
Urban–Rural Differences in Health Care Utilization and COVID-19 Outcomes in Patients With Type 2 Diabetes
ORIGINAL RESEARCH — Volume 19 — July 21, 2022
Annemarie G. Hirsch, PhD1,2; Cara M. Nordberg, MPH1; Karen Bandeen-Roche3; Jonathan Pollak, MPP2; Melissa N. Poulsen, PhD1; Katherine A. Moon, PhD2; Brian S. Schwartz, MD1,2 (View author affiliations)
Suggested citation for this article: Hirsch AG, Nordberg CM, Bandeen-Roche K, Pollak J, Poulsen MN, Moon KA, et al. Urban–Rural Differences in Health Care Utilization and COVID-19 Outcomes in Patients With Type 2 Diabetes. Prev Chronic Dis 2022;19:220015. DOI: http://dx.doi.org/10.5888/pcd19.220015.
PEER REVIEWED
What is already known on this topic?
Studies have reported associations between type 2 diabetes and COVID-19 outcomes, but the extent to which community features modified these associations remains unexplored. Mitigation strategies during the pandemic resulted in type 2 diabetes care disruptions.
What is added by this report?
Community features did not modify the associations between type 2 diabetes and severe COVID-19 outcomes among individuals hospitalized for COVID-19. Community features did modify the disruption to type 2 diabetes care during the COVID-19 pandemic.
What are the implications for public health practice?
Community features modified the trajectories of disruptions in health care utilization during the COVID-19 pandemic and could be used to identify individuals at risk of gaps in type 2 diabetes care.
Abstract
Introduction
Two studies in Pennsylvania aimed to determine whether community type and community socioeconomic deprivation (CSD) 1) modified associations between type 2 diabetes (hereinafter, diabetes) and COVID-19 hospitalization outcomes, and 2) influenced health care utilization among individuals with diabetes during the COVID-19 pandemic.
Methods
The hospitalization study evaluated a retrospective cohort of patients hospitalized with COVID-19 through 2020 for COVID-19 outcomes: death, intensive care unit (ICU) admission, mechanical ventilation, elevated D-dimer, and elevated troponin level. We used adjusted logistic regression models, adding interaction terms to evaluate effect modification by community type (township, borough, or city census tract) and CSD. The utilization study included patients with diabetes and a clinical encounter between 2017 and 2020. Autoregressive integrated moving average time-series models evaluated changes in weekly rates of emergency department and outpatient visits, hemoglobin A1c (HbA1c) laboratory tests, and antihyperglycemic medication orders from 2018 to 2020.
Results
In the hospitalization study, of 2,751 patients hospitalized for COVID-19, 1,020 had diabetes, which was associated with ICU admission and elevated troponin. Associations did not differ by community type or CSD. In the utilization study, among 93,401 patients with diabetes, utilization measures decreased in March 2020. Utilization increased in July, and then began to stabilize or decline through the end of 2020. Changes in HbA1c tests and medication order trends during the pandemic differed by community type and CSD.
Conclusion
Diabetes was associated with selected outcomes among individuals hospitalized for COVID-19, but these did not differ by community features. Utilization trajectories among individuals with diabetes during the pandemic were influenced by community type and CSD and could be used to identify individuals at risk of gaps in diabetes care.
Introduction
Despite early concerns of elevated risk of COVID-19 infection in urban communities (1), studies of infection rates adjusted for socioeconomic factors have shown either no differences or reduced risks between urban and rural areas (2). Urban communities have also been found to have lower risk of severe COVID-19 outcomes (3), attributed to multiple factors, including better access to health care, healthy food, and walkable environments that reduce the risk of severe COVID-19 outcomes (3,4). These same mechanisms may also mitigate risk of severe COVID-19 associated with type 2 diabetes (hereinafter, diabetes).
Reports of associations between diabetes and COVID-19 outcomes have been mixed. Some studies have reported associations with severe COVID-19 (eg, intensive care unit [ICU] admission) and postacute COVID-19 sequelae (5–7), while others have not (8,9). Conversely, most, but not all, studies have reported no association between diabetes and COVID-19 mortality (5,6,9–11). These differences may be due to variation in study design (12) or in study settings from diverse locations around the world. The extent to which community features modify associations between diabetes and COVID-19 outcomes remains unexplored.
The impact of COVID-19 on individuals with diabetes goes beyond COVID-19 infection. Mitigation strategies during the pandemic (eg, suspension of nonurgent care, stay-at-home orders) resulted in diabetes care disruptions (13,14). Health systems serving urban communities have been better able to adapt to COVID-19 mitigation strategies through telehealth technology (15). Thus, gaps in care could be exacerbated in rural communities, which are more likely to have limited access to broadband internet service and greater distances to clinical care settings. The objectives of this study, conducted in geographically diverse communities across 37 Pennsylvania counties, were to determine whether community type and community socioeconomic deprivation (CSD) 1) modified associations between diabetes and COVID-19 hospitalization outcomes, and 2) influenced health care utilization among individuals with diabetes during the COVID-19 pandemic.
Methods
Study population and design
We conducted 2 analyses by using electronic health records (EHRs) from Geisinger, a health system serving central and northeastern Pennsylvania. The goal of the first study (hereinafter, the hospitalization study) was to evaluate associations of diabetes and severe COVID-19 outcomes. We conducted a retrospective cohort study of all patients hospitalized with COVID-19 through December 31, 2020, in 10 Geisinger hospitals. The goal of the second study (hereinafter, the utilization study) was to measure the impact of the COVID-19 pandemic on diabetes care. We included all Geisinger patients with diabetes residing in a 37-county region who had at least 1 clinical encounter between 2017 and 2020 (Appendix Figure 1). We identified individuals with diabetes based on encounter diagnoses, diabetes-relevant medication orders, and laboratory test results, as described previously (16).
