
Baylisascariasis
[Baylisascaris procyonis]
Causal Agent
Baylisascaris procyonis, the raccoon roundworm, is a rare but serious cause of neurologic and ocular disease in humans. Only larval stages are involved in zoonotic infections. It is not known whether other Baylisascaris species, such as B. columnaris of skunks, can cause baylisascariasis in humans.
Life Cycle
Baylisascaris procyonis completes its life cycle in raccoons, with humans acquiring the infection as accidental hosts (dogs serve as alternate definitive hosts, as they can harbor adult worms and shed eggs). Unembryonated eggs are shed in the environment  , where they take 2–4 weeks to embryonate and become infective
, where they take 2–4 weeks to embryonate and become infective  . Raccoons can be infected by ingesting embryonated eggs from the environment
. Raccoons can be infected by ingesting embryonated eggs from the environment  . Many mammals and birds can act as paratenic hosts
. Many mammals and birds can act as paratenic hosts  for this parasite: eggs ingested by these hosts hatch and larvae penetrate the gut wall and migrate into various tissues where they encyst
for this parasite: eggs ingested by these hosts hatch and larvae penetrate the gut wall and migrate into various tissues where they encyst  . The life cycle is completed when raccoons eat these hosts
. The life cycle is completed when raccoons eat these hosts  . The larvae develop into adult worms in the raccoon’s small intestine
. The larvae develop into adult worms in the raccoon’s small intestine  and eggs are passed in raccoon feces. Humans become accidentally infected when they ingest infective eggs from the environment
and eggs are passed in raccoon feces. Humans become accidentally infected when they ingest infective eggs from the environment  . Migration of the larvae through a wide variety of tissues (liver, heart, lungs, brain, eyes) results in visceral (VLM) and ocular larva migrans (OLM) syndromes, but severe neurologic disease with eosinophilic meningoencephalitis may occur following neural larva migrans (NLM)
. Migration of the larvae through a wide variety of tissues (liver, heart, lungs, brain, eyes) results in visceral (VLM) and ocular larva migrans (OLM) syndromes, but severe neurologic disease with eosinophilic meningoencephalitis may occur following neural larva migrans (NLM)  . B. procyonis larvae continue to increase in size up to about 1.8 mm in human hosts, but they are not capable of migrating to the intestine and developing to adulthood. Tissue damage and the signs and symptoms of baylisascariasis are often severe because of the larger size of B. procyonis larvae and their more invasive course of migration.
. B. procyonis larvae continue to increase in size up to about 1.8 mm in human hosts, but they are not capable of migrating to the intestine and developing to adulthood. Tissue damage and the signs and symptoms of baylisascariasis are often severe because of the larger size of B. procyonis larvae and their more invasive course of migration.
Hosts
Raccoons are the major definitive host for B. procyonis. Rarely, domestic dogs and possibly other procyonids (e.g., kinkajous) may have patent infections and shed eggs in feces. The range of paratenic hosts that are susceptible to larva migrans is broad; infections have been recorded in over 150 species of birds and mammals.
Geographic Distribution
Baylisascaris procyonis is widespread among raccoons in the United States and Canada but prevalence varies regionally. Prevalence in raccoons is extremely high in the Northeast, Midwest, and on the West Coast, sometimes in excess of 80%. This roundworm has also been found in raccoons in the Southeast and Southwest regions where it was historically absent, although the prevalence is generally much lower than in northern latitudes. B. procyonis has also been introduced to many parts of Europe, China, and Japan through trade of live raccoons. Most documented human cases have occurred in areas where the parasite is common (e.g., California, Washington, Minnesota, New York).
Clinical Presentation
Infection with Baylisascaris larvae can result in larva migrans syndromes, including visceral larva migrans (VLM), ocular larva migrans (OLM), and neural larva migrans (NLM). B. procyonis-associated NLM is potentially life-threatening. The usual presentation of cerebral baylisascariasis is eosinophilic meningoencephalitis with typical neurologic signs/symptoms. Ocular baylisascariasis has been associated with diffuse unilateral subacute neuroretinitis (DUSN) potentially leading to permanent vision loss.
Some Baylisascaris infections appear to be subclinical or asymptomatic. Antibodies to Baylisascaris have been detected in otherwise healthy individuals, suggesting past infection. The broad-scale prevalence and clinical significance of subclinical infections are not well understood.
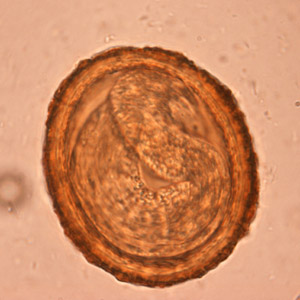
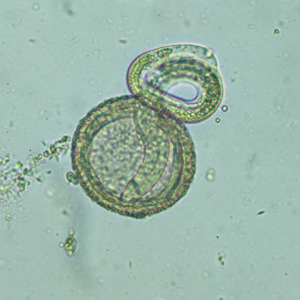
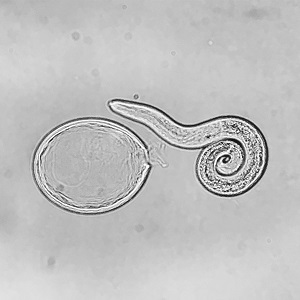
Baylisascaris procyonis eggs.
Baylisascaris procyonis eggs are 80—85 µm by 65—70 µm in size, thick-shelled, and usually slightly oval in shape. They have a similar morphology to fertile eggs of Ascaris lumbricoides, although eggs of A. lumbricoides are smaller (55—75 µm by 35—50 µm) and more coarsely pitted. As humans are not definitive hosts for B. procyonis, eggs are not excreted in human feces. The following images show larval development within the eggs, from a freshly-shed unembryonated egg to eggs containing L1 larvae. Images courtesy of Dr. Cheryl Davis, Western Kentucky University, KY.






