
Cysticercosis
[Taenia solium]
Causal Agent
Cysticercosis is the disease associated with the development of the larval form (cysticercus) of the pork tapeworm, Taenia solium, within an intermediate host. Swine are the usual intermediate host for T. solium but humans, the usual definitive host, can serve as accidental intermediate hosts following ingestion of infectious eggs. Note that cysticercosis is only acquired from the fecal-oral route (ingestion of eggs), not via the ingestion of cysticerci in undercooked pork, which is associated with intestinal taeniasis.
Life Cycle
Cysticercosis is an infection of both humans and pigs with the larval stages of the parasitic cestode, Taenia solium. This infection is caused by ingestion of eggs shed in the feces of a human tapeworm carrier  . These eggs are immediately infectious and do not require a developmental period outside the host. Pigs and humans become infected by ingesting eggs or gravid proglottids
. These eggs are immediately infectious and do not require a developmental period outside the host. Pigs and humans become infected by ingesting eggs or gravid proglottids  ,
,  . Humans are usually exposed to eggs by ingestion of food/water contaminated with feces containing these eggs or proglottids or by person-to-person spread. Tapeworm carriers can also infect themselves through fecal-oral transmission (e.g. caused by poor hand hygiene). Once eggs or proglottids are ingested, oncospheres hatch in the intestine
. Humans are usually exposed to eggs by ingestion of food/water contaminated with feces containing these eggs or proglottids or by person-to-person spread. Tapeworm carriers can also infect themselves through fecal-oral transmission (e.g. caused by poor hand hygiene). Once eggs or proglottids are ingested, oncospheres hatch in the intestine  ,
,  invade the intestinal wall, enter the bloodstream, and migrate to multiple tissues and organs where they mature into cysticerci over 60–70 days
invade the intestinal wall, enter the bloodstream, and migrate to multiple tissues and organs where they mature into cysticerci over 60–70 days  ,
,  . Some cysticerci will migrate to the central nervous system, causing serious sequellae (neurocysticercosis).
. Some cysticerci will migrate to the central nervous system, causing serious sequellae (neurocysticercosis).
This differs from taeniasis, which is an intestinal infection with the adult tapeworm. Humans acquire intestinal infections with T. solium after eating undercooked pork containing cysticerci  . Cysts evaginate and attach to the small intestine by their scolices. Adult tapeworms develop to maturity and may reside in the small intestine for years
. Cysts evaginate and attach to the small intestine by their scolices. Adult tapeworms develop to maturity and may reside in the small intestine for years  .
.
Hosts
Humans are normal definitive host for T. solium; cysticercosis results from humans acting as accidental intermediate hosts for the parasite (this role is normally fulfilled by swine).
Geographic Distribution
Taenia solium is found nearly worldwide. Because pigs are intermediate hosts of the parasite, completion of the life cycle occurs in regions where humans live in close contact with pigs and eat undercooked pork. Poor sanitation leading to environmental fecal contamination is a major factor in transmission. Cysticercosis mainly affects low- and middle-income countries in Africa, Asia (e.g., India, China, and Nepal) and Latin America (e.g., Guatemala, Nicaragua, El Salvador).
It is important to note that human cysticercosis is acquired by ingesting T. solium eggs shed in the feces of a human T. solium tapeworm carrier (e.g. on contaminated food items), and thus can still occur in populations that neither eat pork nor share environments with pigs, as long as the human carrier is present.
Clinical Presentation
The symptoms of cysticercosis vary depending upon the location and number of cysticerci. Cysticerci may develop in skeletal and heart muscle, skin, subcutaneous tissues, the lungs, liver, and other tissues, including the oral mucosa. In most locations, cysticerci cause few symptoms and spontaneously degenerate.
Cysticerci can migrate to the central nervous system and cause neurocysticercosis (NCC), which is associated with serious neurological and epileptic manifestations. Death can occur suddenly. NCC is typically divided into parenchymal and extraparenchymal disease. Parenchymal NCC occurs when cysticerci develop within the brain tissue. Extraparynchymal NCC occurs when cysticerci develop in other parts of the nervous system, such as the subarachnoid space, meninges, ventricles, spine, or eyes. A rare variant called “racemose cysticercosis” involves the development of cysts that are unusually large, multilobular, and clustered; they may lack a scolex. This form usually occurs in extraparenchymal sites (e.g., subarachnoid space, meninges). Mixed parenchymal and extraparenchymal disease can occur.
