Dirofilariasis
[Dirofilaria immitis] [D. repens] [D. tenuis] [D. ursi]
Causal Agents
The genus Dirofilaria includes vector-borne filarial nematodes, which are usually associated with carnivore hosts. Some Dirofilaria spp. are zoonotic; the most commonly seen species in human patients are D. repens, D. tenuis, and D. immitis (the dog heartworm). Rare human infections with other species such as D. striata and D. ursi-like species (D. ursi or D. subdermata) have been reported. Recently, a D. repens-like agent infecting humans was identified in Hong Kong, which has been proposed as a new species D. hongkongensis. Human dirofilariasis is generally divided into pulmonary dirofilariasis (D. immitis) and subcutaneous dirofilariasis (D. repens, D. tenuis, and others).
During a blood meal, an infected mosquito introduces third-stage filarial larvae of Dirofilaria immitis into the skin of the definitive host, where they penetrate into the bite wound  . In the definitive host, the L3 larvae undergo two more molts into L4 and adults. Adults reside in pulmonary arteries, and are occasionally found in the right ventricle of the heart
. In the definitive host, the L3 larvae undergo two more molts into L4 and adults. Adults reside in pulmonary arteries, and are occasionally found in the right ventricle of the heart  . Adult females are usually 230-310 mm long by 350 µm wide; males are usually 120-190 mm long by 300 µm wide. Adults can live for 5 – 10 years. In the heart, the female worms produce microfilariae which circulate in peripheral blood
. Adult females are usually 230-310 mm long by 350 µm wide; males are usually 120-190 mm long by 300 µm wide. Adults can live for 5 – 10 years. In the heart, the female worms produce microfilariae which circulate in peripheral blood  . A mosquito ingests these microfilariae during a blood meal
. A mosquito ingests these microfilariae during a blood meal  . After ingestion, the microfilariae migrate from the mosquito’s midgut through the hemocoel to the Malpighian tubules in the abdomen
. After ingestion, the microfilariae migrate from the mosquito’s midgut through the hemocoel to the Malpighian tubules in the abdomen  . There the microfilariae develop into first-stage larvae
. There the microfilariae develop into first-stage larvae  and subsequently into third-stage infective larvae
and subsequently into third-stage infective larvae  . The third-stage infective larvae migrate to the mosquito’s proboscis
. The third-stage infective larvae migrate to the mosquito’s proboscis  and can infect another definitive host when the mosquito takes a blood meal
and can infect another definitive host when the mosquito takes a blood meal  . In humans
. In humans  , D. immitis larvae tend to follow the same migratory pathway as in the canine host, ending up in the lungs, where they often lodge in small-caliber vessels, causing infarcts and typical “coin lesions” visible on radiographs. Occasionally, D. immitis may cause subcutaneous or other ectopic infections.
, D. immitis larvae tend to follow the same migratory pathway as in the canine host, ending up in the lungs, where they often lodge in small-caliber vessels, causing infarcts and typical “coin lesions” visible on radiographs. Occasionally, D. immitis may cause subcutaneous or other ectopic infections.
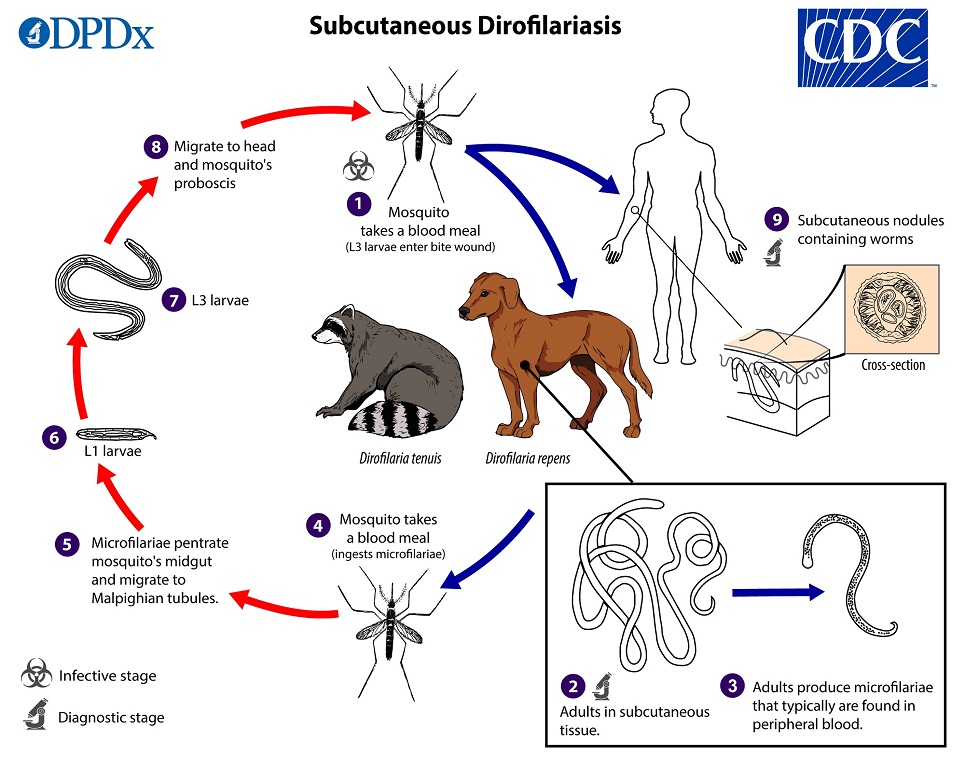
Various Dirofilaria species of the subgenus Nochtiella can cause subcutaneous infections. During a blood meal, an infected mosquito introduces third-stage filarial larvae of Dirofilaria repens or D. tenuis onto the skin of the definitive host (canids and sometimes felids for D. repens, raccoons for D. tenuis), where they penetrate into the bite wound  . In the definitive host, the L3 larvae undergo two more molts into L4 and adults, the latter of which reside in subcutaneous tissues
