
Babesiosis
[Babesia divergens] [Babesia duncani] [Babesia microti] [Babesia MO-1]
Causal Agents
Babesiosis is caused by apicomplexan parasites of the genus, Babesia. While more than 100 species have been reported, only a few have been identified as causing human infections, including B. microti, B. divergens, B. duncani, and a currently un-named strain designated MO-1.
Life Cycle

The Babesia microti life cycle involves two hosts, which includes a rodent, primarily the white-footed mouse, Peromyscus leucopus, and a tick in the genus, Ixodes. During a blood meal, a Babesia-infected tick introduces sporozoites into the mouse host  . Sporozoites enter erythrocytes and undergo asexual reproduction (budding)
. Sporozoites enter erythrocytes and undergo asexual reproduction (budding)  . In the blood, some parasites differentiate into male and female gametes although these cannot be distinguished at the light microscope level
. In the blood, some parasites differentiate into male and female gametes although these cannot be distinguished at the light microscope level  . The definitive host is the tick. Once ingested by an appropriate tick
. The definitive host is the tick. Once ingested by an appropriate tick  , gametes unite and undergo a sporogonic cycle resulting in sporozoites
, gametes unite and undergo a sporogonic cycle resulting in sporozoites  . Transovarial transmission (also known as vertical, or hereditary, transmission) has been documented for “large” Babesia spp. but not for the “small” babesiae, such as B. microti
. Transovarial transmission (also known as vertical, or hereditary, transmission) has been documented for “large” Babesia spp. but not for the “small” babesiae, such as B. microti  .
.
Humans enter the cycle when bitten by infected ticks. During a blood meal, a Babesia-infected tick introduces sporozoites into the human host  . Sporozoites enter erythrocytes
. Sporozoites enter erythrocytes  and undergo asexual replication (budding)
and undergo asexual replication (budding)  . Multiplication of the blood stage parasites is responsible for the clinical manifestations of the disease. Humans are, for all practical purposes, dead-end hosts and there is probably little, if any, subsequent transmission that occurs from ticks feeding on infected persons. However, human to human transmission is well recognized to occur through blood transfusions
. Multiplication of the blood stage parasites is responsible for the clinical manifestations of the disease. Humans are, for all practical purposes, dead-end hosts and there is probably little, if any, subsequent transmission that occurs from ticks feeding on infected persons. However, human to human transmission is well recognized to occur through blood transfusions  .
.
Geographic Distribution
Worldwide, but little is known about the prevalence of Babesia in malaria-endemic countries, where misidentification as Plasmodium probably occurs. In Europe, most reported cases are due to B. divergens and occur in splenectomized patients. In the United States, B. microti is the agent most frequently identified (Northeast and Midwest), and can occur in nonsplenectomized individuals. Babesia duncani has been isolated in patients in Washington and California. MO-1 has been isolated from patients in Missouri.
Clinical Presentation
Most infections are probably asymptomatic, as indicated by serologic surveys. Manifestations of disease include fever, chills, sweating, myalgias, fatigue, hepatosplenomegaly, and hemolytic anemia. Symptoms typically occur after an incubation period of 1 to 4 weeks, and can last several weeks. The disease is more severe in patients who are immunosuppressed, splenectomized, and/or elderly. Infections caused by B. divergens tend to be more severe (frequently fatal if not appropriately treated) than those due to B. microti, where clinical recovery usually occurs.
Babesia sp. in thick blood smears stained with Giemsa.
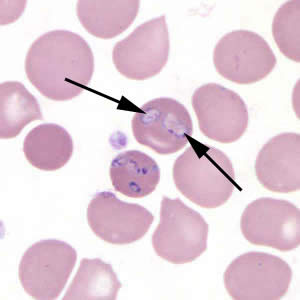
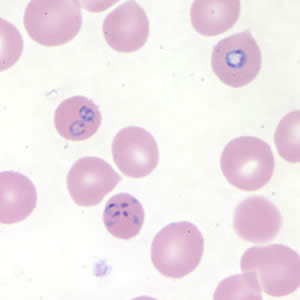
Babesia parasites resemble Plasmodium falciparum, however Babesia has several distinguishing features: the parasites are pleomorphic (vary in shape and size), can be vacuolated, and do not produce pigment.








