Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Guidelines for Safe Work Practices in Human and Animal Medical Diagnostic Laboratories
Recommendations of a CDC-convened, Biosafety Blue Ribbon Panel
Please note: An erratum has been published for this article. To view the erratum, please click here.
The material in this report originated in the National Center for Emerging and Zoonotic Infectious Diseases, Beth P. Bell, MD, MPH, Director.
Corresponding preparer: J. Michael Miller, PhD, Microbiology Technical Services, LLC, Dunwoody, GA 30338. Telephone: 678-428-6319; Fax: 770-396-0955; E-mail: jmm8@comcast.net.
Summary
Prevention of injuries and occupational infections in U.S. laboratories has been a concern for many years. CDC and the National Institutes of Health addressed the topic in their publication Biosafety in Microbiological and Biomedical Laboratories, now in its 5th edition (BMBL-5). BMBL-5, however, was not designed to address the day-to-day operations of diagnostic laboratories in human and animal medicine. In 2008, CDC convened a Blue Ribbon Panel of laboratory representatives from a variety of agencies, laboratory organizations, and facilities to review laboratory biosafety in diagnostic laboratories. The members of this panel recommended that biosafety guidelines be developed to address the unique operational needs of the diagnostic laboratory community and that they be science based and made available broadly. These guidelines promote a culture of safety and include recommendations that supplement BMBL-5 by addressing the unique needs of the diagnostic laboratory. They are not requirements but recommendations that represent current science and sound judgment that can foster a safe working environment for all laboratorians.
Throughout these guidelines, quality laboratory science is reinforced by a common-sense approach to biosafety in day-to-day activities. Because many of the same diagnostic techniques are used in human and animal diagnostic laboratories, the text is presented with this in mind. All functions of the human and animal diagnostic laboratory — microbiology, chemistry, hematology, and pathology with autopsy and necropsy guidance — are addressed. A specific section for veterinary diagnostic laboratories addresses the veterinary issues not shared by other human laboratory departments. Recommendations for all laboratories include use of Class IIA2 biological safety cabinets that are inspected annually; frequent hand washing; use of appropriate disinfectants, including 1:10 dilutions of household bleach; dependence on risk assessments for many activities; development of written safety protocols that address the risks of chemicals in the laboratory; the need for negative airflow into the laboratory; areas of the laboratory in which use of gloves is optional or is recommended; and the national need for a central site for surveillance and nonpunitive reporting of laboratory incidents/exposures, injuries, and infections.
1. Introduction: A Culture of Safety for Diagnostic Laboratories
This report offers guidance and recommends biosafety practices specifically for human and animal clinical diagnostic laboratories and is intended to supplement the 5th edition of Biosafety in Microbiological and Biomedical Laboratories (BMBL-5), developed by CDC and the National Institutes of Health (1). This document was written not to replace existing biosafety guidelines, but to 1) improve the safety of activities in clinical diagnostic laboratories, 2) encourage laboratory workers to think about safety issues they might not previously have considered or addressed, and 3) encourage laboratorians to create and foster a culture of safety in their laboratories. Should any of the guidelines provided herein conflict with federal, state, or local laws or regulatory requirements, the laboratorian should defer to the federal, state, or local requirements. This culture of safety is also supported by the Clinical and Laboratory Standards Institute (2). Work in a diagnostic laboratory entails safety considerations beyond the biological component; therefore, these guidelines also address a few of the more important day-to-day safety issues that affect laboratorians in settings where biological safety is a major focus.
According to the U.S. Bureau of Labor Statistics, in 2008, approximately 328,000 medical laboratory technicians and technologists worked in human diagnostic laboratories in the United States. An estimated 500,000 persons in all professions work in human and animal diagnostic laboratories. Any of these workers who have chronic medical conditions or receive immunosuppressive therapy would be at increased risk for a laboratory-acquired infection (LAI) after a laboratory exposure. Precise risk for infection after exposure is unknown because determining the source or the mode of transmission often is difficult. No national surveillance system is available.
LAIs and exposures have been reported since early in the 20th century, but only in the 1970s were sufficient data available to attempt quantitative assessments of risk. Recent MMWR reports (3–11) have indicated that bacteria account for >40% of infections, with >37 species reported as etiologic agents in LAIs; however, other microbes are often implicated. Hepatitis B has been the most frequent laboratory-acquired viral infection, with a rate of 3.5–4.6 cases per 1000 workers, which is two to four times that of the general population. Any laboratorian who collects or handles tubes of blood is vulnerable (12).
Early surveys of LAIs found that laboratory personnel were three to nine times more likely than the general population to become infected with Mycobacterium tuberculosis (13,14). In a 1986 survey of approximately 4000 workers in 54 public health and 165 hospital laboratories in the United States, 3.5/1000 employee infections occurred in hospital laboratories, and 1.4/1000 employee infections occurred in public health laboratories (15). In a 1994–1995 survey of 25,000 laboratory workers from 397 clinical laboratories in the United Kingdom, the overall rate of LAI was 18/100,000 employees (16).
In a 2005 CDC study of bacterial meningitis in U.S. laboratorians, Neisseria meningitidis accounted for a substantial number of LAIs. The attack rate of this organism in the general population was 13/100,000 persons. The attack rate in the general population aged 30–59 years (the estimated age range of the average laboratorian) was 0.3 per 100,000. The attack rate for microbiologists (aged 30–59 years) was 20/100,000 (17).
LAIs have also included fungal and parasitic infections. The most common agents of laboratory-acquired fungal infections are the dimorphic fungi Blastomyces, Histoplasma, and Coccidioides (18,19); most reported infections were caused by inhalation of conidia. Reported parasite-associated LAIs were caused primarily by Leishmania, Plasmodium, Toxoplasma, Chagas disease organism, and other trypanosomes (20). Of the 52 cases of laboratory-acquired malaria, 56% were vector borne (from mosquitoes used in research, not clinical laboratories). Most infected health-care workers acquired infection from needle sticks during preparation of blood smears or while drawing blood.
In clinical chemistry laboratories, data from 17 New York hospitals listed needle puncture (103 cases), acid or alkali spills (46), glass cuts (44), splash in eye (19), and bruises and cuts (45) as the most frequent exposures (21). Needle puncture, glass cuts, splash in eye, and bruises and cuts have the highest potential for infection from microbes.
In the hematology laboratory, the major causes of injuries are likely to be exposure to blood and body fluids; needle sticks, aerosols from centrifuge or removal of tube stoppers, tube breakage; or contaminated gloves (22). In non-microbiology sections of the diagnostic laboratory, the primary mistake may be assuming that a given specimen contains no infectious agents and then working with little attention to risk for infection. This scenario can be particularly problematic in laboratories developing new technologies, such as molecular and biochemical technologies, and in point-of-care diagnostics performed by staff unaccustomed to testing that requires biosafety considerations and use of barrier techniques such as personal protective equipment.
1.1. Methods
The risks and causes of LAIs have been documented. However, there is a dearth of evidence-based research and publications focused on biosafety; particularly missing are studies documenting safe practices in the day-to-day operations of diagnostic laboratories.
In 2008, CDC convened a Blue Ribbon Panel of laboratory representatives from a variety of agencies, laboratory organizations, and facilities to review laboratory biosafety in diagnostic laboratories. Members of the panel were either selected by the invited national laboratory organization they represented or were invited by CDC because of their roles in biosafety at the national level. The organizations participating in the panel represented the majority of laboratory technologists in the United States. In addition, some members of the panel were representatives of the biosafety community. The Blue Ribbon Panel recommended that biosafety guidelines be developed to address the unique operational needs of the diagnostic laboratory community and that they be science based and made available broadly.
Panel members reviewed the guidelines that were developed and synthesized by the writing team. Official endorsements by the organizations they represented were not required, although each representative was required to submit written approval of the recommendations. Edits and comments from each participant were carefully considered and incorporated where appropriate. The guidelines provided herein are synthesized and supported from systematic reviews of peer-reviewed publications of evidence-based data from which recommendations could be made, justifying common-sense approaches that should be articulated, and where safe procedures have been described and proven. Because of the lack of evidence-based research in much of the current literature on biosafety practices, no attempt was made to weight the evidence and resulting recommendations (i.e., strong or weak). In the absence of supporting evidence-based research and documentation, some recommendations are based on expert opinion by international experts in the field of microbiology and must be appropriately applied until evidence-based research can substantiate their validity. The authors reviewed and approved their own sections and also evaluated how their topics accurately reflected and supported the goals of the entire document.
Each section of recommendations was reviewed both within CDC and by the relevant national organizations whose members would embrace these guidelines. These included the College of American Pathologists, Greater New York Hospital Association Regional Laboratory Task Force, American Society for Microbiology, American Clinical Laboratory Association, Association of Public Health Laboratories, American Society for Clinical Laboratory Science, American Society for Clinical Pathology, American Biological Safety Association, American Association of Veterinary Laboratory Diagnosticians, and individual physicians and subject matter experts. Future research in biosafety practices in the laboratory will contribute to further recommendations and will substantiate others as well as provide opportunities to revise this document.
1.2. Risk
Persons working in clinical diagnostic laboratories are exposed to many risks (1). Whether the patients are humans or animals and whether laboratorians work in microbiology or elsewhere in the laboratory, the human and animal diagnostic laboratory is a challenging environment. The more that laboratorians become aware of and adhere to recommended, science-based safety precautions, the lower the risk. The goal of a safety program is to lower the risk to as close as possible to zero, although zero risk is as yet unattainable as long as patient specimens and live organisms are manipulated. Protection of laboratorians, coworkers, patients, families, and the environment is the greatest safety concern.
1.3. Laboratory Exposures
Laboratory exposures occur more often than is generally suspected. Other laboratory incidents such as minor scrapes or cuts, insignificant spills, or unrecognized aerosols occur even more frequently and might not cause an exposure that results in an LAI. In this report, "laboratory exposures" refer to events that put employees at risk for an LAI and events that result in actual acquisition of LAIs. Except for reporting requirements imposed by CDC's Select Agent Program, which deals with handling of specific, potentially hazardous biological agents and toxins, no national surveillance system is in place to which medical laboratory exposures and subsequent work-related infections are reported. Increased attention has been focused on laboratory biosafety and biosecurity since 2001 but has been largely limited to precautions required for agents of bioterrorism. Other laboratory exposures and LAIs continue to occur, almost always because of a breakdown of established safety protocols. Because of the lack of an official surveillance mechanism for reporting LAIs and because of the fear of punitive action by an oversight agency if injuries are reported, the data needed to determine the extent and cause of LAIs are unavailable. In addition, there is a dearth of science-based insights on prevention of LAIs.
The Blue Ribbon Panel recognizes the need for a voluntary, nonpunitive surveillance and reporting system with the potential for anonymity to be implemented in the United States. Such a system would allow for reporting and evaluation of all LAIs and would potentially lead to training and interventions to facilitate a negligible incidence rate.
1.4. Routes of Laboratory Infection
The five most predominant routes of LAIs are
- parenteral inoculations with syringe needles or other contaminated sharps;
- spills and splashes onto skin and mucous membranes;
- ingestion or exposure through mouth pipetting or touching mouth or eyes with fingers or contaminated objects;
- animal bites and scratches (research laboratories or activities); and
- inhalation of infectious aerosols (1).
The first four routes are relatively easy to detect, but they account for <20% of all reported LAIs (23,24). No distinguishable exposure events were identified in approximately 80% of LAIs reported before 1978 (24–26). In many cases, the only association was that the infected person worked with a microbiological agent or was in the vicinity of a person handling a microbiological agent. The inability to identify a specific event was also reported in a more recent study (27), which found that the probable sources of LAIs were apparent in only 50% of cases. These data suggest that unsuspected infectious aerosols can play a large role in LAIs (1,23,24,28).
1.5. A Culture of Safety
The concept of a "culture of safety," as described in this report, encourages all human and animal diagnostic laboratories to promote an organizational culture of systematic assessment of all work processes and procedures to identify associated risks and implement plans to mitigate those risks. In addition to the often unknown biohazard risk associated with handling diagnostic specimens, each section of the diagnostic laboratory has procedures and processes for handling known infectious agents that convey excessive risk for exposure and possible infection and/or occupational injury. These risks typically are associated with design flaws or lack of or inadequacy of safety procedures and training (1,2). In addition, the day-to-day operations of a human or animal diagnostic laboratory differ markedly from those of an academic or research laboratory and require different biosafety guidelines; these differences prompted the focus of this report on medical laboratory communities, their occupational risks, potential for exposure, and opportunities to mitigate those risks.
Successful establishment of a culture of safety requires that laboratory safety become an integral and apparent priority to the organization, embraced first and foremost by top management and with the concomitant infrastructure support required to foster safe behaviors among its employees (29–31). As required by the Clinical Laboratory Improvement Amendments, the College of American Pathologists, and other accrediting agencies, a laboratory director needs to assume the responsibility for
- establishing and enforcing a policy for a culture of safety within the laboratory;
- identifying as many hazards as possible and specifying practices and procedures that will minimize or eliminate those hazards;
- ensuring that all personnel are instructed in and engaged in performing risk assessments and demonstrating that they can identify laboratory hazards in their individual work environments;
- ensuring that all personnel are trained and competent in the standard practices and techniques that minimize identified workplace hazards;
- providing an avenue for personnel to identify hazards and to present risk-mitigation strategies to management; and
- educating clinicians and nurses about safe specimen procurement and transport to ensure their safety and that of the laboratory personnel who receive the clinical samples.
1.6. Laboratory Design and Architectural Planning for Microbiology
Laboratory design is fundamental to the safety of laboratory workers, hospital staff, and patients. The Clinical and Laboratory Standards Institute document, Laboratory Design; Approved Guideline (32), discusses laboratory design in detail. Because remediating poorly designed laboratory workspace is difficult, or even impossible, design warrants careful planning and consideration of safety issues. The following are suggestions to consider in the design or renovation of the diagnostic laboratory. Although there is no national standard requirement for an amount of space per person working in the laboratory, 300–350 sq. ft/person within a laboratory department is a reasonable figure to provide a safe work area. Ideally, allow a minimum 5-foot space between the worker (at a laboratory chair) and any object behind the worker to provide reasonable maneuverability.
- Design options for the microbiology laboratory should include an enclosed component of the overall laboratory, separated by closable doors from other laboratory sections. Although not required, directional inward airflow from the main laboratory into the microbiology laboratory is also recommended in newly constructed diagnostic laboratories. If the facility is an open design and has no drop ceiling, the microbiology laboratory can have clear glass or Plexiglas walls, which give an appearance of openness but provide a floor-to-ceiling safety barrier from possible aerosol exposures. If a drop ceiling is in place, the clear wall needs to penetrate the deck beyond the ceiling to seal the area. In a previously constructed laboratory without directional room air, the continual operation of biological safety cabinets (BSCs) is encouraged to provide some direction to potential aerosols.
- Directional air is encouraged to provide zones of containment that proceed with increasing negative pressure toward work spaces in which higher-risk laboratory procedures are conducted. Air handling systems within the microbiology laboratory suite must be able to be adjusted and balanced with directional airflow from the corridor into the microbiology laboratory and from the general microbiology laboratory into separate and enclosed tuberculosis, mycology, and virology specialty laboratories.
- For microbiology laboratories, it is critical that the supervisor and laboratory director, along with a biosafety professional, provide input regarding the special needs of a new laboratory facility. Access into the microbiology section must be restricted to staff only. The microbiology section must have a decontamination facility or have a medical waste contract in place, and it must provide a sink for hand washing. Hands-free sinks (foot-pedal operated) are required for biosafety level (BSL)-3 facilities and are recommended for BSL-2 facilities. Bench-tops must be constructed of impervious materials; laminate materials can delaminate and become difficult to disinfect. For BSCs that vent to the outside, air handling should be planned carefully to ensure that the air is vented to the outside after filtration and that the outside vents are placed away from the facility's air intake units. For laboratories that contain multiple classes of BSCs, the hazards that are permitted to be manipulated within the specific unit need to be clearly indicated (by label) to staff (1). The general human and animal microbiology laboratory should be BSL-2.
- If no BSL-3 facilities are available, BSL-2 plus negative airflow and use of respiratory precautions may be used for some agents, provided a risk assessment has been conducted.
- For human laboratories, the separate tuberculosis and virology laboratories that manipulate cultures for identification and characterization would ideally meet BSL-3 requirements. For animal diagnostic virology laboratories in which most manipulated viruses are not human pathogens, the practice is to meet BSL-2 requirements unless a risk analysis indicates a high probability that an agent in a specimen needs BSL-3 containment. Risk assessments should be performed on each facility to include consideration of the specific risks encountered in each laboratory.
- The receiving and set-up areas in microbiology laboratories should be designed with sufficient space to accommodate the greatest number of specimens anticipated. This area needs a Class IIA2 BSC, a sink for hand washing, and an emergency eye wash station. Telephone jacks, computer jacks, and electrical outlets should be built into the module (Use of wireless technologies can reduce the need for telephone or computer wiring in each module.) along with refrigerator space for one or two side-by-side glass-front refrigerators or one double refrigerator to enable easy access by the set-up staff.
- The general laboratory should contain sit-down work spaces designed with adequate space for a computer at each station. Work benches that have storage shelves above the center of the bench might be preferred; these would provide space for supplies without cluttering the work area. Storage shelves need a 1-cm (1/2-inch) lip to ensure chemicals cannot slide off a shelf. Under-shelf lighting is best to illuminate the work area. For convenience, electrical outlets are recommended at each work station, along with telephone and computer jacks. Gas burners are no longer universally recommended.
- If possible, locate carbon dioxide and anaerobic gas tanks outside the actual laboratory (preferably shielded or even installed outside the walls of the building). Placing the tanks outside the laboratory or the building in a locked area will allow easy access for exchange of tanks. Where appropriate, lines that connect gas tanks to specific areas of the laboratory should be made of synthetic tubing to allow future moving if necessary. Accommodations need to be made for daily reading of the gauges in the laboratory unless alarms can be installed. Gas tanks should be individually secured (29).
- If waste will be decontaminated on-site before disposal, the laboratory must have an autoclave large enough to handle its needs. Locate the autoclave in a well-ventilated area, or ensure it is exhausted through a capture hood above it. Ideally, the mycobacteriology laboratory will have its own autoclave. Double-door autoclaves can be installed so that one side opens into the mycobacteriology laboratory and the other side opens into a disposal area used by the laboratory for disposing of other waste. Validation of the autoclave cycles for effective decontamination of the projected loads is recommended in addition to a regular maintenance and quality-assurance program.
- Optimally, the diagnostic laboratory would plan for — a general microbiology laboratory area able to be closed off from the main laboratory, i.e., from other laboratory disciplines; — separate mycobacteriology, virology, and mycology rooms (under negative pressure relative to the general laboratory with a Class IIA2 BSC) with telephone and computer jacks; — adequate space or separate rooms for quality control testing, receipt of supplies, and record storage; and — an extra room for future expansion to offer more services, e.g., molecular or virology testing. The room might need to be renovated to accommodate a Class IIA2 BSC, directional air flow, telephone jacks, and communication devices such as intercoms. The telephone jacks and communication devices should be in all such rooms.
- Ensure that current and future microbiology space is designed for an adequate number of blood culture instruments, automated identification instruments, automated enzyme immunoassays, nucleic acid extraction and testing platforms, and pipetting instruments; refrigerators; automated Gram stainers; automated plate streakers; BSCs; freezers; and additional computer stations for optional use. Some identification instruments require at least 8 feet of footprint space for the unit, printer, and modules. If the laboratory will provide the service, it should plan for a medium-sized anaerobe chamber, about 6 feet of footprint. Risk assessments must include evaluation of the infectious aerosols that might be produced by automated procedural equipment to determine whether containment ventilation is recommended.
- The availability of board-certified laboratory specialists in the laboratory is as important to a medical facility as highly trained, board-certified medical specialists and surgeons. Patients deserve no less if laboratory results are used to guide patient care. Additionally, diplomates of the American Board of Medical Microbiology or the American Board of Medical Laboratory Immunology or equivalent specialists in leadership positions are valuable assets to laboratories that receive and manipulate microbes. Using their skills as laboratory director or as consultant is invaluable and highly recommended. Also, technology specialists should be recruited and retained, particularly in microbiology where interpretive judgment is critical to specimen analysis and ultimately directly affects patient care and outcome.
2. Biological Risk Assessment and Biosafety Guidelines
2.1. Risk Assessment
The laboratory director is ultimately responsible for identifying potential hazards, assessing risks associated with those hazards, and establishing precautions and standard procedures to minimize employee exposure to those risks. Because the identity of an infectious agent is initially unknown in the clinical laboratory, the general recommendation is that the biosafety level (BSL)-2 standard and special practices in Biosafety in Microbiological and Biomedical Laboratories, 5th edition (1) be followed for all work in the clinical laboratory, and the Occupational Safety and Health Administration's (OSHA's) Standard Precautions (gloves, gowns, and protective eyewear) (33) and BSL-2 practices (2) be employed during handling of all blood and body fluids. Other comprehensive resources are available (34,35). Risk assessment, as outlined here and in Section 12, may determine that decreasing or increasing the BSL practices or facilities is warranted (Figure 1).
Qualitative biological risk assessment is a subjective process that involves professional judgments. Because of uncertainties or insufficient scientific data, risk assessments often are based on incomplete knowledge or information. Inherent limitations of and assumptions made in the process also exist, and the perception of acceptable risk differs for everyone. The risk is never zero, and potential for human error always exists.
Identifying potential hazards in the laboratory is the first step in performing a risk assessment. Many categories of microbiological hazards are encountered from the time a specimen is collected until it is disposed of permanently. A comprehensive approach for identifying hazards in the laboratory will include information from a variety of sources. Methods to ascertain hazard information can include benchmarking, walkabouts, interviews, detailed inspections, incident reviews, workflow and process analysis, and facility design.
No one standard approach or correct method exists for conducting a risk assessment; However, several strategies are available, such as using a risk prioritization matrix, conducting a job hazard analysis; or listing potential scenarios of problems during a procedure, task, or activity. The process involves the following five steps:
-
Identify the hazards associated with an infectious agent or material.
-
Identify the activities that might cause exposure to the agent or material.
-
Consider the competencies and experience of laboratory personnel.
-
Evaluate and prioritize risks (evaluate the likelihood that an exposure would cause a laboratory-acquired infection [LAI] and the severity of consequences if such an infection occurs).
-
Develop, implement, and evaluate controls to minimize the risk for exposure.
Standardization of the risk assessment process at an institution can greatly improve the clarity and quality of this process. Training staff in risk assessment is critical to achieving these objectives.
2.1.1. Step 1. Identify the hazards associated with an infectious agent or material.
- The potential for infection, as determined by the most common routes of transmission (i.e., ingestion by contamination from surfaces/fomites to hands and mouth; percutaneous inoculation from cuts, needle sticks, nonintact skin, or bites; direct contact with mucous membranes; and inhalation of aerosols) (Table 1);
- The frequency and concentration of organisms routinely isolated, as determined by specimen type, patient data (of individual or the hospital population), epidemiologic data, and geographic origin of the specimen;
- Intrinsic factors (if agent is known)
— Pathogenicity, virulence, and strain infectivity/communicability;
— Mode of transmission (mode of laboratory transmission may differ from natural transmission);
— Infectious dose (the number of microorganisms required to initiate infection can vary greatly with the specific organism, patient, and route of exposure);
— Form (stage) of the agent (e.g., presence or absence of cell wall, spore versus vegetation, conidia versus hyphae for mycotic agents);
— Invasiveness of agent (ability to produce certain enzymes); and
— Resistance to antibiotics. - Indicators of possible high-risk pathogens that may require continuation of work in a biological safety cabinet (BSC), such as
— Slowly growing, tiny colonies at 24–48 hours with Gram stain showing gram-negative rods or gram-negative coccobacilli;
— Slow growth in blood culture bottles (i.e., positive at ≥48 hours), with Gram stain showing small gram-negative rods or gram-negative coccobacilli;
— Growth only on chocolate agar;
— Rapid growth of flat, nonpigmented, irregular colonies with comma projections and ground-glass appearance;
— Gram stain showing boxcar-shaped, gram-positive rods with or without spores.
2.1.2. Step 2. Identify activities that might cause exposure to the agent or material.
- The facility (e.g., BSL-2, BSL-3, open floor plan [more risk] versus separate areas or rooms for specific activities [less risk], sufficient space versus crowded space, workflow, equipment present);
- The equipment (e.g., in the case of uncertified BSCs, cracked centrifuge tubes, improperly maintained autoclaves, overfilled sharps containers, Bunsen burners);
- Potential for generating aerosols and droplets.
Aerosols can be generated from most routine laboratory procedures but often are undetectable. The following procedures have been associated with generation of infectious aerosols.
— Manipulating needles, syringes and sharps-
Subculturing positive blood culture bottles, making smears
-
Expelling air from tubes or bottles
-
Withdrawing needles from stoppers
-
Separating needles from syringes
-
Aspirating and transferring body fluids
-
Harvesting tissues
-
Flaming loops
-
Cooling loops in culture media
-
Subculturing and streaking culture media
-
Expelling last drop from a pipette (including Eppendorff pipettes)
-
Centrifugation
-
Setting up cultures, inoculating media
-
Mixing, blending, grinding, shaking, sonicating, and vortexing specimens or cultures
-
Pouring, splitting, or decanting liquid specimens
-
Removing caps or swabs from culture containers, opening lyophilized cultures, opening cryotubes
-
Spilling infectious material
-
Filtering specimens under vacuum
-
Preparing isolates for automated identification/susceptibility testing
-
Preparing smears, performing heat fixing, staining slides
-
Performing catalase test
-
Performing serology, rapid antigen tests, wet preps, and slide agglutinations
-
Throwing contaminated items into biohazardous waste
-
Cleaning up spills
-
- Use of animals;
- Use of sharps;
- Production of large volumes or concentrations of potential pathogens;
- Improperly used or maintained equipment;
Examples of possible hazards are decreased dexterity or reaction time for workers wearing gloves, reduced ability to breathe when wearing N95 respirators, or improperly fitting personal protective equipment (PPE). - Working alone in the laboratory.
No inherent biologic danger exists to a person working alone in the laboratory; however, the supervisor is responsible for knowing if and when a person is assigned to work alone. Because assigning a person to work alone is a facility-specific decision, a risk assessment should be conducted that accounts for all safety considerations, including type of work, physical safety, laboratory security, emergency response, potential exposure or injury, and other laboratory-specific issues.
2.1.3. Step 3. Consider the competencies and experience of laboratory personnel.
- Age (younger or inexperienced employees might be at higher risk);
- Genetic predisposition and nutritional deficiencies, immune/medical status (e.g., underlying illness, receipt of immunosuppressive drugs, chronic respiratory conditions, pregnancy, nonintact skin, allergies, receipt of medication known to reduce dexterity or reaction time);
- Education, training, experience, competence;
- Stress, fatigue, mental status, excessive workload;
- Perception, attitude, adherence to safety precautions; and
- The most common routes of exposure or entry into the body (i.e., skin, mucous membranes, lungs, and mouth) (Table 1).
2.1.4. Step 4. Evaluate and prioritize risks.
Risks are evaluated according to the likelihood of occurrence and severity of consequences (Table 2).
- Likelihood of occurrence
— Almost certain: expected to occur
— Likely: could happen sometime
— Moderate: could happen but not likely
— Unlikely: could happen but rare
— Rare: could happen, but probably never will - Severity of consequences
Consequences may depend on duration and frequency of exposure and on availability of vaccine and appropriate treatment. Following are examples of consequences for individual workers.
— Colonization leading to a carrier state
— Asymptomatic infection
— Toxicity, oncogenicity, allergenicity
— Infection, acute or chronic
— Illness, medical treatment
— Disease and sequelae
— Death
2.1.5. Step 5. Develop, implement, and evaluate controls to minimize the risk for exposure.
- Engineering controls
If possible, first isolate and contain the hazard at its source.
— Primary containment: BSC, sharps containers, centrifuge safety cups, splash guards, safer sharps (e.g., autoretracting needle/syringe combinations, disposable scalpels), and pipette aids
— Secondary containment: building design features (e.g., directional airflow or negative air pressure, hand washing sinks, closed doors, double door entry) - Administrative and work practice controls
— Strict adherence to standard and special microbiological practices (1)
— Adherence to signs and standard operating procedures
— Frequently washing hands
— Wearing PPE only in the work area
— Minimizing aerosols
— Prohibiting eating, drinking, smoking, chewing gum
— Limiting use of needles and sharps, and banning recapping of needles
— Minimizing splatter (e.g., by using lab "diapers" on bench surfaces, covering tubes with gauze when opening)
— Monitoring appropriate use of housekeeping, decontamination, and disposal procedures
— Implementing "clean" to "dirty" work flow
— Following recommendations for medical surveillance and occupational health, immunizations, incident reporting, first aid, postexposure prophylaxis
— Training
— Implementing emergency response procedures - PPE (as a last resort in providing a barrier to the hazard)
— Gloves for handling all potentially contaminated materials, containers, equipment, or surfaces
— Face protection (face shields, splash goggles worn with masks, masks with built-in eye shield) if BSCs or splash guards are not available. Face protection, however, does not adequately replace a BSC. At BSL-2 and above, a BSC or similar containment device is required for procedures with splash or aerosol potential (Table 3).
— Laboratory coats and gowns to prevent exposure of street clothing, and gloves or bandages to protect nonintact skin
— Additional respiratory protection if warranted by risk assessment - Job safety analysis
One way to initiate a risk assessment is to conduct a job safety analysis for procedures, tasks, or activities performed at each workstation or specific laboratory by listing the steps involved in a specific protocol and the hazards associated with them and then determining the necessary controls, on the basis of organism suspected (Table 3, Appendix). Precautions beyond the standard and special practices for BSL-2 may be indicated in the following circumstances:
— Test requests for suspected Mycobacterium tuberculosis or other mycobacteria, filamentous fungi, bioterrorism agents, and viral hemorrhagic fevers
— Suspected high-risk organism (e.g., Neisseria meningitidis
— Work with animals
— Work with large volumes or highly concentrated cultures
— Compromised immune status of staff
— Training of new or inexperienced staff
— Technologist preference - Monitoring effectiveness of controls
Risk assessment is an ongoing process that requires at least an annual review because of changes in new and emerging pathogens and in technologies and personnel.
— Review reports of incidents, exposures, illnesses, and near-misses.
— Identify causes and problems; make changes, provide follow-up training.
— Conduct routine laboratory inspections.
— Repeat risk assessment routinely.
2.2. Principles of Biosafety (1)
2.2.1. Containment
"Containment" describes safe methods for managing infectious materials in the laboratory to reduce or eliminate exposure of laboratory workers, other persons, and the environment.
- Primary containment protects personnel and the immediate laboratory environment and is provided by good microbiological technique and use of appropriate safety equipment.
- Secondary containment protects the environment external to the laboratory and is provided by facility design and construction.
2.2.2. Biosafety Levels (Table 4)
BSLs provide appropriate levels of containment needed for the operations performed, the documented or suspected routes of transmission of the infectious agent, and the laboratory function or activities. The four BSLs, designated 1–4, are based on combinations of laboratory practice and techniques, safety equipment (primary barriers), and laboratory facilities (secondary barriers). Each BSL builds on the previous level to provide additional containment. Laboratory directors are responsible for determining which BSL is appropriate for work in their specific laboratories.
- BSL-1 is appropriate for work with agents not known to consistently cause disease in healthy human adults (i.e., laboratories that do not work with disease-causing agents or specimens from humans or animals).
- BSL-2 is appropriate for handling moderate-risk agents that cause human disease of varying severity by ingestion or by percutaneous or mucous membrane exposure (i.e., human and animal clinical diagnostic laboratories).
- BSL-3 is appropriate for work with indigenous or exotic agents that have a known potential for aerosol transmission and for agents that can cause serious and potentially fatal infections (e.g., tuberculosis laboratories).
- BSL-4 is reserved for work with exotic agents that pose a high individual risk for life-threatening disease by infectious aerosols and for which no treatment is available (e.g., laboratories working with Ebola, Marburg, and pox viruses). These high-containment laboratories have complex and advanced facility requirements.
2.3. Material Safety Data Sheets for Organisms and Chemicals
Material Safety Data Sheets (MSDS) for chemicals are available from the manufacturer, supplier, or an official Internet site. The Division of Occupational Health and Safety, National Institutes of Health, has promulgated guidelines for handling genetically manipulated organisms and has additional instructions for accessing MSDS (http://dohs.ors.od.nih.gov/material_safety_data_main.htm).
2.4. Biosafety Manual
- The laboratory director is responsible for ensuring that a laboratory-specific biosafety manual is developed, adopted, annually reviewed, and accessible to all laboratory personnel. All laboratory employees must read this manual, and the director must maintain records of personnel who have read it.
- The manual should be reviewed and updated annually and whenever procedures or policies change. Annual training in biosafety practices is recommended for all personnel who access the laboratory. Recommended topics include the following.
— Institutional and laboratory safety policies
— Management, supervisor, and personnel responsibilities
— Regulations and recommended guidelines
— Routes of exposure in the laboratory
— Risk assessment and reporting of exposures
— Biosafety principles and practices
— Standard precautions for safe handling of infectious materials
— Standard operating procedures
— Hazard communication and biohazard signs
— Engineering controls
— Administrative and work practice controls
— PPE
— When and how to work in a BSC
— Transport of biohazardous materials
— Emergency procedures
— Decontamination and disposal of biohazardous waste
— Training program and documentation
— Medical surveillance and exposure evaluation procedures
3. Fundamental Safety Practices in Diagnostic Laboratories
Many safety procedures, guidelines, and principles apply to all sections of the diagnostic laboratory. The recommendations presented in this section represent a broad view of safety throughout the laboratory. More detailed recommendations can be found in Biosafety in Microbiological and Biomedical Laboratories (BMBL-5) and in the World Health Organization's Laboratory Biosafety Manual (1,36).