Outcomes
The hospitalization study included 5 COVID-19 outcomes: death during hospitalization (up to 120 days after admission) or after hospitalization (up to 220 days) (yes vs no); ICU admission (yes vs no); required mechanical ventilation (yes vs no); D-dimer, a biomarker for thromboembolism (17) (≥0.5 µg/mL vs lower); and troponin level, a biomarker for myocardial damage (18) (elevated vs lower; elevated defined as ≥22 ng/L in men and ≥14 ng/L in women). For the utilization study, we measured weekly rates per 1,000 patients of emergency department (ED) visits, outpatient encounters (including telehealth), hemoglobin A1c (HbA1c) tests, and antihyperglycemic medication orders from January 1, 2018, through December 31, 2020. The denominator for the rates included anyone who was alive at the beginning of the measurement year and met diabetes criteria by the end of the measurement year (2018, 2019, or 2020).
Geocoding and community measurement
We obtained patient addresses from the EHRs and geocoded them to the street level by using ArcGIS World Geocoding Service in ArcMap version 10.4 (Esri) and assigned each patient to an administrative community type based on the residential location. This previously described approach uses boundaries from Pennsylvania’s minor civil divisions and city census tracts to create 3 community types: townships (rural areas to low-density suburbs), boroughs (small towns), and city census tracts, representing a continuum of lower to higher population density (see Appendix) (16). We also classified residential addresses into the US Census Bureau’s categories of urbanized areas, urban clusters, and rural (19).
For each community type, we measured CSD based on 6 indicators from the American Community Survey (2015–2019): percentage of unemployed, with less than a high school education, below poverty level, on public assistance, not in the workforce, and of households without a car (19). A previously described factor analysis demonstrated an adequate model fit of these indicators to a single factor and supported the use of an equally weighted scale based on the sum of the z transformed values of these indicators (20). We have previously reported an association between this CSD measure and diabetes onset (16). We quartiled the index, with the highest quartile representing the most socioeconomically deprived communities.
Statistical analysis — hospitalization study
The analysis goals were to determine the association between diabetes and each of the 5 COVID-19 outcomes and evaluate whether administrative community type and CSD modified these associations. We first evaluated bivariate associations between individual-level characteristics, administrative community type, and CSD and each of the COVID-19 outcomes. Next, we used logistic regression models with a random intercept for community to estimate the odds ratios (ORs) and 95% CIs for the COVID-19 outcomes.
For each outcome, we evaluated a series of 4 models adjusted sequentially to evaluate potential confounders. We then evaluated effect modification of the association between diabetes and COVID-19 outcomes by CSD and community type by adding cross-products between diabetes and administrative community type or CSD to the models. Global test P values were calculated to compare each model with all cross-products to a model with none. The series of models and their covariates are presented below.
Model 1 included age (years; centered linear, quadratic, and cubic terms to allow for nonlinearity), sex (female vs male), race (Black, all other races [Asian, Native Hawaiian or other Pacific Islander, American Indian or Alaska Native] vs White), ethnicity (Hispanic vs non-Hispanic), Medical Assistance, also known as Medicaid in Pennsylvania, as a surrogate for family socioeconomic status (ever vs never) (21), and time period in which hospitalization occurred (early: March to May; middle: June to September; late: October to December 2020). We collapsed races other than White and Black into a single category, all other races, because of small sample sizes. We included time period, based on our hypothesis that COVID-19 outcomes may have improved as the health system learned more about how to manage the disease. In model 2 we added the following comorbid diseases and community features one at a time to both evaluate their associations with COVID-19 outcomes and to determine whether they confounded diabetes associations: chronic kidney disease (vs none); chronic lung disease (vs none); resides in an institutional setting (eg, nursing home) (vs not); administrative community type (borough or city census tract vs township); and CSD (quartiles 2, 3, or 4 vs 1). In model 3 we added both chronic kidney disease and chronic lung disease to model 1 and in model 4 we added institutional setting (nursing homes) to model 3. We conducted sensitivity analyses examining death after discharge and repeated models replacing administrative community type with the urbanicity measure (urbanized areas or urban clusters vs rural).
Statistical analysis — utilization study
The goal was to determine whether administrative community type or CSD modified the impact of the COVID-19 pandemic on diabetes care. We conducted an interrupted time series analysis for each of the 4 utilization outcomes: weekly rates per 1,000 patients of HbA1c tests, antihyperglycemic medications orders, ED visits, and outpatient (including telehealth) visits. An interrupted time series design measures data at multiple time points before and after the introduction of an intervention, in this case the start of the COVID-19 pandemic in Pennsylvania, to examine the effect of the intervention (22).
We first explored multiple iterations of generalized linear models. After observing high dispersion by using Poisson regression models, we used negative binomial models. We conducted diagnostic checks, including serial residual plots and correlograms, which showed nonnegligible serial autocorrelation. We then added harmonic terms to account for seasonal trends in utilization, but diagnostic checks still showed nonnegligible serial autocorrelation. Thus, we used autoregressive integrated moving average (ARIMA) time-series models of utilization rates to account for the autocorrelation (22).