Babesia sp. (tetrad forms) in thin blood smears stained with Giemsa.













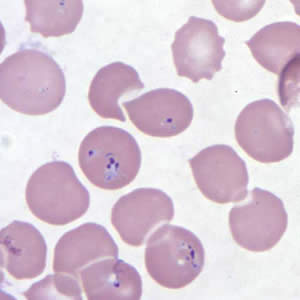


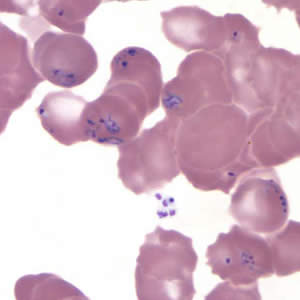

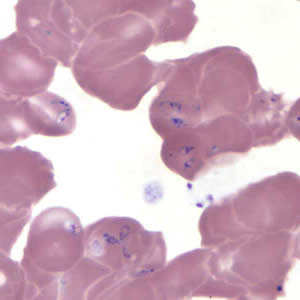
Babesia duncani in a thin blood smear stained with Giemsa.
Babesia parasites resemble Plasmodium falciparum, however Babesia has several distinguishing features: the parasites vary more in shape and in size (pleomorphic), and they do not produce pigment.
Ixodes spp., vectors of babesiosis.



Laboratory Diagnosis
Diagnosis can be made by microscopic examination of thick and thin blood smears stained with Giemsa. Repeated smears may be needed.
Antibody Detection
Diagnosis of Babesia infection should be made by detection of parasites in patients’ blood smears. However, antibody detection tests are useful for detecting infected individuals with very low levels of parasitemia (such as asymptomatic blood donors in transfusion-associated cases), for diagnosis after infection is cleared by therapy, and for discrimination between Plasmodium falciparum and Babesia infection in patients whose blood smear examinations are inconclusive and whose travel histories cannot exclude either parasite.
The indirect fluorescent antibody test (IFA) using B. microti parasites as antigen detects antibodies in 88-96% of patients with B. microti infection. IFA antigen slides are prepared using washed, parasitized erythrocytes produced in hamsters. Patients’ titers generally rise to ≥1:1024 during the first weeks of illness and decline gradually over 6 months to titers of 1:16 to 1:256 but may remain detectable at low levels for a year or more. Specificity is 100% in patients with other tick-borne diseases or persons not exposed to the parasite. Cross-reactions may occur in serum specimens from patients with malaria infections, but generally titers are highest with the homologous antigen.
The extent of cross-reactivity between Babesia species is variable. A negative result with B. microti antigen for a patient exposed on the West Coast may be a false-negative reaction for Babesia infection. Individuals whose exposure could have occurred on the West Coast should be tested also for antibodies to the Babesia duncani, because of the lack of cross-reactivity with B. microti.
Reference:
Krause PJ, Telford S RI, Ryan R, et al. Diagnosis of babesiosis: Evaluation of a serologic test for the detection of Babesia microti antibody. J Infect Dis 1994;169:923-926.
Molecular diagnosis
In some infections with intraerythrocytic parasites, the morphologic characteristics observed on microscopic examination of blood smears do not allow an unambiguous differentiation between Babesia and Plasmodium. Moreover, potential blood donors may have subclinical symptoms and very low parasitemia, undetectable in blood smears. In such cases, the diagnosis can be derived from molecular techniques, such as PCR. In addition, molecular approaches are very valuable in investigations of new Babesia variants (or species) observed in recent human infections in the United States and in Europe.
References:
1. Hojgaard A, Lukacik G, Piesman J. Detection of Borrelia burgdorferi, Anaplasma phagocytophilum and Babesia microti, with two different multiplex PCR assays. Ticks and Tick-borne Diseases 2014 (5):349–351.
2. Bonnet S, Jouglin M, Malandrin L, Becker C, A. Agoulon A, L’Hostis M, Chauvin A. Transstadial and transovarial persistence of Babesia divergens DNA in Ixodes ricinus ticks fed on infected blood in a new skin-feeding technique. Parasitol 2007;134:197–207.
Treatment Information
Treatment information for babesiosis can be found at: https://www.cdc.gov/babesiosis/hcp/clinical-care/index.html
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.