Hospitals, clinical laboratories, state and local health departments, CDC, and the American Society for Microbiology have established and/or published guidelines to follow when suspected agents of bioterrorism have been or could be released in the community. However, routine clinical laboratory testing may provide the first evidence of an unexpected bioterrorism event. Routine clinical specimens also may harbor unusual or exotic infectious agents that are dangerous to amplify in culture. These agents are often difficult to identify, and the routine bench technologist might continue work on the culture by passage, repeated staining, nucleic acid testing, neutralization, and other methods. This continued workup places the technologist and others in the laboratory at risk for infection. Ideally, these specimens are not to be processed or tested in the routine laboratory, and they can be removed from the testing stream if the suspected agent is known. Relationships with the state public health laboratory, and subsequently with the Laboratory Response Network, are critical in this effort.
Once the testing process has begun, the bench technologist must have clear and concise instructions about when to seek assistance from the laboratory supervisor and/or director.
3.1. Specimen Receiving and Log-In/Setup Station
- Microbiology specimens are to be received in uncontaminated containers that are intact and are consistent with laboratory specimen collection policy.
- Use of pneumatic tubes for transport of specimens is acceptable for most specimens but might be contraindicated for specimens without sealed caps, such as urine cups; these are to be delivered by hand (see 3.1.6). Adopt specific standard operating procedures (SOPs) in the event that irreplaceable specimens are considered for transportation using these systems.
- Ideally, all specimens in a biosafety level (BSL) 2 or higher facility are to be processed in a biological safety cabinet (BSC) adhering to safe BSC practices. If a BSC is unavailable in the laboratory, the laboratorian processing intake specimens must wear a laboratory coat and gloves, employ an effective splash shield, and continue to follow universal precautions. Additional precautions may be necessary if warranted by site-specific risk assessments.
- Limit the use of a 4-foot-wide BSC for inoculating plates and preparing smears to one employee at a time, wearing appropriate personal protective equipment (PPE). Six-foot-wide BSCs may accommodate additional testing equipment or materials. Check the manufacturer's recommendations before allowing two employees to work simultaneously in the larger cabinet, and then allow only after a risk assessment.
- Minimal PPE for the general setup area is gown and gloves. In microbiology, a surgical-type mask is recommended, but optional if the BSC is used. For mycobacteriology and virology laboratories where organism manipulation is conducted, workers should wear a fit tested N95 respirator or select other appropriate respiratory protection, as indicated by the risk assessment. An N95 respirator is usually not required for biocontainment levels up through BSL-2, although it provides a higher degree of protection than a surgical mask. Safe BSC practices are to be adhered to at all times. Mycobacterial, fungal, viral, and molecular specimens may require specific additional safeguards.
3.1.1. Leaking containers
- Submit specimens to the laboratory in transport bags that isolate the patient requisition from specimens; always limit bags to one patient to prevent misidentification and cross-contamination.
- Request a new specimen if a container is broken or has spilled its contents. These containers are unacceptable for culture because the contents may have become contaminated. Document the incident, and notify the supervisor if an exposure occurred.
- Visually examine containers for leaks upon arrival and before placing on rockers, in centrifuges, in racks, in closed-tube sampling (cap-piercing probe) systems, in automated aliquot stations or automated slide preparation systems, or on conveyor belts.
- Track and document all incidents of cracked tubes, loose caps, and leaking containers. Increases in documented events may indicate the need to clarify or strengthen specimen acceptance policies or improve specimen collection or transportation practices, or they might identify defective container lot numbers.
- Consider all sputum containers as coming from patients with tuberculosis or pneumonia, and handle with care. External contamination caused by inappropriate lid closure can contaminate the gloves of the laboratorian and all contents of the BSC. If the specimen is leaking or contaminated, consider rejecting it and requesting another specimen if feasible. Change and discard gloves after disinfection and cleanup. (A 1:10 bleach solution or appropriate disinfectant is recommended.) Document the external contamination for reporting purposes.
- Consider all blood culture bottles as coming from patients potentially infected (e.g., with human immunodeficiency virus [HIV] or hepatitis), and handle appropriately. If any concern exists about external contamination, carefully disinfect the outside of the tubes or bottles before inserting them into the blood culture instruments. Change and discard gloves after cleanup and decontamination of the immediate area. Document the external contamination for reporting purposes.
- Leaking stool containers can be a hazard to the technologist, could contaminate the laboratory, or could present an opportunity for specimen comingling and/or contamination that could produce a false result. These should be rejected, if feasible, and a new specimen requested. Otherwise, disinfect the outside of the container before culturing the contents, and change and discard gloves before proceeding. Document the external contamination for reporting purposes.
- Viral specimens with damaged or leaking containers may need to be discarded before opening. Contact the supervisor for instructions on whether or not to continue processing, and be prepared to notify the submitter and request another specimen.
3.1.2. Visible contamination of the outside of containers
- Consider all specimen containers as potentially contaminated.
- Do not rely on visible external contamination to confirm the potential source of contamination.
- Wipe off visible contamination by using a towel or gauze pad moistened with acceptable decontaminant, such as a 1:10 dilution of household bleach, or use the established laboratory disinfectant. Ensure label and bar code are not obscured before advancing specimen for analysis.
3.1.3. Loose caps
- Always grasp the tube or outside of the specimen container, not the stopper or cap, when picking up tubes or specimen containers to prevent spills and breakage.
- Ensure tops are tightly secured on all specimen containers, blood-collection tubes, and sample tubes before advancing for analysis or storage.
3.1.4. Operational procedures
- Ensure that specimen placement, specimen flow, and bench operational workflow are unidirectional (i.e., from clean areas to dirty areas) and uniform for all operators to maximize effective use of engineering controls.
- Determine appropriate PPE on the basis of documented risk and hazard assessments of all the operations performed at each bench. Try to incorporate engineering controls and PPE information in the same location in all procedure manuals, and clearly post the information for each operation carried out at the bench.
- Ensure that workstation procedure manuals include instructions for the organization of all instruments, materials, and supplies in each area as well as instructions for any cleaning and disinfection and the frequency of cleaning and disinfection for all surfaces and instruments.
- Supervisors are to routinely inspect for cleanliness of the bench.
- Have written procedures for nonlaboratory operations, e.g., technical instrument maintenance, in-house or contracted maintenance, emergency response, housekeeping, and construction and utility operations, to mitigate exposures associated with assigned operational tasks.
— Write nonlaboratory operation procedures for nonlaboratory service providers with their input and consultation.
— Document the training and assess the competency of service providers and bench operators for all written nonlaboratory operational bench procedures. - General bacteriology stains may constitute both a chemical or biological hazard.
— Gram stain. Crystal violet, methylene blue, potassium iodide, and ethanol are all irritants; crystal violet is also carcinogenic and somewhat toxic; ethanol is a hepatotoxin.
— Other risks associated with Gram stain procedures include handling live organisms, the potential for creating aerosols, and the potential for skin and environmental contamination.
— Eye protection (safety glasses or chemical splash goggles) and disposable gloves are recommended during staining or preparing stains. Gloves provide protection from the live organisms as smears are prepared and provide protection from unintentional exposure to stain.
— Place contaminated waste in a biohazard bag for disposal. Use biohazard bags only once and then discard them. Never wash or reuse them.
— For all other stains, including fluorescent conjugates, refer to the Material
Safety Data Sheets associated with each stain or chemical. - Equipment decontamination. Examine equipment contaminated with blood or other potentially infectious materials before servicing or shipping, and decontaminate as necessary. Contact the manufacturer for decontamination process.
- If decontamination of equipment or portions of such equipment is not feasible, do the following.
— Label the equipment with a biohazard symbol and a second label specifically identifying which portions remain contaminated.
— Convey this information to all affected employees and servicing representatives before handling, servicing, or shipping so that appropriate precautions will be taken.
3.1.5. Manual removal of sealed caps; specimen aliquotting and pipetting
- Always remove caps behind a bench-fixed splash shield, or wear additional PPE appropriate to protect from splashes and aerosols.
- Place a gauze pad over the cap, and then slowly pry or push the cap off with an away-from-body motion. Never reuse a gauze pad; doing so might contribute to cross-contamination. Several manufacturers market safety devices to help remove caps from tubes and to break open ampoules (e.g., Current Technologies Saf De-Cap [Fisher Health Care, Houston, TX] and the Pluggo [LPG Consulting, Inc., Wood River, IL]).
- Use automated or semiautomated pipettes and safety transfer devices.
3.1.6. Pneumatic tube systems
- Establish SOPs for use and decontamination of the pneumatic tube system (PTS).
- Breakage or leakage of specimens transported using a PTS risks contamination of the transport system itself.
- Base limitations on use of the PTS on a complete risk/hazard assessment. Limit specimen size, volume, weight, and container types sent through the tube system, if warranted. This applies particularly to cytology specimens and certain types of urine containers.
- Place all specimens sent through a PTS in a sealed zip-lock bag.
- Test bags, and ensure they are leakproof under the conditions in the PTS.
- Protect requisition forms by a separate pouch, or enclose them in a separate secondary bag to prevent contamination.
- A zip-lock bag must contain specimens from only one patient.
- Place absorbent wadding between patient bags to help absorb spills and minimize contamination to the outside of the carrier.
- Handle contaminated pneumatic tube carriers in accordance with standard precautions.
- Disinfect contaminated carriers with bleach solution or other disinfectant following the protocol recommended by the manufacturer and approved by the hospital's infection control committee if the system is in use in a hospital.
- Wear gloves when opening PTS carriers containing patient specimens.
- Decontaminate the outside of tube carriers before returning them to patient-care areas. Decontaminate the inside of the carrier if a leak occurs in the specimen container.
- Establish a facility hotline for immediately reporting problems with the PTS.
- Establish an emergency PTS shutdown plan, including roles and responsibilities; include implementation of an alternative specimen transport plan.
- Develop a system to track and analyze incidents of improperly closed carriers, cracked tubes, loose caps, and leaking containers. Increases in documented events may indicate the need to clarify or strengthen PTS-use policies or improve specimen collection practices, and could identify defective carriers and/or container lot numbers.
- Prepare SOPs for both laboratory operators and the nonlaboratory service providers with their input and consultation.
- Document training and competency assessment of service providers and bench operators for PTS maintenance and decontamination procedures. Documented training and assessment of competency will include knowledge of the risks associated with using a PTS and the precautions to be taken to control those risks.
3.2. Personal Precautions.
If engineering controls are in place to prevent splashes or sprays, the requirement for PPE can be modified on the basis of a risk assessment and evidence of the effectiveness of the engineering control to prevent exposure from splashes or sprays. Examples of engineering controls include use of a BSC, having sealed safety cups or heads in centrifuges, and negative air flow into the laboratory.
3.2.1. Work at the open bench
- Because no two workstations are identical, written procedures for each clinical laboratory workstation must include specific work practices and work practice controls to mitigate potential exposures.
- Install a dedicated handwashing sink with hot water in each work area for use after contamination of hands or gloves with blood or other potentially infectious materials. Employees cannot rely solely on a sink in a rest room for washing their hands after work in a technical area. Frequent hand washing is essential. Supply each workstation with alcohol hand rub to facilitate frequent hand cleaning, and with absorbent work pads to contain accidental spills. Make safety glasses, splash shield, respiratory protection, and gloves available for use and when determined necessary by the type of isolate, as described in BMBL-5 (1).
- In the general microbiology laboratory, masks and disposable gloves are not required in the open laboratory but may be voluntarily used. If gloves are used, they can easily become contaminated during routine use; therefore, gloves are not to be washed and reused. Discard gloves, and don a new pair when leaving the workstation.
- Splash guards at workstations are recommended during work at the blood culture bench or at any station at which the potential for splashing exists.
- Notify nearby workers and the supervisor if a splash or spill occurs, regardless of how small.
- Sniffing of bacterial cultures growing on artificial media (to detect characteristic odors supposedly emitted by certain bacteria) is a potentially unsafe laboratory practice that has been associated with multiple types of LAI.
(http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5342a3.htm,
http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5702a2.htm,
http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5702a3.htm, and
http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5532a1.htm)
CDC continues to recommend that sniffing culture plates should be prohibited. Isolates of small gram-negative or gram-variable rods (e.g., gram-negative coccobacilli) should be manipulated within a BSC.
- Do not use open flame burners anywhere in the laboratory. Use disposable loops and needles or use electric incinerators for metal wire devices.
- Locate disinfectant-containing discard containers and sharps containers within easy reach of the work station.
- Use protective covers for computer keyboards at workstations; covers need to be easily cleanable and routinely disinfected along with the bench top, at least at the end of the work shift.
- Place blood culture bottles behind a safety splash shield or in a BSC when tapping with a needle. Gram-negative coccobacilli from blood culture bottles are to be handled within a BSC. Laboratories without the ability to determine or rule out Brucella or Francisella (gram-negative coccobacilli) should consider directly shipping these isolates to a reference laboratory and not try to isolate and identify them.
- Urine remaining from culture activities can be discarded down the sink drain or into the sanitary sewer.
- Discard feces and other specimens such as body fluids and respiratory specimens remaining from culture activities with medical waste, and autoclave if warranted by risk assessment.
- Discard tissue remaining from culture activities of BSL-3 infectious agents into medical waste, and autoclave it.
3.2.2. Personal protective equipment
Engineering controls (2.1.5. Step 5) should always be the first line of defense to minimize exposures.
PPE includes a variety of items, such as gloves, laboratory coats, gowns, shoe covers, boots, respirators, face shields, safety glasses, and goggles, that are designed to protect the laboratory worker from exposure to physical, biological, and chemical hazards. Distributing PPE to each employee as needed helps to ensure access to appropriate PPE.
PPE is often used in combination with BSCs and other devices that contain the agents or materials being handled. In some situations where working in a BSC is impractical, PPE, including splash shields, may form the primary barrier between personnel and hazardous materials (1). (See Section 3.1).
The Occupational Safety and Health Administration (OSHA) defines PPE as "appropriate" if it does not permit blood or other potentially infectious materials to pass through or reach the employee's street clothes, undergarments, skin, eyes, mouth, or other mucous membranes under normal conditions of use (33).
- Sources for PPE standards
— American Society for Testing and Materials (ASTM [now known as ASTM International]) — laboratory coats, hand protection (disposable gloves)
— American National Standards Institute (ANSI) Z87.1-2003 (or earlier ANSI consensus standards) (USA Standard for Occupational and Educational Eye and Face Protection) — eye and face protection.
— Food and Drug Administration (FDA) — hand protection (gloves).
— OSHA–appropriate use of PPE, hand protection, employee training. - Laboratory coats
— Protective laboratory coats, gowns, or uniforms are recommended to prevent contamination of personal clothing. Remove protective clothing before leaving for nonlaboratory areas (e.g., cafeteria, break room, administrative offices). Dispose of single-use protective clothing with other contaminated waste or deposit reusable clothing for laundering by the institution.
— Do not take laboratory clothing and other PPE home for laundering or other uses. The employer must provide laundry service for reusable protective laboratory coats, gowns, uniforms, or scrubs that are potentially or visibly contaminated with blood or other potentially infectious materials at no cost to the employee. - Hand protection
— No ANSI standard exists for gloves, but ASTM standards for disposable gloves are based on the specific type of material with which the glove is made. FDA has indicated that patient examination gloves used during patient care and vascular access procedures meet its adulteration requirements and have a 510(k) medical device registration with this agency. OSHA recommends that selection be based on the tasks performed and the performance and construction characteristics of the glove material. Disposable gloves must be made available in a variety of sizes to ensure that employees are able to select the size that best fits their hands. Provide disposable gloves made of different materials (e.g., nitrile, chloroprene) for employees who have skin sensitivity to either the type of glove material or the accelerants or other chemicals used in the glove manufacturing process.
— Evaluate the employee medical history for evidence of a latex allergy if latex gloves are used in the laboratory.
— Using the hazard assessment for a given operation, laboratory management or an assigned safety officer or safety team should select the most appropriate glove for the task and establish how long it can be worn.
— Before purchasing gloves, laboratory management or an assigned safety officer or safety team should request documentation from the manufacturer that the gloves meet the appropriate test standard(s) for the hazard(s) anticipated. - Eye and face protection
— Eye and face protection (goggles, mask, face shield, or other splatter guard) must be used whenever a splash or spray event could occur. This includes opening containers and pipetting, manipulating, aliquoting, or testing specimens, cultures, biological agents, or other hazardous materials outside the BSC.
— If eye and face protection becomes contaminated, these devices must either be decontaminated before reuse or disposed of with other contaminated laboratory waste.
— Neither eyeglasses nor contact lenses are considered PPE. Laboratory workers who wear contact lenses must use face protection as described above. For those who need corrected vision, wear prescription safety glasses with side shields in the laboratory. In a chemical splash, contact lenses can intensify eye damage because the lens will hold the chemical against the eye for a longer period.
— Surgical masks are not respiratory PPE. - Employee training
— Employers are required by OSHA to train employees to know at least the following (37).-
When PPE is necessary
-
What PPE is necessary
-
How to properly put on, take off, adjust, and wear PPE
-
Limitations of PPE
-
Proper care, maintenance, useful life, and disposal of PPE
-
3.3. Biological Safety Cabinet
- The Class II-A1 or II-A2 BSC is best suited and recommended for the diagnostic laboratory (Table 5) (1). An overview and summary of the different classes and types of BSCs is available in Appendix A of BMBL-5 (1).
- Every diagnostic microbiology laboratory needs one or more BSCs as a primary means of containment for working safely with infectious organisms. The College of American Pathologists requires a BSC in the microbiology laboratory. The lack of a BSC is a Phase II deficiency for microbiology departments that handle specimens or organisms considered contagious by airborne routes. The three basic types of BSCs are designated as Class I, Class II, and Class III.
— The Class I cabinet is similar to a chemical fume hood and is usually hard-ducted to the building exhaust system. It protects personnel and the room environment but is not designed to protect the product inside the cabinet. The Class I BSC could be used in the general laboratory setup area as a second choice of cabinet.
— For most diagnostic laboratories where volatile chemicals and toxins will not be manipulated within the cabinet, the Class II-A2 BSC would be appropriate and easiest to install without a hard duct to the outside. This cabinet can be used at the specimen-processing station; in the mycobacteriology, mycology, and virology laboratories; and in chemistry and hematology if needed. Air can be recirculated back into the room through high-efficiency particulate air (HEPA) filters with little risk if the cabinet is maintained properly and certified annually. The A-1 or A-2 BSC in the mycobacteriology laboratory is also an option with a thimble connection to a building exhaust duct and annual certification. Never hard-duct the Class A BSC to the building exhaust system because building airflow patterns cannot be matched to the cabinet. HEPA filters remove at least 99.97% of 0.3-µm particles, which include all bacteria, viruses, and spores and particles or droplets containing these organisms.
— The Class III cabinet is designed for highly infectious agents, such as Ebola virus and monkey pox virus. - All BSCs must be certified by trained professionals in accordance with Annex F of ANSI/NSF Standard No. 49, at least annually and each time the unit is moved. Moving the cabinet can damage the filter at the glue joint or at the gasket, resulting in dangerous leaks, so filter and cabinet integrity must be tested after each move.
- Proper loading of the BSC and proper access by the laboratorian are described in BMBL-5. Some basic rules are important to highlight.
— Do not sweep your arms into or out of the cabinet. Move arms in and out slowly, perpendicular to the face opening.
— Install the BSC in the laboratory away from walking traffic, room fans, and room doors.
— Do not block the front grill where downflow of air is conducted, or the rear grill where air is removed from the cabinet.
— Let the blowers operate at least 4 minutes before beginning work to allow the cabinet to "purge."
— At the beginning and end of the day, with the blower running, disinfect all surfaces with a 1:10 dilution of household bleach, and remove residual bleach with 70% alcohol, or use another disinfectant appropriate for the organisms encountered.
— Do not use open flames inside the cabinet. First choice: disposable loops; second choice: electric furnaces.
— To decontaminate the BSC before maintenance, engage a BSC certification technician to use either formaldehyde gas, hydrogen peroxide vapor, or chlorine dioxide gas when the BSC is not in use.
— Ultraviolet (UV) lamps are not required in BSCs and are not necessary.
— Open sealed rotors or safety cups on high-speed and ultracentrifuges in a BSC, particularly when respiratory pathogens are manipulated.
— Where safety cups or sealed rotors cannot be used, place centrifuges in a containment device or BSC designed for this purpose.
— Collect medical waste generated inside the BSC in bags or sharps containers. Seal these before removal and place in medical waste containers outside the BSC. - If a person who works at a BSC has an infection that may have involved material manipulated in the cabinet, such as a tuberculin skin test conversion or positive results for a TB interferon gamma release assay in a person working with
Mycobacterium tuberculosis, an evaluation must be performed that includes:
— evaluation and, as needed, repair and recertification of the BSCs in which the implicated work was performed;
— evaluation of procedures to ensure the worker was using proper technique in the BSC and, if needed, reeducation of the worker on proper BSC technique; and
— evaluation (e.g., tuberculin skin testing) of others in the laboratory who work at the same BSCs and, as needed, reeducation of these persons on proper BSC technique.
3.4. Disinfection
3.4.1. Good work practices
- Regardless of the method, the purpose of decontamination is to protect the laboratory worker, the environment, and any person who enters the laboratory or who handles laboratory materials that have been carried out of the laboratory. For detailed information see BMBL-5 Appendix A (1).
- Instructions for disinfecting a laboratory work bench are to be a part of each SOP and must include what PPE to wear, how to clean surfaces, what disinfectant to use, and how to dispose of cleaning materials. Contact time is a critical and necessary part of the instructions. Post the instructions in the bench area for easy reference.
- Routinely clean environmental surfaces before setting up work areas and again before leaving work areas.
- Clean any item (e.g., timer, pen, telephone, thermometer) touched with used gloves.
- Do not use alcohols or alcohol-based solutions alone to disinfect surface areas. These evaporate readily, which substantially decreases efficacy. Use disinfectants recommended for environmental surfaces, such as Environmental Protection Agency (EPA)–registered disinfectants effective against hepatitis B virus, HIV, and other bloodborne pathogens, or use a 1:10 dilution of household bleach. EPA environmental disinfectant product registration information is available at http://www.epa.gov/oppad001/chemregindex.htm.
- Reserve sterilants and high-level disinfectants cleared by FDA for processing reusable medical devices. FDA has identified manufacturers, active ingredients and contact conditions for these products. FDA-cleared sterilants and high-level disinfectants lists are available at http://www.fda.gov/cdrh/ode/germlab.html.
- Clean bench surfaces, stationary racks, clay tiles, rockers, slide staining racks, water/heating baths and all trays whenever a spill occurs. Clean all surfaces at the end of each shift.
- Use of disposable liners may reduce cleaning intervals of the equipment but does not replace the need to clean surface areas or equipment. Clean the underlying bench surface whenever the liner is discarded. The liner must be disinfected or discarded at the end of each shift or if contaminated.
- Disposable, flexible, polyethylene film–backed, nonskid highly absorbent surface liners are available commercially and help to prevent soak-through of most solutions, including dyes and corrosive chemicals. Always discard with medical waste after contamination and at the end of the shift.
- Allow dried blood or body fluid at least 20 minutes' contact with the laboratory-specified decontaminating solution to allow permeation and easy removal (1). Never use a knife or other instrument to scrape dried blood or body fluid from surface areas; doing so can cause percutaneous injury or generate aerosols.
3.4.2. Bleach solutions (sodium hypochlorite) (38)
- Hypochlorite solutions are classified as irritants and corrosives. Undiluted bleach solution is corrosive to stainless steel, and thorough rinsing must follow its use in the BSC and stainless steel sinks to remove the residue. Do not autoclave bleach solutions.
- Never mix different chlorine solutions or store them with cleaning products containing ammonia, ammonium chloride, or phosphoric acid. Combining these chemicals could result in release of chlorine gas, which can cause nausea, eye irritation, tearing, headache, and shortness of breath. These symptoms may last for several hours. A worker exposed to an unpleasantly strong odor after mixing of a chlorine solution with a cleaning product should leave the room or area immediately and remain out of the area until the fumes have cleared completely (see Section 9.1).
- To be an effective disinfectant, working bleach solutions must contain >0.5% but <2% sodium hypochlorite. Hypochlorite concentration in household bleach varies by manufacturer. Many household bleach solutions contain 5.25% sodium hypochlorite, and a 1:10 dilution (5,000 ppm Cl) will produce a 0.53% hypochlorite solution. Use of bleach solutions with lower hypochlorite concentrations might not provide the proper level of disinfection. Each day, prepare a fresh 1:10 household bleach solution.
3.5. Waste Management
A clinical laboratory must establish a waste management plan.
- As part of an on-site waste management plan, the responsibilities of the laboratory management or the designated safety officer or safety team are to
— establish a waste-reduction or minimization program;
— identify and define all categories of waste generated by the laboratory;
— for each category of waste generated, determine applicability of federal, state, and local regulations, including how that category of waste will be segregated, packaged, labeled/color-coded, stored, transported, and tracked within the laboratory, outside the laboratory, and outside the facility to comply with the applicable regulations;
— segregate all regulated waste to prevent access by the public or clients;
— establish a system for reporting and responding to all issues or problems regarding medical waste management; and
— establish treatment and disposal processes (39). Disposal of regulated waste must be by a company meeting state and local licensure requirements.
3.5.1. Decontamination of medical waste before transport and disposal
- "Infectious medical waste" is defined as waste capable of transmitting disease. "Regulated medical waste" is any waste contaminated with substantial amounts of blood or blood products in liquid or semiliquid form or with contaminated sharps. It is considered to confer a higher level of risk, thus warranting regulatory provisions by state or local authorities.
- Clinical laboratories must determine the federal, state, and local laws governing their organization's regulated medical waste and ensure that the organization is in compliance with those laws. State and/or local regulations may require
— permits or registration numbers to generate medical waste;
— development and implementation of a waste management plan; and/or
— specific recordkeeping compliance. - State departments of environmental services (or equivalent) are an excellent resource for assistance in complying with state and local medical-waste laws. To find state laws governing medical waste, visit http://www.epa.gov/epawaste/nonhaz/industrial/medical/programs.htm. Choose the state, then look under the "Primary Materials–Cases, Codes and Regulations." Search the state's "Administrative Codes" or "Statutes" for information about waste management. (Some states use other terms for "infectious medical waste," such as "regulated medical waste" or "special waste.")
- OSHA. 29 CFR Part 1910.1030, Occupational Exposure to Bloodborne Pathogens, provides minimal requirements for labeling and packaging of blood and body fluids when transported or outside a laboratory. Information may be obtained from the local OSHA office or online (33).
- Laboratory management must ensure that employees understand these laws and ensure regulated medical waste is not mixed with nonmedical waste in a facility.
- Document completion of employee training and competency assessment for
— constructing and properly labeling containers for medical waste that require assembly before their use;
— disposing of medical waste in properly labeled containers;
— use of appropriate supplies, e.g., containers, appropriate plastic bags, labeling; and
— following all federal, state, and local regulations regarding waste management, i.e., handling of medical waste, immediate disposal of medical waste, storage of medical waste, transportation of medical waste, which includes any required Department of Transportation labeling (e.g., the word "Biohazard" and the universal biohazard symbol) of transport containers, and final disposal of medical waste.
3.5.2. Management of discarded cultures and stocks
- The laboratory's biosafety level must be considered when discarding cultures and stocks of infectious agents.
- Discarded cultures and stocks of organisms handled under BLS-3 physical containment (e.g., M. tuberculosis) are to be collected and sealed in containers that are closed, leakproof, and posted with the universal biohazard symbol and the word "Biohazard." The containers subsequently need to be autoclaved on-site. Use of other on-site medical waste treatment technologies can be considered if these technologies sterilize the organisms, if they have been properly validated, and if they are recognized as medical waste treatment technologies by the appropriate state environmental regulatory agency.
- Decontaminate discarded cultures and stocks of organisms handled at BSL-2. If this process is done on-site but remote from the microbiology department, place the discarded cultures and stocks into durable, leakproof containers that are secured when they are moved. Decontamination may be done by a medical waste treatment contractor's facility if the waste is placed into medical waste shipping containers and packaged in accordance with applicable regulatory standards. To determine whether these activities can be done in a manner that minimizes possible exposures, conduct a risk assessment. The assessment will determine whether these wastes can be safely managed off-site or should be managed on-site.
3.5.3. Discarding a select agent
- Clinical or diagnostic laboratories and other entities that have identified select agents or toxins contained in a specimen presented for diagnosis or verification are required by regulation (7 CFR 331, 9 CFR 121, and 42 CFR 73) to report the identification within 7 calendar days to the Animal and Plant Health Inspection Service (APHIS) of the U.S. Department of Agriculture or to CDC. In addition, these laboratories or entities are required to report the identification of select agents and toxins from samples received for proficiency testing within 90 days after receipt of the sample.
- Disposal of cultures containing identified select agents such as Brucella spp., Coccidioides immitis, or Yersinia pestis, whether identified in the local facility or by a reference laboratory, falls under the Select Agent Rule (40). The APHIS/CDC Form 4, "Report of the Identification of a Select Agent or Toxin," is used by clinical or diagnostic laboratories and other entities to notify APHIS or CDC of the identification of a select agent or toxin as the result of diagnosis, verification, or proficiency testing, and of the final disposition of that agent or toxin. No further reporting is necessary if the isolate is destroyed within 7 days after identification or shipped to a registered laboratory and CDC is notified of the disposition of the isolate.
- A select agent or toxin can be destroyed by on-site autoclaving. If a medical waste contractor is used for the facility, the cultures containing the identified agent or toxin must first be inactivated by completely immersing the open culture containers in a fresh 1:10 bleach solution overnight before discarding them into medical waste. If the medical waste contractor is registered with the Select Agent Program, the live cultures may be formally transferred to the contractor by using APHIS/CDC Form 2, "Request to Transfer Select Agents and Toxins." Details on the select agent rule and its impact on clinical laboratories can be found in the Clinical Microbiology Newsletter, April 15, 2006 (41).
3.5.4. Autoclave safety
- Gravity displacement steam sterilizers (autoclaves) are frequently used in microbiology (including virology) laboratories. Autoclaves generate substantial heat and pressure, and all users must understand and respect the associated risks.
- Personnel who operate the autoclave must be trained to package, load, and label materials to be autoclaved in accordance with the procedures used to validate the sterilization cycle of the unit. They must also receive training in emergency procedures.
- Do not touch the sides or back of older autoclaves; they have little or no heat shielding and may cause burns.
- Do not stack or store combustible materials (e.g., cardboard, plastic materials) or flammable liquids next to the autoclave.
- Never autoclave materials that contain toxic agents, corrosives (e.g., acids, bases, phenol), solvents or volatiles (e.g., ethanol, methanol, acetone, chloroform), or radioactive materials.
- Place all biomedical waste to be autoclaved in an approved, biohazard-labeled autoclave bag before autoclaving. Not all red or orange bags are capable of being autoclaved. Bags selected for use in autoclaving waste must be specifically manufactured for this purpose. Use only bags designated as appropriate for use in autoclaves when autoclaving medical waste.
- Place all sharps (e.g., needles, scalpels, pipettes, or broken glass) into an approved, leak-resistant, labeled, and rigid sharps container before sterilizing.
- When decontaminating a bag of dry goods, such as bench paper or paper gowns, place 100 mL of water into the autoclave bag to facilitate steam production within the bag.
- Do not overfill bags or the autoclave unit; this might result in inadequate steam circulation, which could interfere with the sterilization process.
- Close autoclave bags loosely with twist ties or other means that allow steam inside.
- Place bags onto stainless steel or polypropylene trays for autoclaving. Do not place bags directly into the autoclave.
- Always allow an autoclave unit to cool before opening. Stand back and open the door slowly to allow the excess steam to escape. Allow the contents to cool before handling. Always use thick, elbow-length, heat-resistant, liquid-impervious gloves to remove hot items from the autoclave.
- After autoclaving, check the autoclave indicator tape to be sure the bars are black. If the indicator tape is not activated, resterilize the load.
- At least weekly, use a biological indicator such as Bacillus stearothermophilus spore strips (or equivalent) to ensure the autoclave is performing properly. Establish and follow a regular maintenance schedule for this equipment that evaluates seals, drains, and other critical aspects.
3.6. Dry Ice
3.6.1. General information
Under certain circumstances, dry ice can be an explosion hazard. Dry ice is solidified carbon dioxide (CO) and it is extremely cold (-109° F [-79° C]). Unlike water-ice, dry ice sublimates (changes directly from solid to gas) as it warms, releasing CO gas. CO vapor is considerably heavier than air; in confined, poorly ventilated spaces, it can displace air, causing asphyxiation.
- Avoid dry ice contact with skin and eyes. Dry ice can cause severe frostbite within seconds of direct contact.
- Never place dry ice into glass or sealed containers. Storage in a sealed container can cause the container to rupture or explode from overpressurization.
- Never handle dry ice with bare hands. Always wear insulated gloves and safety glasses. Use of laboratory coats is also recommended. Use tongs to handle blocks of dry ice. Use scoops to move pelletized dry ice.
- Do not put dry ice into the mouth or otherwise ingest it. If ingested, dry ice can cause severe internal injury. Never put dry ice in beverages to cool them.
- When transporting dry ice, place the container in the trunk of the car or truck bed, and leave the car windows open for fresh air circulation. Never leave dry ice in a parked passenger vehicle. Sublimation of dry ice in a closed passenger vehicle can result in accumulation of dangerous concentrations of asphyxiating CO vapor. When opening a closed cargo area containing dry ice, allow the closed space to ventilate for 5 minutes before entering.
- Do not place dry ice directly on bench tops, tile, laminated countertops, or ceramic sinks. Use an insulating barrier such as double-thickness cardboard or wood. Dry ice can destroy the bonding agent holding the tile or laminated material in place. Dry ice can also cause bench tops and ceramic sinks to crack.
3.6.2. Disposal of dry ice
- Allow the dry ice to sublimate or evaporate to the atmosphere in a well-ventilated area where CO vapor cannot build up.
- Do not dispose of dry ice in sewers, sinks, or toilets. The extreme cold can fracture ceramic fixtures or crack polyvinyl chloride (PVC) piping. If flushed down plumbing, the gas buildup can cause an explosion.