The study period was January 1, 2018, through December 31, 2020, and the intervention period for the models was from March 16, 2020, through the end of 2020. On March 16, 2020, Geisinger implemented restrictions to elective and nonurgent procedures and Pennsylvania implemented statewide mitigation policies, including an initial stay-at-home order. We added linear splines at time points during the intervention period that we predicted could trigger a change in health care utilization: March 16, 2020; May 4, 2020, when Geisinger reinstated elective and nonurgent procedures; July 13, 2020, which marked an increase in state and national COVID-19 infection rates; and November 30, 2020, when Geisinger reinstated elective procedure restrictions, as ordered by the Pennsylvania Department of Health.
We fit ARIMA models that did and did not account for seasonal utilization trends and models with and without the splines after the start of the intervention period. Based on Bayesian Information Criterion, model fit was better for nonseasonal ARIMA models when the intervention period splines were included; thus, our final models were nonseasonal ARIMA models with 4 linear splines. Data preparation was done by using Stata version 16 (StataCorp LLC). Analyses were performed in R version 4.0.3 (R Core Team).
For each utilization outcome, we evaluated effect modification separately by administrative community type and CSD. Models included a main effect term for each level of the community variable and interaction terms with the following variables: an indicator for the intervention period, study week, and spline terms. ARIMA models allow for 3 parameters: “p,” the number of autoregressive lags incorporated; “d,” the number of past values subtracted (“differenced”) from the current value; and “m,” the number of lags over which errors from prior observations are incorporated in the current error. The auto.arima() function in R’s forecast package (version 8.15) was used to determine the best ARIMA(p,d,q) order for the main effect models for each of the 4 utilization outcomes; d equaled 0 in all cases because no differencing was applied. We then applied the same ARIMA order to the models that evaluated effect modification by administrative community type and CSD. In sensitivity analyses, we repeated the models for each outcome, stratified by the urbanicity measure (urbanized areas or urban clusters vs rural).
Results
Hospitalization study
In 2020, 2,751 patients were hospitalized for COVID-19 and 1,020 of these patients met criteria for diabetes before admission (Table 1). Among those hospitalized, 458 died in the hospital and 105 died after discharge. During hospitalization, 650 patients were admitted to the ICU and 342 required mechanical ventilation. Among 2,300 patients with a troponin measure, 1,346 had an elevated level. Among 2,134 patients who had a D-dimer measure, 1,879 had levels ≥0.5 µg/mL.
Diabetes was associated with higher odds of ICU admission and elevated troponin levels in all models, but was not associated with death, mechanical ventilation, or elevated D-dimer levels (Table 2). Chronic kidney disease, chronic lung disease, institutional residence, and age were associated with higher odds of elevated troponin levels and death (Table 2). Age and female sex were also associated with higher odds of elevated troponin levels, while Hispanic ethnicity and hospitalization later in 2020 were associated with lower odds. The only factor associated with mechanical ventilation was the hospitalization time period, with the middle (June–September) and late (October–December) months associated with lower odds of ventilation. No comorbid diseases studied were associated with elevated D-dimer. We found no consistent evidence of effect modification of the associations between diabetes and COVID-19 outcomes by administrative community type or CSD. Administrative community type was not associated with death after discharge and the urbanicity measure was not associated with any of the outcomes (not shown).
Utilization study
A total of 93,401 patients, with a mean age of 57 years, met the criteria for the utilization study (Table 3). Consistent with the demographics of the region that Geisinger serves, individuals were predominately White (93.6%) and the majority resided in townships (55.2%). We present findings based on visual inspection of trends for each outcome for ease of interpretation (Figure 1 and Figure 2). These trends were supported by model coefficients and tests of statistical significance, unless otherwise noted.
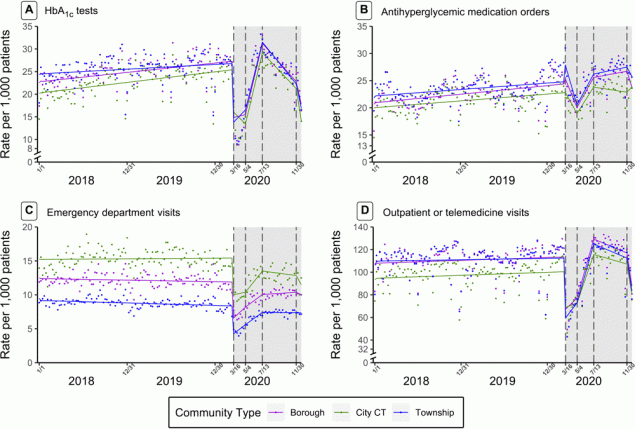
![]()
Figure 1.
Nonseasonal autoregressive integrated moving average time-series models with linear splines at 4 dates in 2020 (March 16, May 4, July 13, and November 30) of weekly utilization rates per 1,000 patients with type 2 diabetes of hemoglobin A1c(HbA1c) tests (A), antihyperglycemic medication orders (B), emergency department visits (C), and outpatient or telehealth visits (D). All plots were stratified by administrative community type. The gray shading indicates the intervention period: March 16, 2020–December 31, 2020. [A text version of this figure is available.]
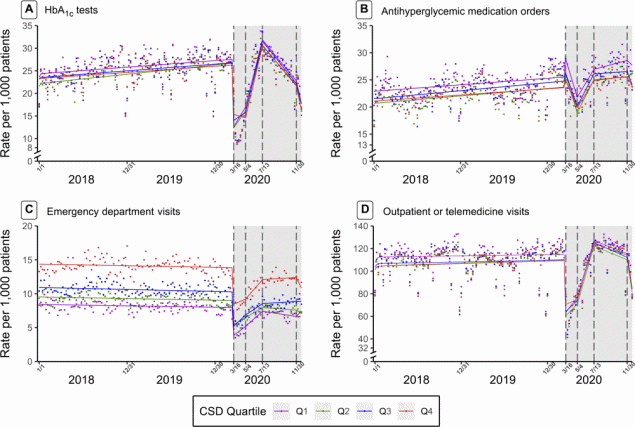
![]()
Figure 2.
