
Angiostrongyliasis, Neural
[Angiostrongylus cantonensis]
Causal Agent
The nematode (roundworm) Angiostrongylus (=Parastrongylus) cantonensis, also called the rat lungworm, is a common cause of human eosinophilic meningitis (neural angiostrongyliasis) and occasionally ocular disease.
Angiostrongylus costaricensis is the causal agent of abdominal angiostrongyliasis and is discussed here.
Life Cycle
 . The females lay eggs that hatch in the terminal branches of the pulmonary arteries, yielding first-stage larvae. The first-stage larvae migrate to the pharynx, are swallowed, and passed in the feces. They penetrate or are ingested by a gastropod intermediate host
. The females lay eggs that hatch in the terminal branches of the pulmonary arteries, yielding first-stage larvae. The first-stage larvae migrate to the pharynx, are swallowed, and passed in the feces. They penetrate or are ingested by a gastropod intermediate host  . After two molts, third-stage larvae are produced
. After two molts, third-stage larvae are produced  which are infective to mammalian hosts. When the infected gastropod is ingested by the definitive host, the third-stage larvae migrate to the brain where they develop into young adults
which are infective to mammalian hosts. When the infected gastropod is ingested by the definitive host, the third-stage larvae migrate to the brain where they develop into young adults  . The young adults return to the venous system and then the pulmonary arteries where they become sexually mature. Of note, various animals act as paratenic (transport) hosts: after ingesting the infected snails, they carry the third-stage larvae which can resume their development when the paratenic host is ingested by a definitive host. Humans can acquire the infection by eating raw or undercooked snails or slugs infected with the parasite; they may also acquire the infection by eating raw produce that contains a small snail or slug, or part of one
. The young adults return to the venous system and then the pulmonary arteries where they become sexually mature. Of note, various animals act as paratenic (transport) hosts: after ingesting the infected snails, they carry the third-stage larvae which can resume their development when the paratenic host is ingested by a definitive host. Humans can acquire the infection by eating raw or undercooked snails or slugs infected with the parasite; they may also acquire the infection by eating raw produce that contains a small snail or slug, or part of one  . There is some question whether or not larvae can exit the infected gastropods in slime (which may be infective to humans if ingested, for example, on produce). Infection may also be acquired by ingestion of invertebrate paratenic hosts containing L3 larvae (e.g. crabs, freshwater shrimp). In humans, larvae migrate to the brain, or rarely to the lungs, where the worms ultimately die
. There is some question whether or not larvae can exit the infected gastropods in slime (which may be infective to humans if ingested, for example, on produce). Infection may also be acquired by ingestion of invertebrate paratenic hosts containing L3 larvae (e.g. crabs, freshwater shrimp). In humans, larvae migrate to the brain, or rarely to the lungs, where the worms ultimately die  . Larvae may develop to fourth or fifth stage in the human host, but seem to be incapable of maturing fully.
. Larvae may develop to fourth or fifth stage in the human host, but seem to be incapable of maturing fully.
Learn more about Angiostrongylus cantonensis in this new motion graphic video.
Hosts
A. cantonensis is known to use multiple rat species as definitive hosts, including black rats (Rattus rattus), brown rats, (Rattus norvegicus), and cotton rats (Sigmodon hispidus). Gastropod (snail and slug) intermediate hosts are numerous and include Achatina spp., Biomphlalaria spp., Bulinus spp., Lymnaea spp., Pomaecea spp., and more. Apart from humans, aberrant infections have occurred in several species of wild and domestic mammals and birds.
Geographic Distribution
Most cases of A. cantonensis-associated eosinophilic meningitis originate from Southeast Asia and Pacific Islands. The parasite has also been reported from Africa, the Caribbean, Australia, Hawaii, and recently the southern United States.
Clinical Presentation
The most notable manifestation of A. cantonensis infection is eosinophilic meningitis caused by the presence of larvae in the brain and resultant local host reactions. Somatic symptoms (e.g. headache, fever, malaise) as well as varying degrees of neurological dysfunction are usually noted. A. cantonensis infection may occasionally prove fatal. Ocular angiostrongyliasis is associated with uveitis, blurred vision, and a substantial loss of visual acuity.



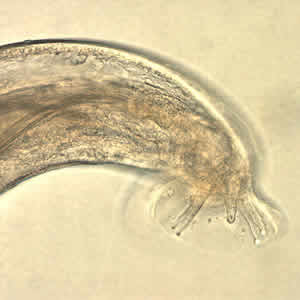





L3 larvae are infective to humans, who serve as incidental hosts. They are around 0.5 mm long, possess a pointed terminal projection on the end of the tail, and may be encased in cuticle (exuviae) molted from previous stages. The following photographs show L3 larvae derived from slug tissues.






Clinical and Morphologic Diagnosis
In eosinophilic meningitis with A. cantonensis, the cerebrospinal fluid (CSF) is abnormal (elevated pressure, proteins, and leukocytes; eosinophilia). On rare occasions, larvae have been found in the CSF or in sections of brain tissue.
Molecular Diagnosis
The CDC offers a real-time PCR assay for the specific detection of A. cantonensis in human CSF specimens. The CSF should be kept cold (ice packs) and shipped to CDC as soon as possible; alternatively it can be kept frozen until shipment is arranged. Shipment on dry ice is optional. Recommended volume of CSF is at least 2 mL. Volumes less than 1 mL may lead to reduced test sensitivity.
Diagnostic References:
Qvarnstrom Y, da Silva AQA, Teem JL, Hollingsworth R, Bishop H, Graeff-Teixeira C, and da Silva AJ: Improved Molecular Detection of Angiostrongylus cantonensis in Mollusks and Other Environmental Samples with a Species-Specific Internal Transcribed Spacer 1-Based TaqMan Assay. Appl. Envir. Microbiol.; 2010; 76: 5287 – 5289.
Qvarnstrom Y, Sullivan JJ, Bishop HS, Hollingsworth R, da Silva AJ: PCR-based detection of Angiostrongylus cantonensis in tissue and mucus secretions from molluscan hosts. Appl. Environ. Microbiol.; 2007; 73:1415-1419.
Eamsobhanaa P, Limb PE, Solano G, Zhange H, Ganf X, Yongc HS: Molecular differentiation of Angiostrongylus taxa (Nematoda: Angiostrongylidae) by cytochrome c oxidase subunit I (COI) gene sequences. Acta Tropica; 2010; 116: 152–156.
Laboratory Safety
Observe standard universal precautions for handling CSF and other bodily fluids.
Suggested Reading
Barratt, J., Chan, D., Sandaradura, I., Malik, R., Spielman, D., Lee, R., Marriott, D., Harkness, J., Ellis, J. and Stark, D., 2016. Angiostrongylus cantonensis: a review of its distribution, molecular biology and clinical significance as a human pathogen. Parasitology, 143(9), pp.1087-1118.
Tseng, Y.T., Tsai, H.C., Sy, C.L., Lee, S.S.J., Wann, S.R., Wang, Y.H., Chen, J.K., Wu, K.S. and Chen, Y.S., 2011. Clinical manifestations of eosinophilic meningitis caused by Angiostrongylus cantonensis: 18 years’ experience in a medical center in southern Taiwan. Journal of Microbiology, Immunology and Infection, 44 (5), pp.382-389.
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.