- Do not place dry ice in trash cans or similar containers. The extreme cold and resulting condensation can destroy these receptacles.
3.7. Electrical Safety
- Electrical hazards can be categorized into two main types: those that can result in an electrical shock and those that can cause fires and/or explosions.
- Electrical shocks can be avoided by ensuring that equipment and electrical cords and plugs are in good repair, grounded outlets are used, and ground-fault interrupt outlets or circuit breakers are used near sinks, eyewashes, emergency showers, or other water sources.
- Do not overload electrical circuits. Minimize or eliminate the use of multi-outlet power strips. When power strips are necessary, the safety office of the facility or a licensed electrician must approve their use.
- Disconnect equipment attached to high-voltage or high-amperage power sources from the source, or provide a lockout device on the breaker box to prevent circuit activation before maintenance is performed.
- Because electrical devices can generate sparks, do not use them near flammable or volatile gases or liquids.
- Never place flammable liquids in a household refrigerator. The spark generated by the door-activated light switch can ignite fumes trapped in the unit, causing an explosion and fire.
- Specialized refrigerators must be used when storing chemicals that have explosion potential.
3.8. Gases in the Laboratory: Compressed Gas Cylinders
Compressed CO cylinders are often used to provide gases for CO incubators; the risks associated with these incubators are minimal as long as the room is well ventilated.
3.8.1. Hazards
- Gas cylinders pose three major safety hazards:
— Gas cylinders are heavy; thus, a falling cylinder can cause injury.
— The valve attached to the cylinder is relatively fragile compared with the cylinder; if the valve is broken off, the cylinder can become a dangerous projectile.
— Faulty valves or regulators can leak, allowing toxic or flammable gases to enter the room. - In the electron microscopy laboratory, nitrogen is used to bring vacuum chambers to atmospheric pressure, and critical point driers use CO as a transitional fluid in the drying process for scanning electron microscopy specimens.
- Argon is used in sputter coaters, and some laboratories carry out plasma ashing of biological specimens, which requires oxygen (42).
3.8.2. Minimizing hazards
Many of these potential hazards can be minimized by adoption of safe handling practices.
- Cylinders must be securely anchored to the wall with chains or straps to prevent falling. Cylinders <18 inches tall may be secured in approved stands or wall brackets.
- When installing a new cylinder, leave the protective valve cap in place until the cylinder is secured. Replace the protective valve cap before the straps or chains are removed from the cylinder.
- Special regulators and threading are designed for each gas type. Do not try to force the threads or use the wrong regulator on a tank.
- Regulators are normally supplied with instructions for routine maintenance and periodic checking to ensure safe operation. Follow these instructions and checks carefully.
- Always use specially designed cylinder carts when moving cylinders. Cylinders must be secured to the cart and the valve covers must be attached when moving them. They are not to be dragged, rolled, or physically carried. Do not pick cylinders up by the cap.
3.9. Liquid Gases (Cryogens)
Cryogenic liquids are liquefied gases that have a normal boiling point below -238°F (-150°C). Liquid nitrogen is used in the microbiology laboratory to freeze and preserve cells and virus stocks. The electron microscopy laboratory, frozen section suites, and grossing stations for surgical pathology frequently use liquid nitrogen; some laboratories also use liquid helium. The principal hazards associated with handling cryogenic fluids include cold contact burns and freezing, asphyxiation, explosion, and material embrittlement.
3.9.1. Cold contact burns and freezing
- Liquid nitrogen is dangerously cold (-320°F [-196°C]), and skin contact with either the liquid or gas phase can immediately cause frostbite. At -450°F (-268°C), liquid helium is dangerous and cold enough to solidify atmospheric air.
- Always wear eye protection (face shield over safety goggles). The eyes are extremely sensitive to freezing, and liquid nitrogen or liquid nitrogen vapors can cause eye damage.
- Do not allow any unprotected skin to contact uninsulated piping, hoses, tongs, spargers, specimen box storage racks, or other metal objects because these become extremely cold when exposed to liquid nitrogen. Skin will stick to the metal, tearing the flesh when one attempts to withdraw from it.
- When filling cryogenic dewars, wear long-sleeved shirts or laboratory coats, long trousers (preferably without cuffs which could trap the liquid), closed shoes (never sandals or open shoes), and insulated cryogloves labeled as appropriate for use with cryogenic liquids. Do not tuck pant legs into shoes or boots; doing so could direct liquid into the foot coverings and trap the cryogenic liquid against the skin.
- Wear loose-fitting thermal gloves with elbow-length cuffs when filling dewars. Ensure that gloves are loose enough to be thrown off quickly if they contact the liquid.
- Never place gloved hands into liquid nitrogen or into the liquid nitrogen stream when filling dewars. Gloves are not rated for this type of exposure. Insulated gloves are designed to provide short-term protection during handling of hoses or dispensers and during incidental contact with the liquid. Use special cryogenic liquid tongs when retrieving items from liquid nitrogen.
- Liquid nitrogen confers a high risk of splattering; jets of liquid nitrogen can be generated when canes, canisters, and other objects that are at much higher temperatures are placed into liquid nitrogen. These activities can present a freezing hazard.
- Do not insert a hollow tube into the liquid nitrogen because liquefied gas may spurt from the tube.
3.9.2. Asphyxiation hazards
- Although nitrogen is nontoxic and inert, it can act as an asphyxiant by displacing the oxygen in the air to levels below that required to support life. Inhalation of nitrogen in excessive amounts can cause dizziness, nausea, vomiting, loss of consciousness, and death without warning.
- When liquid cryogens are expelled into the atmosphere at room temperature, they evaporate and expand to 700–800 times their liquid volume. Even small amounts of liquid can displace large amounts of oxygen gas and decrease the oxygen content of the atmosphere below a safe level (23,38,42,43).
- Do not store dewars or nitrogen containers in a confined space. The venting gas could displace enough oxygen to become a hazard.
- If enclosed spaces must be used, install oxygen monitors. Train personnel to leave the area immediately if the alarm sounds. The alarm must be audible both inside and outside the room to prevent anyone from entering the room.
3.9.3. Explosion hazards
- Liquid gases, even those considered inert, can present explosion hazards.
- Heat flux into the cryogen is unavoidable regardless of insulation quality. Cryogenic fluids have small latent heats and will expand 700–800 times as they warm to room temperature. Therefore, even a small heat input can create large pressure increases within the vessel.
- Dewars must be moved carefully. Sloshing liquid into warmer regions of the container can cause sharp pressure rises.
- Do not drop, tip, or roll containers on their sides; doing so could damage the vessel and/or cause a sharp increase in internal pressure.
- Cryogenic containers are equipped with pressure relief devices designed to control the internal pressure. Cryogenic containers will periodically vent gases. This is normal. Do not plug, remove, or tamper with any pressure relief device.
- Vents must be protected against icing and plugging. When all vents are closed, the expanding gas can cause an explosion. Vents must be maintained open at all times.
- Always use special ultralow-temperature containers to hold liquid nitrogen. Never place liquid nitrogen into domestic thermos flasks because they are not designed to withstand the large and rapid temperature changes that occur when liquid nitrogen is placed in the vessel (42,43).
- Fill liquid nitrogen dewars slowly to minimize the internal stresses of cooling. Excessive stress could damage the vessel and cause it to fail.
- Liquid helium is cold enough to solidify atmospheric air. Only helium is to be introduced or allowed to enter the helium volume of a liquid helium dewar. Take precautions to prevent air from back-diffusing into the helium volume.
- Liquid nitrogen and liquid helium have boiling points below that of liquid oxygen, and they can condense oxygen from the atmosphere. Repeated replenishment of the system can cause oxygen to accumulate as an unwanted contaminant. Similar oxygen enrichment can occur where condensed air accumulates on the exterior of cryogenic piping. An explosion could occur if this oxygen-rich liquid is allowed to soak insulating or other materials that are not compatible with oxygen. In addition, some oils can form an explosive mixture when combined with liquid oxygen.
3.9.4. Cryotube explosions
- PPE includes an ANSI-specification, impact-resistant face shield, heavy gloves, and a buttoned laboratory coat during removal of cryotubes and ampoules from nitrogen tanks.
- Cryotubes and glass ampoules used for freezing cells and viruses can explode without warning when removed from cryogenic storage. These tube explosions are presumed to be caused by entry of liquid nitrogen into the tube through minute cracks; as the tube thaws, the rapidly expanding gas causes the tube to explode, scattering the contents of the tube (23).
- Whenever possible, store ampoules in the gaseous phase rather than submerging in the liquid nitrogen of the cryogenic dewar. An imperfectly sealed ampoule will pick up less nitrogen in the gaseous phase.
- Nitrogen outgassing from an imperfectly sealed vial will sometimes produce a hissing noise before the vial explodes. The absence of hissing does not mean the vial is safe. Place cryotubes and ampoules onto gauze or paper toweling in an autoclavable, heavy-walled container immediately after removal from the nitrogen tank, and close the lid of the heavy-walled container quickly. If an explosion occurs, autoclave the entire vessel.
3.9.5. Embrittlement
- Never pour cryogenic liquids down the drain. Laboratory plumbing is one of many ordinary materials that become brittle at cryogenic temperatures and easily fracture.
- Wood and other porous materials may trap oxygen at low temperatures and will explode when subjected to mechanical shock (42).
3.9.6. Infectious disease hazards
- Liquid nitrogen can become contaminated when ampoules are broken in the dewar, and contaminants can be preserved in the nitrogen (23). These potentially infectious contaminants can contaminate other vials in the dewar and generate an infectious aerosol as the liquid nitrogen evaporates.
- Plastic cryotubes rated for liquid nitrogen temperatures are recommended for liquid nitrogen storage because they appear to be sturdier than glass ampoules and are less likely to break in the nitrogen.
3.10. Slip, Trip, and Fall Hazards
Slips, trips, and falls can cause a laboratory worker to drop or spill vessels containing infectious agents or dangerous chemicals. They can also lead to skin punctures and abrasions that make laboratory workers more vulnerable to LAIs.
Good housekeeping is the most fundamental means for reducing slips, trips, and falls. Without good housekeeping, any other preventive measures (e.g., installation of sophisticated flooring, specialty footwear, or training on techniques of walking and safe falling) will never be fully effective.
3.10.1. Slips
- Common causes of laboratory slips include wet or oily surfaces; loose, unanchored rugs or mats; and flooring or other walking surfaces that do not have some degree of traction in all areas.
- Water on the floor is the major slip hazard. Remove any water on the floor promptly.
- Paraffin from tissue mounting and cutting can accumulate in tissue processing areas and can make the floor slick despite regular cleaning unless special floor care measures are taken.
- Mineral oils, mounting fluids, stainless steel cleaners, and other laboratory chemicals and/or reagents create slip hazards if they get on the floor. Clean up with soap and water as soon as such spills are discovered.
- Do not use alcohols to clean floors; alcohols will dissolve floor wax, creating areas with different degrees of traction.
- Mats can present a slip hazard if they are not properly anchored to the floor.
- Walking on paper, cardboard, or packaging materials can present a slip hazard.
3.10.2. Trips
- Common causes of tripping include obstructed view, poor lighting, clutter in the walkway, mats or other items in the walkway, uncovered cables, open drawers or cabinets, and uneven walking surfaces. Permeable mats and rugs are not recommended in microbiology, except as noted later in this section.
- Keep drawers and cabinets closed except when they are being accessed.
- Clutter and items that protrude from kneehole spaces can injure workers as they move down aisles. Keep clutter to a minimum. Make sure that boxes and other items do not protrude into aisles.
- Do not run cords or cables across aisles or other walkways.
- The safety officer or laboratory management must assess the use of ergonomic antifatigue mats in other sections of the laboratory before employing in a specific laboratory area. Concerns to be aware of before using such mats include the following:
— These mats are somewhat thick, and the raised surface presents a trip hazard.
— The mats are obstructions for carts and chairs and may cause them to tip.
— They make spill cleanup difficult.
— They make cleaning and disinfecting the floors difficult for the custodial staff.
— They may also place custodial staff at risk if they pick up or move mats that have been inadvertently contaminated with chemicals or infectious agents.
— Liquids will often wick under the mat, hiding potential contamination problems.
— Mats present a trip/fall hazard that could impede egress from the laboratory in an emergency.
3.11. Ultralow-Temperature Freezers
Wear thermally resistant gloves and a laboratory coat when handling items stored at ultralow temperatures. Specimens stored at ultralow temperatures are extremely cold [-70°C to -85°C]), and paradoxically, direct contact with the skin can cause severe burns.
3.12. Ultraviolet light
- Short-wave UV light has had several applications in the laboratory, including use in fluorescent microscopes, as a terminal disinfectant in some type 1 water systems, and for visualizing nucleic acid bands in ethidium bromide–stained gels.
- Exposure to short-wave UV light has been linked to skin cancers, corneal scarring, and skin burns. These effects can result from direct or reflected UV light exposure (44).
- Do not use UV lights for decontaminating BSCs. Organisms in cracks, shadows, and on the underside of equipment are not affected by UV light treatment. In addition, the radiation and ozone produced by these lights will attack plastic and rubber items in and around the BSC, shortening their lifespan. This exposure can affect mechanical pipette calibrations and other sensitive equipment functions.
- If UV lights must be used for other reasons in BSCs, provide a means to monitor them throughout their life with intensity sensors. Calibrated UVC sensors are a reliable and cost-effective way to monitor UVC radiation levels in BSCs. Monitor these lights because germicidal UV lights have an expected life of about 9,000 hours.
- Germicidal UV irradiation for longer than 15 minutes is counterproductive because it produces no additional germicidal benefit and it accelerates equipment degradation.
- The UV lamp must never be on while an operator is working in the cabinet. Not all protective eyewear will protect laboratory workers from deleterious UV light exposure. Make sure the protective eyewear is rated for UVC protection.
- Wear UV safety glasses when performing routine lamp maintenance or when potential exists for direct or indirect (reflected light) exposure.
- Wear gloves, long-sleeved laboratory coat, and full-face shield when working with UV view boxes lacking protective filter shields.
- In areas where UV light is used, display placards stating "Caution, Ultraviolet Light, Wear Protective Eyewear."
3.13. Vacuum devices
Vacuum-assisted filtration devices and side-arm suction flasks are used routinely in the general laboratory, whereas the electron microscopy laboratory uses vacuum-assisted evaporators, freeze-driers, freeze-fracture, and sputter coater units. Vacuum-assisted devices present implosion hazards and risk aerosol generation.
3.13.1. Implosion safety
- Implosions can occur when the pressure differential exceeds the specifications of the vessel.
- Implosions can scatter sharp glass debris in all directions and seriously injure anyone in the vicinity (42,43). They will also disperse any infectious agents that are present in the vessel.
- Heavy-walled, side-arm suction flasks are generally rated to withstand a pressure differential of one atmosphere (14.7 PSI); house vacuum systems or vacuum pumps that provide pressure differentials exceeding that level must be regulated with an in-line pressure regulator.
- Cracks, chips, and scratches in vacuum flasks and bell jars can weaken the glass and cause an implosion even when the proper differentials are provided by pressure regulators.
- Care must be taken to prevent damage to bell jars and suction flasks caused by excessive wear or impact with hard objects.
- Implosion guards made of plastic mesh or plastic boxes have been used with suction flasks to contain glass pieces if the vessel fails. When infectious agents or blood or blood products are being handled, the use of plastic flasks is strongly recommended.
3.13.2. Aerosol generation
- Vacuum-assisted aspiration traps consist of one or two suction flasks plumbed together in series with an in-line HEPA filter (e.g., Vacushield Vent Device, Pall Life Sciences, Port Washington NY, or equivalent device) to prevent contamination of the vacuum pump or house vacuum system (1).
- When using a dedicated vacuum pump, many laboratories also include a suction flask containing coarse Drierite (W.A. Hammond Drierite Co., Ltd, Xenia, OH) or an equivalent desiccant to remove moisture from the air, thereby protecting the pump. Aspiration traps are used in virology to remove culture media from tubes, shell vials, and other vessels before refeeding or other cell manipulations. Aspiration systems are also used in enzyme-linked immunoassay (ELISA) plate washers.
- All these devices generate aerosols by agitating the fluid and placing the fluid surface under reduced pressure (23). Aerosols can deposit infectious agents on the immediate surfaces, and finer aerosols can be inhaled.
3.13.3. Aerosol protection measures
- Use aspiration devices in a BSC to contain any aerosols.
- Operators are to wear a disposable laboratory coat and gloves to protect themselves from infectious droplets.
- When a culture aspiration is complete, allow the BSC blower to run for 5 minutes to purge any airborne aerosols; decontaminate the work surfaces in the normal manner.
- Replace the in-line HEPA filters every 6 months or when they become wet or noticeably blocked.
3.13.4. Disposal of liquid wastes from vacuum-assisted aspiration traps
- Never pour infectious wastes down the sink.
- Decontaminate liquid wastes from aspiration traps with bleach before disposal.
- When using an aspiration trap attached to an individual vacuum pump, laboratories usually pass the vapors through an activated charcoal trap to protect the pump from chlorine vapor corrosion.
- A variety of suction trap configurations is possible, and the ultimate configuration will depend upon workflow and individual laboratory practice. The following procedures apply to all configurations.
— Change vacuum flasks when they are three-fourths full to prevent overfilling. Some laboratories prefer to mark the maximum fill volume on the flask and add a sufficient volume of bleach at the beginning of the day to produce a 1:10 bleach solution when the aspirated fluids reach the maximum fill mark.
— Disinfect the hose by aspirating 10–50 mL of a freshly made bleach solution into the trap. Lift the hose to allow all the bleach to enter the trap. Wait 20 minutes, then remove the trap from the BSC.
— Once decontaminated, the fluid is considered noninfectious and may be poured down the sanitary sewer.
Note for the virology laboratory: Bleach will reduce the phenol red dye in cell culture media, and the solution will go from red to colorless. If this color change does not occur, the fluid has not been decontaminated and sufficient bleach must be added to decontaminate the vessel.
3.14. Biological Hazards
3.14.1. Punctures and cuts
Skin punctures and cuts can directly introduce an infectious agent into the body and can provide a route whereby a secondary agent can enter.
- Needle sticks (45)
— Clinical laboratories must establish a needlestick and sharps injury prevention program.
— Limit the use of needles and syringes to procedures for which there are no alternative methods. Needlestick injuries occur most often when needles are returned to their protective sheathes after use.
— Do not resheathe needles. If resheathing is absolutely required, the procedure must utilize a needle resheathing device to minimize injury and accidental inoculation.
— Do not use needle-cutting devices because they can produce infectious aerosols. After use, place needles and syringes in leak- and puncture-resistant containers appropriately labeled with the word "Biohazard" and the universal biohazard symbol for decontamination and disposal.
— Do not bend, shear, recap, or remove needles from disposable syringes, or otherwise manipulate by hand before disposal.
— Microtome/cryostat blades used to cut frozen sections are another potential sharp that must be handled carefully. Wear cut-resistant gloves during disassembly of the potentially contaminated blade for cleaning and disinfection. - Breakage
— Never pick up broken glass with gloved or bare hands. Use forceps, disposable plastic scoops, tongs or hemostats to pick up broken glass; dispose of the broken glass into a sharps container. Place a broom or hand brush and dustpan in various laboratories or in the utility closet for picking up noncontaminated glassware.
— Do not use broken or chipped glassware. Discard it in the appropriate sharps container labeled for broken glassware.
— When handling broken containers with spilled infectious substances, adhere to the following guidelines (1).-
Wear appropriate gloves for this procedure (based on risk assessment and protection needed).
-
Cover the broken container and spilled infectious substance with a cloth or with paper towels.
-
For the routine BSL-2 laboratory, pour a disinfectant or a fresh 1:10 household bleach solution over the covered area and leave for a minimum of 20 minutes. It would take 23 minutes to clear the air of airborne M. tuberculosis from a spill at 99% removal efficiency if the room had 12 room air changes per hour, and 35 minutes for this removal with 99.9% efficiency (46). Given the variability of the number of room air changes per hour in diagnostic laboratories, the wait time has to be carefully evaluated.
-
The cloth or paper towels and the broken material should be cleared away into biohazard sharps receptacles. Fragments of glass are to be handled with forceps, not gloved hands. (Using wadded up tape with forceps facilitates this procedure). Small HEPA vacuum cleaners are also available for removal of fine glass particulates.
-
- If laboratory forms or other printed or written matter is contaminated, the information on the forms or written matter is to be copied onto another form and the original discarded into the biohazard waste container.
- Pasteur pipettes
— Whenever possible, substitute plastic or evaluate the procedure to determine if a newer or better technique is now available.
— Both the top and the bottom of a Pasteur pipette can cause puncture wounds.
— Before handling a glass Pasteur pipette, examine the top of the pipette to see if it is broken or cracked. Broken pipettes can produce puncture wounds.
— When seating glass Pasteur pipettes into suction lines, hold the pipette at the top and do not allow your hand to extend below the tip. Improper technique while seating the pipette can produce puncture wounds if the hand slips or the pipette breaks.
— Dispose of used Pasteur pipettes in leak- and puncture-resistant containers. In most locations, contaminated Pasteur pipettes are considered sharps and must be disposed of as such. - Other sharp devices
— Knives, scissors, and tissue homogenizers are frequently used to dissociate tissue specimens before testing. These items must be handled carefully in order to prevent cuts and skin punctures that could injure or inoculate laboratory workers with infectious materials.
— Pointed forceps are often used for fine dissociation and for removing coverslips from shell vials. These forceps can puncture the unwary user, causing injury and/or infection.
— Glass slides can break and puncture skin.
— Culture tubes and shell vials can crack or shatter if caps are forced. The resulting shards can easily penetrate latex gloves and skin.
— The lip of glass vessels may be chipped, and these chipped surfaces can cut unwary laboratory workers. Discard chipped glassware at the earliest opportunity. - Sharps disposal
— Carefully place used disposable needles, syringes, scalpels, blades, pipettes, and similar objects into properly labeled leak- and puncture-resistant containers made for disposal. Most authorities require needles and syringes to be disposed of in such sharps containers, whether used or not.
— Locate sharps disposal containers in or near the area where the sharps are used in order to prevent environmental contamination and injuries associated with accumulating sharps at the point of generation and moving sharps from one place to another
— Replace sharps containers that are two-thirds to three-fourths full. Sharps containers must close securely for transport to decontamination areas. Injuries can occur when laboratory personnel try to forcibly close full containers. Overfilled containers can pop open again, creating a hazard for other workers.
— Place nondisposable sharps into a covered leak-resistant, hard-walled container for transport to a processing area for decontamination, preferably by autoclaving (1).
— Place materials to be decontaminated off-site into a medical waste shipping container, and secure for transport in accordance with applicable state, local and federal regulations (1).
— Place clean, uncontaminated sharps (e.g., clean broken glassware, chipped clean pipettes) into rigid, puncture-resistant containers for disposal in the normal trash stream. Tape containers shut to prevent accidental opening and potential injuries.
— Never place sharp items directly into the regular trash. They could injure custodial or other staff members when the trash bags are removed from rigid trash containers.
3.14.2. Ingestion and contact with infectious agents
- Refrain from touching eyes, nose, mouth, and lips while in the laboratory.
- Do not place pens, pencils, safety glasses, or other laboratory items in the mouth or against the lips.
- Do not store food or beverages for human consumption in the laboratory.
- Mouth pipetting is prohibited; mechanical pipetting devices must be used.
- Eating, drinking, smoking, handling contact lenses, and applying cosmetics are not permitted in the laboratory.
- Wash hands after working with potentially hazardous materials and before leaving the laboratory. The laboratory must have a sink for handwashing, preferably located near the laboratory exit.
- Gloves must be worn to protect hands from exposure to hazardous materials. In the molecular biology area, gloves also protect the specimen from nucleases that are on the skin.
— Change gloves when they are contaminated, integrity has been compromised, or when otherwise necessary
— Remove gloves and wash hands when work with hazardous materials has been completed and before leaving the laboratory.
— Do not wash or reuse disposable gloves.
— Never touch your face, mouth, eyes, or other mucous membranes when wearing gloves in the laboratory
— Because gloves worn in the diagnostic laboratory are considered potentially contaminated, place them into biohazard disposal containers when discarding.
— Remove gloves when answering the telephone or using common equipment like computers. - To prevent contamination of ungloved hands, design the laboratory so that it can be easily cleaned.
— Decontaminate work surfaces with an appropriate disinfectant after completion of work and after any spill or splash of potentially infectious material (see Section 3.4, Disinfection).
— Bench tops must be impervious to water and resistant to heat, organic solvents, acids, alkalis, and other chemicals.
— Chairs used in laboratory work must be covered with a nonporous material that is easily cleaned. Uncovered cloth chairs are inappropriate. - Telephones are a potential vehicle for transferring infectious agents to the face and mucous membranes, and they should be used with this in mind.
— Never pick up or dial a telephone with gloved hands.
— Disinfect telephones regularly with disinfectants. (Alcohols do not inactivate nonenveloped viruses or destroy DNA.)
— Use the hands-free or speaker phone features whenever possible to avoid touching the telephone handset to the face. - Never bring briefcases, purses, backpacks, books, magazines, and other personal items into the laboratory. These items are difficult to disinfect.
3.14.3. Spills and splashes onto skin and mucous membranes
- It is the responsibility of all laboratory workers to perform all procedures in a manner that minimizes the creation of splashes and aerosols.
- All splashes to the eye must be flushed for a minimum of 15 minutes. If a laboratory worker wearing contact lenses receives a splash to the eye, the eye must be flushed with water, the lens removed, and the eye flushed again. Discard disposable contact lenses. Disinfect reusable contact lenses before returning them to the eye. Consultation with the manufacturer may be warranted.
- Specimen containers, culture tubes, shell vials, and other cylindrical vessels used in the laboratory are easily tipped and could roll when placed on the bench top. Glass vessels can break if dropped. Secure these vessels in racks whenever possible to prevent opportunities for breakage.
3.14.4. Aerosols and droplets
Any procedure that imparts energy to a microbial suspension can produce infectious aerosols (1,23). Procedures and equipment frequently associated with aerosol production include pipetting, mixing with a pipette or a vortex mixer, and use of blenders, centrifugation, and ultrasonic devices (sonicators) (1,23,47). These procedures and equipment generate respirable particles that remain airborne for protracted periods. When inhaled, these tiny particles can be retained in the lungs.
These procedures and equipment also generate larger droplets that can contain larger quantities of infectious agents. The larger droplets settle out of the air rapidly, contaminating, work surfaces as well as the gloved hands and possibly the mucous membranes of persons performing the procedure.
Respirable particles are relatively small and do not vary widely in size distribution. In contrast, hand and surface contamination is substantial and varies widely (1,48). The potential risk from exposure to larger-size droplets requires as much attention in a risk assessment as the risk from respirable particles.
- Pipetting
Pipettes and pipetting processes can be an appreciable source of infectious aerosols and environmental contamination. Therefore, it is prudent to wear gloves, eye protection, and a laboratory coat with knit cuffs when pipetting and to perform pipetting operations in a BSC. The following guidelines are categorized into those for serologic or mechanical pipettes.
— Serologic pipettes. When the last drop of fluid is forcibly expelled out of the pipette tip, small and large droplet aerosols are formed that can contaminate the hands and the environment (23). To minimize aerosol generation, place the pipette tip against the inside wall of tubes, flasks, or other vessels, and gently expel the last drops of fluid.
— When dispersing cell clumps, virologists frequently draw fluids into and out of the pipette to homogenize specimens and cell suspensions. A substantial amount of aerosolization can occur during this process, especially when the fluid is forcibly expelled from the pipette tip. Aerosols are generated in a similar manner during "pipette mixing" of culture dilutions. Closed-cap vortex mixing is the preferred method for this type of mixing. If pipette mixing is required, keep the pipette tip below the surface of the fluid and do not eject the entire fluid volume from the pipette. This will reduce aerosolization and bubble formation.
— Vigorous pipetting (rapid aspiration of fluid into the pipette) can generate aerosols within pipettes. Some of the aerosols will be trapped by the cotton plug at the proximal end of the pipette. However, some aerosols can travel through the cotton plug and contaminate the pipetting device. Certain mechanical pipetting devices have HEPA filters that minimize contamination of the handset. Replace these filters regularly and whenever they become wet. Decontaminate pipette bulbs regularly and whenever they become contaminated.
— Mechanical pipettes. Hand-held mechanical pipetting devices are used for enzyme immunoassay (EIA) testing, molecular diagnostics, and other activities that require precision.
— In molecular diagnostics, pipette contamination is the most frequent cause of false-positive results (49). Pipette contamination can occur from aerosols, from touching the outside of the pipette to a contaminated surface, and from contaminating the inside of the pipette during the pipetting process.
— Expelling the last remaining fluid in the tip will result in droplet splatter and aerosol formation. These aerosols and droplets can contaminate the other samples and the environment. Most mechanical pipetting devices have two stops on the plunger — the "To Deliver" stop and the "Expel" stop. Pipette volumes are calibrated for accuracy at the "To Deliver" stop, and there is no need to expel the final amount of fluid to preserve pipetting accuracy.
— Touch pipette tips to the inside of the well or tube before pressing the delivery plunger. Never direct the pipetting stream into the middle of the well because this will cause splashing and contamination.
— Care must be exercised when ejecting used tips into discard containers because the remaining fluid can splash and splatter widely.
— The outside of the pipette barrel can become contaminated through splatter, aerosols, or by touching the barrel to contaminated objects. Do not extend the barrel of the pipette into a reagent, sample or discard container. If normal-length tips cannot reach the fluid in the tube, use extended-reach pipette tips to prevent barrel contamination.
— Disinfect mechanical pipettes regularly following the manufacturer's instructions or with a 1:10 household bleach dilution followed by 70% alcohol to remove as much bleach as possible.
— Use of aerosol-resistant pipette tips can substantially reduce nucleic acid contamination inside the pipette. Aerosol-resistant tips contain a hydrophobic microporous filter that is bonded onto the walls of the pipette tip. The microporous filter traps aerosols before they can contaminate the barrel of the pipette. These filters can also prevent contamination of the specimen when a contaminated pipette is inadvertently used (49).
— When an accidental falling drop from a pipette tip encounters a hard surface, it generates aerosols and a series of small droplets, some of which may be large enough to fall and repeat the process (50). Greater contamination ensues when drops fall a greater distance onto a hard surface.
— Many laboratories use commercial plastic-backed bench paper in BSCs and on laboratory work benches to contain or absorb contamination from falling drops.
— When faced with the inevitability of a falling drop, it is best to lower the tip of the pipette and allow the drop to fall a short distance onto an absorbent towel. This procedure will minimize the kinetic energy of the drop and its capacity to splatter. - Tubes and other vessels
— Thin films sometimes form in the neck of culture tubes, shell vials, microcentrifuge tubes, specimen vials, and other containers. Breaking or popping this film produces aerosols and microdroplet splatter that can contain infectious agents, nucleic acids, or other potential contaminants (23).
— Containers with thin films in the neck are to be recapped and centrifuged whenever possible to disrupt the film or cause it to merge with the fluid in the vessel.
— If centrifugation is not possible (e.g., with culture flasks), place gauze or another absorbent material over the opening and insert a pipette into the flask to disrupt the film. Dispose of the pipette and the absorbent material with other contaminated materials.
— Microcentrifuge and other plug-topped tubes will often produce aerosols and splatter when opened. Screw-cap microcentrifuge tubes can reduce this risk.
— To minimize the amount of fluid on the cap, subject microcentrifuge tubes to a quick "pulse" centrifugation before they are opened.
— Open microcentrifuge tubes in a BSC whenever possible.
— When opening plug-seal microcentrifuge tubes, cover the top of the tube with absorbent material (e.g., alcohol-moistened gauze) to catch any splatter that might occur. Dispose of the absorbent material with other contaminated materials. - Lyophilized materials, serum vials and ampoules
Opening vials of freeze-dried (lyophilized) material can be hazardous because these fine dry powders are easily dispersed into the atmosphere when air rushes into the evacuated vessel (23). The following procedure may be used to safely open a serum vial containing lyophilized material.
— Move the vial and the suggested diluent (water or medium as appropriate) to a BSC.
— Wear gloves and laboratory coat when opening lyophilized vials.
— Use a hemostat to remove the aluminum crimp from the vial. Discard the crimping material into the sharps container.
— Cover the stopper with a moistened gauze pad, and carefully lift the edge of the stopper and allow air to slowly enter the vial. Do not disturb the contents of the vial.
— Once the vacuum has been released, remove the stopper completely and place it upside down on absorbent paper.
— Add the appropriate amount of diluent to the vial using a sterile pipette.
— Replace the stopper and allow the vial contents to hydrate for several minutes.
— Discard the gauze, stopper, and absorbent paper with other contaminated materials.
— Using a pipette, transfer the contents of the vial to an appropriate container.
— Discard the original vial with other contaminated materials.
— Needle and syringe methods for removing infectious agents from serum vials are not recommended because they can contaminate the environment and because they provide opportunities for needlestick injuries. Use forceps, not needles, to remove serum separator tubes that are stuck in centrifuge carriers. - Glass ampoules
— Once opened, glass ampoules can present a risk for cuts and punctures.
— Safety ampoule breakers can prevent injuries by covering the ampoule during the breaking process.
— For ampoules containing infectious materials, cover the score line with gauze moistened with disinfectant; then break as usual using the safety ampoule breaker.
— Place the ampoule breaker into a beaker containing a 1:10 bleach solution after removing the ampoule.
3.15. Ultrasonic Devices
- Ultrasonic devices are principally used to lyse bacteria and viruses and to clean glassware and laboratory equipment.
- Use the lowest effective power setting to minimize aerosol generation.
- Cover bath sonicators while the device is in use.
- Articles destined for ultrasonic cleaning must be properly decontaminated before cleaning to prevent aerosolization of infectious agents (23).
- Always conduct organism lysis and homogenization procedures in closed containers.
- Change bath fluids frequently to prevent aerosolization of bacterial and fungal contaminants present in the bath.
