Cyclosporiasis
[Cyclospora cayetanensis]
Causal Agents
Cyclospora cayetanensis is a coccidian protozoan. It appears that all human cases are caused by this species; no animal reservoirs for C. cayetanensis have been identified.
Life Cycle
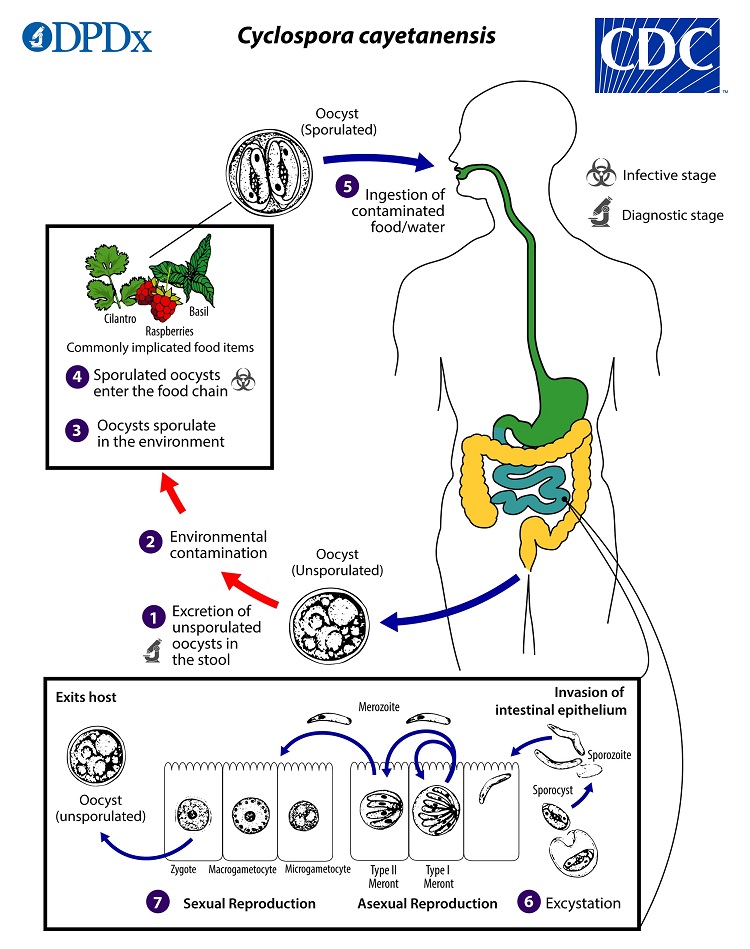
When freshly passed in stools, the oocyst is not infective  (thus, direct fecal-oral transmission cannot occur; this differentiates Cyclospora from another important coccidian parasite, Cryptosporidium). In the environment
(thus, direct fecal-oral transmission cannot occur; this differentiates Cyclospora from another important coccidian parasite, Cryptosporidium). In the environment  , sporulation occurs after days or weeks at temperatures between 22°C to 32°C, resulting in division of the sporont into two sporocysts, each containing two elongate sporozoites
, sporulation occurs after days or weeks at temperatures between 22°C to 32°C, resulting in division of the sporont into two sporocysts, each containing two elongate sporozoites  . The sporulated oocysts can contaminate fresh produce and water
. The sporulated oocysts can contaminate fresh produce and water  which are then ingested
which are then ingested  . The oocysts excyst in the gastrointestinal tract, freeing the sporozoites, which invade the epithelial cells of the small intestine
. The oocysts excyst in the gastrointestinal tract, freeing the sporozoites, which invade the epithelial cells of the small intestine  . Inside the cells they undergo asexual multiplication into type I and type II meronts. Merozoites from type I meronts likely remain in the asexual cycle, while merozoites from type II meronts undergo sexual development into macrogametocytes and microgametocytes upon invasion of another host cell. Fertilization occurs, and the zygote develops to an oocyst which is released from the host cell and shed in the stool
. Inside the cells they undergo asexual multiplication into type I and type II meronts. Merozoites from type I meronts likely remain in the asexual cycle, while merozoites from type II meronts undergo sexual development into macrogametocytes and microgametocytes upon invasion of another host cell. Fertilization occurs, and the zygote develops to an oocyst which is released from the host cell and shed in the stool  . Several aspects of intracellular replication and development are still unknown, and the potential mechanisms of contamination of food and water are still under investigation.
. Several aspects of intracellular replication and development are still unknown, and the potential mechanisms of contamination of food and water are still under investigation.
Hosts
Humans appear to be the only major host for C. cayetanensis. Occasionally, cysts are recovered from animal feces, but it is likely that this represents spurious passage following coprophagy.
Geographic Distribution
Cyclosporiasis has been reported in many countries, but is most common in tropical and subtropical areas. In the United States, the majority of cases are reported during the spring and summer months. Outbreaks have been identified nearly every year since the mid-1990s.
Clinical Presentation
After an average incubation period of one week, symptomatic infections typically manifest as watery diarrhea of varying severity. Other manifestations include complications of dysentery, further abdominal symptoms, and sometimes non-specific systemic symptoms (e.g. headache, low-grade fever). Untreated infections typically last for 10–12 weeks and may follow a relapsing course. The duration of symptoms and associated weight loss are greater in individuals with HIV or possibly other immunosuppressive conditions. Infections can be asymptomatic in disease-endemic regions.