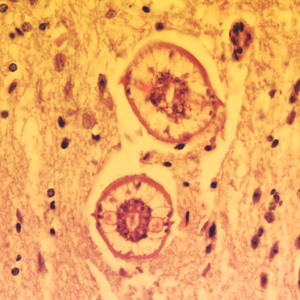
Larvae of Baylisascaris spp. in tissue.



B. procyonis adults.
Baylisascaris procyonis adults are found only in the definitive host, the raccoon (rarely dogs). They have the typical large and robust form of ascarid worms, and lack the prominent cervical alae of adult Toxocara species. Males measure 6—9 cm in length and demonstrate the curved posterior end similar to Ascaris. Females are very large and measure 8—18 cm in length.


Laboratory Diagnosis
Human infections are difficult to diagnose, and often the diagnosis is by exclusion of other causes. Results from complete blood count (CBC) and cerebrospinal fluid (CSF) examination often reveal eosinophilia. Examination of tissue biopsies can be extremely helpful if a section of larva is contained, but removing a piece of tissue in which there are larva present can be problematic. Ocular examinations revealing a migrating larva, larval tracks, or lesions consistent with a nematode larva are often the most significant clue to infection with Baylisascaris.
CDC has developed an immunoblot for detection of antibodies to recombinant Baylisascaris antigen (rBpRAG-1) in serum or CSF, which can be performed for cases of clinical neurologic disease when the index of suspicion is high*.
* Rascoe, L.N., Santamaria, C., Handali, S., Dangoudoubiyam, S., Kazacos, K.R., Wilkins, P.P. and Ndao, M., 2013. Interlaboratory optimization and evaluation of a serological assay for diagnosis of human baylisascariasis. Clinical and Vaccine Immunology, 20 (11), pp.1758-1763.
Laboratory safety
Standard protocols for the processing of histologic, serum, and CSF samples apply.
Suggested reading
Kazacos, K.R., 2016. Baylisascaris larva migrans: US Geological Survey circular 1412. Reston (VA): US Geological Survey.
Sircar, A.D. et al. 2016. Raccoon roundworm infection associated with central nervous system disease and ocular disease—six states, 2013–2015. MMWR. Morbidity and Mortality Weekly Report, 65 (35); pp. 930–933
Sapp, S.G.H., Rascoe, L.N., Wilkins, P.P., Handali, S., Gray, E.B., Eberhard, M., Woodhall, D.M., Montgomery, S.P., Bailey, K.L., Lankau, E.W. and Yabsley, M.J., 2016. Baylisascaris procyonis roundworm seroprevalence among wildlife rehabilitators, United States and Canada, 2012–2015. Emerging Infectious Diseases, 22(12), pp. 2128–2131.
DPDx is an educational resource designed for health professionals and laboratory scientists. For an overview including prevention, control, and treatment visit www.cdc.gov/parasites/.
