
Fasciolopsiasis
[Fasciolopsis buski]
Causal Agents
The trematode Fasciolopsis buski, the largest intestinal fluke of humans.
Life Cycle

Immature eggs are discharged into the intestine and stool . Eggs become embryonated in water
. Eggs become embryonated in water  , eggs release miracidia
, eggs release miracidia  , which invade a suitable snail intermediate host
, which invade a suitable snail intermediate host  . In the snail the parasites undergo several developmental stages (sporocysts
. In the snail the parasites undergo several developmental stages (sporocysts 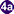 , rediae
, rediae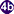 , and cercariae
, and cercariae 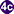 ). The cercariae are released from the snail
). The cercariae are released from the snail  and encyst as metacercariae on aquatic plants
and encyst as metacercariae on aquatic plants  . The mammalian hosts become infected by ingesting metacercariae on the aquatic plants. After ingestion, the metacercariae excyst in the duodenum
. The mammalian hosts become infected by ingesting metacercariae on the aquatic plants. After ingestion, the metacercariae excyst in the duodenum  and attach to the intestinal wall. There they develop into adult flukes (20 to 75 mm by 8 to 20 mm) in approximately 3 months, attached to the intestinal wall of the mammalian hosts (humans and pigs)
and attach to the intestinal wall. There they develop into adult flukes (20 to 75 mm by 8 to 20 mm) in approximately 3 months, attached to the intestinal wall of the mammalian hosts (humans and pigs)  . The adults have a life span of about one year.
. The adults have a life span of about one year.
Geographic Distribution
Asia and the Indian subcontinent, especially in areas where humans raise pigs and consume freshwater plants.
Clinical Presentation
Most infections are light and asymptomatic. In heavier infections, symptoms include diarrhea, abdominal pain, fever, ascites, anasarca and intestinal obstruction.
Fasciolopsis buski eggs.





Fasciolopsis buski adults.


Intermediate hosts of F. buski.


Laboratory Diagnosis
Microscopic identification of eggs, or more rarely of the adult flukes, in the stool or vomitus is the basis of specific diagnosis. The eggs are indistinguishable from those of Fasciola hepatica.
Morphology
Treatment Information
Treatment information for fascioliasis can be found at: https://www.cdc.gov/fasciolopsis/hcp/clinical-care/index.html
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.