3.16. Clean versus Dirty Areas of the Laboratory
In the microbiology laboratory, all the technical work areas of the department are considered dirty. The same concepts of demarcation and separation of molecular testing areas that are described in this section can be used to establish clean and dirty areas in other parts of the diagnostic laboratory.
3.16.1. Clean areas
- Wear different color laboratory coats in clean and dirty areas of the laboratory (have them available at entrance to clean areas), or require no laboratory coats in clean areas.
- Decontaminate reusable materials and devices (e.g., telephone, clocks, computers, tissue boxes, work books) brought into the clean area unless they are known to be new, and immediately apply laboratory-designated, color-coded tape.
- A visual reminder on small objects such as workbooks, tissue boxes, and pens can easily identify items located to a clean area.
- Demarcate separation of dirty and clean floor areas with tape (tape must stand up to floor cleaning) to clearly denote clean/dirty area boundaries.
- Develop a policy for cleaning and maintaining clean areas.
- Train all personnel (including service personnel) regarding how to identify and maintain clean areas and to recognize the significance of the demarcation tape and other means of area identification.
- Document training and assess competency in use of and maintaining clean areas.
3.16.2. Offices
Offices (e.g., of supervisors and laboratory director) that open into the clinical laboratory represent hybrid areas within the laboratory. These offices are not typically designed or maintained in a manner that allows for easy or efficient disinfection.
- Keep a supply of hand disinfectant gel in all office and work areas and use the gel frequently.
- Components of offices that should remain clean but may be overlooked include
— laboratory documents, reports, and records; small equipment; pens; procedure manuals and other items that have been in the laboratory and could have been handled with gloved hands;
— carpets and chairs that are difficult to disinfect;
— books, journals, and other reference materials that can be taken into the laboratory or taken for use outside the laboratory;
— personal items (e.g., photographs, awards, briefcases, coats, boots, backpacks, purses, personal electronic devices) that are difficult to disinfect and would not be allowed in the general laboratory; and
— food items. - Designating office areas as "clean" does not necessarily make or keep them uncontaminated, especially when potentially contaminated items are brought into the office and reference materials and documents move freely between the office and laboratory. The following procedures can help reduce the risk of contamination in laboratory office areas.
— Never bring specimens, cultures, proficiency samples, and similar items into office areas.
— Remove PPE before entering the offices, and wash hands before entering these areas.
— Establish a dedicated and protected clean area for personal items (e.g., purses, briefcases, and similar items).
— Disinfect desks and personal workspaces, telephones, and computer keyboards in office areas regularly.
— Refrain from touching eyes, nose, mouth, and lips while in office areas.
— Do not place pens, pencils, eyeglass bows, or other items in the mouth or against the lips.
— Do not apply or permit cosmetics in office areas.
— Do not store food in the office.
— Wash hands after working in the office and before entering common areas such as rest rooms, administrative areas, cafeteria, and the library.
— Avoid clutter in office areas as much as possible. Boxes, papers, and other items make the office difficult to clean and decontaminate.
— Laboratory directors and supervisors are responsible for assessing the exposure risks associated with use of laboratory documents and reference materials in the dirty areas of the laboratory and developing use policies to minimize those risks.
3.16.3. Dirty areas
- All areas of the working laboratory — including all equipment, keyboards, waste, and surfaces — are considered "dirty" areas.
- No standards are currently available that describe operating procedures within dirty areas of the laboratory. Laboratorians must be vigilant in recognizing the potential or risk of transmitting an etiologic agent by touching items in these areas.
3.17. Instrumentation
Whether automated or manual, procedures with the potential for producing specimen aerosols and droplets (e.g., stopper removal, vortexing, opening or piercing evacuated tubes, using automatic sample dispensers) require PPE and engineering controls designed to prevent exposure to infectious agents.
3.17.1. Water baths and water (humidification) pans in CO incubators
- Clean regularly even if disinfectants are added to the water.
- To reduce bioburden, add disinfectant such as a phenolic detergent, fungicides, or algaecides, to the water as needed. Avoid using sodium azide to prevent growth of microorganisms because it forms explosive compounds with certain metals.
- Raise the temperature to 90°C or higher for 30 minutes once a week for decontamination purposes.
- Immediately clean after a spill or breakage.
- Water baths and humidification pans in CO incubators can harbor bacteria, algae, and fungi that become aerosolized when the water bath lid or incubator doors are opened. These aerosols can contaminate cultures and the environment.
- Empty and clean water baths and humidification pans regularly to minimize organism buildup and the production of biofilms that are notoriously difficult to remove.
3.17.2. Centrifuges and cytocentrifuges
- Centrifuges can be extremely dangerous instruments if not properly cleaned, maintained and operated. Laboratory staff must be trained in centrifuge operation and the hazards associated with centrifugation.
- Current regulations governing the manufacture of centrifuges ensure that operators are safeguarded against some potential injuries and exposures by the fitting of lid locks that prevent opening of the lid while the rotor is still spinning, imbalance detectors, and devices to prevent rotor overspeed, and that construction materials can withstand any rotor failure. Older centrifuges without these safeguards must be operated with extreme caution, and laboratories should have documented risk assessments and operating manuals that specifically provide operating instructions to mitigate these specific hazards.
- Each particular centrifuge type must have an operation manual.
- Operators are to have documented training and competency assessments on each type of centrifuge they operate. Documented instruction for each centrifuge type includes proper instrument startup and shutdown, emergency procedures and shutdown, balancing of tubes, use of safety cups and covers, rotor and container selection, requirements for high-speed and ultracentrifuges, and container fill-height limitations.
- Operate all high-speed and ultracentrifuges on a stable, resonance-free surface (floor, bench top, or heavy table) with at least 6-inch clearance at the sides and 4 inches at the rear of the centrifuge.
- In BSL-2 or higher areas, rotors need aerosol containment ("O-rings") and gasketed safety cups.
- Load and unload rotors in a BSC, particularly in virology and mycobacteriology sections.
- Manufacturer instructions for use and care of centrifuges, and especially rotors, are to be strictly enforced to prevent the serious hazards and potential exposures associated with rotor failure. Store rotors in a dedicated clean space and in an environment specified by the manufacturer.
- Clean centrifuges at the end of each shift and immediately after a spill.
- Never operate centrifuges with visible spills of blood or body fluid present.
- Rotors need annual stress testing and a complete certified analysis; most centrifuge manufacturers offer this service.
- Maintain a complete and comprehensive rotor log for every high-speed and ultracentrifuge rotor to include all user names, run dates, durations, speeds, total rotor revolutions, and any notes on rotor condition.
- Retire rotors after the manufacturers' recommended revolutions or years of service, whichever comes first, except where an annual stress test (from Magnaflux [Glenview, IL] or other professionally recognized analysis) proves an absence of structural flaws. Long-term budgetary planning for this event is important.
- During normal operations, air issues from centrifugation ventilation ports at high speeds, and any infectious particles present in the airflow will disperse rapidly and widely (47).
- Tube breakage during centrifugation presents the greatest risk for contamination because large aerosol clouds are produced. Occult contamination can occur when centrifuging tubes without gasketed safety caps.
- The airflow rushing around the tubes can create a venturi effect that can draw fluids from the threads of screw-capped tubes. The high-velocity airflow can also aerosolize dried or liquid materials that might be present on the outside of the tube.
- Consistent use of gasketed centrifuge safety cups and sealed rotors can substantially reduce the risks associated with centrifuging infectious or potentially infectious materials.
- Centrifugation equipment must be properly maintained to prevent malfunctions and aerosols within the centrifuge.
- Provide a centrifuge spill kit containing a disinfectant compatible with the centrifuge materials, puncture-resistant gloves, tweezers or forceps, cotton, hemostats, broom, hand brush, and dustpan.
- If a specimen tube breaks within the plastic screw-capped canister or bucket in a centrifuge, take the following steps.
— Turn the motor off and allow time for aerosols to settle before opening the centrifuge.
— Remove the canister and place in a BSC.
— Notify a supervisor or senior person in charge and other colleagues working in the area.
— While wearing protective clothing, open the canister under the BSC.
— Pour a 1:10 dilution of bleach or a noncorrosive disinfectant into the canister to decontaminate all surfaces; let the canister soak in bleach or disinfectant solution for 20 minutes. Clean the canister thoroughly.
— Do not pick up broken glass with gloved hands. Use forceps or cotton held in forceps, or tongs or hemostats, and dispose into a biosafety sharps container.
— Discard all nonsharp contaminated materials from canister into a red biohazard bag for biohazard waste disposal.
— Swab or wipe unbroken capped tubes with the same disinfectant; then swab or wipe again, wash with water, and dry.
— All materials used during the cleanup must be treated as infectious waste.
Note: If the specimen tube breaks in a centrifuge that does not have individual canisters but does have a biohazard cover and sealed rotor, follow the manufacturer's instructions for cleaning and decontamination.
3.17.3. Automated analyzers
- Automated analyzers frequently have added features to help reduce operator exposures, but these do not totally eliminate the potential for exposure. A common feature in newer systems is closed system sampling.
- Sample probes that move quickly or deliver fluid rapidly might generate aerosols and droplets.
- Always use instruments according to manufacturer instructions.
- Ensure instrument safety shields and containment devices are in place at time of use.
- Limit the amount of hand movement near the sample probe and liquid-level sensors.
- Wear gloves and use gauze pads with impermeable plastic coating on one side on instruments for which the operator is required to wipe sample probes after sampling.
- Newer instruments have automatic probe wash cycles, eliminating this source of exposure.
- Handle sample trays and sample plates with caution, and cover them when not being sampled to prevent spillage.
- Fill sample cups and aliquot tubes using mechanical devices; never decant them.
- Effluents of clinical analyzers are to be considered contaminated, and disposal must comply with state and local regulations.
- Follow manufacturer instructions for routine cleaning and trouble-shooting specimen spills on or within an instrument, including the appropriate PPE and type of cleaning solution to be used.
- When manufacturer instructions do not include spill containment and cleanup instructions, collaborate with the manufacturer to develop an SOP that will effectively protect the operator and maintain and extend the instrument's operational life.
- Safety guidelines for cell sorters have been published (51). Consider adding bleach to the waste receptacle so that a full receptacle would contain about 10% bleach.
3.17.4. Vacuum-assisted aspiration devices (See Section 3.13.)
3.17.5. ELISA plate washers in microbiology
- ELISA plate washers can create aerosols and droplets by agitating the fluid and placing the fluid surface under reduced pressure. Large-particle droplets generated by the washing and aspiration process can deposit infectious agents on the immediate surfaces, and finer aerosols can travel greater distances and can be inhaled.
- Handle ELISA plates with gloves at all times, and consider them to be contaminated.
- Disinfect ELISA plate washers and the area around the washer each day of use.
- Whenever possible, place aerosol containment covers over ELISA plate washers to minimize aerosol contamination of laboratory workers and the environment.
3.17.6. Identification, blood culture, and PCR instruments
Bacterial identification and antimicrobial susceptibility instruments, blood culture instruments, PCR instruments, and other laboratory instruments and devices are to be cleaned or disinfected according to the manufacturer's directions or recommendations. The routine and emergency cleaning procedure for each instrument must be a part of the safety component of the procedure manual.
3.18. Rapid Tests (Kits)
- Whether a rapid test is conducted in the laboratory or at the point of care, used testing kits are considered contaminated and should be disposed of appropriately.
- Limit the use of rapid testing kits to a specific area of the laboratory to maximize efficiency of environmental controls that can prevent aerosol exposures when manipulating reagents, samples, and control organisms.
- Disposable, flexible, polyethylene film–backed, nonskid, highly absorbent surface liners are recommended to contain spills and minimize contamination of test kit materials and boxes.
- Wipe the outside of test kits with appropriate laboratory disinfectant before returning them to the storage area.
3.19. Unidirectional Work Flow and Separation of Work Areas
- The reagent preparation area is the cleanest area, followed by the specimen preparation area, and finally the product detection area.
- Leave transportable items (e.g., pens, tape, scissors, glove boxes) in each designated area.
- Change laboratory coats and gloves and wash hands before entering each area.
- Follow this emergency response procedure if a potentially infectious aerosol release occurs outside a BSC.
— All persons must immediately vacate the laboratory unit where the spill occurred.
— Exposed persons are to be referred for medical advice and evaluation.
— Inform the laboratory supervisor and biosafety officer of the situation immediately.
— No one is to enter the room for at least 30 minutes to allow aerosols to be carried away and heavier particles to settle.
— If the laboratory does not have a central air exhaust system, entrance is to be further delayed (e.g., up to 24 hours).
— Post signs indicating that entry is forbidden.
— After the appropriate time, proceed with decontamination, supervised by the biosafety officer. Appropriate PPE must be worn, which may include respirators.
4. Tuberculosis Laboratory
Tuberculosis (TB) resulting from exposure to infectious aerosols remains a major risk for laboratorians. There is no safe level of exposure since exposure to as few as 1–10 organisms can cause disease. An estimated 8%–30% of laboratorians may experience tuberculin conversions (52). To reduce exposures to Mycobacterium tuberculosis, a hierarchy of controls must be employed, including safe work practices, use of containment equipment, and specially designed laboratory facilities (1). Tuberculosis laboratories need to be separate and isolated from the main microbiology laboratory. Develop all policies and practices related to safety using a risk assessment process that is documented in the laboratory's biosafety manual.
- It is the laboratory director's responsibility to ensure that every new employee receives safety training that includes proper and safe handling practices, use of safety equipment such as the biological safety cabinet (BSC), personal protective equipment (PPE), decontamination procedures, spill cleanup, use of the autoclave, waste disposal, knowledge of tuberculosis disease symptoms, and reporting illnesses and exposures.
- Most importantly, adherence to biosafety practices must be monitored and an annual competency assessment be completed.
- Employees and laboratory management must be familiar with the engineering components of the TB laboratory, such as how many air exchanges occur per hour, how negative pressure is measured, whether doors automatically lock, and how the intercom works in the suite.
4.1. Specimen Receiving and Log-In/Setup Station
In most clinical laboratories specimens are first received in the main microbiology laboratory (biosafety level [BSL]-2), where they are logged in and processed for other bacteriologic testing. The specimens submitted for TB analysis are moved to the TB laboratory for further processing specific for TB.
4.1.1. Specimen receiving in the main microbiology laboratory
- A wide variety of specimens are received for tuberculosis testing, including sputum, urine, tissue, cerebrospinal fluid, and gastric washings.
- Procedures on clinical specimens that do not produce an aerosol can be performed in a BSL-2 laboratory. Propogation and culture manipulation are performed in the BSL-3 laboratory.
- All clinical samples submitted specifically for TB testing must be handled by persons using PPE consisting of a laboratory coat and gloves, and the work must be conducted in a BSC (53) that is certified at least annually.
- Before opening any TB specimen container, regardless of the presence of visible contamination, disinfect the outside by wiping it with gauze soaked in a tuberculocidal disinfectant.
- Move the specimens to the tuberculosis laboratory, where all procedures for TB specimen decontamination, culture propagation, and subsequent manipulation of the cultures are performed in BSL-3 facilities and with the use of containment equipment and practices and respiratory protection (1).The BSL-3 facility must be properly maintained, and the door to the laboratory kept closed.
- Retrofitting a BSL-2 facility to accommodate a BSL-3 laboratory is not an option for some facilities that must test for M. tuberculosis. Biosafety in Microbiological and Biomedical Laboratories (BMBL-5) (1) has removed the language suggesting that BSL-3 procedures could be done in a BSL-2 laboratory when working with M. tuberculosis if a BSC were used and the air exhausted to the outside of the building. It is recommended that this alternative be used for laboratories without a BSL-3 facility only if three conditions can be documented:1) a risk assessment determines that work with M. tuberculosis can be conducted safely in a separate, closed BSL-2 laboratory using BSL-3 practices and procedures; 2) exhaust air from the laboratory room is vented to the outside of the building; and 3) the laboratory director approves the practice. If any of these conditions cannot be met, a BSL-3 facility is recommended for culture manipulation.
4.1.2. Specimen receiving in other laboratory sections
- When processing specimens with a suspicion of tuberculosis in a surgical pathology suite wear an N95 particulate respirator during frozen sectioning. Do not use propellant to flash-freeze tissue.
- When performing autopsy procedures, bone saws must have a vacuum attachment to minimize dispersal of bone dust. If tuberculosis is suspected, wear an N95 particulate respirator or powered air-purifying respirator (PAPR) during the procedure, and do not remove it until sufficient time has elapsed after the procedure for effective removal of airborne particles by the ventilation system, as indicated in the laboratory biosafety manual.
4.1.3. Leaking containers
- Collect specimens for processing into a leakproof container, and transport the specimens to the laboratory in a sealable leakproof plastic bag. An appropriate container ensures that handling of the specimen can begin without external contamination.
- The transport bag is opened inside a BSC to guard against the creation of aerosols, spray and splatter.
4.1.4. Visible contamination on the outside of container
- Specimens that leak during transport must be rejected and a new specimen requested. Before opening a transport bag, observe the specimen for leakage.
- If the outside of the container is grossly contaminated with the contents of the container, reject the specimen, document the rejection, and request another specimen.
- When examination of the exterior of the specimen container demonstrates minor or superficial contamination, clean the exterior with an appropriate disinfectant before further handling.
- Open a specimen container carefully because splashing or splattering may contaminate the outside of the container.
- Wipe the exterior of the container with gauze soaked in a tuberculocidal disinfectant after removing and replacing caps.
4.2. Stains and Disposal
Prepare smears in a BSC because aerosols, droplets and splatters can be generated. Unstained smears may contain viable tubercle bacilli and are to be handled with caution.
4.2.1. Gram stain
Specimens submitted for routine cultures, especially sputum and other respiratory specimens, may contain tubercle bacilli and must be handled with care regardless of whether or not acid-fast bacillus (AFB) cultures were ordered.
4.2.2. Acid-fast stains — Kinyoun, Ziehl-Neelsen, auromine–rhodomine (fluorescent)
- Before removing smears from the BSC, heat-fix the slide on an electric slide warmer with the temperature set between 149° and 167°F (65° and 75°C) for 2 hours. Monitor and record the temperature of the slide warmer each day of use. Even after heat-fixing, the slide may contain viable tubercle bacilli and should be treated as contaminated.
— For laboratories that do not process AFB cultures but wish to make a direct smear, the smear can be made in the BSL-2 laboratory.
— Use of a slide-warming tray rather than a flame is recommended for fixation of slides. Liquefaction and concentration of sputa for acid-fast staining may be conducted safely on the open bench by first treating the specimen in a BSC with an equal volume of 5% sodium hypochlorite solution (undiluted household bleach) and waiting 15 minutes before processing (1).
4.3. Culture Reading and Acceptable Activities at the Open Bench
- Only those activities that are solely observational and do not risk creation of aerosols can be performed at the open bench. Any manipulation of colonies of growth is performed within the BSC (54). Only closed, non-glass containers of culture with the outside of the container properly disinfected can be brought out of the BSC for spectrophotometer or other observational readings. However, the preference is that all work with cultures be conducted inside a certified BSC.
- Procedures must be in place to address the possibility of culture breakage (See Section 4.7, Spill Cleanup).Viable cultures must be transported securely using racks, safety carriers and/or carts to prevent breakage.
- Work surfaces are to be decontaminated each day testing is performed in the laboratory.
4.4. Personal Precautions and Work Practices
Precautions and work practices are selected with regard to the potential quantity of tubercule bacilli encountered in the procedure being performed. Hence, specimens have a lower concentration than a culture, in which the number of organisms is amplified. Because aerosols are generated whenever energy is imparted into the specimen, all protocols in the TB laboratory are evaluated through the risk assessment process for the potential to generate aerosols. Common aerosol-generating procedures are pouring liquid cultures and supernatant fluids, using fixed-volume automatic pipetters, and mixing liquid cultures with a pipette.
- Laboratorians who handle specimens in which M. tuberculosis is a suspected pathogen and/or perform diagnostic testing for M. tuberculosis must undergo at least annual testing for tuberculosis infection. This can be accomplished by a tuberculin skin test (TST) or interferon gamma release assay (IGRA). If the TST is performed, a two-step process is used upon hiring, and followed thereafter by a one-step TST. More frequent screening for TB may be necessary if a laboratory incident with risk of exposure to tuberculosis or a documented conversion occurs. Do not place a TST if the laboratorian has a history of either BCG vaccine or previous positive TST, in which case, an IGRA would be performed.
- Personnel must be aware that certain changes in health, e.g., receiving chemotherapy, may place them at increased risk for tuberculosis if exposure occurs.
4.4.1. Personal protective equipment
- A solid-front, disposable gown with snug (knit) cuffs is routinely used as protection against sprays and splatter.
- Gloves are to be worn at all times when working in the BSL-3 facility and must be long enough to externally overlap the sleeves of the gown. In the BSL-2 environment, wearing of gloves is dependent on the laboratory's routine practice that is guided by a risk assessment.
— In general, gloves are worn whenever there is reasonable risk of contamination of skin from spray, splatter or droplets during aerosol-generating procedures. Gloves are used starting with the initial work of observing the outside of the container for external contamination.
— Gloves are not required when observing cultures outside the BSL-3 environment. - As routine work practice, the laboratorian should remove all outer protective clothing when leaving the laboratory. Regardless of whether gloves are worn, thorough washing of hands after completion of procedures is required.
4.4.2. Respiratory protection
- Wearing a respirator, such as N95, is highly recommended for protecting the laboratorian when processing and manipulating specimens or TB cultures. Surgical masks are less effective because they are designed to contain aerosols expelled by the user, not to protect from aerosols. Personnel must be medically evaluated and fit tested before using an N95 respirator. If a person cannot be successfully fit tested for an N95 respirator (e.g., a person with facial hair), an acceptable alternative is a PAPR.
- No BSC is 100% effective. Failures do occur and respirators provide added protection.
- Personnel working in the TB laboratory must adhere to the facility's respiratory protection program meeting OSHA requirements (55).
— The components of the respiratory protection program are a written standard operating procedure, training, storage of the respirator if it is to be reused, inspection of the respirator before use, medical review, and program evaluation.
— Eligibility to participate in the respirator program includes a medical review and a pulmonary function test.
— Before use, the respirator must be fit tested to determine the size of respirator that best fits the worker and ensures a tight seal to the face.
— The annual fit testing is an opportunity for personnel to demonstrate proper donning of the respirator. - The PPE requirements for laboratory personnel must also be followed by outside service technicians. Do not permit servicing, cleaning, or checking of equipment in a BSL-3 facility unless a trained technical or professional person is present to ensure that adequate safety precautions are followed.
- PPE worn in the BSL-3 is to be removed before exiting the laboratory. Hands are always thoroughly washed after removal of PPE. Likewise, PPE worn in the BSL-2 must be removed before exiting the laboratory. Laboratory coats used while working in the laboratory are never worn outside the laboratory.
4.5. Disinfection
- A disinfectant for the TB laboratory is selected on the basis of its tuberculocidal activity and categorized as intermediate activity level (1). Compounds commonly selected are phenolics, iodophors, chlorine compounds, or alcohols. The killing time of germicides is never instantaneous, and exposure times and matrix of contaminated material must be considered when choosing an appropriate disinfectant.
- Daily disinfection of all surfaces in the TB laboratory is required because M. tuberculosis is very resistant to drying and can survive for long periods on solid surfaces.
- A good disinfection practice is to soak a gauze pad or paper towel in disinfectant and place it on the work surface inside the BSC while processing specimens.
- When decanting fluids in the BSC, use a splashproof container. Disinfectant must be added to a splashproof container before use. If the splashproof container has a funnel, rinse it with disinfectant after use.
- Use a loop incinerator device or an alcohol sand flask to remove large clumps of organisms from wire loops or spades.
4.6. Decontamination and Disposal of Laboratory Waste
- Provide an autoclave in the mycobacteriology laboratory so that generated waste can be sterilized before being transported from the laboratory. Adhere to the scheduled quality control and maintenance procedures for the autoclave.
- If an autoclave is not available or for items that cannot be autoclaved, all waste from the mycobacteriology laboratory must be securely contained in leakproof containers. Package waste so that the outside of the container can be disinfected before it leaves the laboratory.
- Chemically disinfect waste materials before removing them from the BSC.
4.7. Spill Cleanup
- The response in the event of a spill depends on the amount of aerosols produced. The decision to follow a minimal aerosol or major aerosol spill response procedure is made in conjunction with the supervisor and the safety officer and in accordance with the biosafety manual (Table 6).
— If minimal aerosols are produced, such as from a spilled specimen, cover the spill with absorbent paper towels and flood with tuberculocidal disinfectant. Leave the laboratory until at least 99% of airborne particles have been removed (Table 6), as determined by the safety officer. Let the disinfectant stand on the spill until re-entry. Disinfect floors and countertops.
— In the event of a major aerosol-producing spill or breakage, such as a liquid culture containing M. tuberculosis, immediately evacuate the laboratory. No one may reenter the area until enough air exchanges have occurred to remove 99%–99.9% of droplet nuclei from the environment, as determined by the safety officer using guidance in Table 6. The supervisor or safety officer may determine it is necessary to decontaminate the laboratory with formaldehyde gas or another agent. Appropriate respirator protection and other PPE must be worn to clean up spills or broken material. Do not pick up broken glass with hands.
4.8. Clean versus Dirty Areas of the Laboratory
- The laboratory work area must remain uncluttered and be arranged so that the flow of material is from a clean area to a dirty area of the laboratory.
- Inside the BSC, the immediate work area is to be covered with a tuberculocidal disinfectant–soaked pad to capture any drops or splatter that may result from manipulation of the specimen, pipettes, loops, tubes, slides or other instruments. If the gauze pad dries during work processes, rewet it.
4.9. AFB Blood Cultures
- Do not process blood submitted for mycobacteria analysis with routine blood cultures. The specimen of choice is whole blood. Process it in a BSL-3 laboratory.
- If it is necessary to process a routine blood culture for mycobacteria, conduct all work on a positive blood culture in a BSL-3 facility.
4.10. Instrumentation
- An aerosol-proof centrifuge with a safety-shield rotor is required for centrifugation of a specimen that may contain live tubercule bacilli.
- Decontaminate specimen tubes and place them into domed O-ring–sealed safety cups inside the BSC before transporting to the centrifuge, or place the decontaminated tubes into a rack and carry to the centrifuge. After centrifugation, keep the unopened tubes in the carrier until they are inside the BSC; then decant into a splashproof container.
- Install a sink equipped with either an automated motion-detecting faucet or knee or foot controls.
4.11. Testing
4.11.1. Rapid testing (direct molecular test kits)
- Perform all work in a BSL-3 laboratory and within the BSC.
- Once the sample on which a rapid test will be performed has been inactivated or genetic material extracted, further testing can be performed in a BSL-2 laboratory setting.
4.11.2. Molecular testing
- All work involving processing specimens suspected of containing tubercle bacilli and manipulation of mycobacterial cultures must be performed in a BSL-3 laboratory and within the BSC.
- Once the sample on which a molecular test will be performed has been inactivated or genetic material extracted, further testing can be performed in a BSL-2 laboratory setting.
5. Autopsy/Necropsy, Surgical Pathology
- Autopsy (human cadaver examination). The infectious and hazardous risks to the laboratory worker performing an autopsy are higher than those for any other health-care professionals because of the procedures used, the population being assessed, and performance of work in an open area. Use of scalpels, saws, and needles as well as exposure to sharp objects within the body, bone fragments, fractured metal, and/or needles, can result in cuts and percutaneous injuries. Manipulation of large organs that results in body fluid and blood splashes, and use of instrumentation such as hoses and saws create aerosols in an open area that can result in inhalation, direct contact, or contact with contaminated items in the environment. Those involved in the autopsy directly as well as others in the room are at risk for exposures. Other potential safety risks include exposure to chemicals such as formalin, therapeutic radiation beads, and retained electrical hardware. An alert and well-trained worker, good facility design, optimally fitting and user-friendly personal protective equipment (PPE), appropriate surgical procedures during manipulation of the body and dissected material, and subsequent disinfection and sterilization procedures are all critical in minimizing biosafety risk during autopsy and embalming.
- Necropsy (animal cadaver examination). The risk of laboratory-acquired infection is very different when working with human cadavers (where infectious agents in essentially all the cadavers are infectious to humans) versus animal cadavers (where infectious agents in most cadavers are not human pathogens). Regardless, animal cadavers can harbor zoonotic agents, and risk assessment to determine whether zoonotic infectious agents may be present in a cadaver, as outlined in Section 12, is critically important for establishing appropriate animal necropsy biosafety procedures. The guidelines in this section are combined biosafety best practices for both human autopsy and human surgical pathology and animal necropsy and veterinary surgical pathology. When necessary, biosafety guidelines specific for human or animal diagnostic laboratory settings are highlighted.
5.1. Autopsy/Necropsy–Associated Infections
The source of most laboratory-acquired infections and hazardous exposures that occur during autopsy/necropsy is unknown, and all autopsies and necropsies are to be considered risky (1,56).
- Human autopsy facilities would function safest at biosafety level 3 (BSL-3) for optimal protection of those involved directly with the autopsy and for personnel in the surrounding area (1,56–58). If a BSL-3 facility is not available, autopsies can be performed using the barrier precautions of BSL-2 plus the negative airflow and respiratory precautions of BSL-3 (56).
- Animal necropsy facilities can function at BSL-2 with an option for BSL-3 practices when warranted by a case-by-case risk assessment (considering, for example availability of Class II biological safety cabinet (BSC), downdraft necropsy tables, and appropriate PPE, such as eye and face protection). Only if a risk assessment indicates a high probability for the presence of a high-consequence livestock pathogen (USDA livestock select agent or toxin [See Section 12.1]) would BSL-3 facilities be required.
- The Medical Director, or in the case of animal necropsy, the attending pathologist, is responsible for risk assessment and for consideration of limited autopsy/necropsy procedures and subsequent acceptable risk level to personnel before each autopsy/necropsy (1,2,56,59)
- Select a staff member to be trained in safety procedures, and give this person oversight of safety procedures and risk analysis in the pathology suite.
5.1.1. Bloodborne pathogens
Human-health–care workers involved in performance of autopsies are at high risk for occupationally acquired bloodborne pathogens because of both the injuries sustained and the population undergoing autopsy. Transmission risk is highest per exposure for hepatitis B virus, then hepatitis C virus and human immunodeficiency virus, respectively. These infections have been documented from autopsies as well as during embalming (1,2,56,60–62).
5.1.2. Other infections
Specific data for other bloodborne pathogens, such as cytomegalovirus, are lacking, but infectious transmission is possible and risk may be higher especially for pregnant (serologically negative) or immunocompromised workers. Assess persons at higher risk for infection on a case-by-case basis and allow them to consent to participating in the autopsy only after being counseled (2,63).
5.1.3. Infectious aerosols
Autopsies/necropsies of cadavers with suspected zoonotic agents generate potentially infectious aerosols. Although Mycobacterium tuberculosis is the prototypical pathogen most noted to be transmitted by aerosolization, persons who had meningococcemia, anthrax, rickettsiosis and legionellosis are other examples. Manipulation of infectious tissue can result in both airborne particles in a size (<5 µm) that floats on air currents for extended periods and can subsequently reach the pulmonary alveoli and small-droplet particles (>5 µm) that settle more quickly. Contamination may occur from fluid-aspirating hoses, from spraying the cadaver, and from oscillating saws. The aerosols created stay within the autopsy area and can result in subsequent contact with mouth and eyes, inhalation, or ingestion and can contaminate inanimate surfaces such as computers, telephones and camera equipment (56,57).
5.1.4. Organisms that require additional safety practices
- No cases of autopsy-acquired Creutzfeldt-Jakob disease (CJD) have been documented. However, because the prion infectious particle cannot be rendered noninfectious by normal decontamination and sterilization methods, enhanced precautions are mandatory. Transmissibility of the prion is retained in formalin-fixed paraffin blocks (64–67).
- The only natural animal prion disease with known zoonotic infection potential is classical bovine spongiform encephalopathy (BSE). Necropsy guidelines for cattle with suspected BSE are published elsewhere and focus primarily on avoiding skin puncture, reducing splashes onto mucous membranes, decontaminating facilities and equipment, and disposing of carcasses (68).
5.1.5. Other biosafety exposures
- Cyanide, metallic phosphides and organophosphate pesticides
— Specific precautions are required and may include use of a fume hood or class II type B2 BSC that is ducted to the outside, fume respirators, limiting the autopsy/necropsy, and limiting the time of exposure (56). - Diagnostic radioactive beads or therapeutic scans
— Consult the radiation safety officer of record for recommended appropriate measures for limiting the exposure of radiation, transferring the body, and postexposure testing of personnel. - Electrical and other hardware hazards (pacemakers, indwelling catheters)
— Deactivate pacemakers before the autopsy continues. Discharge of electrical current is possible when defibrillators are present. If hardware is present, it should be noted and then removed so as not to cause cutting injuries during the autopsy.
5.1.6. Reporting to the mortician
Report known bloodborne pathogens or other suspected aerosolization danger to the mortician and others potentially handling the body to limit subsequent transmissions that may occur during transport or embalming (69).
5.1.7. Necropsy remains of animals
Dispose of animal cadavers with potential zoonotic infectious agents by appropriate decontamination (e.g., incineration, alkaline digestion or other methods), and do not return them to animal owners for private burial.
5.2. The Autopsy/Necropsy Suite
5.2.1. Inspect the body/carcass
- Search for implanted items retained after death. These are to be noted and clamped or covered before transport to reduce body fluids oozing from the body. Clean the body of visible bloody/body fluids. Cover the autopsy table with a plastic sheet to retain the majority of fluids, or alternatively, use tables with drains so that fluids may be collected in buckets or floor drains.
- Appropriately dispose of fluids and tissues from necropsy of animals with suspected zoonotic agents using methods that provide adequate decontamination, depending upon the specific suspected infectious agent (e.g., incineration, rendering, composting).
5.2.2. Safety guidelines for the suite
- Use the universal biohazard symbol to mark the autopsy suite as a biohazard area at the entrance.
- Secure access to the autopsy suite, and grant access only to those personnel trained in the biosafety procedures specific to this area.
- Protect vacuum hoses with liquid disinfectant traps and HEPA filters or their equivalent.
