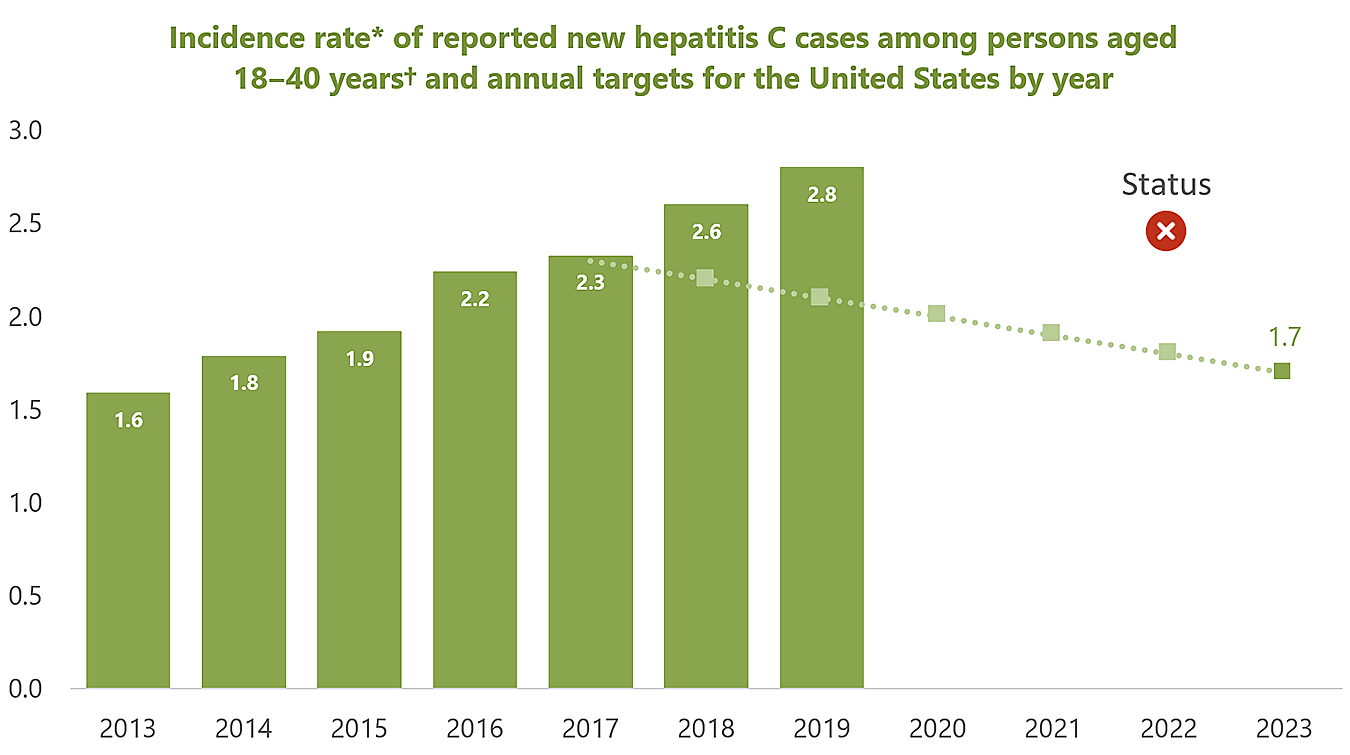
National Progress Report 2025: Reduce reported rate* of new hepatitis C virus infections among persons who inject drugs† by ≥25%
 |
Met or exceeded current annual target |  |
Moving toward annual target, but annual target was not fully met |  |
Annual target was not met and has not changed or moved away from annual target |
Source: CDC, National Notifiable Diseases Surveillance System (1)
*Rate per 100,000 population.
†Persons aged 18–40 years were used as a proxy for persons who inject drugs.