Nonseasonal autoregressive integrated moving average time-series models with linear splines at 4 dates in 2020 (March 16, May 4, July 13, and November 30) of weekly utilization rates per 1,000 patients with type 2 diabetes of hemoglobin A1c(HbA1c) tests (A), antihyperglycemic medication orders (B), emergency department visits (C), and outpatient or telehealth visits (D). All plots were stratified by quartile of community socioeconomic deprivation (quartile 4 = most deprived). The gray shading indicates the intervention period: March 16, 2020–December 31, 2020. [A text version of this figure is available.]
Administrative community type
Prepandemic rates of ED and outpatient visits differed by community type and urbanicity, such that cities (vs townships) (Figure 1) and urbanized areas and urban clusters (vs rural) (Appendix Figure 2) had higher rates of ED encounters and lower rates of outpatient visits. This disparity persisted throughout the pandemic, as the trajectory of ED and outpatient visits after March 2020 did not differ by administrative community type.
Before the pandemic, weekly rates of HbA1c tests were lower in cities than in townships, but rates were increasing faster in cities than in townships. The drop in weekly HbA1c tests in March was greater in cities than in townships. Statistical output from the ARIMA models indicated that HbA1c tests declined at a faster rate in cities than in townships in March and recovered at a faster rate in cities than in townships in May. However, an inspection of Figure 1A reveals that this finding may be, in part, an artifact of the model, as the nadir in utilization appears to have occurred slightly later than March 16, and hence the modeled results (lines) do not fully reflect the observed data between March and May. All administrative community types experienced the same rate of decline in HbA1c utilization from July through the end of 2020.
Similarly, before the pandemic, weekly rates of antihyperglycemic medication orders were lower in cities than in townships and in urbanized areas than in rural areas. In the week before March 16, there was an increase in the rate of medication orders in townships and boroughs that was not observed in cities, as indicated in Figure 1B by the peaks in rates before the intervention period (shaded gray). In March there was a decline in medication order rates in all administrative community types, but that decline was slower in cities than in townships. Rates started to increase in May, again at a slower rate in cities than in townships. After July, rates continued to increase in townships and boroughs, but started to decrease again in cities.
Community socioeconomic deprivation
Prepandemic rates of ED and outpatient visits and antihyperglycemic medication orders differed by CSD, such that patients from more deprived communities (quartiles 2, 3, or 4 vs quartile 1) had higher rates of ED encounters, lower rates of outpatient or telemedicine visits, and lower rates of medication orders (Figure 2). These disparities persisted throughout the pandemic, as the trajectory of visits and medication orders did not differ by level of CSD. Statistical output from the ARIMA models indicated that the frequency of HbA1c tests declined at a faster rate in the most deprived community (vs least deprived) in March and recovered at a faster rate in the most deprived community (vs least deprived) in May. However, an inspection of Figure 2A reveals that this finding may be an artifact of the model, as the nadir in utilization appears to have occurred slightly later than March 16, and hence the modeled results (lines) do not fully reflect the observed data between March and May. The rate of decline in HbA1c tests in July through the end of 2020 was the same across levels of CSD.
Discussion
We evaluated how the COVID-19 pandemic influenced diabetes for both hospitalization outcomes and health care utilization, with a focus on whether these impacts differed by community features. We evaluated 5 hospitalization outcomes (death, ICU admission, ventilator use, elevated troponin levels, and elevated D-dimer levels) and 4 features of health care utilization (HbA1c tests, antihyperglycemic medication orders, ED visits, and outpatient and telehealth visits). We observed that persons with diabetes had higher odds of ICU admission and elevated troponin levels, but these associations were not modified by community features. In contrast, the impacts of the pandemic on the patterns of HbA1c tests and antihyperglycemic medication orders among individuals with diabetes showed important differences by community type, urbanicity, and CSD, providing evidence that clinical care for persons with diabetes during the pandemic was affected by residential setting.
Consistent with prior studies, we observed associations of diabetes with some, but not all, indicators of severe COVID-19 outcomes (5–11). Specifically, patients with diabetes had increased risk of ICU admission and elevated troponin levels. Elevated troponin levels have been associated with mortality among patients with COVID-19, but we did not find an association between diabetes and mortality (18). Elevated troponin among individuals with diabetes may be a marker of existing chronic heart damage rather than damage related to COVID-19 infection (23).
Early in the pandemic, reports from China implicated diabetes as a risk factor for severe COVID-19 outcomes (24). Thus, the elevated risk of ICU admission among persons with diabetes could be due to more severe disease in diabetes or because health systems were more proactively moving individuals with diabetes to ICU settings. Other conditions identified as high risk for poor outcomes early in the pandemic were not associated with ICU admission in our study, providing evidence that ICU admission may have been driven by a need for more intensive care among those with diabetes.
Community type, urbanicity, and CSD were not associated with COVID-19 hospitalization outcomes, nor did they modify associations between diabetes and these outcomes. Prior studies reported that the risk of severe COVID-19 outcomes was reduced in urban communities (2,3) and that the risk of severe COVID-19 was higher in more deprived communities (25) than in the general population. By studying patients hospitalized for COVID-19, our study sample was restricted to those experiencing more severe disease. The mechanisms through which community features influence risk of COVID-19 hospitalization and death in the general population (eg, better access to health care, walkable environments) may have less influence on hospitalization outcomes among those who already have serious disease (ie, are already hospitalized for COVID-19).