- Use hand saws whenever possible to reduce aerosols. Moisten bone before cutting. The pathologist may choose to use oscillating bone saws with a vacuum attachment; use these in a closed area if possible. These vacuum attachments are difficult to keep clean and are to be handled as a potential risk of infection. Cover any subsequent jagged edges of exposed bone with towels. When cutting the skull during autopsy when prions are suspected, bag the head.
- Do not leave used needles on the table. Do not detach or resheathe needles. Discard the whole unit into sharps containers. Make sure that sharps containers are available in the work area, and inspect them periodically to ensure that they are never more than two-thirds full. Seal off and replace them when they reach this level.
- Limit the number of personnel working on the human body at any given time to the prosector and/or physician and circulator. Allow only one person to cut at a given time. The same number limitation should apply to small animal necropsy. Large animal necropsy generally requires multiple prosectors working together in a way that will avoid accidental lacerations.
- Prepare multiple scalpels before autopsy so blade changes while hands are slippery and contaminated can be avoided. Alternatively, change outer gloves before changing blades. Use blunt-ended scissors when possible instead of scalpels, and use a magnet to pick instruments from the table if they become slippery.
- Do not pass sharp objects such as scalpels or scissors to another person. Place them on the table for another person to pick up.
- Place specimen containers (e.g., blood culture bottles) on a clean surface for inoculation. Use a rack if possible. Do not hold in the hands while inoculating.
- Examination of organs in the body and evisceration technique must be considered so as to limit exposure to blood, body fluids and cuts.
- For unfixed tissue that will be removed from the autopsy table, do the following.
— Place on a tray or in a bucket to avoid splashing or dripping fluids.
— After examination, cutting, and/or photography, return the tissue to the autopsy table to be replaced in the body and/or fixed.
— Place specimens that will be submitted for culture or other laboratory tests in a primary container that is surface decontaminated on the outside and then placed into a secondary leakproof container and labeled as biohazard.
— Large organs will have to be removed and cut into multiple sections (breadloaved) so that adequate permeation of the tissue for fixation will occur.
— Unfixed tissue that will not be returned to the body is considered biohazard waste and is to be kept to a minimum and subsequently disposed of in a manner that will allow appropriate decontamination.
— For autopsy, either suture or staple the body closed. Hold skin flaps with forceps, not hands, when suturing.
— Review of any unfixed tissue requires use of the same PPE as that used in the autopsy. - Use hands-free or foot-activated recording devices during dictation, and a hands-free speaker phone to minimize contamination of inanimate surfaces.
- Provide a hands-free sink at the exit for washing.
- Provide an eyewash station and shower (2).
5.3. Chemicals (Formaldehyde)
Formaldehyde (3.7%–4.0%) used for specimen preservation is the most common toxic chemical to which autopsy workers are exposed. The chemical is volatile and toxic and causes irritation to the eyes, mucous membranes, and skin and is associated with increased risk for all cancers. Occupational Safety and Health Administration (OSHA) regulations specify an exposure limit of 0.75 ppm as an 8-hour time-weighted average, and 2.0 ppm for short-term (15-minute) exposures (70). If formaldehyde can be detected by smell, it likely means exposure is occurring at a concentration beyond acceptable limits.
Limit exposure to formaldehyde in the following manner.
- Cover all specimen buckets where organs may be deposited for fixation.
- Collect discarded formalin-soaked towels and other formalin-soaked waste in a bag at the grossing table. Periodically spray a formalin-neutralizing agent on the waste as it is filled. Seal off the bag when it is filled.
- Discard bagged formalin-soaked towels and other waste in a lined container that can be opened and closed with a foot pedal.
- Cut large fixed organs in a fume hood or downdraft table.
- Monitor workers and resident pathologists with formaldehyde monitoring badges for 8-hour periods, and at least 15-minute periods periodically, to assess formaldehyde exposure.
- Ensure that tissue grossers are competent in proper tissue grossing technique.
5.4. Spills
- Use neutralizing, absorbent mats for small spills. Neutralizing reagents provide a convenient, cost-effective method for disposal of hazardous formaldehyde, glutaraldehyde, and other aldehyde solutions. They convert hazardous aldehydes into a nonhazardous, noncorrosive, nontoxic polymer and water. The polymer produced is not a hazardous waste, as defined by U.S. Title 40 Code of Federal Regulations (71). These neutralizing agents tend to reduce disposal costs and contribute to a safer work environment. In some cases, after formaldehyde waste treatment with crystal products, the resulting solid waste may be discarded in approved laboratory solid waste streams.
- Wear appropriate protective gloves and protective clothing to prevent skin exposure. Wear protective eyeglasses or chemical safety goggles or use full-face shields as described by OSHA's eye and face protection regulations (72).
5.5. Protective Equipment
5.5.1. Safety equipment
- Biological safety cabinets (2,56) are not common in autopsy suites because of their limitations in accommodating the volume and size of material being manipulated.
- Use necropsy facilities that have class II BSCs when practical (for small animals) for necropsies of cadavers with suspected zoonotic agents, as indicated by a case-by-case risk analysis. However, because necropsy of large animal cadavers with suspected zoonotic agents is not practical in BSCs, use PPE, engineering controls, and procedures that have been specifically developed for clinical laboratories.
- For optimal protection when there is a known risk of exposure to bloodborne pathogens and to agents transmitted by aerosols, all autopsy/necropsy facilities should be able to use BSL-3 work practices and physical containment or reference the work to a facility that does (56,59).
- Provide unidirectional airflow from clean areas to dirty areas.
- Ensure that the room is under negative pressure relative to other surrounding rooms, with 11–12 air exchanges per hour.
- Air can be directly vented to the outside or recirculated into the room through HEPA filters, but do not allow the air to recirculate into any clean surrounding areas because this has been associated with outbreaks of M. tuberculosis infection.
- In the event of redesign of the autopsy/necropsy suite, consider placement of a BSC to mitigate exposure to infectious tissues (56,73).
5.5.2. PPE for autopsy/necropsy personnel
- Clothing
— Fluid-resistant (surgical) jumpsuit or shirt and pants that cover from neck to feet and arms
— Waterproof apron and waterproof sleeves
— Closed-toe shoes covered with fluidproof shoe covers or booties
— Surgical cap or hood bonnet that covers head entirely (autopsy only) - Protection from splashes and aerosols
— For general autopsy, or for necropsy when a risk assessment indicates a high likelihood for zoonotic agents in the cadaver and the necropsy cannot be conducted in a BSC, use a transparent face shield that covers eyes, mouth and neck, and a fit tested N95 respirator. Eye glasses and contact lenses alone are not adequate for protection.
— Persons unable to wear N95 respirators must wear powered air-purifying respirators (PAPRs).
— Surgical masks do not protect autopsy participants from inhaling airborne contaminants such as infectious respirable aerosols or hazardous chemical vapors. (56,57,69,73,74). - Gloves
— Double-gloving is recommended throughout the autopsy/necropsy, with glove changes every hour. Eight percent of gloves are punctured during autopsy, and about one-third of punctures remain undetected until after gloves are removed (56,75).
— For autopsy, select a cut-resistant glove of fine-woven steel to prevent cuts from bone and scalpels, and cover this with a rubber glove for slip resistance. Cut-resistant gloves do not protect from needle sticks.
— Select gloves specific to the particular task and circumstances. For example, use heavy gloves for chemical exposure, or gloves that resist accidental puncture if needles will be used.
— Change gloves immediately upon recognizing a puncture, and wash hands with soap and water before resuming the autopsy/necropsy. - Cover cuts or abrasions with waterproof occlusive bandages.
- Provide PPE for personnel present but not directly participating in the autopsy, i.e., medical students/observers. Observers are to use the same PPE as those participating in the autopsy/necropsy.
- All surfaces are to be considered contaminated.
5.6. Disinfection and Cleaning Procedures for Equipment and Instruments
5.6.1. Human autopsy
- Clean any spills of body fluids or tissues immediately. Cover the spill with paper towels and saturate the area with a disinfectant or a detergent solution, or use a mop followed by disinfection of the spill area with 1:10 bleach solution.
- For decontamination of the body after autopsy, wash the body with a detergent solution, followed by an antiseptic solution or diluted bleach. Place the body in a leakproof shroud, and label as biohazard. Direct notification of the mortician is appropriate in cases of known bloodborne pathogens or the presence of M. tuberculosis.
5.6.2. Human autopsy/animal necropsy
The following guidelines for disinfection and cleaning following an autopsy or necropsy apply to both types of procedures.
- Anyone involved with the body, room, or instrument cleanup must wear the appropriate PPE.
- Wash all work surfaces and floors with a detergent solution followed by disinfection with a 1:10 bleach solution or other suitable commercial disinfectant.
- Remove visible gross tissue from nondisposable instruments (knife handles, pans, aprons) before chemical disinfection or autoclaving. Place sharp objects in puncture-resistant containers and wash with detergent solution to remove any remaining gross tissue. Rinse with water and wet thoroughly with a 1:10 bleach solution or appropriate commercial disinfectant, the volume depending on amount of blood and body fluid present. Consider that bleach will corrode stainless steel and aluminum devices. Remove the disinfectant or bleach by rinsing with water.
- Place disposable items, paper products, aprons, sponges, and similar items, into a biohazard container or bag for terminal treatment or autoclaving.
- Place reusable clothing in a leakproof biohazard bag for laundering according to hospital protocol.
-
Wash reusable, nonlaunderable items such as aprons with a detergent solution, decontaminate with bleach solution, rinse with water and allow to dry before next use.
- No dirty items or anything used in the autopsy is to be removed from the room, including clothing and laboratory coats.
- Keep camera, telephones, computer keyboards, and other items as clean as possible, but consider them to be contaminated and handle them with gloves. Wipe the items with detergent solution and 1:10 bleach solution or appropriate disinfectant after each use.
- Wash hands with soap and water upon removal of gloves when exiting the room.
5.7. Waste management
5.7.1. Human tissue
Either incinerate all pathological waste, since this is considered hazardous material and is regulated by the U.S. Department of Transportation (DOT), or transport pathological waste to on-site or off-site treatment facilities in clearly labeled, dedicated, leakproof containers or carts that meet DOT requirements. DOT sharps waste containers need to be puncture-proof in addition to meeting these requirements. State, local, and regional regulations may also apply and need to be addressed.
5.7.2. Animal tissue
Dispose of all animal necropsy waste (tissues or postnecropsy cadaver) using an appropriate method as determined by the case-by-case risk analysis assessment (incineration, autoclaving and standard waste disposal, rendering, composting, cremation, private burial).
5.7.3. Other waste
Shred autoclave red-bag waste if appropriate. State, local, and regional regulations may also apply and need to be addressed. (See Section 3.5, Waste Management.)
5.8. Clean versus Dirty Areas
Clean areas might include an administrative area and bathrooms with showers. Air from these areas should be exhausted differently than from the autopsy suite (56,76,77). All other areas are considered dirty, and appropriate PPE is required.
5.9. Surgical Pathology
- Surgical pathology includes gross dissection and frozen sectioning. Histology and cytology are "hybrid" areas of autopsy and the clinical diagnostic laboratories. Each laboratory section has its own unique safety issues; data on standardized biosafety practice or on reported biosafety incidents are very limited. Documentation and research in surgical pathology safety are necessary for future guidelines and recommendations (78,79).
- Surgical pathology risks are associated with manipulating large amounts of fresh tissues from unknown infectious sources, which may result in puncture, cuts, and splashes of blood and body fluids, similar to the autopsy where fresh organs must be viewed at a grossing table and cut into sections thin enough for fixation (i.e., "breadloaved"). Other risks include the use of cryostat cutting equipment or freezing spray that generates infectious aerosols when sectioning frozen tissue, and exposures to large volumes of formaldehyde.
- Tissue that will be used for slide examination is fixed in formalin; subsequently, small tissue sections of interest are dissected with scalpels, placed in cassettes, replaced in specimen vats with formalin, and transported to histology.
- Histology then embeds the formalin-fixed tissue in paraffin and cuts these sections with microtome-bladed instruments to make slides for viewing using specific stains and immunochemistry. Although most formalin-fixed specimens are noninfectious, both M. tuberculosis and CJD agent can persist in transmissible form in formalin-fixed, paraffin-embedded material. Exposure to toxic formaldehyde is common.
- Cytology receives large- and small-volume body fluids, bone marrow samples, or needle aspirate specimens, most of which are received in fixative but others that must be processed by aliquoting or pouring off large-volume body fluids, which can result in splashing and spills. Procedures such as centrifugation and cytospin processing can produce aerosols. Air-dried slides can be a source of contamination until they are fixed and stained.
5.9.1. Specimen receiving and log-in
- Handle specimens with standard precautions, and wear gloves when receiving and accessioning (2,57). Receipt areas in all surgical pathology laboratories are considered dirty areas, and all exterior containers considered contaminated.
- Submit specimens, including skin and gastrointestinal biopsies, delivered from the operating room, autopsy, or from outside collection sites in leakproof containers or place in fixative specimen containers at the site of collection Place the specimen in a secondary leakproof bag or container and label as a biohazard. Place the requisition in an outside pocket where it will avoid contamination with the specimen.
- Leaking specimens or visibly contaminated specimens
Specimens may be contaminated with fresh tissue, blood, or formalin. Handling is based on whether the specimen is fresh or formalin-fixed.
— If the specimen is submitted in fixative and the secondary container is not leaking, tighten the specimen cap and place the primary container in a clean bag; wipe any formalin from the bench top.
— If the specimen is submitted as fresh tissue and the exterior container is leaking, place the specimen into another container and process it using appropriate PPE. Specimens are then to be transferred to a clean container and labeled.
— If the requisition is contaminated, discard it as biohazardous waste and replace it.
5.9.2. Work at the open bench
- Separate the log-in room and administrative areas from the grossing room if fresh tissue or cryostats are used in the same open area (56,76,77).
- Handling of fresh tissue
— Examine fresh tissue in a BSC if possible, or in a room separated from the rest of the surgical pathology grossing stations. Use an N95 mask and other PPE similar to autopsy conditions, depending on the volume of blood and likelihood of body fluid exposure (56). Fresh tissue should never be handled without gloves. PPE as used in autopsy procedures is advocated for handling fresh tissue (2,56,57,80,81).
— For human pathology, store fresh tissue not undergoing fixation or unable to be adequately fixed (e.g., teeth or foreign bodies) in a double, sealable, leakproof container; label as biohazard and store in a refrigerator or freezer (2).
— Standard precautions against bloodborne infections are to be used irrespective of the clinical history (79). - Frozen sections
— Frozen sectioning is performed on fresh tissue and is a high-risk procedure for infectious exposure. Freezing tissue does not kill organisms, and the use of the cryostat cutting blade creates potentially dangerous aerosols. Discuss the true clinical necessity for frozen sectioning with the surgical team.
— Although some cryostat instruments have a downdraft into the instrument, aerosols are dispersed into the room where the cutting takes place. Do not use freezing propellant sprays, which speed the freezing process by a few seconds and cause aerosolization of not only the tissue being frozen but also the tissues from previously cut specimens that are at the base of the instrument. Such procedures generate aerosol and droplet contamination, posing an infectious risk to all personnel in the area (56,79,82). The Clinical and Laboratory Standards Institute and others have recommended discontinuation of freezing sprays because they are not recommended by the manufacturers of cryostat instrumentation (2,79).
— Ideally, use cryostats in a closed room that has air vented directly to the outside or recirculated through a HEPA filter to avoid contamination to the rest of the surgical pathology suite.
— Certain cryostats have ultraviolet lights, but these are not a substitute for terminal cleaning of the instrument and have been ineffective in killing mycobacteria.
— In human pathology laboratories, gloves, face shield or goggles, and N95 mask must be worn when processing. - Bone cutting. See Autopsy Section 5.2.2.
- Fixed tissue
— Fix tissue in 10% formalin in a concentration 10 times the volume of tissue to ensure effective fixation and to reduce potential of infectious contamination. However, the viability of infectious organisms is dependent on a host of variables that have not been clearly identified, and fixed tissue has been shown to remain infectious (78). M. tuberculosis has been transmitted from fixed specimens as well as grown from fixed specimens, and CJD is not inhibited by the routine concentration of formalin. Grossing stations where formalin-fixed specimens are cut are designed for decreasing the fumes of formaldehyde but are not BSCs (2,57,83–85). Formalin-fixed specimens must be handled with gloves.
— Use standard precautions and appropriate PPE in all work with fixed tissue. Face shields are optimal for full face and neck protection if splashing with formalin is likely. Shields offer the most comfort for extended periods while allowing the user to wear prescription eyeglasses or to do dictation.
— Eyeglasses and contact lenses are not a substitute for eye protection.
— Goggles and fluid-resistant mask are an alternate choice but not preferred.
— Use face shields/goggles that can be decontaminated or that are disposable.
— Keep containers with tissues in formalin closed to reduce exposure fumes. - Cytology specimens
— PPE is dependent on the specimen. Gloves and laboratory coat are required for all specimens until slides are fixed and stained.
— Pour off or aliquot large-volume specimens with potential for splashing and/or aerosolization inside a BSC, and wear fluid-resistant clothing, apron, and two pairs of gloves.
— Open small-volume body fluids or aspirates submitted in tubes in a BSC, or use a splash guard or a face shield and aliquot with a disposable pipette rather than pouring to avoid splashing and spill.
— Handle specimens received in fixative with gloves because of the toxicity of the fixatives and possible external contamination (57). If slide preparation will use a cytospin preparation, the system must include a bowl with safety lid and outside cover.
— Consider all slides, impression smears, cytological preparations, and bone marrow smears as infectious until fixed and stained.
Use a safety centrifuge with safety cups with O-rings and sealable tops for centrifugation of fluids (51). - Decontamination
— Cryostat. Instrument shavings generated by cutting are considered contaminated. Collect accumulated cuttings and discard them as biohazardous waste. Defrost the instrument and decontaminate daily with 70% alcohol. Decontaminate weekly with a tuberculocidal disinfectant or after a known case of M. tuberculosis infection. Wear stainless steel mesh gloves while cleaning the microtome knives.
— Microtome. Consider a similar schedule as with cryostats.
— Consider all surfaces, computer, telephone, and counters as contaminated if any person using gloves touches these items. Disinfect equipment and bench tops daily.
— Remove gloves and wash hands with soap and water before exiting the various laboratory rooms.
5.9.3. Clean versus dirty areas of the laboratory
All of the surgical pathology specialty areas (cytology, histology, grossing or frozen section rooms) are considered dirty areas if fresh specimens or body fluids are received or processed in an open room (not in a BSC or separately vented area).
5.9.4. Tissue stains
Multiple staining procedures are performed in histology and cytology. The most common are included here. Some of these stains are prepared with ethanol and some with methanol, which can have an impact on management options for their waste. Provide material safety data sheets (MSDS) for each component in the laboratory.
- Hematoxylin stain
Hematoxylin stain is not hazardous under Environmental Protection Agency regulations. Drain disposal is recommended with the permission of local wastewater treatment authorities. Follow federal, state, and local regulations. - Giemsa stain
The preferred disposal method for Geimsa stain is incineration at a permitted hazardous waste treatment facility. Localities may restrict the amounts of alcohols that may be flushed down the drain; consult appropriate authorities. Ensure compliance with all local, state, and federal government regulations. - Wright stain
Wright stain contains methanol, a listed hazardous waste. Sewer disposal of listed hazardous wastes is not acceptable or permitted. The preferred disposal method is incineration. Ensure compliance with all government regulations.
5.9.5. Fixatives
- Formalin (HCHO)
— Formaldehyde may be purchased as a 37%–40% HCHO solution. The most common formaldehyde preparation is 10% formalin (3.7%–4% formaldehyde), which is available commercially. Diluting formalin or formaldehyde is discouraged to limit exposure. Local governments often restrict the amounts of aldehydes that may be flushed down the drain. Each laboratory must comply with all government regulations.
— Neutralization is the preferred disposal method for formaldehyde. Neutralizing reagents provide a convenient, cost-effective method for disposal of formaldehyde, glutaraldehyde, and other aldehyde solutions. They convert hazardous aldehydes into a nonhazardous, noncorrosive, nontoxic polymer and water. The polymer produced is not a hazardous waste, as defined by U.S. Code of Federal Regulations, Title 40 (71). These neutralizing agents tend to reduce disposal costs and contribute to a safer work environment. In some cases, after formaldehyde waste treatment with crystal products, the resulting solid waste may be discarded in approved laboratory solid waste streams. Before engaging in sewer disposal of neutralized formalin solutions, be sure to have formal approval of applicable local wastewater authorities.
— Wear appropriate protective gloves and protective clothing to prevent skin exposure. Wear protective eyeglasses, chemical safety goggles, or full-face shields, as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 (72) or European Standard EN166. Do not wear contact lenses when working with formalin. Maintain eye wash fountain and drench facilities in the work area. - Other pathology fixatives
— Fixatives are classified as regulated waste. OSHA's Standard, Bloodborne Pathogens, 29 CFR Part 1910, 1048 and 1030, mandates that pathology specimens be placed in secondary containers that are constructed to contain all contents and prevent leakage of fluids during handling, storage, transport or shipping (33,70).
— Label the secondary containers as biohazards, and close the container securely prior to removal to prevent spillage of contents during handling, storage, transport and shipping. - Xylenes and alcohols
— Xylene is categorized by EPA as a hazardous waste under the Resource Conservation and Recovery Act (RCRA) and has been assigned EPA hazardous waste no. U239. Waste xylene can be separated from dissolved paraffin by distillation, and commercially available recycling units can accomplish this separation effectively to produce reusable xylene. Xylene also may be disposed of by a properly permitted hazardous waste contractor if the xylene is contained in an organic metallic or organic laboratory pack that meets the requirements of 40 CFR 264.316 or 265.316 (71).
— Xylene that cannot be saved for recovery or recycling must be handled as hazardous waste and sent to an RCRA-approved incinerator or disposed in an RCRA-approved waste treatment facility. Processing, use, or contamination of this product may change the waste management options. State and local disposal regulations may differ from federal disposal regulations. Dispose of the container and unused contents in accordance with federal, state and local requirements.
— The disposal guidelines for alcohols (ethyl alcohol, isopropyl alcohol, methyl alcohol) are identical to those for xylene. - Testing for formalin and xylene exposure
Conduct periodic testing with chemical badges for an 8-hour period and at least one 15-minute period periodically to assess formaldehyde exposure for persons who are routinely exposed (e.g., technologists, tissue grosser, resident pathologists), and rotate among various areas so that every work station is assessed.
5.10. Engineering Controls and Facility Renovations
When updating or renovating autopsy and other areas of the anatomic pathology laboratory that process fresh tissue and body fluids, the following should be considered.
- Designing closed specimen-receiving/administrative areas for receiving specimens if BSCs are not available for processing fresh specimens or if cryostats are in the same area as administrative personnel.
- Providing rooms with negative air flow relative to surrounding rooms.
- Installing a BSC for processing fresh tissues.
- Providing a separate room for cryostats and bone saws apart from the remainder of the surgical grossing suite.
5.11. Creutzfeldt-Jakob Disease
Special precautions for autopsy and autopsy suite decontamination, brain-cutting, and histologic tissue preparation procedures are required when processing cases of possible CJD (1,56,65,67,86).
5.11.1. Autopsy
Perform autopsies using BSL-2 precautions augmented by BSL-3 facility ventilation and respiratory precautions. Wear standard autopsy PPE. Limit the autopsy to brain removal. Restrict participants to only those who are necessary. Double-bag the brain and place it in a plastic container for freezing or fix it in 3.7%–4% formaldehyde after sectioning. Formaldehyde fixation occurs for 10–14 days before histologic sections are collected.
5.11.2. Histologic preparations
- Prions remain transmissible after formalin fixation and paraffin embedding. (56,64,66,86).
- PPE includes, gloves, laboratory coat, apron, and face protection.
- Before histologic slide preparation, small blocks of brain tissue <5 mm thick are soaked in 95%–100% formic acid for 1 hour, followed by soaking in fresh 4% formaldehyde for at least 48 hours. Such tissue may be processed either by hand or by machine. Blocks and slides may then be handled routinely. If the laboratory prefers to process by hand, the procedures are the same as for untreated tissues.
- If the tissue has not been pretreated with formic acid before embedding, double gloves and eye protection are to be worn at all times, including during sectioning. All solutions, including water washes, are collected and treated with equal volumes of fresh, undiluted bleach or 1N NaOH for 60 minutes. Disposable supplies are either incinerated or autoclaved at appropriate temperature and pressure (1,56,86). Glassware and tools are generally soaked in undiluted bleach for 1 hour and then rinsed well. If appropriate for the tool, they may autoclaved at appropriate temperature and pressure. Equipment that cannot be soaked or autoclaved should be wiped with undiluted bleach and allowed to sit for 30 minutes.
- Care should be taken to collect all scraps of paraffin and unused sections on a disposable sheet. The microtome itself may be wiped with bleach or sodium hydroxide solution, but it cannot be thoroughly decontaminated. If frequent possible CJD cases are handled, laboratories may wish to dedicate an old microtome to this purpose.
- All waste is to be collected and disposed. Discard the knife blade with the sharps.
- Slides containing untreated tissue are stained by hand in disposable specimen cups or Petri dishes. The slides are labeled as infectious CJD and stored/filed in an appropriately labeled container. The sectioned block is sealed with paraffin.
- Tissue remnants are discarded as infectious hospital waste for incineration. Fluids are diluted 1:1 with undiluted bleach for 1 hour before disposal.
- Dedicated equipment may be required depending on local jurisdictions.
6. Parasitology Laboratory
Exposure to infectious parasites during diagnostic procedures may result from handling specimens, drawing blood, performing various types of concentration procedures, culturing organisms, and conducting animal inoculation studies. Relevant parasites and their possible routes of infection are listed in Table 7 and Box 1. Table 8 contains information on resistance to antiseptics and disinfectants.
6.1. Specimen Receiving and Log-In/Setup Station
- Fresh specimens (feces, other gastrointestinal tract specimens, urine, blood, tissues, cerebrospinal fluid, other body fluids, arthropods) represent a potential source of infectious parasites.
- Safety precautions include proper labeling of fixatives; designating specific areas for specimen handling (biological safety cabinets may be necessary under certain circumstances); proper containers for centrifugation; acceptable discard policies; appropriate policies for no eating, drinking, or smoking within the working areas; and, if applicable, correct techniques for organism culture and/or animal inoculation.
- Collect or transfer every specimen into a leakproof primary container with a secure lid (avoid snap-top closure).
- Use disposable plastic bags with separate pockets for the requisition slip and specimen when possible (2,87).
6.1.1. Leaking containers
- Visually inspect all specimen containers for leakage.
- Contaminated primary containers must be decontaminated before further manipulation. In some circumstances, it might be necessary to transfer the contents to a clean container or collect another specimen before submission to the testing area.
- Discard contaminated requisitions as biohazardous waste and replace them.
6.1.2. Loose caps
- Blood specimens are submitted in tubes, usually Vacutainer tubes (with either lavender or green tops). If the stopper appears to be loose or there is evidence of blood on the outside of the tube, push the stopper into the tube for a secure fit, and request the sender to submit another specimen. If the specimen cannot be repeated, decontaminate the outside before processing the specimen.
6.2. Stains and Reagents (88,89)
6.2.1. Trichrome stain
- Wheatley's modification of the Gomori tissue trichrome stain is considered nonhazardous waste but must be disposed of in accordance with federal, state and local environmental control regulations.
- Drain disposal is recommended with the permission of local wastewater treatment authorities.
6.2.2. Hematoxylin stain
- Hematoxylin stain is not hazardous under Environmental Protection Agency (EPA) regulations.
- Drain disposal is recommended with the permission of local wastewater treatment authorities. Canadian disposal regulations generally parallel those in the United States.
- Follow federal, state and local regulations.
6.2.3. Iodine
- Dispose of all liquid and iodine-contaminated parasitology-related material in Department of Transportation (DOT)–approved waste containers.
- Incineration for liquids is the suggested method of disposal.
- Comply with all federal, state and local regulations for disposal.
6.2.4. Acid-fast stains (modified)
- Dispose of container and unused contents in accordance with applicable federal, state and local requirements.
- State and local disposal regulation may differ from federal disposal regulations.
6.2.5. Giemsa stain
- The preferred disposal method is incineration at a permitted hazardous waste treatment facility.
- Localities may restrict the amounts of alcohols that may be flushed down the drain. Ensure compliance with all government regulations.
6.2.6. Wright stain
- The preferred disposal method is incineration in an approved facility.
- Localities may restrict the amounts of alcohols that may be flushed down the drain. Ensure compliance with all applicable government regulations.
6.2.7. Formalin (HCHO)
- Formaldehyde is normally purchased as a 37%–40% HCHO solution; however, for dilution, it should be considered to be 100%. Two concentrations are commonly used: 5%, which is recommended for preservation of protozoan cysts, and 10%, which is recommended for helminth eggs and larvae. Although 5% is often recommended for all-purpose use, most commercial manufacturers provide 10%, which is more likely to kill all helminth eggs. The most common formalin preparation is 10% formalin.
- Incineration is the preferred disposal method for formaldehyde.
- Local governments often restrict the amounts of aldehydes that may be flushed down the drain. Each laboratory will need to comply with all government regulations.
- Use neutralizing reagents to dispose of hazardous formaldehyde. Commercial compounds and safe aldehyde solutions should be employed. They convert glutaraldehyde and other more hazardous aldehydes into a nonhazardous, noncorrosive, nontoxic polymer and water. The polymer produced is not a hazardous waste, as defined by U.S. Title 40 CFR 261.24(a) (71). These neutralizing agents tend to reduce disposal costs and contribute to a safer work environment.
- In some cases after formaldehyde waste treatment with crystal products, the resulting solid waste may be discarded in approved laboratory solid waste streams. Before engaging in sewer disposal of neutralized formalin solutions, get formal approval from the local wastewater treatment authority.
- Wear protective gloves and protective clothing to prevent skin exposure. If a biological safety cabinet (BSC) is not being used when removing specimens from formalin-containing vials, wear protective eyeglasses or chemical safety goggles, and use face shields as described by the Occupational Safety and Health Administration's (OSHA) eye and face protection regulations in 29 CFR 1910.133 (72) or European Standard EN166.
- Maintain American National Standards Institute (ANSI)–specification eyewash station and drenching facilities in the work area.
6.2.8. Mercury-based fixatives
- The use of mercury in chemical analysis can be phased out in most, if not all parasitology laboratories. Excellent, safe commercial products can now substitute for fixatives, such as Zenker solution, histologic fixatives, and other products that previously required the use of mercury. Some substitutes, such as copper, tin, zinc, and chromium compounds also have some risk, but less than that associated with mercury.
- Recycling companies may or may not accept mercury-containing waste, including laboratory solutions. The companies differ in the type of mercury waste accepted and the transportation requirements (90–98).
6.2.9. Zinc-based fixatives (containing formalin)
- Zinc-based fixatives are generally not considered hazardous waste by EPA (under the Resource Conservation and Recovery Act [RCRA]) and are now accepted as good substitutes for those containing mercury-based compounds These mercury substitutes must not be disposed of with solid waste unless they have been neutralized with one of the commercially available detoxification products (71,99).
- Dispose of materials in accordance with federal (40 CFR 261) (71), state, and local requirements.
- The recommended cleansing agent is water.
- Zinc formalin is toxic because of its formaldehyde content. Dispose by using a licensed waste hauler. Do not mix waste streams unless instructed to do so by your waste hauler. Some wastewater treatment authorities may grant permission for drain disposal of limited amounts if the zinc content is <600 ppm.
- Zinc formalin is recyclable and can be neutralized with commercially available detoxification products.
- Zinc-based fixatives are now available that do not contain formalin. Universal fixatives such as Total-Fix do not contain mercury, polyvinyl alcohol, or formalin and can be used for concentration, permanent stained smears, and fecal immunoassays, except for the Entamoeba histolytica and the Entamoeba histolytica/E. dispar group, which require fresh or frozen specimens for testing. Giardia and Cryptosporidium spp. tests can be run on fresh, as well as preserved specimens. Dispose of materials in accordance with federal (40 CFR 261) (71), state, and local requirements.
6.2.10. Copper-based fixatives (containing no formalin)
- Many localities restrict the amount of copper compounds that may be flushed down the drain.
- Ensure compliance with all government regulations.
6.2.11. Xylene and alcohols
- Xylene is categorized as a hazardous waste under RCRA and has been assigned EPA hazardous waste no. U239. Use of one of the current commercial substitutes for xylene will avoid the need to address the special handling required for hazardous waste management described here.
- Xylene may be disposed of in an organometallic or organic laboratory pack that meets the requirements of 40 CFR 264.316 or 265.316 (71). Xylene can be distilled by using a variety of commercially available recycling units to produce reusable xylene.
- Xylene that cannot be saved for recovery or recycling is to be handled as hazardous waste and sent to an RCRA-approved incinerator or disposed in an RCRA-approved waste facility.
- Processing, use, or contamination of this product may change the waste management options. State and local disposal regulations may differ from federal disposal regulations. Dispose of the container and unused contents in accordance with federal, state and local requirements.
- The disposal guidelines for alcohols (ethyl alcohol, isopropyl alcohol, methyl alcohol) are identical to those for xylene.
6.3. Working at the Bench
- In general, the same precautions and practices that are used in microbiology laboratories are applicable to diagnostic parasitology procedures, particularly regarding aerosol transmission (2,33).
- Guidelines for specimen collection and processing include standard precautions as well as use of recommended equipment according to approved methods (e.g., BSCs, fume hoods, centrifuges, sharps, glassware) (2,100,101).
6.4. Personal Precautions
6.4.1. Biological safety cabinet versus fume hood
- Although use of a fume hood is not mandatory when processing stool specimens containing formalin, use an OSHA-compliant formalin monitoring program.
- Even with the substitution of dehydrating reagents other than xylene, fume hoods are recommended in order to eliminate fecal and solvent odors. A small, table-top model is acceptable.
- A BSC is not required for processing fecal specimens in the parasitology laboratory; however, some laboratories use Class I (open-face) or in many cases, a Class II-A2 (laminar-flow) BSC for processing all unpreserved specimens.