. In the definitive host, the L3 larvae undergo two more molts into L4 and adults, the latter of which reside in subcutaneous tissues  . D. repens adult females are usually 100—170 mm long by 460—650 µm wide; males are usually 50—70 mm long by 370—450 µm wide. D. tenuis is slightly smaller; adult females are usually 80-130 mm long by 260—360 µm wide; males are usually 40—50 mm long by 190—260 µm wide. Females produce microfilariae that are released into the peripheral blood
. D. repens adult females are usually 100—170 mm long by 460—650 µm wide; males are usually 50—70 mm long by 370—450 µm wide. D. tenuis is slightly smaller; adult females are usually 80-130 mm long by 260—360 µm wide; males are usually 40—50 mm long by 190—260 µm wide. Females produce microfilariae that are released into the peripheral blood  . A mosquito ingests the microfilariae during a blood meal
. A mosquito ingests the microfilariae during a blood meal  . After ingestion, the microfilariae migrate from the mosquito’s midgut through the hemocoel to the Malpighian tubules in the abdomen
. After ingestion, the microfilariae migrate from the mosquito’s midgut through the hemocoel to the Malpighian tubules in the abdomen  . There the microfilariae develop into first-stage larvae
. There the microfilariae develop into first-stage larvae  and subsequently into third-stage infective larvae
and subsequently into third-stage infective larvae  . The third-stage infective larvae migrate to the mosquito’s proboscis
. The third-stage infective larvae migrate to the mosquito’s proboscis  and can infect another definitive host when the mosquito takes a blood meal
and can infect another definitive host when the mosquito takes a blood meal  . In humans
. In humans  , D. repens usually manifests as either a wandering worm in the subcutaneous tissue or a granulomatous nodule, although there are reports of pulmonary dirofilariasis with this species. D. tenuis follows a similar presentation, but may also be found around the eye or on the conjunctiva. Because of this, the infection in humans was first known as Dirofilaria conjunctivae.
, D. repens usually manifests as either a wandering worm in the subcutaneous tissue or a granulomatous nodule, although there are reports of pulmonary dirofilariasis with this species. D. tenuis follows a similar presentation, but may also be found around the eye or on the conjunctiva. Because of this, the infection in humans was first known as Dirofilaria conjunctivae.
Subcutaneous infections with Dirofilaria striata, D. ursi, and possibly D. subdermata have been reported. Characteristics visible in clinical specimens have not been sufficient to definitively distinguish D. ursi and D. subdermata, so suspected infections are usually reported as “D. ursi-like”. These less common species presumably follow a similar life cycle, but with different hosts (see below).
Hosts
The most typical hosts for D. immitis are domestic dogs, coyotes, jackals, and wolves. Adult worms are occasionally found in others species such as domestic cats, bobcats, ferrets, and foxes. However, microfilaremia is usually low or absent in these aberrant hosts and thus they are not major reservoirs. Vectors include mosquitoes from several genera (Aedes, Culex, Anopheles, Mansonia).
D. repens definitive hosts include primarily wild and domestic canids, and occasionally felids. The only known natural definitive host for D. tenuis is the raccoon. Both are by various mosquito vectors from the genera Aedes, Culex, and Anopheles.