Consistent with prior studies (13,14), we observed decreased HbA1c tests, ED and outpatient visits, and antihyperglycemic medication orders at the start of the pandemic, when mitigation measures were implemented at the health system and state level. Utilization was rebounding by May 2020, when mitigation measures were lifted. By July 2020, many mitigation measures had been eased, with all Pennsylvania counties moving to the green phase (lowest risk of infection) on July 3, 2020 (26). Yet we observed that the trend of increasing utilization slowed in July for antihyperglycemic medication orders and ED visits, and for HbA1c tests and outpatient visits, rates started to decline. This could potentially be explained by increased national infection rates starting in mid-July, with cases doubling in 19 US states (27), news that may have influenced local care-seeking behaviors. Thus, individuals with diabetes experienced disruptions in care during multiple phases of the COVID-19 pandemic, including periods of strict mitigation policies and periods of elevated infection rates.
Changes in antihyperglycemic medication order rates differed by community type. The more gradual decline in medication orders in cities may be driven by the peak in medication order rates that occurred in townships and boroughs, but not cities, immediately before the pandemic. The peak in medication orders early in the pandemic has been previously attributed to “panic buying” because of concerns about possible medication shortages (28). Individuals residing in townships and boroughs may have more proactively prepared for a potential disruption in medication supplies, obtaining medications in early March 2020.
ED utilization differences persisted by community type and urbanicity. In contrast with a national report of higher ED utilization in rural, versus urban, communities (29), we found higher ED visits among patients in city census tracts (vs townships) and urbanized areas (vs rural). In prior work in our study region (16), associations between urbanicity and diabetes onset have also differed from national trends, potentially reflecting geographic differences that indicate a need for more localized research on the impact that community features have on health.
This research had numerous strengths. First, our measure of CSD used a spatial scale that is behaviorally relevant, rather than suboptimal scales based on census tract or county boundaries (20). Second, by studying a single health system serving a geographically diverse region, our findings were less vulnerable to confounding by health system factors (eg, treatment protocols) that could differ by community features.
This study had some limitations. First, the study population was predominately White individuals. Findings may not be generalizable to populations with different sociodemographic characteristics, though findings are likely generalizable to the region studied. Second, patients missing D-dimer or troponin measures were excluded from the analysis of these outcome measures. Third, the administrative community type is challenging to replicate in states without similar municipality boundaries.
In a large, geographically diverse region of Pennsylvania, diabetes was associated with more severe COVID-19 outcomes among individuals hospitalized for COVID-19. These outcomes did not differ by community features, and the higher odds of ICU admission and elevated troponin levels among persons with diabetes was not influenced by community features. Diabetes care was disrupted during periods when COVID-19 mitigation policies were in place and when infection rates were elevated nationally. Community features modified the trajectories of health care utilization during these phases of the pandemic and could be used to identify individuals at risk of gaps in diabetes care. It is important to evaluate the impact of these utilization differences on diabetes outcomes.
Acknowledgments
This publication was made possible by cooperative agreement no. DP006296, funded by the Centers for Disease Control and Prevention, Division of Diabetes Translation. The study sponsor did not have a role in collection, analysis, interpretation of data, writing the report, or in the decision to submit the article for publication.
The authors have no conflicts of interest to report.
Author Information
Corresponding author: Annemarie G. Hirsch, PhD, MPH, Department of Population Health Sciences, Geisinger, 100 N Academy Ave, Danville, PA 17822. Telephone: 267-626-8110. Email: aghirsch@geisinger.edu.
Author Affiliations: 1Department of Population Health Sciences, Geisinger, Danville, Pennsylvania. 2Department of Environmental Health and Engineering, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland. 3Department of Biostatistics, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland.