- A BSC is recommended, especially if the laboratory is processing fresh specimens or performing cultures for parasite isolation (Table 7).
6.4.2. Personal protective equipment
- Use appropriate hand hygiene (washing and antiseptics) for laboratory work in a diagnostic parasitology laboratory.
- Wear disposable gloves of latex, vinyl, or nitrile during accessioning and processing of all specimens for parasitologic examination, especially when handling blood, body fluids, and stool specimens (100,101); this recommendation applies whether the clinical specimens are fresh or are submitted in fecal preservatives. Always wash hands after glove removal.
6.4.3. Immunization
- The Advisory Committee on Immunization Practices recommends that all persons whose work-related activities involve exposure to blood or other potentially infectious body fluids in a health-care or laboratory setting receive hepatitis B vaccine. Booster doses are not recommended (102).
- Documentation, including signed statements and records of hepatitis B vaccination or declination, must be kept.
6.4.4. Disinfection
General recommendations for the microbiology laboratory are sufficient for use in the diagnostic parasitology section; these would include guidelines for disinfection of countertops, telephones, computers, equipment, and hands-free telephones.
6.5. Dirty versus Clean Areas of the Laboratory
General guidelines for the microbiology laboratory also apply for the parasitology section of the laboratory. No special recommendations are necessary.
6.6. Instrumentation
Safety requirements for the use of instruments are the same as those used for a general microbiology laboratory and are primarily involved with specimen handling.
6.7. Antibody and Antigen Parasitology Testing
Safety requirements for antibody and antigen testing are the same as those used for a general microbiology or immunology laboratory and are primarily involved with specimen handling.
7. Mycology Laboratory
Although not a strict requirement, it is recommended that mycology laboratories that culture for filamentous fungi and manipulate those organisms be separate and isolated from the main microbiology laboratory with negative air pressure moving into the room from the main laboratory. Direct access to a Class II biological safety cabinet (BSC) is critical for this activity whether mycology work is conducted in a separate room or in an isolated section of the main laboratory. Most mycology diagnostic work can be conducted in the biosafety level (BSL)-2 laboratory.
7.1. Specimen Receiving and Log-In/SetUp Station
7.1.1. Leaking containers
Guidelines for the general microbiology laboratory apply also for the mycology laboratory. No special recommendations are necessary.
7.1.2. Visible contamination on outside of container
Guidelines for the general microbiology laboratory apply also for the mycology laboratory. No special recommendations are necessary.
7.1.3. Loose caps
Guidelines for the general microbiology laboratory apply also for the mycology laboratory. No special recommendations are necessary.
7.2. Stains and Disposal
7.2.1. Gram stain
The Gram stain is not the optimum stain for fungus, but if used particularly for yeast, the same guidelines that apply to bacteriology/clinical microbiology are followed for mycology.
7.2.2. Mycology stains
- Calcofluor white is not considered a hazardous substance; no special safety measures are required. Use routine laboratory procedures: i.e., after skin contact, wash with soap and water; after eye contact, rinse eye well with water; seek medical attention if illness is reported after inhalation or swallowing.
- Follow accepted laboratory procedures applied to infectious materials for handling and disposing of slides. Fungal cells stained with calcofluor white can remain viable.
- If potassium hydroxide (KOH) is used with the calcofluor white, more stringent precautions must be taken. Refer to the material safety data sheet for guidance (103).
— KOH 10%–15% solution is corrosive. Handle it with care. KOH may cause burns or irritation to skin, eyes, and respiratory tract. Avoid eye/skin contact and inhalation or ingestion. Use gloves and eye protection if there is a danger of splashing or aerosol formation.
— KOH is not listed as hazardous waste under the Resource Conservation and Recovery Act (RCRA). The small amount used on a slide with calcofluor white can be disposed of with slides containing infectious materials. If disposing of larger amounts, do so in accordance with federal, state and local requirements (104–106). - Lactophenol cotton blue (lactophenol aniline blue or Poirrier's blue)
— In amounts commonly used in a clinical laboratory, lactophenol cotton blue is acidic; avoid contact with skin, eyes, and clothing.
— Rinse thoroughly with water if spilled.
— For fungal slides stained with the fluid, follow accepted laboratory procedures for handling and disposing of infectious materials.
— If larger amounts are to be produced or disposed of, the product is considered more hazardous, i.e., toxic by inhalation and contact with the skin, and especially if swallowed. Use only with adequate ventilation.
— Dispose as hazardous waste in accordance with federal, state, and local regulations (107–109). - India ink
— No special personal protection is required under normal use conditions; however, the yeast cells can remain viable.
— India ink is not a regulated hazardous waste. Dispose in accordance with applicable federal, state, and local regulations, and dispose of slides safely (110). - Acid-fast stain
Guidelines for the tuberculosis laboratory apply also for the mycology laboratory. No special recommendations are necessary. - Gomori methenamine silver stain
— Ingredients are toxic, corrosive, and harmful; avoid contact with skin and eyes.
— Use with adequate ventilation; do not inhale.
— Dispose of as a hazardous waste in accordance with applicable federal, state, and local regulations (111). - Giemsa stain
— Because of its methanol content, Giemsa stain is toxic by inhalation, absorption, or ingestion. Protective gloves and safety goggles are not required but are recommended.
— Dispose of in accordance with federal, state, and local regulations. The preferred method is incineration at an approved facility (112).
7.3. Culture Reading at the Bench (1,113)
- A separate, closable room for mycology activities is recommended but not required.
- Conduct all culture manipulations in a BSC whether in a separate room or within a designated space in the open microbiology laboratory. All mould colonies (filamentous, fuzzy, cottony) must be handled in a Class II BSC. This applies as well to moulds growing on bacteriology plates. See section 7.6 for further information.
- Use shrink seals or tape to seal petri plates (especially if mould begins to grow) in order to prevent accidental opening and spread of hyphal segments, conidia, or spores.
- Never use petri plates if Coccidioides immitis is suspected or if a filamentous culture is to be mailed or otherwise transported to another laboratory. Use slants in screw-cap tubes.
— Observe all plates and slants for growth before opening. - In general, cultures growing yeast-like colonies can be read on the open bench in a BSL-2 laboratory; but if the isolate is suspected of being Cryptococcus neoformans (moist, mucoid colonies) or any dimorphic fungus, move it to a Class IIA2 BSC.
- Never sniff a fungal culture to determine whether it has an odor. Do not open plates containing moulds on the open bench, even if it is a bacteriology work station.
7.4. Personal Precautions
7.4.1. Biosafety cabinet
- The Class IIA2 BSC is recommended for some mycology work; i.e., all moulds (fuzzy, wooly, cottony, powdery, or velvety) must be handled in the BSC, never on the open bench.
- For mycology, follow the same BSC guidelines that apply to bacteriology.
7.4.2. Personal protective equipment
- Guidelines for the general microbiology laboratory apply also for the mycology laboratory.
- Wear gloves and remove watches and bracelets when manipulating a mould culture having the possibility of being a dermatophyte. When the task is completed, wash hands and wrists well.
7.4.3. Disinfection
Recommendations for the general microbiology laboratory are sufficient for use in the mycology laboratory; these include guidelines for disinfection of countertops and items such as telephones, computers, equipment, and hands-free telephones.
7.4.4. Decontamination and disposal of laboratory waste
- Follow the same guidelines that apply to clinical microbiology.
- If an autoclave is unavailable and medical waste is handled off-site, open plates and tubes containing Coccidioides spp. and completely immerse them in a 1:10 bleach solution overnight before disposing.
7.5. Clean versus Dirty Areas of the Laboratory
Guidelines for the general microbiology laboratory apply also for the mycology laboratory.
7.6. Select Agents and Pathogenic Moulds
- Handle all mould-like colonies in a BSC.
- Make a wet preparation of all cultured moulds before setting up a slide culture in order to detect structures that may indicate the possibility of the isolate being a highly pathogenic systemic fungus (113).
- Refrain from setting up slide cultures of isolates that on wet preparation are suggestive of Histoplasma capsulatum, Blastomyces dermatitidis, Coccidioides immitis, C. posadasii, Paracoccidioides brasiliensis, Penicillium marneffei, or Cladophialophora bantiana. Make every attempt to identify them by well-prepared wet preps and DNA probes if available (113).
- If a laboratory-isolated organism is identified as H. capsulatum, B. dermatitidis, Coccidioides spp., or Paracoccidioides brasiliensis, BSL-3 practices and facilities are recommended for handling mould-form cultures and environmental samples likely to contain infectious conidia (1).
- Coccidioides spp. are the only fungi currently classified as a select agent requiring registration with CDC/U.S. Department of Agriculture for possession, use, storage and/or transfer (1,114). This may change in the future.
- To decontaminate Coccidioides isolates when autoclaving is unavailable (i.e., when culture plates are picked up by a contractor for off-site autoclaving), soak the plates and tubes, overnight in a fresh 1:10 bleach solution that completely immerses the opened tubes and plates prior to disposal (see Section 3.5.3).
7.7. Blood Culture Bench
- Guidelines for the general microbiology laboratory apply also for the mycology laboratory.
- Plates growing mould-like colonies must be examined in a BSC, not on the open bench.
- If a small-celled yeast (~3 µm) is detected, consider the possibility of H. capsulatum, and handle under BSL-2 conditions in a Class 2 BSC (113).
7.8. Instrumentation
Instruments used for mycology studies are most commonly those for continuously monitored blood culture and for yeast identification. Follow the same guidelines that apply to bacteriology/clinical microbiology.
7.9. Rapid Testing (Kits)
- Apply the clinical microbiology guidelines for monomorphic yeasts in mycology.
- If the isolate is a mold, it must be handled in the BSC.
7.10. Molecular Testing
Follow the clinical microbiology safety guidelines for mycology with the additional advisory that mold isolates must be handled in a BSC during extraction of nucleic acids.
8. Virology Laboratory
8.1. Specimen Processing and Log-In Bench
8.1.1. Biohazards associated with specimen receiving and log-in
The clinical virology laboratory receives a wide variety of clinical specimens for virus detection. Because the infectious nature of this material is largely unknown, special care must be taken to prevent contamination of personnel, the environment, and other clinical specimens.
- Handle all clinical specimens under biosafety level (BSL)-2 conditions (1). Some special pathogens and select agents must be handled under more stringent biosafety conditions.
- Wear a laboratory coat, gloves, and eye protection whenever clinical specimens are handled. This includes the specimen receiving and log-in processes.
- Conduct culture setup and all other specimen manipulations in a Class II or higher biological safety cabinet (BSC).
- Compile a "Do Not Process/Do Not Test" list containing the names of suspect agents that are not to be opened or processed (e.g., smallpox, Ebola virus, vesicular stomatitis virus, foot and mouth virus, swine fever viruses, CDC select agents). Such specimens received for testing need to be promptly and properly packaged and shipped to an appropriate reference laboratory. Contact the appropriate reference laboratory in advance of any forwarding.
- If the laboratory has a BSL-3 facility, compile a list of suspect agents that must be processed and tested under BSL-3 conditions.
8.1.2. Leaking containers
- Do not process leaking containers because they can be a hazard to the technologist, could contaminate the laboratory, or could present an opportunity for specimen comingling and/or contamination that could produce a false result.
- Inform the attending physician or submitting veterinarian or organization regarding why testing will not be performed, and request a new specimen.
- Place leaking specimen containers into a biohazard bag and decontaminate by autoclaving or another approved method.
8.1.3. Visible contamination on outside of container
Specimens with a small amount of contamination (e.g., a dried blood spot) on the outside of the container are to be brought to the attention of the laboratory director. The director can examine the specimen and determine if it is suitable for testing and whether it constitutes a hazard to laboratory personnel.
8.1.4. Special precautions for suspicious specimens
- Accept specimens transported to the laboratory by enforcement officials in accordance with local and state regulations and following chain-of-custody procedures. However, the purpose of the testing and the suspect agent need to be determined before testing is started. Ensure that appropriate procedures for chain of custody are in place and followed even if the sample is not processed.
- The specimen login and processing personnel are responsible for checking the suspect agent "Do Not Process/Do Not Test" list before the specimen is sent on for testing (see 8.1.1). Notify the laboratory director if the suspect agent is on the list.
- If the laboratory has a BSL-3 facility, provide the specimen processing and log-in personnel a list of tests and suspect agents that must be handled only in the BSL-3 laboratory.
- Do not process or test unapproved or unusual specimen types as described in the laboratory accessioning standard operating procedures.
- Do not accept any specimen that the laboratory is not certified to test.
8.2. Stains, Chemicals, and Disposal
- Chemical hazards in the virology and electron microscopy laboratory will vary depending on the extent of service provided by the laboratory. The potential hazards presented in this section are intended to be illustrative, not inclusive of all chemicals used, and educational in nature and are not intended to replace material safety data sheet (MSDS) information or state, local, or institutional policies.
- The chemical fume hood used in the laboratory must be certified annually and checked daily for appropriate operation specifications.
8.2.1. Alcohols
Ethanol, methanol, isopropyl alcohol, and alcohol blends are used in the virology laboratory to fix cells, for nucleic acid extraction and precipitation, and as a disinfectant.
- Store concentrated alcohols in a cabinet rated for flammable storage. Isopropyl and methanol alcohol vapors can be toxic. Use these products only in well-ventilated areas. Isopropyl alcohol can also cause contact dermatitis. Alcohols are effective disinfectants for enveloped viruses, but they have little effect on nonenveloped viruses.
- Do not use alcohols in closed spaces. Exposure to solvent fumes can cause eye, nose and throat irritation, drowsiness, headaches, and skin dryness. When possible, use these in a fume hood.
- Do not use alcohols around open flames or instruments that cause sparks.
- Wear latex, vinyl, or nitrile gloves when handling alcohols to minimize skin exposure.
- Some alcohols will cloud plastics; care must be taken when wiping down plastic instrument faces with alcohols.
8.2.2. Antibiotics
Antibiotics in routine use include penicillin, streptomycin, gentamicin, ciprofloxacin, kanamycin, tetracycline, amphotericin B, and neomycin. These antibiotics can be found in culture media and viral transport media.
Concentrated antibiotic mixtures are frequently used to increase the antibiotic concentrations in samples containing large numbers of bacteria or fungi. Concentrated antibiotic solutions can be purchased at 50 times (50×) and 100 times (100×) the working concentration.
Although the risks associated with antibiotic preparation and use are relatively low in the virology laboratory, antibiotic preparation and handling has been associated with hypersensitivity reactions and contact dermatitus (115,116) and asthma (116–120) in hospital, pharmaceutical, and animal workers.
- Always wear gloves, mask, and eye protection when handling antibiotic powders and when preparing or dispensing concentrated antibiotic solutions. Respiratory protection (e.g., fume hood, mask, or positive pressure respirator) may be required in some instances.
- Prevent aerosol generation when working with antibiotic powders and solutions because these aerosols can contaminate the environment, sensitize other laboratory workers (115), and present a hazard to antibiotic-sensitive individuals (115,116).
- Do not allow laboratory personnel with known antibiotic sensitivities to prepare concentrated antibiotic solutions.
- It may also be prudent to exclude pregnant employees from preparing concentrated antibiotic solutions because antibiotics might have adverse or unknown effects on the developing fetus.
8.2.3. Bleach solutions (see 3.4.2)
8.2.4. Cycloheximide
Cycloheximide is used as an antibiotic, protein synthesis inhibitor, and plant growth regulator. In the virology laboratory, cycloheximide is used in Chlamydia re-feed media.
- Cycloheximide powders and solutions are irritants, causing redness, itching and burning. Animal studies have shown that cycloheximide causes adverse reproductive effects, including birth defects, sperm toxicity, and testicular damage. It is not known if cycloheximide can cause similar reproductive effects in humans, so handle it as if it were a reproductive toxin. Consult MSDS documents for more information.
- The highest potential for exposure in the laboratory is during the weighing of cycloheximide powders and during the preparation of cycloheximide solutions. Cycloheximide may enter the body as an aerosol, and orally through dust exposure. Exposure can also occur through hand contamination of food, beverages, or cosmetics, or directly by touching the mouth with contaminated hands.
- Handle cycloheximide powder in a chemical fume hood.
- Wear personal protective equipment (PPE), including laboratory coat and gloves, when handling cycloheximide powders and solutions in order to prevent skin contamination, skin absorption, and/or hand-to-mouth exposure. Wash hands with soap and water after glove removal.
- Cycloheximide disposal
Cycloheximide is inactivated by alkaline solutions (pH >7.0). Aspirating cycloheximide-containing culture fluids into vacuum traps containing a 1:10 bleach solution will inactivate the chemical. Most soaps and detergents are alkaline, and these agents will also inactivate cycloheximide.
8.2.5. Dimethyl sulfoxide
Dimethyl sulfoxide (DMSO) is used as a cryoprotectant when freezing cell cultures. DMSO is a powerful solvent and can penetrate skin and latex gloves.
- Minimize contact with skin and mucous membranes.
- Wear laboratory coat and eye protection when handling DMSO solutions. Double-gloving may be prudent because the chemical will eventually penetrate latex gloves.
- Nitrile gloves, which are commonly used in chemical laboratories, are rapidly dissolved by DMSO.
- DMSO easily penetrates the skin, and substances dissolved in DMSO may be quickly absorbed. This property has been used as a drug delivery system to allow antifungal medications to penetrate skin, toenails and fingernails. In the laboratory however, DMSO exposure could facilitate the absorption of contaminants. When DMSO comes into contact with the skin, some people report that they can quickly taste an oyster- or garlic-like flavor.
- DMSO is mutagenic for mammalian somatic cells, bacteria, and yeast. Long-term exposure may cause damage to blood, kidneys, liver, skin, mucous membranes, and eyes. See MSDS documents for more information.
8.2.6. Electron microscopy stains, fixatives, and buffers
- Osmium tetroxide and glutaraldehyde are used as electron microscopy fixatives. Liquid and vapor components are strong fixatives and will quickly fix the skin, mucous membrane and eye tissues of laboratory personnel.
— Open vials in the chemical fume hood. Keep vials in double bottles and seal the tops with parafilm.
— Handle vials with disposable gloves.
— Use eye protection, gloves, and disposable laboratory coats when handling the fixative and when fixing tissues. - Uranyl acetate, phosphotungstic acid, and ammonium molybdate are used as negative stains in the electron microscopy laboratory. All of these compounds contain heavy metals and are very toxic if inhaled, ingested or introduced through cuts or abrasions. Uranyl acetate is weakly radioactive, and powders need to be kept in a metal container. Phosphotungstic acid is corrosive and causes burns on exposed skin and mucous membranes. Ammonium molybdate is very dangerous in case of eye contact, ingestion, and inhalation.
— Laboratory workers must use PPE, including laboratory coat, gloves, and eye protection, when handling powders and solutions. See MSDS documents for more information.
— Prepare these stains in a chemical fume hood to prevent inhalation.
— Care must be taken to prevent contamination of work areas with powders. - Electron microscopy buffers such as sodium cacodylate and veronal acetate contain arsenic and sodium barbital, respectively. These buffers must be handled with caution.
— Prepare buffers in a chemical fume hood to prevent inhalation of powders.
— Use PPE, including laboratory coat, gloves, and eye protection, when handling powders and solutions. See MSDS documents for more information.
8.2.7. Electron microscopy embedding media
(Meth)acrylates and epoxy-based materials are frequently used to embed biological samples for electron microscopy. Epoxy products include Epon, Araldite, Spurr resin, and Maraglas. Formvar (polyvinyl formal) is used as a support film for electron microscopy grids and for making replicas. Many of these compounds are toxic, carcinogenic or potentially carcinogenic and are known to cause skin irritation, dermatitis, and skin sensitization. Consult individual MSDS documents for more information.
- Use these compounds in well-ventilated areas, preferably in a chemical fume hood. Many of these chemicals are dissolved in flammable solvents, and they must be kept away from heat and ignition sources.
- Plastic monomers will quickly penetrate latex and vinyl gloves; change these gloves frequently when embedding with plastics (121–124).
- Wear gloves when handling or trimming plastic-embedded blocks. All the monomers may not be polymerized, and unpolymerized monomers will retain their toxic properties.
- Cover working areas with paper towel or plastic-lined absorbent pads, and clean up spills immediately with alcohol.
- Use soap and water to remove any resins that come into contact with skin. Do not use alcohol to remove resins from skin because alcohol increases penetration of the resin.
- Disposal of embedding media and film-making solutions. Embedding materials are generally less hazardous when polymerized or hardened.
— Never pour plastic-containing solutions (e.g., propylene oxide–Epon mixture) down the drain. They will harden in the drain and can plug it.
— Harden all waste before disposal. Store hardened waste in a fume hood and dispose of the container as hazardous waste.
— Store discarded containers, beakers, vials, pipettes, and any instruments that have been in contact with resins or support films in puncture-resistant containers in the fume hood until they can be sent off as hazardous waste.
8.2.8. Ethidium bromide
Ethidium bromide (EtBr) is a DNA intercalating agent that is commonly used as a nonradioactive marker for visualizing nucleic acid bands in electrophoresis and other gel-based separations. EtBr is a potent mutagen, toxic after acute exposure, and is an irritant to the skin, eyes, mouth and the upper respiratory tract.
- Handle pure EtBr in a chemical fume hood because the powder can easily contaminate the entire laboratory.
- Designate an area where EtBr work is going to be performed, and use EtBr solutions only in that area.
- Cover surfaces within the designated area with a plastic-lined absorbent pad. Replace the pad on a scheduled basis or when it becomes contaminated.
- Use PPE, including laboratory coat, eye protection and gloves when handling EtBr solutions and gels.
Note: Latex gloves provide little protection against EtBr. Nitrile gloves provide an effective short-term barrier. Double-gloving provides increased protection. - Wash hands thoroughly after removing gloves.
- Application of sodium hypochlorite solutions to spent solutions of EtBr will deactivate the ethidium bromide, but the reaction products are mutagenic, according to the Ames test (125). Use an alternative deactivation method, use or a permitted hazardous waste treatment facility to dispose of these spent solutions.
- Use of sodium hypochlorite solutions in work areas of EtBr use is also not recommended.
- EtBr waste management
— Collect and manage even small volumes or concentrations of EtBr waste as hazardous waste or follow local regulations.
— Bag materials coming into contact with EtBr, and dispose of as hazardous chemical waste.
— Minimize EtBr solution volumes by adding activated charcoal. The charcoal can be collected by filtration and placed into leak-resistant containers for hazardous waste disposal.
— Place agarose gels containing EtBr into a leak-resistant plastic container and dispose as hazardous waste.
8.2.9. Evans blue
Evans blue is used as a counterstain during fluorescence microscopy. Evans blue powders and solutions are skin irritants, but there is no known flammability, carcinogenicity, or teratogenicity warning associated with this compound.
- The highest potential for exposure in the laboratory is during the weighing of Evans blue powders and during the preparation of solutions. Breathing powders can cause respiratory irritation. Skin and mucous membrane irritation can also occur.
- Handle Evans blue powder in a chemical fume hood to prevent inhalation.
- Wear PPE, including laboratory coat, eye protection, and gloves when handling Evans blue powders and solutions to prevent skin contamination.
8.2.10. Guanidinium solutions
Guanidinium chloride, guanidinium thiocyanate, and guanidinium isothiocyanate are chaotropic agents used to disrupt cells and denature proteins (particularly RNases and DNases) during nucleic acid extraction procedures.
These chemicals are strong irritants, and eye exposure can result in redness, irritation and pain. They are toxic if ingested and may cause neurologic disturbances. If inhaled, guanidinium compounds can cause respiratory tract irritation coughing, and shortness of breath.
- Handle guanidinium powders in a chemical fume hood to prevent inhalation.
- Wear PPE, including laboratory coat, gloves, and eye protection when handing powders and solutions.
- Do not add bleach to any sample waste containing guanidinium thiocyanate because of the production of toxic fumes. Guanidinium compounds are reactive with acids and other oxidizers, producing toxic fumes including cyanide vapors (thiocyanate and isothiocyanate derivatives), hydrochloric acid vapors (guanidinium hydrochloride), and nitrogen oxides (all forms).
8.2.11. Neutral red
Neutral red is a pH indicator and a vital stain used in some plaque assays. It may be harmful if swallowed, inhaled, or absorbed through the skin and can cause irritation to the skin, eyes, and respiratory tract.
- Handle neutral red powder in a chemical fume hood to prevent inhalation.
- Wear PPE, including laboratory coat and gloves, when handling neutral red powders and solutions to prevent skin contamination.
- The highest potential for exposure in the laboratory is during the weighing of neutral red powders and during the preparation of solutions.
8.2.12. Merthiolate (thimerosal)
Merthiolate, or thimerosal, is a mercury-containing antiseptic and antifungal agent used as a preservative in some laboratory solutions. Concentrated thimerosal is very toxic when inhaled, ingested, and in contact with skin.
- Wear PPE, including laboratory coat and gloves, when handling merthiolate powders and solutions to prevent skin and mucous membrane exposure.
- The low quantities used in some commercial reagents are relatively safe, but thimerosal exposure can have cumulative effects. In the body, merthiolate is metabolized or degraded to ethylmercury (CHHg+) and thiosalicylate. Ethylmercury clears from blood with a half-time of about 18 days, and from the brain in about 14 days.
8.2.13. Organic solvents
- Acetone is the principal organic solvent used in the virology laboratory and is primarily used as a fixative for cell smears.
- Acetone is flammable and it is classified as an irritant, causing eye damage and skin and respiratory tract irritation. Long-term exposure can result in reproductive, nervous system, kidney, liver and skin damage.
- Store acetones in a flammable storage cabinet and keep away from sources of heat, sparks, or flame. Do not store or use acetone in a refrigerator that is not rated as explosion proof. Sparks from the refrigeration pump and the door-actuated light switch could ignite acetone fumes and cause an explosion.
- Use acetones in a well-ventilated area (or chemical fume hood) to prevent respiratory irritation.
- Do not use vinyl exam gloves for handling acetones and other aggressive organic solvents because vinyl gloves can be dissolved by these agents.
- Latex gloves will eventually dissolve in acetone, and double-gloving is recommended for short-term acetone usage.
- Acetones will dissolve or cloud many plastics, and care must be taken to protect plastic devices from acetone exposure.
- Electron microscopy
— Many volatile solvents, including ethane, propylene oxide, and ethers, are used in the electron microscopy laboratory. These solvents are extremely flammable and are fire and explosion hazards. Care must be taken to prevent static discharges that could ignite the chemicals.
— Use these solvents in a chemical fume hood to prevent respiratory irritation and minimize the buildup of explosive vapors.
— Wear PPE, including laboratory coat, eye protection, and chemically resistant gloves when handling these chemicals.
— It may be prudent to disconnect flammable gas lines to electron microscopy laboratories to discourage the use of open flames.
— Store small quantities in a well-ventilated flammable storage cabinet, and keep these reagents away from sources of heat, sparks, or flame.
8.2.14. Sodium azide
Sodium azide is a common preservative in many laboratory reagents, including monoclonal antibodies, buffers, and enzyme immunoassay reagents.
- Sodium azide is an acute toxin and a mutagen. Reduce all contact with this substance to the lowest possible level.
- Sodium azide and hydrazoic acid (HN, which is formed from NaN in water) are known to produce hypotension (low blood pressure) in laboratory animals and humans and to form strong complexes with hemoglobin, thereby blocking oxygen transport in the blood.
- Wear PPE, including gloves, laboratory coat, and eye protection when handling solutions containing sodium azide.
- Sodium azide is not explosive except when heated near its decomposition temperature (300°C) or combined with metals.
- Never flush solid or concentrated sodium azide solutions down the drain since this practice can cause serious incidents when the azide reacts with lead or copper in the drain lines and explodes.
- Dilute solutions (0.1%) found in most laboratory reagents may be flushed down the sink with copious volumes of water to prevent metal azide buildup. Sodium azide reacts with heavy metals (such as silver, gold, lead, copper, brass, or solder in plumbing systems) and metal salts to form highly explosive compounds such as lead azide and copper azide. These metal azides can explode when the plumbing is repaired or jarred.
- A "skin" designation has been assigned to the Occupational Safety and Health Administration (OSHA) Permissible Exposure Limits because of the ability of sodium azide to readily penetrate intact skin. Any dermal exposure can substantially contribute to the overall exposure to sodium azide.
- Sodium azide is not compatible and may react violently with chromyl chloride, hydrazine, bromine, carbon disulfide, dimethyl sulfate, dibromomalonitrile, strong acids (such as hydrochloric, sulfuric and nitric), and acid chlorides.
8.3. Handling Cell Cultures at the Bench
- All cell cultures, whether inoculated with clinical specimens or not, are potentially infectious. Unintended or adventitious viral agents have been found in many cell lines, diploid cells, and primary cultures (Table 9). Adventitious agents may be spread during cell culture manipulations and can originate from
— latently or persistently infected primary tissue, secondary cultures, and cell lines;
— animal products such as fetal calf serum and trypsin; or
— transforming agents (human papilloma virus [HPV], SV-40, herpesviruses, retroviruses, adenoviruses, and others) used to immortalize cells. - Many of these agents can go undetected because they do not produce cytopathic effects; therefore, cell passage and archiving can perpetuate these agents for generations.
8.3.1. Cell lines
- Primary cultures of human (and potentially animal) tissue, cells, and blood present the greatest risk for harboring unintended or adventitious infectious agents that can infect humans. As a result, OSHA included human cell lines in its Final Rule on Bloodborne Pathogens (33). Even though OSHA Standards CFR 29, Bloodborne Pathogens, refer to human blood and tissue, adherence to these standards in the veterinary laboratory is advisable. Conduct tissue culture procedures in a Class II BSC.
- Primate cells and tissues also present risks to laboratory workers. SV-5 and SV-40 are common contaminants of primary rhesus monkey kidney cells, and cultures from macaques and other Old World monkeys may be latently infected with Herpesvirus simiae (B-virus). B-virus infection presents an often fatal hazard for personnel handling these animals and their tissues.
- Cultures from nude and severe combined immune deficient (SCID) mice pose a special risk of harboring agents that could cause subclinical or chronic infections in laboratorians.
- Certain cell lines were immortalized with viral agents such as SV-40, Epstein-Barr virus, adenovirus or HPV. These cells may produce infectious virus or they may have viral genomic material within the cells. Other cell lines may carry viral genetic elements that were introduced purposefully during experiments or inadvertently during culture manipulation. These viruses and virus genetic elements survive freezing and may be present in archived culture materials. Many cell lines are also persistently infected with broad-host-range retroviruses that can present an infection hazard for laboratory workers.
- Tumorigenic human cells may present a potential hazard from self-inoculation (126–129). Tumors or tumor cells that have been inoculated into nude mice may acquire additional adventitious agents such as lymphocytic choriomeningitis virus (LCMV).
- Many of the adventitious viruses do not produce cytopathic effects or alter the cell phenotype, and many can survive freezing and storage in liquid nitrogen for long periods.
- When infected tumor cells are cultured, the supernatant fluids can contain infectious virus, and the laboratory may optionally elect to treat this as biohazardous waste. LCMV presents a special problem for pregnant women because the virus can be transmitted to the fetus, causing fetal death or severe central nervous system malformation.
8.3.2. Cell culture practices
Workers who handle or manipulate human or animal cells and tissues are at risk for possible exposure to potentially infectious latent and adventitious agents that may be present in those cells and tissues. CDC/National Institutes of Health recommended cell culture practices (1) include the following.
- Strictly follow BSL-2 recommendations for PPE, such as laboratory coats, gloves and eye protection.
- Handle human and other primate cells using BSL-2 practices and containment.
- Perform all work in a Class II or higher BSC. Class I safety cabinets and clean benches are never used for cell culture manipulation because they do not provide adequate protection for the operator.
- Autoclave or disinfect all material coming into contact with cell cultures before discarding.
- Enroll all laboratory staff working with human cells and tissues in an occupational medicine program specific for bloodborne pathogens, and work under the policies and guidelines established by the institution's exposure control plan.
- Determine if laboratory staff working with human cells and tissues need to provide a baseline serum sample (if this is institutional policy based on a risk assessment), be offered hepatitis B immunization and be evaluated by a health-care professional following an exposure incident (1). Similar programs for persons working with nonhuman primate blood, body fluids, and other tissues are recommended.
8.3.3. Biohazards associated with cell culture reading
- Dried medium on the lip of culture tubes could present a contamination hazard. Handle all culture vessels as if they were contaminated.
— Wear gloves and a lab coat when handling viral cultures.
— Use eye protection if there is a potential splash hazard.
— Perform all culture manipulations in a BSC.
— Decontaminate culture tubes and other materials that come into contact with cell cultures before disposal, using an effective method (usually autoclaving or chemical disinfection) (1). - Shell vial, tube, and other cultures go through multiple manipulations (e.g., media aspiration, refeeding, inoculation) that can generate aerosols. These aerosols and manipulations can contaminate the outside of the culture vessel.
- The occasional leaking tube on another shelf could also contaminate the outside of vessels on lower shelves and may not leave visible evidence of the contamination.
8.3.4. Biohazards associated with liquid nitrogen use
Liquid nitrogen can become contaminated when ampoules are broken in the dewar, and contaminants can be preserved in the nitrogen (23). These potentially infectious contaminants can contaminate other vials in the dewar and generate an infectious aerosol as the liquid nitrogen evaporates. Plastic cryotubes rated for liquid nitrogen temperatures are recommended for liquid nitrogen storage because they appear to be sturdier than glass ampoules and are less likely to break in the nitrogen. Glass ampoules are not recommended.
Ampoules and cryotubes can explode when removed from liquid nitrogen creating infectious aerosols and droplets. See Section 3.9 for additional information.
8.4. Personal Precautions
No amount of safety engineering can reduce the physical, chemical, and biological risks in a laboratory environment if personal precautions are not employed consistently and rigorously. All laboratory workers and visitors are responsible for following established procedures regarding personal precautions. Directors and supervisors should periodically review their biosafety responsibilities (1).