Among the rarer causes of subcutaneous dirofilariasis, D. ursi is a parasite of bears including American black bears (Ursus americanus), grizzly bears (U. arctos), and Asian black bears (U. thiabetanus), and is transmitted by black flies (Simulium spp.), rather than the mosquito vectors of most other Dirofilaria species. The North American porcupine (Erethizon dorsatum) is the definitive host for D. subdermata. D. striata has been described from some wild Nearctic felids (e.g. bobcats, pumas, ocelots).
Geographic Distribution
Dirofilaria immitis is cosmopolitan in dogs in North and South America, Australia, Japan and Europe, and is particularly prevalent in warmer areas where transmission is sustained for most of the year.
Dirofilaria repens is exclusive to the Old World, infecting dogs and cats across Europe, Asia, and Africa. In Europe, the Mediterranean region is considered highly D. repens-endemic. The European range of D. repens seems to be expanding, with transmission has been documented as far north as Finland and East into European Russia.
Among species occurring in the New World, the range of D. tenuis is not fully characterized, but it is prevalent in raccoons in Florida. Dirofilaria striata is a parasite of wild felids in North, Central, and South America; the only confirmed human infection (subcutaneous) occurred in North Carolina. Dirofilaria ursi is mostly a North American species, having been found in bears from Canada and the northern United States, but some reports exist from Japan.
Clinical Presentation
Dirofilaria immitis most often causes pulmonary disease in the human host, but can also uncommonly cause nodules in other tissues. Humans are suboptimal hosts and larvae that migrate to the heart usually die. Dead worms produce infarcts when they lodge in pulmonary vessels; these infarcts are usually referred to as “coin lesions” on chest radiography, which may be mistaken for malignancy. Following embolization, patients may present with vague systemic symptoms (e.g. malaise, fever, chills) and respiratory distress. There is often a mild eosinophilia.
Species of Dirofilaria in the subgenus Nochtiella usually (but not always) manifest as subcutaneous nodules. These nodules are often tender, and may be fixed or migratory. D. repens-associated lesions can occur in a variety of locations, the most typical being in exposed sites (e.g. scalp, arms, legs, eyelids, chest), but occasionally have been found in deeper tissue such as the breast, epididymis, spermatic cord, and subconjunctiva. Many reports of D. tenuis involve the facial region (e.g. ocular and periocular sites, oral mucosa, cheek) and breast nodules. Unlike D. immitis, some of these species may achieve a degree of development beyond the larval stage in the human host, as adult-stage D. repens and D. tenuis are often discovered in human infections. However, most do not develop to sexual maturity. There are very rare reports of patent D. repens infections in humans with detectable microfilariae in the blood.
Dirofilaria spp.
The genus Dirofilaria consists of many species that infect a wide range of hosts worldwide, including carnivores, rodents and primates. Humans are incidental hosts for several of these; the most frequent include the dog parasites D. immitis and D. repens, and the raccoon parasite D. tenuis. The worms usually die before completing their development in the human host, but tend to follow the same migratory pathway as in the natural definitive host; D. tenuis and D. repens most often remain in the subcutaneous tissue where the dying worms produce a localized granulomatous nodule, or, in the case of D. immitis, pulmonary infarcts and/or nodules often appearing as coin lesions on X-ray.












Laboratory Diagnosis
Dirofilaria immitis is usually diagnosed by the finding of the distinctive coin lesions on chest X-rays. The species that produce subcutaneous nodules are diagnosed by the finding of adult worms in biopsy specimens of these nodules. The morphologic features of the cuticle, musculature, and lateral chords are important criteria for identifying Dirofilaria. Worms removed from the eye are usually diagnosed by external features of the cuticle; worms may also be processed by routine histologic methods to study the internal structures. There are currently no serologic or molecular methods available in the United States for diagnosis of human dirofilariasis.
Laboratory Safety
Standard protocols for the processing and examination of histological specimens apply. No infectious stages of Dirofilaria spp. are encountered in a clinical diagnostic setting.
Suggested Reading
Winkler, S., Pollreisz, A., Georgopoulos, M., Bagò-Horvath, Z., Auer, H., To, K.K.W., Krücken, J., Poppert, S. and Walochnik, J., 2017. Candidatus Dirofilaria hongkongensis as causative agent of human ocular filariosis after travel to India. Emerging Infectious Diseases, 23 (8), p.1428.
Simón, F., Siles-Lucas, M., Morchón, R., González-Miguel, J., Mellado, I., Carretón, E. and Montoya-Alonso, J.A., 2012. Human and animal dirofilariasis: the emergence of a zoonotic mosaic. Clinical Microbiology Reviews, 25 (3), pp.507-544.
Orihel, T.C. and Eberhard, M.L., 1998. Zoonotic filariasis. Clinical Microbiology Reviews, 11 (2), pp.366-381.
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.