References
- Frumkin H. COVID-19, the built environment, and health. Environ Health Perspect 2021;129(7):75001. CrossRef PubMed
- Kim B, Rundle AG, Goodwin ATS, Morrison CN, Branas CC, El-Sadr W, et al. COVID-19 testing, case, and death rates and spatial socio-demographics in New York City: an ecological analysis as of June 2020. Health Place 2021;68:102539. CrossRef PubMed
- Wali B, Frank LD. Neighborhood-level COVID-19 hospitalizations and mortality relationships with built environment, active and sedentary travel. Health Place 2021;71:102659. CrossRef PubMed
- Hamidi S, Ewing R, Sabouri S. Longitudinal analyses of the relationship between development density and the COVID-19 morbidity and mortality rates: early evidence from 1,165 metropolitan counties in the United States. Health Place 2020;64:102378. CrossRef PubMed
- Raghavan A, Nanditha A, Satheesh K, Susairaj P, Vinitha R, Chandrasekaran S, et al. Profile and prognosis of patients hospitalized for COVID-19 virus infection with and without diabetes — an observational study from South India. Diabetes Metab Syndr 2021;15(4):102143. CrossRef PubMed
- Longmore DK, Miller JE, Bekkering S, Saner C, Mifsud E, Zhu Y, et al. ; International BMI-COVID Consortium. Diabetes and overweight/obesity are independent, nonadditive risk factors for in-hospital severity of COVID-19: an international, multicenter retrospective meta-analysis. Diabetes Care 2021;44(6):1281–90. CrossRef PubMed
- Su Y, Yuan D, Chen DG, Ng RH, Wang K, Choi J, et al. ; ISB-Swedish COVID-19 Biobanking Unit. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell 2022;185(5):881–895.e20. CrossRef PubMed
- Suleyman G, Fadel RA, Malette KM, Hammond C, Abdulla H, Entz A, et al. Clinical characteristics and morbidity associated with coronavirus disease 2019 in a series of patients in metropolitan Detroit. JAMA Netw Open 2020;3(6):e2012270. CrossRef PubMed
- Feldman EL, Savelieff MG, Hayek SS, Pennathur S, Kretzler M, Pop-Busui R. COVID-19 and diabetes: a collision and collusion of two diseases. Diabetes 2020;69(12):2549–65. CrossRef PubMed
- Petrilli CM, Jones SA, Yang J, Rajagopalan H, O’Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ 2020;369:m1966. CrossRef PubMed
- McGurnaghan SJ, Weir A, Bishop J, Kennedy S, Blackbourn LAK, McAllister DA, et al. ; Public Health Scotland COVID-19 Health Protection Study Group; Scottish Diabetes Research Network Epidemiology Group. Risks of and risk factors for COVID-19 disease in people with diabetes: a cohort study of the total population of Scotland. Lancet Diabetes Endocrinol 2021;9(2):82–93. CrossRef PubMed
- Selvin E, Juraschek SP. Diabetes epidemiology in the COVID-19 pandemic. Diabetes Care 2020;43(8):1690–4. CrossRef PubMed
- Fragala MS, Kaufman HW, Meigs JB, Niles JK, McPhaul MJ. Consequences of the COVID-19 pandemic: reduced hemoglobin A1c diabetes monitoring. Popul Health Manag 2021;24(1):8-9. PubMed
- Ikesu R, Miyawaki A, Sugiyama T, Nakamura M, Ninomiya H, Kobayashi Y. Trends in diabetes care during the COVID-19 outbreak in Japan: an observational study. J Gen Intern Med 2021;36(5):1460–2. CrossRef PubMed
- Patel SY, Rose S, Barnett ML, Huskamp HA, Uscher-Pines L, Mehrotra A. Community factors associated with telemedicine use during the COVID-19 pandemic. JAMA Netw Open 2021;4(5):e2110330. CrossRef PubMed
- Schwartz BS, Pollak JS, Poulsen MN, Bandeen-Roche K, Moon K, DeWalle J, et al. Association of community types and features in a case–control analysis of new onset type 2 diabetes across diverse geography in Pennsylvania. BMJ Open 2021;11(1):e043528. CrossRef PubMed
- Mishra Y, Pathak BK, Mohakuda SS, Tilak TVSVGK, Sen S, P H, et al. Relation of D-dimer levels of COVID-19 patients with diabetes mellitus. Diabetes Metab Syndr 2020;14(6):1927–30. CrossRef PubMed
- Wibowo A, Pranata R, Akbar MR, Purnomowati A, Martha JW. Prognostic performance of troponin in COVID-19: a diagnostic meta-analysis and meta-regression. Int J Infect Dis 2021;105:312–8. CrossRef PubMed
- United States Census Bureau. 2010 Census urban and rural classification and urban area criteria. 2010. https://www.census.gov/programs-surveys/geography/guidance/geo-areas/urban-rural/2010-urban-rural.html. Accessed December 5, 2019.
- Liu AY, Curriero FC, Glass TA, Stewart WF, Schwartz BS. Associations of the burden of coal abandoned mine lands: three dimensions of community context in Pennsylvania. International Scholarly Research Notices 2012;2012:1–11. CrossRef
- Casey JA, Pollak J, Glymour MM, Mayeda ER, Hirsch AG, Schwartz BS. Measures of SES for electronic health record–based research. Am J Prev Med 2018;54(3):430–9. CrossRef PubMed
- Turner SL, Karahalios A, Forbes AB, Taljaard M, Grimshaw JM, Cheng AC, et al. Design characteristics and statistical methods used in interrupted time series studies evaluating public health interventions: protocol for a review. BMJ Open 2019;9(1):e024096. CrossRef PubMed
- Ceriello A, Standl E, Catrinoiu D, Itzhak B, Lalic NM, Rahelic D, et al. ; “Diabetes and Cardiovascular Disease (D&CVD)” Study Group of the European Association for the Study of Diabetes (EASD). Issues for the management of people with diabetes and COVID-19 in ICU. Cardiovasc Diabetol 2020;19(1):114. CrossRef PubMed
- Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. ; China Medical Treatment Expert Group for Covid-19. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020;382(18):1708–20. CrossRef PubMed
- Hawkins RB, Charles EJ, Mehaffey JH. Socio-economic status and COVID-19–related cases and fatalities. Public Health 2020;189:129–34. CrossRef PubMed
- Governor Tom Wolf. Reopening phase orders updated to include last county moving to green on July 3. https://www.governor.pa.gov/newsroom/reopening-phase-orders-updated-to-include-last-county-moving-to-green-on-july-3/. Accessed December 23, 2021.
- Chan C, Shumaker L. U.S. records over 25,000 coronavirus deaths in July. July 31, 2020. https://www.reuters.com/article/us-health-coronavirus-usa-july/u-s-records-over-25000-coronavirus-deaths-in-july-idUSKCN24W1G1. Accessed December 21, 2021.