Taenia solium cysticerci (also called “cysticercus cellulosae”) are fluid-filled cystic structures consisting of a thin bladder wall and parenchymatous portion containing a single invaginated scolex surrounded by a convoluted spiral canal. The hooks of the armed scolex may be visible in tissue sections. Cysticerci are typically 5—15 mm long, but may reach up to 5 cm.




A rare variant of cysticercosis (racemose cysticercosis or “cysticercus racemosus”) involves the development of a spreading, multilobular cyst that may lack a scolex, typically in the subarachnoid region.

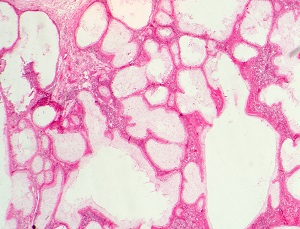
Laboratory Diagnosis
Diagnosis of neurocysticercosis involves a careful history and physical exam, neuroimaging studies (both a magnetic resonance imaging [MRI] and a non-contrast computed tomography [CT] scan of the brain), and serologic testing with enzyme-linked immunotransfer blot. All patients strongly suspected of having cysticercosis or neurocysticercosis should have confirmatory testing by immunoblot.
Antibody Detection
The immunoblot assay based on T. solium antigens is the test of choice for confirming a clinical and radiologic presumptive diagnosis of cysticercosis and can be used to test both serum and/or CSF. Note that currently available antibody detection tests for cysticercosis do not distinguish between active and inactive infections and thus have not been useful in evaluating the outcomes and prognoses of medically treated patients.
Antigen Detection
Antigen testing is available at CDC in specific instances and after consultation with CDC (Public Inquiries).
Microscopic Diagnosis
The definitive diagnosis consists of demonstrating the cysticercus in the tissue involved. Cysticercosis can be diagnosed by the demonstration of larval Taenia solium cysts (cysticerci) in tissue sections.
Laboratory Safety
Standard safety precautions for the processing of tissue or serum samples apply.
Suggested Reading
Cantey, P.T., Coyle, C.M., Sorvillo, F.J., Wilkins, P.P., Starr, M.C. and Nash, T.E., 2014. Neglected parasitic infections in the United States: cysticercosis. The American Journal of Tropical Medicine and Hygiene, 90(5), pp.805–9. DOI: 10.4269/ajtmh.13-0724
Coyle C.M., Mahanty S., Zunt J.R., Wallin M.T., Cantey P.T., White Jr. C., O’Neal S.E., Serpa J.A., Southern P.M., Wilkins P., McCarthy A.E., Higgs E.S., and Nash T.E., 2012. Neurocysticercosis: neglected but not forgotten. PLoS Neglected Tropical Diseases, 6(5), e1500. DOI:10.1371/journal.pntd.0001500
Garcia H.H., Nash T.E., Del Brutto O.H., 2014. Clinical symptoms, diagnosis, and treatment of neurocysticercosis. Lancet Neurology, 13(12), pp. 1202–15. DOI:10.1016/S1474-4422(14)70094-8
Gripper L.B and Welburn S.C., 2017. Neurocysticercosis infection and disease—A review. Acta Tropica, 166, pp. 218–224. DOI:10.1016/j.actatropica.2016.11.015
World Health Organization, 2018. Fact Sheet on taeniasis/cysticercosis (updated February 2018). Weekly Epidemiological Record, 93(46), pp. 630–2. Available from: http://apps.who.int/iris/bitstream/handle/10665/275876/WER9346.pdf?ua=1
White Jr. A.C., Coyle C.M., Rajshekhar V., Singh G., Hauser W.A., Mohanty A., Garcia H.H., Nash T.E., 2018. Diagnosis and treatment of neruocysticercosis: 2017 clinical practice guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of tropical Medicine and Hygiene (ASTMH). Am J Trop Med Hyg, 98(4), pp. 945–66. DOI:10.4269/ajtmh.18-88751
Winkler A.S. and Richter H., 2015. Landscape analysis: management of neurocysticercosis with an emphasis on low- and middle-income countries. Geneva: World Health Organization; WHO/HTM/NTD/NZD/2015.05. Available from: http://apps.who.int/iris/bitstream/handle/10665/152896/WHO_HTM_NTD_NZD_2015.05_eng.pdf?sequence=1
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.
