDF – 14 pages], Report of Medical Examination and Vaccination Record, must be completed in its entirety, signed, and placed in a sealed envelope for submission to the Department of Homeland Security (DHS), Citizenship and Immigration Services (USCIS) office. This includes assigning a classification on the I-693 if an applicant is Class A or Class B for syphilis.
For applicants requiring syphilis treatment, the civil surgeon is required to document the following on Form I-693 at Part 8, Civil Surgeon Worksheet, Section 1.B., Communicable Diseases, Syphilis:
- Results and dates of treponemal and non-treponemal tests, including titers for nontreponemal tests. Type of test used should be noted.
- Result of the second treponemal test, if applicable, should be recorded under Remarks in the syphilis section.
- Stage of syphilis based on history and clinical findings
- Drug regimen received (including doses, dosage units, and administration routes of all medications), start date, completion date, and any periods of interruption.
- Clinical course, if applicable, such as clinical improvement or lack of improvement during and after treatment for primary or secondary syphilis, including resolution of symptoms and signs and any drug reactions.
| Acronym | Full Phrase |
|---|---|
| CDC | Centers for Disease Control and Prevention, United States |
| CIA | Chemiluminescence immunoassay |
| CSF | Cerebral spinal fluid |
| DHS | Department of Homeland Security |
| EIA | Enzyme immunoassay |
| FTA-ABS | Fluorescent treponemal antibody absorbed |
| HIV | Human immunodeficiency virus |
| RPR | Rapid plasma reagin |
| STI | Sexually transmitted infection |
| TP-PA | T. pallidum passive particle agglutination assay |
| USCIS | United States Citizenship and Immigration Services |
| U.S. FDA | U.S. Food and Drug Administration |
| VDRL | Venereal Disease Research Laboratory |