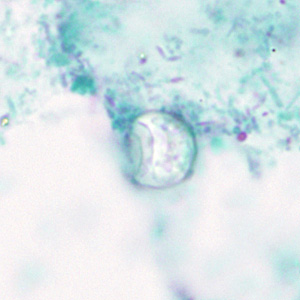
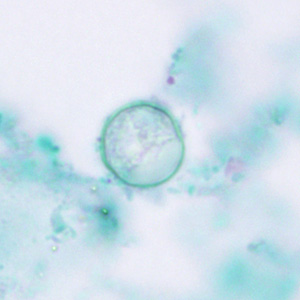
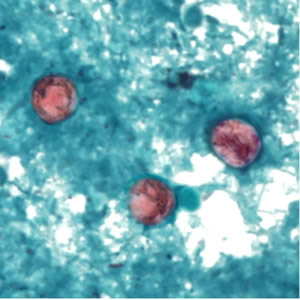
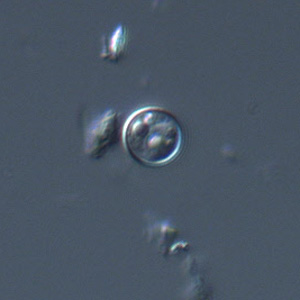
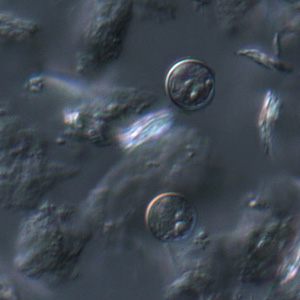
Cyclospora cayetanensis oocysts in wet mounts.











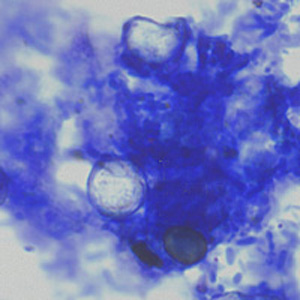
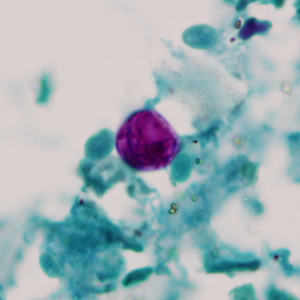
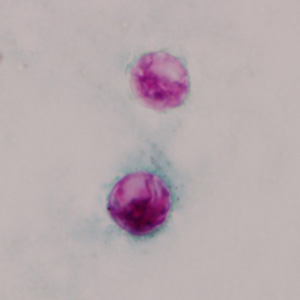
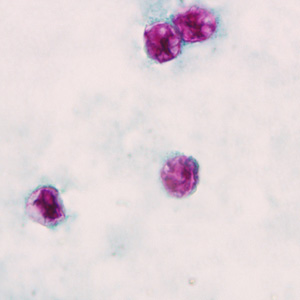









Laboratory Diagnosis
Unpreserved stool collected in enteric transport media (e.g., Cary-Blair) is commonly used for culture independent diagnostic tests (CIDTs) and can be used for confirmatory testing by microscopy and/or PCR if needed. Unpreserved specimens should be refrigerated and sent to the diagnostic laboratory as rapidly as possible.
Cyclospora oocysts can be excreted intermittently and in small numbers. Thus:
- A single negative stool specimen does not rule out the diagnosis; three or more specimens at 2- or 3-day intervals may be required.
- Concentration procedures should be used to maximize recovery of oocysts. The method most familiar to laboratorians is the formalin-ethyl acetate sedimentation technique (centrifuge for 10 minutes at 500 × g). Other methods can also be used (such as the Sheather’s flotation procedure).
Microscopic Examination
The sediment can be examined microscopically with the following techniques:
- Wet mounts (by conventional light microscopy, which can be enhanced by UV fluorescence microscopy or differential interference contrast [DIC, Nomarsky])
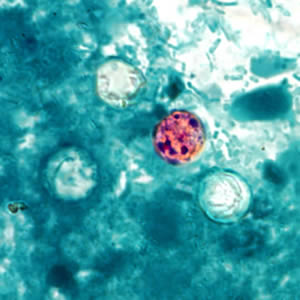
- Stained smears (using modified acid-fast stain or a modified safranin stain). Both modified acid-fast stains and modified safranin staining will stain Cyclospora cysts from a pink to brilliant red. Staining may be more variable using modified acid-fast.
- Other common stains including Giemsa and trichrome are not adequate for the reliable detection of Cyclospora.
Molecular Diagnosis
Several conventional and real-time PCR protocols have been developed to specifically detect Cyclospora cayetanensis in stool. Commercial, FDA-approved panels (that include other enteric pathogens) are available and are highly sensitive.
Laboratory Safety
Standard precautions for the processing of stool samples apply.
Cyclospora oocysts are not immediately infectious when passed, however, precautions should still be taken to avoid exposure to other pathogens that may be present in stools.
Suggested Reading
Ortega, Y.R. and Sanchez, R., 2010. Update on Cyclospora cayetanensis, a food-borne and waterborne parasite. Clinical Microbiology Reviews, 23 (1), pp.218–234.
Casillas, S.M., Bennett, C. and Straily, A., 2018. Notes from the Field: Multiple Cyclosporiasis Outbreaks—United States, 2018. Morbidity and Mortality Weekly Report, 67(39), p.1101.
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.