8.4.1. Biological safety cabinet
- Conduct all culture manipulations in the virology laboratory in a Class II BSC. These manipulations include, but are not limited to, culture inoculation, feeding, passage, hemadsorption and hemagglutination testing, virus dilutions and titrations, cell fixation, immunofluorescent staining, and preparing controls and control slides.
- Follow appropriate BSC setup and operation procedures (1). See Section 3 and Table 5 for guidelines on use of BSCs and descriptions of the characteristics and uses of BSC types.
- Certify BSCs according to NSF/ANSI Standard 49 annually, or after cabinet is moved, HEPA filters are replaced or disinfected, or the unit has undergone any major repairs that could affect the seating or performance of the HEPA filtration system.
- Remove the contents of the BSC and disinfect the interior of the BSC daily or after a spill or contamination event.
— Do not use alcohols as a primary surface disinfectant in BSCs because alcohols have little or no effect on nonenveloped viruses. Vapors from isopropyl alcohol can be toxic and may cause contact dermatitis. A 1:10 bleach solution provides the best disinfecting activity, but care must be taken to remove the residual chlorine with water because the chlorine will eventually corrode the stainless steel surfaces.
— Remove the floor plate of the cabinet and the front grate monthly or bimonthly for cleaning. Disinfect the floor plate, grate, and the plenum below the floor plate.
— Wipe work surfaces, interior walls and the interior surface of the of the window with a 1:10 solution of household bleach (1) followed by one wiping with water to remove the residual chlorine, and one wiping with 70% ethanol (EtOH). Remove residual chlorine because it will eventually corrode stainless steel surfaces.
— Wipe down any items that will be returned to the BSC.
— Let the blower run for at least 4 minutes to remove any particulates.
— Some laboratories leave the sash up and blowers running at all times, whereas other laboratories turn off the blower and close the sash (if so equipped) at the end of the day.
8.4.2. Personal protective equipment
- Most recommendations for the general microbiology laboratory are appropriate for the virology laboratory (Section 3).
- Gloves must be worn to protect hands from exposure to hazardous materials and extreme temperatures. In the molecular biology area, gloves are also used to protect the specimen from nucleases that are on the skin. See Sections 8.1–4 for guidelines on use of gloves or other PPE when handling specific stains or chemicals.
8.4.3. Disinfection
Disinfection guidelines for the general microbiology laboratory are applicable to the virology laboratory (Section 3).
8.5. Decontamination and Disposal of Laboratory Waste
- Disinfect reagents or materials coming into contact with clinical specimens, cell cultures, or virus cultures (including gloves and PPE) before they are placed into the medical waste stream. Fluids may be disinfected by treating with a 1:10 household bleach (Section 3.4.2 and 3.5) or by autoclaving. Disinfect all other materials by autoclaving before they are placed into the medical waste.
- Decontaminate specimens, reagents, cultures and equipment that come into contact with specimens from patients with suspected Creutzfeldt-Jakob or other prion-associated disease according to local protocols, usually by autoclaving for 1 hour in the presence of 20,000 ppm hypochlorite solutions or 1 N NaOH (1).
8.6. Clean versus Dirty Areas of the Laboratory
- In the virology laboratory, the distinction between dirty and clean areas is a misnomer; all areas within the laboratory present increased opportunities for encountering infectious, chemical, and physical hazards. Laboratory precautions are applicable to all areas of the laboratory.
- Cell culture preparation and reagent preparation areas are often called "clean" areas because no specimens, amplified nucleic acids, or control materials are allowed in these areas. The goal of establishing and policing these "clean" areas is to prevent reagent and cell culture contamination that could produce false results. These are laboratory areas, and laboratory precautions still apply.
8.7. Early Recognition of High-Risk Organisms
Routine clinical laboratory testing may provide the first evidence of an unexpected bioterrorism event, and routine clinical specimens may also harbor unusual or exotic infectious agents that are dangerous to amplify in culture. Early recognition of these possible high-risk organisms is critical, as is adherence to all fundamentals of laboratory safety. Events that require intervention by a supervisor or laboratory director are listed (Table 10). Although the majority of events are caused by inadvertent actions and pose no risk, laboratory technologists and directors should be aware that multiple high-risk causes are possible. How the laboratory responds to these trigger events will depend upon whether the laboratory has a BSL-3 facility and the capabilities of the state and local laboratory response network (LRN).
8.7.1. Fluorescent antibody testing bench
- Dim staining of cells when control smears and other positive specimens stain strongly can be due to poor antigen expression, sampling crusted lesions, and cellular degradation. Dim staining can also occur when the antibody reagent cross-reacts with a similar antigenic epitope arising from antigenic drift, the presence of a viral subspecies, or an unsuspected virus. This type of reaction occurs more frequently when staining with polyclonal antibodies.
- An unusual pattern of staining (e.g., speckled cytoplasmic staining with a reagent that usually produces nuclear staining) can indicate the presence of an unsuspected virus.
- Staining in an unusual cell type (e.g., staining of squamous cells rather than ciliated respiratory epithelial cells) could indicate the presence of a different virus or an expected virus with an altered tropism.
- Notify the laboratory supervisor or director of these findings.
8.7.2. Suspicious or unusual results
- Known agents of bioterrorism or public health emergencies, such as foot and mouth disease, flaviviruses, smallpox, alpha viruses, and hemorrhagic fever viruses, will grow in routine cell cultures used in the clinical laboratory. If a culture produces a cytopathic effect (CPE) pattern that is consistent with one of these agents, do not open for further testing. Contact the attending physician or veterinarian for more information on the patient/animal, and contact the state or local LRN laboratory for further instructions.
- Cultures that produce a familiar CPE pattern in an unusual cell type and/or an unexpected CPE result from a specific specimen source (e.g., an eye specimen producing hypertrophic rounding in nearly every cell type) might indicate the presence of an altered or unexpected virus. Contact the physician or veterinarian for more information, and move the specimen into the BSL-3 laboratory for any additional testing.
- Confirmatory test failure can be caused by antigenic drift or operator error during testing. Alternative confirmatory tests such as polymerase chain reaction (PCR) can be used because PCR methods generally present fewer risks than additional fluorescent antibody staining. If PCR testing fails to identify the virus, move the culture into the BSL-3 laboratory for further testing.
- If a BSL-3 laboratory is not available, contact the state or local LRN laboratory for assistance in identifying the viral agent.
8.7.3. Nucleic acid testing
- Specimens that produce an altered melting curve shape, too many peaks, and/or an altered melting temperature (Tm) when controls and other patient curves are normal could indicate poor extraction, the presence of primer dimers, or genetic changes under the primers or probes. These results can also be caused by the presence of an unexpected or unusual (chimeric, recombinant or reassortant) pathogen. Re-extraction and retesting present a modest additional risk. If the results continue to be unusual, the specimen should be referred to the LRN laboratory for additional testing.
- PCR specimens with altered amplification slopes may be the result of genetic variations under the primers and/or probe or the presence of an unusual pathogen. Re-extraction and retesting present a modest additional risk. If the results continue to be unusual, the laboratory should refer the sample to their state or local LRN for additional characterization.
8.8. Hazards Associated with the Electron Microscopy Laboratory
Diagnostic electron microscopy can be a relatively simple and rapid method for morphologic identification of agents in a specimen. Electron microscopy procedures can serve as a general screen to detect novel organisms or organisms that have altered genetic or immunologic properties that render them undetectable by nucleic acid or immunoassay protocols (192). Electron microscopy laboratories share many of the physical, chemical, and biological hazards described for the virology laboratory but also have some unique features.
8.8.1. Flammable and combustible liquids
The electron microscopy laboratory uses a wide variety of flammable solvents, and the use of open flames is discouraged (see Section 8.2.13).
- Place solvents requiring refrigeration in special flammable-storage refrigerators that minimize exposed electrical connections and reduce the opportunity for spark generation.
- Store flammable liquids in flammable-storage cabinets in accordance with local and state regulations.
- Handle flammable liquids in fume hoods to minimize vapor buildup. Ultrasonic cleaning of Wehnelt cap assemblies in an acetone baths must be done in a chemical fume hood.
- Never pour flammable liquids down the drain because they can cause an explosion.
8.8.2. X-ray hazards
The electron microscope will generate dangerous levels of X-rays within the microscope as high-energy electrons strike the metal components. Modern electron microscopes have sufficient shielding and lead-impregnated glass viewing ports that minimize dangers to the operator. However, modifications to the instrument, adding and removing accessories, and some maintenance procedures can compromise the shielding.
- Use a calibrated thin-window Geiger-Müller (GM) survey meter to verify the shielding effectiveness, and monitor radiation levels
— initially at the time of installation;
— whenever the microscope is modified; and
— periodically as indicated by state, local, or institutional policies. - Radiation levels are to be <0.5 mR at 5 centimeters from the unit.
8.8.3. Electrical hazards
See Section 3.7 for information regarding routine electrical safety in the electron microscopy laboratory.
- Special high-voltage and high-amperage electrical sources are used to power electron microscopes and other equipment in the laboratory. Only trained technicians are to service this equipment. Install safety interlocks and power lockouts to prevent activation of electrical circuits while the instrument is being serviced.
- Changing the filament in the high-voltage electron gun can present an electrical hazard if the grounding rod does not make contact with the Wehnelt cap. A substantial charge can build up on the cap, and the charge must be relieved before touching the cap.
8.8.4. Chemical hazards
Several heavy metal stains and aggressive fixatives are used in the electron microscopy laboratory. See Section 8.2 and the MSDS materials provided by the manufacturers for guidelines for handling stains and fixatives. Embedding and filmmaking materials are chemical hazards, and many of these materials are dissolved in flammable organic solvents.
- Limit acute and long-term exposure to these chemicals.
- Place embedding ovens in a chemical fume hood to minimize exposure to potentially hazardous chemicals.
- Latex gloves are not appropriate for all chemicals, and appropriate glove usage must be emphasized for all laboratory personnel.
8.8.5. Cryogens and compressed gases
The most commonly used cryogens used in the electron microscopy laboratory are liquid nitrogen and liquid helium. Compressed helium, CO and nitrogen are also used. Hazards and safety measures associated with these gases are summarized in Sections 3.8 and 3.9.
8.8.6. Specialized equipment
The electron microscopy laboratory uses a number of specialized instruments whose use can be hazardous. For example, evaporators, freeze-driers, freeze-fracture, and sputter coater units use vacuum, and the vessels could implode. Implosion hazards are reviewed in Section 3.13.1.
- To prevent eye damage during evaporation, use welder's goggles to view the source.
- Allow the components to cool before touching them.
- Do not breathe any of the evaporated metal that may flake off from the surface.
- Critical point dryers can be quite dangerous because of the high pressures generated within the "bomb" (121,193). Follow the operating and safety procedures described in the operator's manual for safe operation.
— Place a one-half-inch thick lexan shield between the operator and the bomb.
— Do not secure the shield directly over the window of the bomb where it would receive the full force of an explosion. That force could shatter the shield.
— A polycarbonate face shield is also recommended when observing the contents of the bomb.
— Vent freons or freon substitutes from critical point dryers directly outside or through a fume hood.
8.8.7. Biological hazards
The biological hazards of the electron microscopy laboratory are similar to those of the virology laboratory, and good laboratory practices must be followed.
- Conduct primary specimen handling in a biological safety cabinet to prevent aerosols and contamination of the laboratory.
- Negative staining solutions may not inactivate microorganisms and viruses. Osmium tetroxide is an effective sterilant, but it cannot be used for all specimens.
8.9. Rapid Testing (Kits)
Several FDA-approved, rapid immunodiagnostic tests for viral antigens and antibodies are available. Originally designed for point-of-care or near point-of-care testing, many of these tests are being used for testing in clinical virology laboratories. The following biosafety recommendations are based upon CDC biosafety guidance for handling clinical specimens or isolates containing 2009-H1N1 influenza A virus (194).
- Procedures that involve only simple steps such as inserting a swab into medium or pipetting specimens and reagents and are not reasonably expected to generate aerosols may be performed on the bench top using only splash protection. Use the BSC for testing procedures that require centrifugation, vortexing, vigorous mixing, or other methods that could generate aerosols.
- Perform bench top testing in a manner that will prevent splashes and generation of aerosols. The appropriate PPE for this type of testing consists of a laboratory coat, gloves, eye protection and a face mask, such as a surgical, dental, medical procedure, isolation, or laser mask. A splash shield providing protection of the entire face fulfills the need for separate eye protection and face mask.
- Decontaminate work surfaces and equipment with a 1:10 bleach solution as soon as possible after specimens are processed.
- Rapid immunodiagnostic testing, when performed in a Class II BSC, does not require use of additional of eye protection and a face mask.
8.10. Molecular Laboratory
Molecular virology laboratories share many of the physical, chemical and biological hazards described for the virology laboratory, but they also present some unique hazards.
8.10.1. Electrical hazards
See Section 3.7 for information regarding routine electrical safety in the molecular virology laboratory. Special high-voltage power sources are used in electrophoresis and nucleic acid sequencing equipment.
- Never use high-voltage electrical equipment near flammable liquids and gases because an arc could cause an explosion or fire.
- Disconnect the power to electrophoresis equipment before disassembling the apparatus.
- Only trained technicians should service high-voltage electrical equipment.
- Never tamper with or defeat safety interlocks and power lockouts on electrophoresis equipment.
8.10.2. Ultraviolet light hazards
- Short-wave ultraviolet (UV) view boxes are often used to visualize nucleic acid bands in gels. Special care must be taken to protect eyes and completely cover the skin when visualizing and photographing gels on a UV view box.
- Wear gloves, long-sleeved laboratory coat, and a UV-resistant full-face shield when working with UV view boxes.
- Use only face shields rated for short-wave UV light for this purpose.
- Locate UV view boxes out of the normal traffic flow of the laboratory so that bystanders or persons passing by are not exposed to the ultraviolet light. Other UV light hazards are discussed in Section 3.12.
8.10.3. Chemical hazards
The chemical hazards unique to the molecular virology laboratory include chloroform, ethidium bromide (Section 8.2.8) and guanidinium-based extraction reagents (Section 8.2.10). Avoid acute and long-term exposure to these.
8.10.4. Biological hazards
The biological hazards in the molecular virology laboratory are similar to those of the virology laboratory, and good laboratory practices must be followed.
- Conduct primary specimen handling in a biological safety cabinet to prevent aerosols and contamination of the laboratory.
- Extracted nucleic acids may or may not be sterile. An inadequately performed extraction procedure may not completely remove or inactivate the infectious agents in the specimen. The genome of positive-stranded RNA viruses (poliovirus) is suspected to be infectious, and once introduced into the cell, the viral genome requires no virus-coded proteins or accessory components for virus replication. Although the risks of infection in this manner are very small, it is prudent to handle extracted viral nucleic acids as if they were infectious. This practice mirrors specimen-handling procedures used in other areas of the laboratory and supports a unified specimen-handling policy for the entire laboratory.
9. Chemistry Laboratory
All specimens of human and animal origin tested by the chemistry, toxicology, or drug-testing laboratory may contain infectious agents. It is imperative to understand and minimize the risk of exposure to patient specimens through surface contact, aerosolization, or penetrating injury. Risk mitigation of laboratory-acquired infections is discussed in Sections 2 and 3.
9.1. Automated Analyzers (see also 3.17.3 and 10.6.3)
Automated analyzers frequently have added features to help reduce operator exposures, but they do not totally eliminate the potential for exposure. A common feature in newer systems is closed system sampling.
- Chemistry/toxicology analyzers often have high-velocity robotic arms and samplers that might cause skin punctures and lacerations. Operate analyzers only with the cover closed.
- Sample probes that move quickly or deliver fluid rapidly may generate aerosols and droplets.
- Always use instruments according to manufacturer instructions.
- Ensure instrument safety shields and containment devices are in place at time of use.
— Limit the amount of hand movement near the sample probe and liquid-level sensors.
— When using instruments for which the operator is required to wipe sample probes after sampling, wear disposable gloves and use gauze pads with impermeable plastic coating on one side. Newer instruments have automatic probe wash cycles eliminating this source of exposure. - To prevent spillage, handle sample trays and samples with caution, and keep them covered when not being manipulated.
- Assume that the outside of blood tubes is contaminated, and be prepared to wipe the outside of the tube with the laboratory disinfectant or with a solution of 1:10 household bleach.
- Fill sample cups and aliquot tubes using mechanical devices (e.g., transfer pipettes), and never decant (pour) them.
- Consider effluents of clinical analyzers to be contaminated with pathogens; some may also be contaminated with hazardous chemicals. Their disposal must comply with state and local regulations. Investigate these effluents and consult applicable standards before discharging them in the sewer.
- Follow manufacturer instructions for routine cleaning and trouble-shooting specimen spills on or within an instrument, including the appropriate personal protective equipment (PPE) and type of cleaning solution to be used.
— When manufacturer instructions do not include spill containment and clean-up instructions, collaborate with the manufacturer to develop a standard operating procedure (SOP) that will effectively protect the operator and maintain and extend the instrument's operational life.
— Have fresh 1:10 household bleach solution on hand in case of an emergency spill or breakage after being assured by the manufacturer that chlorine will not damage instrument components. - Perform daily cleaning of fluidic systems and sampler following manufacturer instructions.
- Collect waste into a waste container that contains fresh concentrated household bleach in sufficient quantity to achieve a final concentration of 10% bleach when the flask is full. Do not collect effluents containing compounds not compatible with bleach (Table 11) into bleach to avoid formation of chlorine gas.
- Do not mix sodium hypochlorite (bleach) with any other chemical unless adequate engineering controls and PPE are in place. Accidental mixing may cause dangerous conditions that could result in injury to personnel and/or damage to property or the environment.
9.2. Tissue Preparation for Chemical/Toxicological Analysis
- Consider all unfixed tissues as biohazardous, regardless of the patient diagnosis or the test(s) ordered.
- The use of a fixative is not always sufficient to eliminate all types of biohazards.
- Use a biological safety cabinet (BSC) or PPE, including gown and gloves. with a fixed containment device for sample aspiration.
- Automated sample loading systems reduce sample handling and also perform sample vortexing in an enclosure that prevents operator exposure to aerosols and splashes.
— Take special care when loading samples onto the instrument tube racks.
— For unfixed samples, load instrument tube racks inside a BSC or wear PPE to protect from splashes and aerosols.
9.3. Specific Analyzer Risks
To adequately assess the risk of active biohazards in analyzer effluents or processes, risk analysis should begin with assessment of procedures that occur prior to the use of specific analyzers. Sample preparation protocols may fully inactivate viruses and bacteria so that the risk of biohazardous aerosol generation in the analyzer effluent is essentially zero. One example is the use of protein-precipitation techniques or protein denaturing solvents in liquid chromatography, which would negate biohazard concerns in aerosols or effluents generated by the analyzer.
9.3.1. Graphite furnaces
- Completely dry samples before vaporization.
- Adequately ventilate devices that use heat to vaporize specimens to ensure that infectious agents are not escaping into ambient air.
- Keep instrument covers and panels closed and secure while instrument is in use, in accordance with manufacturer's recommendations.
9.3.2. Mass spectrometers
- When mass spectrometers are used as detection devices that are programmed to monitor selected ions from the effluent of liquid chromatography (LC-MS), exposure to infectious agents in the effluent could occur if a risk assessment before the analyzer process shows that sample preparation procedures do not inactivate infectious agents.
- Devices that use nondestructive (soft) techniques to ionize samples, e.g., sonic spray ionization, may present a risk of exposure to operators if pre–analyzer process risk assessment shows that sample preparation procedures do not inactivate infectious agents.
10. Hematology and Phlebotomy Laboratory
10.1. Specimen Receiving and Log-In/Setup Station
Biosafety guidelines for the hematology laboratory are the same as those for the microbiology laboratory and are described in Section 3.1.
10.2. Work at the Open Bench
See Section 3.2.1.
10.2.1. Standard operating procedures
Standard operating procedures are described in Section 3.1.4.
10.2.2. Manual removal of sealed caps and specimen aliquotting/pipetting
See Section 3.1.5.
10.2.3. Unfixed specimens
- Microscopes
— Disinfect the stage, eyepieces, knobs and any other contaminated parts daily.
— Select a disinfectant that will be noncorrosive to the microscope and appropriate for potential infectious agents. - Slide preparations
— Avoid waving slides in the air or using electric fans at an open bench to air-dry slides.
— An electric fan may be used to air-dry slides in a fume hood and, only if necessary, in a biological safety cabinet (BSC), not on the bench top. Using a fan in the BSC will disrupt airflow and will render the BSC unsuitable for other laboratory work until the airflow has been stabilized. - Buffy coat smears
Use disposable Wintrobe tubes in place of capillary tubes to avoid cutting glass tubes and possible exposure to blood and shards of glass. - Hemacytometer
Use extreme caution when using glass hemacytometers and glass coverslips to avoid punctures from glass shards. Plastic hemacytometers are commercially available and offer repeatable and reliable measurements and analysis. Several hemacytomer designs eliminate the use of coverslips and allow for exact volume control. - Bleeding times (195)
— Wear gloves (see Sections 3.2.2 and 8.4.2).
— Use appropriate pediatric/adult template safety devices.
— Ensure devices are reviewed annually in compliance with Occupational Safety and Health Administration (OSHA) Bloodborne Pathogens Standards (33) and CDC guidelines (196).
— Use disposable razors when removal of arm hair from test site is required.
— Dispose of used bleeding time device and razor in sharps container, and filter paper in medical waste container. - Bone marrow aspirates
— Include appropriate hospital infection control policies and procedures for patient care settings in the laboratory policy and procedure for collection of bone marrow aspirates when collected in these settings.
— Limit what is brought into patient rooms. Bone marrow safety tray kits are marketed by several manufacturers. Use of disposable trephine and aspiration needles is recommended.
— Collect specimens and prepare squash smears and slides in the laboratory.
— If smears must be prepared outside the laboratory, e.g., in patient care settings, use Standard Precautions; use risk and hazards assessment to determine what and when additional personal protective equipment (PPE) might be appropriate; use a flat secure surface to prepare smears and slides; ensure cover/caps on additional specimens are tightly secured before transport; and ensure slides, squash smears, and additional specimens are transported in leakproof and breakage-resistant receptacles. - Document training and assess competency of laboratory personnel. Include knowledge of, and adherence to, hospital infection control policies/procedures in patient settings, and the concept of Standard Precautions in all documented training and competency assessments.
10.3. Personal Precautions
Guidelines for personal precautions, including use of a BSC (Section 3.3), PPE (Section 3.2.1), and disinfection (Section 3.4) are described in Section 3.
10.4. Decontamination and Disposal of Laboratory Waste (39)
See Section 3.5 for guidelines for decontamination and disposal of laboratory waste.
10.5. Dirty versus Clean Areas of the Laboratory
See Section 3.16.
10.6. Instrumentation
Whether automated or manual, procedures with the potential for producing specimen aerosols and droplets (e.g., stopper removal, vortexing, opening or piercing evacuated tubes, automatic sample dispensers) require either PPE or engineering controls designed to prevent exposures to infectious agents.
10.6.1. Waterbaths
See Section 3.17.1.
10.6.2. Centrifuges
See Section 3.17.2.
10.6.3. Automated hematology/hemostasis analyzers
Automated analyzers frequently have added features to help reduce operator exposures, but these do not totally eliminate potentials for exposure. A common feature in newer systems is closed system sampling. See Sections 3.17.3, 9.1, and 11.6.3 for additional information.
- Sample probes that move quickly or deliver fluid rapidly can generate aerosols and droplets.
- Always use instruments according to manufacturer instructions.
- Ensure instrument safety shields and containment devices are in place at time of use.
- Limit the amount of hand movement near the sample probe and liquid-level sensors.
- Wear gloves and use gauze pads with impermeable plastic coating on one side when using instruments for which the operator is required to wipe sample probes after sampling. Newer instruments have automatic probe wash cycles eliminating this source of exposure.
- Handle sample trays and sample plates with caution, and cover them when not being sampled to prevent spillage.
- Fill sample cups and aliquot tubes using mechanical devices, and never decant (pour) them.
- Assume that effluents of clinical analyzers are contaminated, and dispose in compliance with applicable federal, state, and local environmental regulations.
- Follow manufacturer instructions for routine cleaning and trouble-shooting specimen spills on or within an instrument, including the appropriate PPE and type of cleaning solution to be used.
- When manufacturer instructions do not include spill containment and cleanup instructions, collaborate with the manufacturer to develop a standard operating procedure that will effectively protect the operator and maintain and extend the instrument's operational life.
10.6.4 Flow cytometers (see Section 3.17.3)
Occupational exposures in a routine flow cytometry (FCM) laboratory arise either from sample handling or, more specifically, from aerosols and droplets generated by the flow itself. Flow cytometric applications, e.g., phenotypic analysis, calcium flux evaluations, and apoptosis measurements of unfixed cells, when performed using jet-in-air flow cytometers with extremely high pressure settings can expose operators to potentially hazardous aerosols.
- FCM biosafety procedures should specifically focus on aerosol containment, waste management and equipment maintenance (197).
- Consider all unfixed materials (peripheral leukocytes, bone marrow, various body fluids, cultured cells, and environmental samples) as biohazardous.
— Use of a fixative is not always sufficient to eliminate all types of biohazards.
— Use a BSC or PPE with an equivalent fixed containment device for sample aspiration.
— Always wear disposable gloves and protective clothing when operating a flow cytometer.
— Allow only operators with documented training and experience (professional consensus is 2 years) to perform potentially biohazardous cell sorting (51).
— Restrict access by allowing only essential personnel during sorting. Post a notice at the entrance that cell sorting is in process. Personnel wishing to monitor the sort must wear the same PPE as the operator of the cell sorter. - Cell-sorters are equipped with a nozzle to form a jet of microdroplets; this step is likely to generate aerosols.
- Instrument failures such as clogged sort nozzle or air in the fluidic system can drastically increase aerosol formation.
- Newly designed safety attachments for cell sorters have become commercially available. Some enclosed fluid system flow cytometers perform cell sorting using a fluid switching mechanism. These cytometers, in contrast to jet-in-air cell sorters, do not generate aerosols during cell sorting.
- Efficiency of aerosol control measures on sorter instruments is to be tested periodically following manufacturer instructions, particularly when unfixed human cells and known biohazardous samples are acquired or sorted.
- Simpler bead-based technology for measuring efficiency has been developed.
- Newer flow cytometers have added biosafety features — e.g., enclosed flow cells, droplet containment modules, and automated samplers — for reducing risk of operator exposure to instrument-generated sample droplets and aerosols.
- Ensure the instrument is used in a manner specified by the manufacturer.
- Open and close the instrument cover with care.
- Keep instrument covers and panels closed and secure while instrument is in use.
- Do not disable safety interlocks and/or sensors.
- Place sample tubes securely into the sample introduction port. Otherwise it could be blown off once it is pressurized and splash sample onto the operator.
— Some sample ports contain a metal sip tube that can damage gloves when the tube is not inserted carefully.
— For better splash protection, wearing safety glasses or chemical splash goggles during acquisition is recommended.
— Automated sample loading systems reduce sample handling and also perform sample vortexing in an enclosure that prevents operator exposure to aerosols and splashes. Take special care when loading samples onto the instrument tube racks with these systems. For unfixed samples, load instrument tube racks inside a BSC or wear PPE to protect from splashes and aerosols. - Perform daily cleaning of fluidic systems and sampler following manufacturer instructions.
- Collect waste into a waste container that contains fresh concentrated household bleach in sufficient quantity to achieve a final concentration of 10% when the container is full.
Note: Adding chlorine to a waste container where ammonium chloride Tris buffer is used as a lysing agent can release dangerous chlorine gas. Use a broad-spectrum idophor instead.
10.6.5. Automated slide stainers
- Ensure the instrument is used in a manner specified by the manufacturer.
- Keep instrument covers and panels closed and secure while instrument is in use.
10.6.6. Total or semiautomated hematology test systems
- Conduct hazard and risk assessments to identify critical operations that pose a risk for exposure.
- Standard operating procedures should include
— instructions for troubleshooting tube breakage and specimen spills on conveyor tracts, belts, sorter, aliquot, and cap-piercing probe stations;
— instructions requiring at least daily cleaning and disinfection and cleaning after tube breakage and/or specimen spills; and
— appropriate PPE to be worn when cleaning and troubleshooting robotics.
Note: HEPA filters or wadded tape held with forceps are helpful for removing fine glass particles. - If instructions are not provided by the manufacturer, collaborate with the manufacturer to develop trouble-shooting and cleaning procedures that will protect the operator and be compatible with and extend the life of the robotic equipment.
10.7. Rapid Testing (Kits) (Section 3.18)
Consider used testing kits to be contaminated, and dispose of them appropriately in accordance with applicable local and state environmental regulations.
10.8. Molecular Testing (198)
- The Clinical and Laboratory Standards Institute has published standards for nucleic acid amplification assays for hematopathology (199).
- Unidirectional work flow and spatial separation of work areas must be strictly adhered to in addition to standard laboratory safety guidelines for open bench and instrument operations.
— Reagent preparation is the cleanest area, then specimen preparation area, and finally product detection area.
— Leave transportables, e.g. pens, tape, scissors, glove boxes, in each designated area (see Section 3.19). Color-coding each area and using color-coded tape and color-coded laboratory coats helps enforce spatial separations of work areas and retain transportables in their designated areas.
— Change laboratory coats and gloves and wash hands before entering each area (see Section 3.19).
10.9. Phlebotomy
- Evaluate, select, and use engineered sharps injury prevention devices that are acceptable for clinical care and provide optimal protection against injuries. Evaluation of engineered sharps injury prevention devices and consideration of their prospective use should involve employees who use sharps.
- Establish a process providing annual evaluation and selection of sharps injury prevention devices (196; Section 3.14).
10.9.1. General recommendations (200–203)
- Ensure sharps disposal containers are easily accessible in patient rooms and in patient drawing areas; are never more than three-fourths full; and are included in the annual evaluation, selection, and use of sharps injury prevention devices (204).
- Ensure all applicable patient care and infection control polices and procedures are strictly adhered to in patient drawing areas.
- Wash hands with simple soap and water or antimicrobial solution to protect against external and internal exposure to bloodborne pathogens. Ensure hands are washed before gloving; after gloves are removed; after contact with each patient or patient sample; before leaving the laboratory, drawing station, or patient room; before eating; and after hands have touched a possibly contaminated surface.
- Establish a process and procedure for specimen transport within and, if applicable, outside the facility (93).
— If patient specimens are transported from the drawing station to another area of the facility, ensure they are transported in a secondary container that has a tight-fitting latchable cover and is constructed of material to contain blood spills.
— The phlebotomy service procedure manual must include spill response and spill cleanup instructions for all areas of the hospital where there is potential for specimen spills (including such areas as elevators and stairwells).
— Ensure courier services employed by the laboratory enforce the laboratory policy for transportation of specimens, and ensure documentation of personnel training and competency assessment regarding specimen transport (including procedures for spill response, cleanup, and incident reporting).
10.9.2. Dirty versus clean areas in the laboratory
See Section 3.16.
10.9.3. Pneumatic tube systems
See Section 3.1.6.
10.9.4. Personal precautions
See Section 3.2.
10.9.5 Disinfection of work space
See Section 3.4.1.
Regardless of the method, the purpose of decontamination is to protect the phlebotomist, the patient and the environment, and anyone who enters a patient room/drawing station or who handles materials that have been carried into or out of the patient room/drawing station.
- Limit the phlebotomy materials brought into a patient room.
- Routinely clean environmental surfaces before setting up the patient room/drawing station work area, and again before leaving the patient room or after each patient in the drawing station work area.
- Disinfect any item (e.g., pen, telephone) touched with used gloves.
- Do not use alcohols or alcohol-based solutions to disinfect surface areas because they evaporate readily, which significantly decreases efficacy. Instead use aqueous disinfectants such as 1:10 dilution of household bleach or the hospital-recommended disinfectant.
- Use disinfectants recommended for environmental surfaces, such as 1:10 dilution of household bleach or other EPA- registered disinfectants effective against hepatitis B virus, human immunodeficiency virus, and other bloodborne pathogens (205).
10.9.6. Disinfecting patient room work areas and drawing stations
See Section 3.4.1.
- Include instructions in the procedure manual regarding what PPE to use, how to clean, what disinfectant to use, and how to dispose of the materials. Post the instructions in the phlebotomy office for reference.
- Allow dried blood or body fluid at least 20 minutes' contact with the tuberculocidal disinfectant to allow permeation and easy removal.
- Never use a knife or other instrument to scrape dried blood or body fluid from surface areas because this can generate aerosols.
10.9.7. Documentation of training and competency assessment in phlebotomy
Assessment includes knowledge of, and adherence to, any applicable hospital infection control policies/procedures in patient settings and the concept of Standard Precautions.
11. Blood Bank
11.1. Transfusion-Transmitted Diseases
Many infectious agents are transmitted through transfusion of infected blood; these include hepatitis B virus, hepatitis C virus, human immunodeficiency viruses 1 and 2, human T-cell lymphotropic viruses (HTLV-I and II), cytomegalovirus, parvovirus B19, West Nile virus , dengue virus, trypanosomiasis, malaria, and variant Creutzfeldt-Jakob disease. The AABB provides information on transfusion-transmitted diseases as well, available at http://www.aabb.org/Pages/Homepage.aspx.
11.2. Bloodborne Pathogen Standard
The Occupational Safety and Health Administration's (OSHA) Bloodborne Pathogen Standard, 29 CFR 1910.1030 must be adhered to in the blood bank laboratory (33).
11.3. Specimen Receiving and Log-In/Setup Station
Guidelines for receiving and logging specimens and handling specimen containers are described in Section 3.1.
11.4. Work at the Open Bench
Written procedures for blood bank include specific work practices and work practice controls to mitigate potential exposures. Standard operational procedures (SOPs) and procedure manuals are described in Section 3.1.4.
11.4.1. Unfixed specimens
- Microscopes
— Disinfect the stage, eyepieces, knobs and any other contaminated parts after use or according to a specified schedule determined by the laboratory.
— Select an appropriate disinfectant that will be noncorrosive to the microscope (see Section 3.4.1). - Slide preparations
— Replace glass with plastic where possible.