- Engstrom T, Baliunas DO, Sly BP, Russell AW, Donovan PJ, Krausse HK, et al. Toilet paper, minced meat and diabetes medicines: Australian panic buying induced by COVID-19. Int J Environ Res Public Health 2021;18(13):6954. CrossRef PubMed
- Greenwood-Ericksen MB, Kocher K. Trends in emergency department use by rural and urban populations in the United States. JAMA Netw Open 2019;2(4):e191919. CrossRef PubMed
Tables
| Variable | Type 2 diabetes, n = 1,020 (37.1%) | No type 2 diabetes, n = 1,731 (62.9%) |
|---|---|---|
| Sociodemographics and habits | ||
| Age at first hospitalization, mean (SD), y | 69.9 (13.2) | 65.6 (19.4) |
| Sex, female | 467 (45.8) | 841 (48.6) |
| Race, Black | 40 (3.9) | 89 (5.1) |
| Hispanic | 70 (6.9) | 131 (7.6) |
| Medical Assistance, >0% of time | 139 (13.6) | 187 (10.8) |
| Institutionalized housing | 144 (14.1) | 168 (9.7) |
| Tobacco use, ever | 555 (54.4) | 761 (44.0) |
| Selected outcomes | ||
| Died in hospital | 198 (19.4) | 260 (15.0) |
| Died after hospital | 34 (3.3) | 71 (4.1) |
| Deceased total | 232 (22.8) | 331 (19.1) |
| Admitted to intensive care unit | 277 (27.2) | 373 (21.6) |
| Required mechanical ventilation | 137 (13.4) | 205 (11.8) |
| Hospital readmissions | 65 (6.4) | 87 (5.0) |
| Selected comorbid conditions | ||
| Chronic kidney disease | 417 (40.9) | 348 (20.1) |
| Chronic lung disease | 217 (21.3) | 229 (13.2) |
| Selected laboratory measurements | ||
| Troponin, plasma, any hospitalizationb | ||
| Missing | 127 (12.5) | 324 (18.7) |
| Elevated level | 606 (59.4) | 740 (42.8) |
| Maximum value, mean (SD) | 92.6 (441.4) | 62.2 (325.8) |
| D-dimer, plasma, any hospitalization | ||
| Missing | 197 (19.3) | 420 (24.3) |
| ≥0.5 µg/mL | 723 (70.9) | 1,156 (66.8) |
| Maximum value, mean (SD) | 3.28 (4.77) | 3.02 (4.49) |
| Community measures | ||
| Residential location by administrative community type | ||
| Township | 490 (48.0) | 891 (51.5) |
| Borough | 302 (29.6) | 499 (28.8) |
| City census tract | 228 (22.4) | 341 (19.7) |
| CSD quartiles | ||
| 1st (least disadvantaged) | 166 (16.3) | 337 (19.5) |
| 2nd | 202 (19.8) | 374 (21.6) |
| 3rd | 297 (29.1) | 500 (28.9) |
| 4th (most disadvantaged) | 355 (34.8) | 520 (30.0) |
Abbreviation: CSD, community socioeconomic disadvantage.
a Data are shown as n (%) unless otherwise indicated.
b Cut-point for elevated troponin level differs by sex: men, 22 ng/L; women, 14 ng/L.
| Variable | Death, total vs not deceasedc (n = 2,751) | ICU, any vs not (n = 2,751) | Ventilator, any vs none (n = 2,751) | Troponin ≥22 ng/L vs lower (n = 1,346) | D-dimer ≥0.5 µg/mL vs lower (n = 1,879) |
|---|---|---|---|---|---|
| Model 1: base model | |||||
| Sex, female vs male | 0.77 (0.63–0.95) | 0.76 (0.63–0.93) | 0.85 (0.66–1.10) | 1.31 (1.12–1.53) | 0.83 (0.71–0.98) |
| Race and ethnicity | |||||
| Non-White vs White | 0.56 (0.35–0.89) | 0.62 (0.44–0.87) | 0.83 (0.55–1.27) | 1.02 (0.70–1.49) | 0.93 (0.69–1.26) |
| Hispanic vs non-Hispanic | 0.54 (0.28–1.05) | 0.79 (0.54–1.14) | 1.03 (0.66–1.61) | 0.54 (0.35–0.82) | 1.06 (0.76–1.47) |
| Medical Assistance, >0% time vs no time | 1.39 (0.95–2.04) | 1.02 (0.76–1.37) | 0.90 (0.60–1.35) | 1.25 (0.90–1.73) | 1.28 (0.95–1.71) |
| Time period for hospitalization (2020)d | |||||
| Middle months vs early months | 0.51 (0.35–0.75) | 1.13 (0.82–1.57) | 0.35 (0.22–0.55) | 0.70 (0.51–0.96) | 0.53 (0.39–0.72) |
| Late months vs early months | 0.49 (0.40–0.62) | 0.50 (0.40–0.62) | 0.43 (0.33–0.56) | 0.76 (0.61–0.96) | 0.80 (0.63–1.03) |
| Type 2 diabetes | 1.15 (0.94–1.41) | 1.21 (1.01–1.45) | 1.06 (0.85–1.32) | 1.87 (1.56–2.25) | 0.96 (0.80–1.15) |
| Models 2a–e: adjusted associations of new variables added one at a time to base model | |||||
| 2a. Chronic kidney disease vs none | 1.46 (1.18–1.80) | 1.00 (0.80–1.24) | 1.04 (0.84–1.46) | 2.60 (2.10–3.23) | 0.95 (0.78–1.15) |
| 2b. Chronic lung disease vs none | 1.35 (1.06–1.73) | 1.10 (0.87–1.38) | 1.07 (0.79–1.43) | 1.64 (1.29–2.09) | 0.99 (0.78–1.25) |
| 2c. Institutionalized vs not | 1.63 (1.23–2.16) | 1.04 (0.77–1.42) | 1.14 (0.77–1.69) | 2.23 (1.67–2.97) | 0.92 (0.70–1.22) |
