
Oesophagostomiasis
[Oesophagostomum spp.]
Causal Agents
Bursate nematodes in the genus, Oesophagostomum. Oesophagostomum bifurcum is the most-common species infecting humans in Africa.
Life Cycle
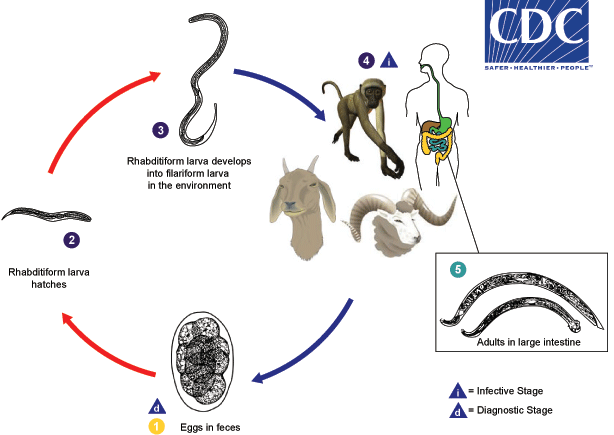
Common livestock such as sheep, goats, and swine, as well as non-human primates, are the usual definitive hosts for Oesophagostomum spp., but other animals, including humans and cattle, may also serve as definitive hosts. Eggs are shed in the feces of the definitive host  , and may be indistinguishable from the eggs of Necator and Ancylostoma. Eggs hatch into rhabditiform (L1) larvae in the environment
, and may be indistinguishable from the eggs of Necator and Ancylostoma. Eggs hatch into rhabditiform (L1) larvae in the environment  , given appropriate temperature and level of humidity. In the environment, the larvae will undergo two molts and become infective filariform (L3) larvae
, given appropriate temperature and level of humidity. In the environment, the larvae will undergo two molts and become infective filariform (L3) larvae  . Worms can go from eggs to L3 larvae in a matter of a few days, given appropriate environmental conditions. Definitive hosts become infected after ingesting infective L3 larvae
. Worms can go from eggs to L3 larvae in a matter of a few days, given appropriate environmental conditions. Definitive hosts become infected after ingesting infective L3 larvae  . After ingestion, L3 larvae burrow into the submucosa of the large or small intestine and induce cysts. Within these cysts, the larvae molt and become L4 larvae. These L4 larvae migrate back to the lumen of the large intestine, where they molt into adults
. After ingestion, L3 larvae burrow into the submucosa of the large or small intestine and induce cysts. Within these cysts, the larvae molt and become L4 larvae. These L4 larvae migrate back to the lumen of the large intestine, where they molt into adults  , . Eggs appear in the feces of the definitive host about a month after ingestion of infective L3 larvae.
, . Eggs appear in the feces of the definitive host about a month after ingestion of infective L3 larvae.
Geographic Distribution
Oesophagostomum spp. are widely distributed wherever livestock is raised, but more common in the tropics and subtropics. The highest incidence in humans is in the northern regions of Togo and Ghana, where O. bifurcum (primarily a monkey parasite) appears to cycle naturally in the human populations. Sporadic cases in humans have also been recorded in Brazil, Malaysia, Indonesia, French Guiana, and West Africa
Clinical Presentation
Acute abdomen is the most-common manifestation in humans, mimicking an appendicitis. A low-grade fever and tenderness in the lower-right quadrant are the most-common symptoms; vomiting, anorexia, and diarrhea are less-common. Intestinal obstruction may also occur, mimicking a hernia. Patients may also present with large, painless cutaneous masses in the lower abdominal region. In rare instances, Oesophagostomum spp. will perforate the bowel wall, causing purulent peritonitis or migrate to the skin, producing cutaneous nodules.
Eggs of Oesophagostomum spp.


L3 infective larvae of Oesophagostomum spp.




Adults of Oesophagostomum spp.






Oesophagostomum spp. in tissue specimens.



Laboratory Diagnosis
Diagnosis is difficult during routine ova and parasite (O&P) examinations of stool, due to the similarity of Oesophagostomum eggs to the eggs of Necator and Ancylostoma. Eggs tend to be shed in greater numbers during cases of oesophagostomiasis than hookworm infection, however. Finding an intact worm during surgery or in a biopsy specimen can provide a definitive diagnosis.
Treatment Information
For information about treatment please contact CDC-INFO.
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.