Summary of Findings
The rate of new hepatitis C cases reported to CDC among persons aged 18–40 years has increased steadily each year since 2013 to 2.8 cases per 100,000 population in 2019, above the 2019 target rate of 2.1 per 100,000 population. Injection-drug use is the most common risk reported for persons with new hepatitis C virus infection, and increases in hepatitis C incidence, particularly among persons aged 18–40 years, are temporally associated with increases in this risk factor (1).
Reduction needed to meet 2025 goal: A 39.3% reduction from the 2019 rate of reported new hepatitis C cases is needed to meet the 2025 goal of 1.7 cases per 100,000 population.
This reduction can best be achieved by
- Supporting local, state, and federal public health surveillance and other data-collection initiatives to detect where HCV transmission is occurring and provide evidence to guide strategies aimed at reducing hepatitis C incidence among groups at higher risk of infection, particularly persons who inject drugs (PWID).
- Providing hepatitis C-related health services, including routine hepatitis C testing for PWID and appropriate care and curative hepatitis C treatment for PWID living with hepatitis C.
- Implementing audience-friendly, culturally competent educational campaigns to encourage testing among PWID.
- Supporting implementation of comprehensive community-level programs for PWID (e.g., access to syringe services programs, linkage to medication-assisted treatment programs, testing, and treatment).
- Conducting prevention research to improve the effectiveness of hepatitis C prevention interventions and decrease hepatitis C incidence among PWID.
- Building partnerships to promote implementation of prevention strategies in settings that serve PWID to increase availability and utilization of hepatitis C prevention services in these settings.
Technical Notes
Data Sources: CDC, National Notifiable Diseases Surveillance System (NNDSS) and CDC/National Center for Health Statistics/US Census Bureau, Bridged-race Population Estimates
Numerator: Number of acute HCV infections reported annually among persons aged 18–40 years
Denominator: Total population of persons aged 18–40 years in reporting jurisdictions
Indicator Notes: (1) The NNDSS is a nationwide collaboration that enables all levels of public health to share notifiable-disease-related health information. Surveillance for viral hepatitis through NNDSS is based on case definitions developed and approved by the Council of State and Territorial Epidemiologists (CSTE) and CDC. Reported cases of acute viral hepatitis C are required to meet specific clinical and laboratory criteria. Only laboratory-confirmed cases of acute viral hepatitis are presented in this report. Acute hepatitis C is reportable in all jurisdictions. Health care providers, hospitals, and/or laboratories report cases to the local or state health department, and states voluntarily submit reports or notify CDC of newly diagnosed cases of hepatitis C that meet the CSTE/CDC surveillance case definition. Case rates per 100,000 population are calculated based on the projected resident population of the United States as of July 1 during each data-collection year.
Goal Setting: The 2025 goal of 1.7 per 100,000 population is consistent with CDC’s Division of Viral Hepatitis 2025 Strategic Plan and HHS’s 2021-2025 Viral Hepatitis National Strategic Plan. Annual targets assume a constant (linear) rate of change from the observed baseline (2017 data year) to the 2025 goal (2023 data year).
Limitations: Viral hepatitis is largely underreported in the NNDSS. Based on a simple, probabilistic model for estimating the proportion of patients who were symptomatic, received testing, and were reported to health officials in each year, the actual number of acute hepatitis C cases is estimated to be 13.9 times the number reported to CDC. (1–2) Additionally, rates of reported cases may vary over time based on changes in public and provider awareness, laboratory and diagnostic techniques, and the definition of the condition.
References
- Centers for Disease Control and Prevention. Viral Hepatitis Surveillance—United States, 2019. Atlanta: US Department of Health and Human Services, Centers for Disease Control and Prevention; 2021. Available at: https://www.cdc.gov/hepatitis/statistics/2019surveillance/index.htm.
- Klevens RM, Liu S, Roberts H, Jiles RB, Holmberg SD. Estimating acute viral hepatitis infections from nationally reported cases. Am J Public Health. 2014;104(3):482-7.