| 2d. CSD, 1st vs 4th quartile (least versus most deprived) | 0.74 (0.54–1.01) | 0.85 (0.64–1.14) | 0.96 (0.66–1.39) | 0.79 (0.61–1.02) | 0.97 (0.73–1.29) |
| 2e. Administrative community type | |||||
| Borough vs township | 0.92 (0.73–1.15) | 1.03 (0.81–1.31) | 1.12 (0.85–1.49) | 1.18 (0.94–1.48) | 0.80 (0.64–1.01) |
| City census tract vs township | 1.05 (0.78–1.40) | 0.81 (0.63–1.05) | 0.88 (0.65–1.20) | 0.96 (0.76–1.21) | 0.85 (0.66–1.08) |
| Models 3 and 4: fully adjusted diabetes associations (model 3: CKD and CLD added to base model; model 4: institutionalized added to Model 3) | |||||
| 3. Type 2 diabetes vs none | 1.05 (0.85–1.29) | 1.21 (1.003–1.45) | 1.04 (0.83–1.30) | 1.57 (1.30–1.90) | 0.97 (0.78–1.16) |
| 4. Type 2 diabetes vs none | 1.02 (0.83–1.25) | 1.21 (1.0004–1.45) | 1.03 (0.82–1.30) | 1.54 (1.28–1.86) | 0.97 (0.80–1.17) |
Abbreviations: CKD, chronic kidney disease; CLD, chronic lung disease; CSD, community socioeconomic deprivation; ICU, intensive care unit.
a Adjusted for age: linear, quadratic, and cubic.
b All data are shown as odds ratio (95% CI).
c Death total = death during and after hospitalization.
d Time period: early (March to May); middle (June to September); late (October to December), all in 2020.
| Variable | Full cohort (N = 93,401) | 2018 cohort (n = 81,393) | 2019 cohort (n = 85,812) | 2020 cohort (n = 87,612) |
|---|---|---|---|---|
| Sociodemographics | ||||
| Age, mean (SD), y | 57.0 (14.4) | 56.8 (14.2) | 56.6 (14.2) | 56.4 (14.2) |
| Sex, female | 44,897 (48.1) | 39,538 (48.6) | 41,507 (48.4) | 42,269 (48.2) |
| Race, non-White | 5,993 (6.4) | 4,592 (5.6) | 5,364 (6.2) | 5,822 (6.6) |
| Hispanic | 3,962 (4.2) | 3,159 (3.9) | 3,606 (4.2) | 3,854 (4.4) |
| Medical assistance, >0% of time | 12,898 (13.8) | 10,857 (13.3) | 11,786 (13.7) | 12,390 (14.1) |
| Community measures | ||||
| Residential location by administrative community type | ||||
| Township | 51,564 (55.2) | 45,181 (55.5) | 47,461 (55.3) | 48,337 (55.2) |
| Borough | 27,302 (29.2) | 23,800 (29.2) | 25,034 (29.2) | 25,587 (29.2) |
| City census tract | 14,535 (15.6) | 12,412 (15.2) | 13,317 (15.5) | 13,688 (15.6) |
| Community socioeconomic deprivation quartiles | ||||
| 1st (least deprived) | 19,634 (21.0) | 17,204 (21.1) | 18,132 (21.1) | 18,478 (21.1) |
| 2nd | 21,674 (23.2) | 18,941 (23.3) | 19,936 (23.2) | 20,383 (23.3) |
| 3rd | 25,954 (27.8) | 22,504 (27.6) | 23,757 (27.7) | 24,262 (27.7) |
| 4th (most deprived) | 26,139 (28.0) | 22,744 (27.9) | 23,987 (28.0) | 24,489 (28.0) |
a Individuals were included in measurement year if they were alive at the start of the year and met type 2 diabetes criteria by the end of the year.
b Data are shown as n (%) unless otherwise indicated.
Appendix
Appendix Figure 1 and Appendix Figure 2 are available at https://www.geisinger.edu/research/departments-and-centers/environmental-health-institute/diabetes.
| US Census Bureau categories | Township | Borough | City census tract |
|---|---|---|---|
| Median population density, per square mile | 62 | 1,713 | 5,537 |
| Rural, n | 32,153 | 2,979 | 30 |
| Column % | 62.4 | 10.9 | 0.21 |
| Row % | 91.4 | 8.4 | 0.09 |
| Urban cluster, n | 8,572 | 9,945 | 2,948 |
| Column % | 16.6 | 36.4 | 20.3 |
| Row % | 39.9 | 46.3 | 13.7 |
| Urbanized area, n | 10,839 | 14,378 | 11,557 |
| Column % | 21.0 | 52.7 | 79.5 |
| Row % | 29.5 | 39.0 | 31.4 |
a The row percent reflects the percentage of each of the 3 rows: rural, urban, urbanized cluster. Among people living in rural areas, for example, 91.4% live in townships, 8.4% live in boroughs, and 0.09% live in city census tracts. The column percent reflects the percentage of each of the 3 columns: townships, boroughs, city census tracts. In the townships column, for example, 62.4% of townships are rural, 16.6% of townships are in urban clusters, and 21.0% of townships are in urbanized areas.
Error processing SSI fileThe opinions expressed by authors contributing to this journal do not necessarily reflect the opinions of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors’ affiliated institutions.