
Angiostrongyliasis
[Angiostrongylus cantonensis] [Angiostrongylus costaricensis]
Causal Agents
The nematode (roundworm) Angiostrongylus cantonensis, the rat lungworm, is the most common cause of human eosinophilic meningitis. In addition, Angiostrongylus (Parastrongylus) costaricensis is the causal agent of abdominal, or intestinal, angiostrongyliasis.
Life Cycle

Learn more about Angiostrongylus cantonensis in this new motion graphic video.
The life cycle of Angiostrongylus (Parastrongylus) costaricensis is similar, except that the adult worms reside in the arterioles of the ileocecal area of the definitive host. In humans, A. costaricensis often reaches sexual maturity and release eggs into the intestinal tissues. The eggs and larvae degenerate and cause intense local inflammatory reactions and do not appear to be shed in the stool.
Geographic Distribution
Most cases of eosinophilic meningitis have been reported from Southeast Asia and the Pacific Basin, although the infection is spreading to many other areas of the world, including Africa and the Caribbean. Abdominal angiostrongyliasis has been reported from Costa Rica, and occurs most commonly in young children.
Clinical Presentation
Clinical symptoms of eosinophilic meningitis are caused by the presence of larvae in the brain and by local host reactions. Symptoms include severe headaches, nausea, vomiting, neck stiffness, seizures, and neurologic abnormalities. Occasionally, ocular invasion occurs. Eosinophilia is present in most of cases. Most patients recover fully. Abdominal angiostrongyliasis mimics appendicitis, with eosinophilia.
Angiostrongylus costaricensis eggs and larvae in tissue stained with hematoxylin and eosin (H&E).
In humans, Angiostrongylus eggs and larvae are not normally excreted but remain sequestered in tissues. A. costaricensis infections are predominantly abdominal; both eggs and larvae (occasionally adult worms) can be identified in biopsy or surgical specimens of intestinal tissue where the eggs and larvae are engulfed in giant cells and/or granulomas. A. cantonensis infections are predominantly cerebral, being one of the most common causes of eosinophilic meningitis, although developing but immature adult worms can on occasion migrate to the lungs. No A. cantonensis eggs or larvae have been recognized in human tissues.
The larvae of A. costaricensis need to be distinguished from larvae of Strongyloides stercoralis. A. costaricensis first stage (L1) larvae tend to be slightly smaller in diameter than Strongyloides and have a single lateral ala, whereas Strongyloides third stage (L3) larvae have minute double lateral alae. The alae can be difficult to discern in most histologic sections. However, the presence of granulomas containing thin shelled eggs and/or larvae generally serve to distinguish A. costaricensis infections from Strongyloides infections.








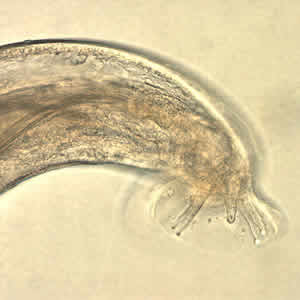



A. cantonensis larvae recovered from slugs.






Laboratory Diagnosis
Morphologic Diagnosis
In eosinophilic meningitis with A. cantonensis, the cerebrospinal fluid (CSF) is abnormal (elevated pressure, proteins, and leukocytes; eosinophilia). On rare occasions, larvae have been found in the CSF. In abdominal angiostrongyliasis with A. costaricensis, eggs and larvae can be identified in biopsy specimens.
Molecular Diagnosis
The CDC offers a real-time PCR assay for the specific detection of A. cantonensis in human CSF specimens. The CSF should be kept cold and shipped to CDC on ice pack as soon as possible; alternatively it can be kept frozen until shipment is arranged. Shipment on dry ice is optional. Recommended volume of CSF is at least 2 ml. Volumes less than 1 ml may lead to reduced test sensitivity.
No specific molecular tests are available for A. costaricensis. A conventional PCR followed by DNA sequencing can be performed for A. costaricensis on a case-by case basis. Acceptable specimen type is tissue biopsies.
References
Qvarnstrom Y, da Silva AQA, Teem JL, Hollingsworth R, Bishop H, Graeff-Teixeira C, and da Silva AJ: Improved Molecular Detection of Angiostrongylus cantonensis in Mollusks and Other Environmental Samples with a Species-Specific Internal Transcribed Spacer 1-Based TaqMan Assay. Appl. Envir. Microbiol.; 2010; 76: 5287 – 5289.
Qvarnstrom Y, Sullivan JJ, Bishop HS, Hollingsworth R, da Silva AJ: PCR-based detection of Angiostrongylus cantonensis in tissue and mucus secretions from molluscan hosts. Appl. Environ. Microbiol.; 2007; 73:1415-1419.
Eamsobhanaa P, Limb PE, Solano G, Zhange H, Ganf X, Yongc HS: Molecular differentiation of Angiostrongylus taxa (Nematoda: Angiostrongylidae) by cytochrome c oxidase subunit I (COI) gene sequences. Acta Tropica; 2010; 116: 152–156.
Treatment Information
Treatment information for angiostrongyliasis can be found at: Clinical Treatment of Rat Lungworm | Rat Lungworm Disease (Angiostrongylus) | CDC
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.