— Avoid waving slides in the air or using electric fans at an open bench to air-dry slides.
— If other instruments are used to dry slides, have a risk assessment performed to measure aerosol risks. - Manual cell washing
Perform a risk/hazard assessment.
— For saline washing of cell suspensions, use of automated cell washers reduces some, but not all, of the hazards associated with this procedure.
— Dumping saline washes into pour-off containers can generate splashes and aerosols. Splashing can be minimized by using semi-automated pipettes and keeping liquid levels in pour-off containers below one-fourth full. Wear fixed shields or appropriate water-resistant personal protective equipment (PPE) to protect from splashes.
— When shaking small test tubes for resuspending red cell pellets and reading end-point agglutination, use fixed bench-top shields or wear appropriate PPE to protect from splashes and sprays. - Perform procedures using a plasma extractor or expressor with appropriate face and eye protection to protect from sprays and possible explosion of the blood/component bags.
- Use appropriate face and eye protection when cutting donor segments during either confirmatory testing or other testing.
- Wipe outer surfaces of blood bags and components with a towel moistened with appropriate disinfectant before release for infusion, ensuring that the disinfectant will not compromise the plastic bag.
- If engineering controls are in place to prevent splashes or sprays at blood bank workbenches, the requirement for PPE may be modified on the basis of an assessment and evidence of the effectiveness of the engineering control to prevent exposure to splashes or sprays.
11.4.2. Biological safety cabinet
A Class II biological safety cabinet (BSC; see Section 3.3) is required for all aerosol-generating processes.
11.4.3. Personal protective equipment
See Section 3.2.2.
11.4.4. Disinfection
See Section 3.4.
11.4.5. Decontamination and disposal of laboratory waste
See Section 3.5 for discussion, including a waste management plan.
11.5. Clean versus Dirty Areas of the Laboratory
See Section 3.16.
11.6. Instrumentation
See Section 3.17.
11.6.1. Refrigerators and freezers
For all refrigerators and freezers in the blood bank, establish a cleaning and maintenance protocol that will minimize contamination and extend the life of the equipment and also maintain the sophisticated cooling systems blood bank refrigerators require to provide uniform and quick temperature recovery when needed. (also see Section 3) Most newer blood bank laboratory refrigerators and freezers are stainless steel and have painted finishes and removable trays, which make cleaning and sanitizing an easier process.
- Manufacturer instructions for use and care of blood bank refrigerators and freezers usually include recommended PPE and type of disinfectant.
- Collaborate with the manufacturer to establish SOPs for cleaning and maintenance if instructions are not provided with the equipment.
- Clean up blood spills immediately.
- Clean refrigerator handles and outside doors around handles at the end of each shift.
- Label the refrigerator with the universal biohazard symbol and the word "Biohazard."
11.6.2. Automated blood bank analyzers
Automated or semi-automated instruments are now available that are adapted either to donor collection settings or patient transfusion settings. Although these instruments have the potential to replace much of the open bench testing in blood banks and donor collection settings, manual testing is still being used for some antibody detection and verification procedures and in smaller laboratories. All blood bank automated analyzers currently approved for use in the United States have added features to help reduce operator exposures, but they have not totally eliminated potential for exposure.
- Sample probes that move quickly or deliver fluid rapidly may generate aerosols and droplets.
- Ensure instruments are used according to manufacturer instructions.
- Ensure instrument safety shields and containment devices are in place at time of use.
- Limit the amount of hand movement near sample probes and liquid-level sensors.
- Wear gloves and use gauze pads with impermeable plastic coating on one side when using instruments for which the operator is required to wipe sample probes.
- Handle sample trays, sample cards and sample plates with caution, and cover when not being sampled to prevent spillage.
- Fill aliquot tubes using mechanical devices, and never decant (pour).
- Consider effluents of clinical analyzers to be contaminated, and dispose in compliance with state and local regulations.
- Follow manufacturer instructions for routine cleaning and trouble-shooting of specimen spills on or within an instrument, including the appropriate PPE and type of cleaning solution to be used.
- When manufacturer instructions do not include spill containment and cleanup instructions, collaborate with the instrument manufacturer to develop an SOP that will effectively protect the operator and maintain and extend the instrument's operational life.
11.6.3. Total or semiautomated test systems
See Section 10.6.6.
11.7. Test Kits and Reagent Trays
See Section 10.7.
11.8. Donor Blood Collection, Apheresis, and Disposition
Donor collection and apheresis areas are considered patient care settings, and all applicable hospital patient care and infection control polices/procedures must be strictly adhered to.
- Establish and maintain processes and procedures to control the quality of infectious disease testing and safe disposition and transport of all collected blood and blood products.
- Use Standard Precautions.
- Use risk and hazard assessments to determine what or when additional PPE might be appropriate.
- Ensure cover/caps on any additional specimens are tightly secured before transport.
- Place blood or blood products that are being transported from the collection site to another location in a secondary container which, in addition to maintaining a specified temperature range, also has a tight-fitting cover and is constructed of material to contain blood spills.
- Place single-donor units or components issued for transfusion within a secondary container to contain spills, especially when environmental conditions might cause rupture (e.g., pneumatic tube systems).
- Select and use engineered sharps injury prevention devices that are acceptable for clinical care and provide optimal protection against injuries. See Section 3.14.1 for discussion of preventing punctures and cuts.
- Establish a process to provide annual evaluation of use and selection of sharps injury prevention devices (196).
- Provide easy access to sharps disposal containers; never fill them more than three-fourths full; and include them in the annual evaluation, selection, and use of sharps injury prevention devices.
- Do not store donor blood and components with patient specimens and reagent trays.
- Document training and assess competency of laboratory staff. Include knowledge of, and adherence to, hospital infection control policies/procedures in patient settings and the concept of Standard Precautions in all documented training.
- Discard outdated blood, blood components, and tissue in compliance with federal, state and local regulations.
12. Veterinary Diagnostic Laboratory
12.1. Introduction
This section provides practical guidelines for work practices that minimize biosafety hazards from veterinary diagnostic specimens. Many of the biosafety practice guidelines for human clinical microbiology laboratories are applicable in veterinary diagnostic laboratories. Similar to human clinical microbiology laboratories, the nature of the work performed in veterinary diagnostic laboratories puts these laboratorians, too, at risk for laboratory-acquired infections. Sixty percent of infectious diseases in humans are due to multihost pathogens that move across species lines (206,207), and during the past 30 years, 75% of the emerging human pathogen diseases (e.g., West Nile virus fever, highly pathogenic avian influenza, Lyme disease) have been zoonotic, i.e., transmitted between humans and animals (208). All nonhuman diagnostic specimens are potentially infectious to humans, although the degree of risk is less so than with handling and examination of human diagnostic specimens. Potential infectious agents in human diagnostic specimens are by definition human pathogens. Conversely, not all potential infectious agents in animal diagnostic specimens are human pathogens. The key to managing biosafety risk in veterinary diagnostic laboratories depends not only upon good general biosafety practices but, more importantly, on a practical risk assessment of the "unknown" diagnostic specimen.
In general, veterinary diagnostic laboratories use biosafety level (BSL)-2 practices and facilities for general veterinary diagnostic work and do practical risk assessment of incoming accessions to determine whether decreased (BSL-1) or increased (BSL-3) biosafety practices or facilities are warranted. Where biosafety risk and practices differ between handling of human and animal diagnostic specimens, those differences are highlighted in this section.
12.2. Biological Risk Classification and Assessment
12.2.1. Risk classification
Two classifications of risk groups have been developed to facilitate the assessment of risk from various microbes and to recommend appropriate safety practices for the handling of those microbes (1). The World Organization for Animal Health (OIE) and World Health Organization (WHO) list four groups of biohazardous agents for humans and animals based upon level of risk and availability of effective treatment and prevention (Table 12) (209). CDC/National Institutes of Health (CDC/NIH) guidelines propose four biosafety levels and recommendations for appropriate containment practices for agents known to cause laboratory-acquired infections (Tables 12,13) (1). The two lists of risk groups are roughly equivalent, and neither makes allowance for persons who are particularly susceptible to infections by pre-existing conditions, such as a compromised immune system or pregnancy. In both risk group classification systems, increasing risk levels (numbers) imply increasing occupational risk from exposure to an agent and the need for additional containment for work with that agent.
- Generally, work in routine veterinary diagnostic laboratories assumes that clinical specimens contain group 2 agents and operate with BSL-2 practices, unless a risk assessment indicates otherwise. On occasion, veterinary diagnostic laboratories might encounter group 3 agents and use BSL-3 practices. Only under extraordinary circumstances would veterinary diagnostic specimens contain risk group 4 agents; these are not included here. Examples of risk group 2 and group 3 agents commonly encountered in veterinary diagnostic laboratories are listed (Box 2).
- In addition to zoonotic agents, veterinary diagnostic laboratories must assess for the suspected presence of "high-consequence livestock pathogens" during risk assessment. High-consequence livestock pathogens are defined by the U.S. Department of Agriculture (USDA), Animal and Plant Health Inspection Service (APHIS) Agricultural Select Agent program in accordance with select agent and toxin regulations published in 2008 in the U.S. Federal Register by the U.S. Department of Health and Human Services (42 CFS part 73) and by USDA (9 CFS part 121, and 7 CFS part 331) (40). Criteria used to classify high-consequence livestock pathogens included severity of effect on animal products, virulence and transmissibility of the agent, and availability of effective treatment. Although not necessarily zoonotic agents, high-consequence livestock pathogens can have severe detrimental economic impact on agricultural animal health and require handling using BSL-3 practices and facilities to prevent environmental dispersement and contamination (Box 3).
12.2.2. Risk assessment
See Section 2 for detailed risk assessment guidelines.
- There is no official standard approach, method, or one correct way to conduct a risk assessment, but several strategies are available, such as using a risk prioritization matrix, conducting a job hazard analysis, or simply listing the potential scenarios of what could go wrong while conducting a procedure, task, or activity. Risk assessment in veterinary diagnostic laboratories takes into account the likelihood of various risk group or BSL microorganisms being present in unknown clinical samples, plus the likelihood that routine processing of the clinical samples would expose laboratory workers to infectious agents in those samples. A risk assessment will consider the source of the clinical sample (including host species and clinical history), the suspected pathogen within a specimen with its inherent risk group characteristics, the work activity during diagnostic workup of the clinical sample in the laboratory, and the competencies and experience of the laboratory personnel.
- The most critical risk assessments for veterinary diagnostic laboratories are consideration of host species, the known medical condition and clinical history of the patient, clinical signs of the patient, and endemic local geographic conditions. (Many laboratories receive samples from wide and diverse geographic areas.)
- Certain risk group 3 agents are endemic to specific geographic regions or specific species and clinical syndromes and would warrant increasing BSL practices to appropriate levels. Examples include tularemia causing sudden death in western gray squirrels in the western United States, anthrax causing sudden death in cattle in the north central United States, Coxiella burnetii causing ovine abortions, and psittacosis resulting in respiratory or enteric disease in aviary birds. Alternatively, risk assessment might indicate a reduction of biosafety practices from routine BSL-2 practices. An example would be animal blood samples submitted for serologic analysis. Unlike human blood samples that can harbor bloodborne human pathogens such as human immunodeficiency virus or hepatitis virus, animal serum in general does not contain zoonotic bloodborne pathogens and could often, based upon a risk assessment, be handled using BSL-1 practices.
- The assessment of clinical history and other data provided on a laboratory accession/submission form depends upon professional judgment and is to be conducted or overseen by a qualified veterinarian familiar with the zoonotic and select agents (Boxes 2,3) and the diseases caused by those agents. If questions arise during case accessioning and log-in that cannot be clarified from the accession paperwork, contact the submitting veterinarian by telephone. Knowledge regarding typical clinical signs, host range, basic epidemiology and geographic distribution of diseases caused by these agents is essential.
- Biosafety risk assessment for veterinary diagnostic specimens is critical because the pathogenic potential for humans in veterinary diagnostic specimens is different from that in human specimens.
12.3. General Biosafety Guidelines
See Section 3 for extensive and detailed biosafety guidelines generally applicable to all subdiscipline areas within a veterinary diagnostic laboratory.
- The person most at risk of exposure and laboratory-acquired infection is the laboratorian working to identify a suspect infectious agent within the diagnostic specimen; therefore, the choice of laboratory work practices to prevent personal exposure is one of the most important decisions in designing a laboratory biosafety plan.
- In the laboratory, the routes of exposure are limited and include inhalation of fine-droplet infectious aerosols by the airborne route, direct contact on skin or mucous membranes or ingestion of large-droplet infectious material, or percutaneous transmission by needles or other sharps (Table 1). The National Research Council Committee on Hazardous Biological Substances in the Laboratory in 1989 recommended seven basic prudent biosafety practices to avoid exposure to infectious agents via the most common routes of laboratory infection (210). These practices, although identified as the most important, are to be supplemented by additional practices, equipment and facility design whenever there is an increased risk of exposure to a biosafety hazard or the possibility of exposure to a BSL-3 agent.
12.3.1. Hand washing
- Hand washing is the most important procedure to reduce the duration of exposure to an infectious agent and prevent dissemination of the infectious agent. Hand contamination occurs during manipulation of specimens and contact with work surfaces, telephones and equipment.
- Laboratory personnel must wash their hands
— immediately after removing gloves
— after obvious contamination;
— after completion of work;
— before leaving the laboratory; and
— before hand contact with nonintact skin, eyes or mucous membranes.
12.3.2. Personal Protective Equipment
- For routine work in veterinary diagnostic and clinical laboratories personal protective equipment (PPE) must be provided, used, and maintained in the laboratory workspace. Train laboratory workers in the use of PPE for specific tasks and to know the limitations of PPE and the appropriate procedures for maintaining and disposing of PPE. The level of PPE use in routine veterinary diagnostic laboratory work should be sufficient for the BSL practices appropriate to the suspected or identified risk, and, at a minimum, include gloves and protective clothing.
- Gloves protect the wearer from exposure to potentially infectious material and are usually thin latex, vinyl or nitrile. They are to be changed frequently. Protective clothing includes fully closable long-sleeved coats or gowns that extend below the level of the workbench. Open-toe shoes are not to be worn in the laboratory to prevent accidental spillage on bare skin. Do not wear protective clothing outside the laboratory or take home for cleaning or laundering.
- Use face and eye protection when splashes or sprays of potentially infectious material might be generated during laboratory processing, including during laboratory cleanup. Face and eye protection equipment could include splash goggles, face shields or bench top splash guards.
- Use respiratory protection devices (respirators) if risk assessment indicates BSL practices appropriate to prevent inhalation of potentially infectious aerosols. The decision to use respirators in the laboratory may come either from the inherent risk of potential infectious agents in a clinical specimen (e.g., BSL-3 or risk group 3 agents) or from laboratory manipulations necessary for agent identification that may produce infectious aerosols. The type of respiratory protection depends upon the specific hazard. If an N95 or higher-rated respirator cannot be used (e.g., because of facial hair or asthma), wear a powered air purifying respirator (PAPR) when respiratory protection is required. Surgical masks are not effective respiratory protection.
12.3.3. Staff training
- Biosafety training and education of workers about potential hazards and safe work practices are essential to creating a safe work environment. The size of the safety training program will vary with needs but should include
— Standard Precautions;
— selection, use and limitations of PPE;
— management of biohazardous waste;
— postexposure management, reporting and investigation of incidents;
— bloodborne pathogen information;
— basic understanding of risk groups and risk assessment; and
— procedures for biohazardous spills. - Document biosafety training (date and content of training) in the employee's training record, and maintain the record for 3 years. Evaluate the effectiveness of laboratory safety training periodically. Safety assessments could include safety audits, inspections by outside agencies, review of incident reports, and observations and suggestions made by employees. Although management provides resources to address and correct safety deficiencies, the efforts of laboratorians working at the bench top provide the foundation of a safe work environment.
12.3.4. Biological spill management
- Management of biological spills in clinical laboratories must account for the specific infectious agent (if known), the volume of infectious material spilled, and the presence of aerosols. Aerosols may readily transmit in spills involving BSL-3 or risk group 3 agents. Thus, occupants must evacuate the areas immediately, close doors and not re-enter the area for 30–60 minutes.
- PPE for biological spills includes puncture-resistant gloves, N95 respirators (BSL-2 or 3), fluid impenetrable shoe covers, coats or gowns, and facial protection.
- For BSL-3 agents, use a respirator or HEPA-filtered respirator. Another option is to call a designated spill emergency response team. Remove any broken glass in a spill area, and discard without contact with the hands (use [e.g.] broom, forceps, tongs). A typical biological spill clean procedure involving a possible aerosol should include the following:
— Alert personnel in area and evacuate.
— Close doors and do not re-enter area for 30–60 minutes (post sign forbidding entry to the area), based on the number of air exchanges.
— Alert laboratory supervisor.
— Don PPE appropriate for type of spill.
— Remove and discard broken glass or other objects (without contact with hands).
— Absorb the spill with absorbent material.
— Discard contaminated material in a biohazardous waste container.
— Clean spill site with aqueous detergent.
— Decontaminate area with appropriate disinfectant.
— Rinse spill site with water and allow site to dry.
— Copy contaminated laboratory forms and discard into the biohazard waste container.
— Place all disposable contaminated cleanup material in the biohazard bag and treat as infectious waste.
— Wash hands.
— Prepare a spill/incident report, identify cause of spill, and determine remedial action. - If a spill occurs in a biological safety cabinet (BSC), do not turn off the cabinet fan. Minor spills in a BSC can be absorbed with absorbent paper. If infectious material flows into the grille, wipe all items in the cabinet with disinfectant and remove them. Close the drain valve and pour disinfectant onto the surface and through the grille into the drain pan. Allow appropriate contact time, then drain, rinse and dry.
- When breakage occurs in a centrifuge (which inherently would produce aerosols), keep the centrifuge tightly closed for 30 minutes before decontamination commences.
- If a specimen tube breaks within the plastic screw-capped canister in a centrifuge:
— Turn the motor off.
— Remove the canister immediately and place in a BSC.
— Notify senior person in charge and other colleagues working in the area.
— While wearing protective clothing, open the canister under the safety cabinet.
— Pour a 1:10 dilution of bleach or a noncorrosive disinfectant into the canister to decontaminate all surfaces. Let the canister soak in bleach or disinfectant solution for 10 minutes. Clean canister thoroughly.
— Do not pick up broken glass with gloved hands. Use forceps or cotton held in forceps, or tongs or hemostats, and dispose into a biosafety sharps container.
— Discard all nonsharp contaminated materials from canister into a red biohazard bag for biohazard waste disposal.
— Swab unbroken capped tubes with the same disinfectant; then swab again, wash with water and dry.
— Treat all materials used during the cleanup as infectious waste.
Note: If the specimen tube breaks in a centrifuge that does not have individual canisters but does have a biohazard cover and sealed rotor, follow the manufacturer's instructions for cleaning and decontamination.
12.3.5. Immunization
- The Advisory Committee on Immunization Practices, in addition to recommending immunization of health-care personnel with vaccines recommended for all adults (influenza, measles/mumps/rubella, varicella, and tetanus/diphtheria/pertussis), recommends meningococcal or hepatitis B vaccination for those at risk for occupational exposure (211). These agents are not present in animal diagnostic specimens. In veterinary diagnostic laboratories, it is not recommended that laboratorians be immunized routinely against potential risk group 3 or BSL-3 pathogens. The only situation in which immunization is to be considered is against rabies for laboratorians processing a large number of specimens potentially containing rabies virus (e.g., routinely processing central nervous tissues from animals with neurologic disease compatible with rabies) (Table 14) (212).
12.4. Pathology (Necropsy and Surgical Pathology)
See Section 5 for detailed biosafety guidelines applicable to necropsy, surgical pathology, and histology working areas in a veterinary diagnostic laboratory.
12.5. Parasitology
See Section 6.
12.6. Mycology
See Section 7.
12.7. Virology
See Section 8.
12.8. Toxicology
See Section 9.
12.9. Hematology/Serology
See Section 10.
12.10. Molecular Diagnostics and Rapid Tests
Biosafety guidelines to be followed when conducting molecular diagnostic testing (i.e., polymerase chain reaction [PCR]) or using rapid tests such as enzyme-linked immunosorbent assay (ELISA) can be specific to the particular testing being conducted. These are discussed in Section 3 and Sections 4, 5, 6, 7, 8 and 10, which deal with specific types of pathogens and testing. Section 8.10 provides the most thorough biosafety guidelines for molecular diagnostic testing.
12.11. Storage, Packaging, and Shipping
See Section 13 for detailed biosafety guidelines applicable to functions within a veterinary diagnostic laboratory regarding storage, packaging and shipping of infectious or diagnostic specimens.
12.12. Biosafety Education/Training
See Section 15 for practical guidelines regarding biosafety training within a veterinary diagnostic laboratory.
12.13. Biosafety Quality Improvement
See Section 16 for guidelines regarding continual improvement of biosafety within a veterinary diagnostic laboratory.
13. Storing, Packaging, and Shipping Infectious Substances
13.1. Storage of Infectious Substances
Infectious substances in a clinical microbiology laboratory are encountered as fresh and processed patient specimens, cultures and subcultures, stored isolates, and serum or plasma. Invariably, all of these substances must occasionally be stored in some form and for some length of time, and many of these substances will be manipulated, relocated, and otherwise touched by laboratory workers. Therefore, storage of infectious substances is an important and integral component of worker safety in clinical microbiology laboratories. Handle all stored infectious substances using Standard Precautions and aseptic technique. Organisms responsible for external contamination of the storage vial will remain viable during storage and can be transmitted by manipulating the vial.
- Use primary containers with tight-fitting lids.
- Store as far as possible from common walkways, laboratory cart, human traffic, and reagents.
- Ensure restricted access to the storage site.
- Ensure storage at temperatures appropriate to maintain viability of microorganisms.
- Use sturdy racks, buckets, or boxes that will ensure the item will remain upright.
- Provide storage cabinets or refrigerators with latching doors.
- Use separate refrigerators to store long-term cultures archives, subcultures, and processed patient specimens.
13.2. Packing and Shipping Infectious Substances
- Use of reference laboratories for routine and specialized testing has increased in recent years. Some reference laboratories may not be able to send a courier to collect specimens from the submitting laboratory. Therefore, knowledge of specimen transportation standards and requirements needed to forward specimens using common air and ground carriers has become progressively more important for diagnostic laboratories.
- The American Society for Microbiology has published comprehensive guidelines for packing and shipping infectious substances (213–215). They are based on guidelines issued by the International Air Transport Association (IATA) and the U.S. Department of Transportation (DOT). The information presented here is an abridged version of these guidelines and is not intended to be an all-inclusive guide to packing and shipping infectious substances. Permits may be needed in addition to these requirements.
Note: The requirements and regulations governing the transport of infectious substances change frequently. Shippers are responsible for being aware of these changes, adhering to current regulations, obtaining permits in advance of shipping, and interpreting applicable regulations for themselves and their facilities. Persons shipping these substances are advised to check the web sites of the respective appropriate agencies.
13.2.1. Governing authorities and regulations
- The most recognized and used packing and shipping guidelines in the world are those in the annual IATA publication Dangerous Goods Regulations (216).
- In the United States, DOT regulates the commercial transportation of dangerous goods (e.g., explosives, gases, flammable liquids, infectious substances, and radioactive materials) by both air and ground carriers (93). The DOT regulations are in substantial agreement with IATA.
13.2.2. Importance of regulations
The purpose of the regulations is to protect the public, emergency responders, laboratory workers, and personnel in the transportation industry from accidental exposure to the infectious contents of the packages. An important non–safety-related benefit of adherence to these regulations and requirements is minimizing the potential for damage to the contents of the package during transport and reducing the exposure of the shipper to criminal and civil liability associated with improper shipment of dangerous goods.
13.2.3. Exceptions
- Transportation of small quantities of non–Category A infectious substances (usually specimens being transported for clinical, diagnostic, or other patient care purposes; see Section 13.3) is exempt from most DOT regulations if the specimens are transported by courier, i.e., private or contract carrier in a motor vehicle used exclusively to transport such substances (93).
- Pack and secure non–Category A infectious substances inside the vehicle according to DOT regulations.
- These DOT regulations are usually less stringent than Occupational Safety and Health Administration (OSHA) regulations and state that the substances need only be in leakproof containers, sealed securely, and secured within the vehicle during transport. The usual OSHA regulations still apply during courier transportation of infectious substances. Refer to DOT and IATA websites for updates.
13.2.4. Specific regulations
- The safe and legal transport of infectious substances is based on the following mandated activities:
— training every 2 (IATA) or 3 (DOT) years for persons handling infectious substances for shipment;
— classification and naming of infectious substances;
— selection of correct packaging materials;
— packing shipments correctly;
— placing appropriate markings and labels onto the outer package; and
— documenting relevant aspects of each package and its contents.
13.2.5. U.S. Postal Service
The U.S. Postal Service publishes its own regulations in the USPS Domestic Mail Manual (96). The USPS regulations for mailing hazardous materials generally adhere to DOT regulations; however, consult the USPS Domestic Mail Manual for specific needs and requirements.
13.3. Classification of Infectious Substances
13.3.1. Classification
All shipped goods must be classified using a three-step process to define dangerous goods that are shipped by commercial carriers. Classification allows the shipper to select the proper IATA packing instructions and directions to use, and provides information necessary to complete required documentation (a Shipper's Declaration for Dangerous Goods) if the substance is a Category A infectious substance.
13.3.2. Steps of classification
- The material is classified into one of the nine IATA-specified classes (Class 1 through Class 9) of dangerous goods. Infectious and toxic substances are Class 6 dangerous goods; dry ice is a Class 9 dangerous good. Class 6 and Class 9 substances usually are the only dangerous goods shipped by laboratorians.
- Class 6 substances must be categorized into either Division 6.1 (toxic substances) or Division 6.2 (infectious substances).
- Division 6.2 infectious substances must be classified into one of nine IATA-specified types of infectious substances: Category A infectious substance, Category B infectious substance, patient specimens, exempt human or animal specimens, genetically modified organisms, exempt substances, biological products, infected animals, or medical waste (Table 15).
- If the substance is determined to be either a "patient specimen" or an "organism" and is not obviously a Category A or Category B substance but meets the criteria of or has characteristics of a Category A or Category B substance, the shipper must classify it as a Category A or Category B substance. Otherwise, the substance must be classified as an "exempt human or animal specimen" or a "genetically modified organism" (Class 9), respectively.
13.3.3. Category A infectious substances
A Category A substance is "an infectious substance which is transported in a form that, when exposure to it occurs, is capable of causing permanent disability, or life-threatening or fatal disease to otherwise healthy humans or animals" (93).
- Category A substances are specifically designated and listed by IATA and DOT. The list of Category A substances is not all-inclusive; the shipper is allowed by IATA to perform a thorough risk assessment and to use discretion and professional judgment when deciding if a substance meets Category A criteria. Category A substances must be assigned UN number UN2814 or UN2900 (Figure 2).
Note: Certain Category A infectious substances are considered Category A only if the substance is in culture form, i.e., concentrated; this distinction is clearly indicated in the specific IATA list. - Certain Category A pathogens have been designated as agents of bioterrorism and are known as select agents (40). Federal regulations require shippers to have special registration and permits to possess, use, transfer, and receive select agents.
13.3.4. Category B infectious substances
A Category B substance is "an infectious substance that does not meet the criteria for inclusion in Category A" (93). Category B substances are not in a form generally capable of causing disability, life-threatening illness, or fatal disease. Category B substances must be assigned UN number UN3373 (Biological Substance, Category B). Following are examples of possible Category B substances:
- Typical clinical, diagnostic, or patient specimens, e.g., blood, biopsies, swab specimens, excreta, secreta, body fluids, or tissues (a) being shipped for routine culturing or screening testing for non–Category A infectious microorganism(s), or (b) suspected of containing a non–Category A microorganism(s);
- Typical clinical laboratory cultures (usually on solid or in liquid media) of non–Category A microorganisms routinely encountered and manipulated in clinical microbiology laboratories (Figure 2).
13.3.5. Exempt human (or animal) specimens
Exempt human or animal body site specimens are those for which there is "minimal likelihood there are pathogens present" (93). Examples of such specimens include urine or serum to be tested for glucose, cholesterol, hormone levels, prostate-specific antigen, and analytes used to evaluate heart and kidney function.
- Professional judgment and knowledge of patient medical history may used to determine if the specimen is an infectious risk or contains pathogens.
- Exempt human or animal specimens have less stringent packaging requirements than do Category A and Category B substances. IATA requires outer packages containing exempt human or animal specimens to be clearly labeled as "Exempt Human Specimen" or "Exempt Animal Specimen." DOT does not require this label on outer packages (Figure 2).
13.3.6. Exempt substances
Many substances commonly encountered in clinical laboratories are exempt from strict infectious substance shipping requirements (Figure 2). Examples of such substances are
- Substances that do not contain infectious substances or are unlikely to cause disease in humans and animals;
- Most environmental samples (e.g., food, soil);
- Substances that contain neutralized or inactivated microorganisms;
- Substances to be tested for alcohol or drugs, pregnancy indicators, cancer, and antibodies;
- Samples submitted for forensic analysis;
- Dried blood spots and fecal occult blood screen specimens;
- Blood and blood components collected for the purpose of transfusion or transplantation;
- Food and Drug Administration (FDA)-approved and FDA-licensed biological products; and
- <30 mL of 10% formalin per primary container when the formalin is used as a preservative.
13.3.7. Patient specimens
- A "patient specimen" is material collected directly from humans or animals for diagnostic, treatment, prevention, investigational, or research purposes.
- Patient specimens that meet Category A or Category B criteria must be classified as Category A or Category B substances.
- Patient specimens that meet neither Category A nor Category B criteria must be treated as exempt human or animal specimens (Figure 2).
13.3.8. Genetically modified organisms
Genetically modified organisms usually meet either Category A or Category B criteria. If this is not the case, the organism must be classified as a "genetically modified microorganism" (Class 9, Miscellaneous Dangerous Goods) and packed and shipped as such.
13.3.9. Biological products
Virtually all commercially available biological products are exempt from regulations for packing and shipping infectious substances. Examples of biological products include bacterial typing sera, vaccines, bacterial antigens, antimicrobial agents, reagents for identifying bacteria, and reagents used in antimicrobial susceptibility testing.
13.3.10. Infected animal
- A live, intentionally infected animal that is known or reasonably expected to contain an infectious substance cannot be transported by air unless the substance cannot be transported by any other means. An exemption from DOT will be required.
- Consultation with individual commercial carriers is advised if either live or dead infected animals need to be shipped.
13.3.11. Medical waste
- Medical waste that contains Category A or Category B infectious substances must be packed and shipped as such and assigned number UN2814, UN2900, or UN3373.
- Medical waste that is reasonably believed to have a low probability of containing infectious substances must be packed and shipped as "medical waste not otherwise specified" (n.o.s) (UN3291).
13.4. Naming Category A and Category B Substances
- After classifying the substance, the shipper must identify (officially name) the Category A and Category B infectious substances by assigning the substance one of the >3,000 IATA-specified and internationally recognized UN numbers and proper shipping names listed in the blue pages section of the IATA Dangerous Goods Regulations (216).
- This list provides 14 informational items for each of the proper shipping names; the items correspond to the information needed to complete the Shipper's Declaration for Dangerous Goods. Fortunately, only seven of the 3,000 proper shipping names are used by most clinical microbiology laboratories:
— Two for Category A substances that affect humans;
— Two for Category A infectious substances that affect animals;
— One for a Category B infectious substance;
— One for genetically modified organisms;
— One for dry ice. - The blue pages provide proper shipping names, UN numbers, packing instructions, quantity limits, and other information related to packing and shipping substances.
13.5 Packing Instructions and Packing Substances
13.5.1. Packing instructions and directions
- IATA packing instructions (PI) describe the minimum standards for safe transport of infectious substances.
- The instructions used by clinical laboratories are those that relate to shipping Category A infectious substances (PI 620), Category B infectious substances (PI 650), and dry ice (PI 954).
- There are no specifically PI-numbered instructions for specimens classified as "exempt human or animal specimens"; however, IATA provides directions that must be followed (Table 16).
13.5.2. Marking and labeling outer packages
- Shippers are responsible for the proper marking and labeling of the outer shipping container (commonly, a cardboard box).
- The markings and labels communicate essential information regarding the shipper and consignee of the package, nature and weight of the contents of the package, the potential hazard of the substance, how the substance is packed, and information to be used in case of an emergency (Figures 3,4).
13.5.3. Specific markings and labels
The following list cites the situations requiring a marker or label, and the specific markings or labels for that situation.
- Shipper and consignee — shipper's and consignee's name and address;
- Responsible person — name and telephone number of someone who can answer general questions about the shipment;
- Category A Substance — a Class 6 diamond-shaped "Infectious Substance" label, and a proper shipping name (not the technical name), UN number, and quantity label;
- Category B Substance — "Biological Substance, Category B" and "UN3373" labels;
- Dry ice — Class 9 "Miscellaneous Dangerous Goods" and weight of dry ice label;
- Package orientation — arrows on opposite sides of packages containing >50 mL of a liquid or frozen liquid;
- Exempt patient specimens — "Exempt Human Specimen" or "Exempt Animal Specimen" label;
- Outer package containing Category A infectious substance — a "UN" label; a UN inside of a circle, and a series of letters and numbers that indicate the type of package, class of goods the package is designed to carry, manufacturing date, authorizing agency, and manufacturer.
13.6. Documentation
13.6.1. Shipper's Declaration for Dangerous Goods
- A Shipper's Declaration is a legal contract between the shipper and carrier. It is required to document the shipment of Category A infectious substances and must be accurate and legible.
- Essentially all of the IATA-specified technical information required to complete the "Nature and Quantity of Dangerous Goods" section of the Declaration can be found in the blue pages of IATA Dangerous Goods Regulations (216).
13.6.2. Emergency response telephone number
- DOT (but not IATA) regulations state that an emergency response telephone number must be provided on Shipper's Declarations that accompany shipments of Category A infectious substances.
- The number must be monitored at all times by a person (not an answering machine, message service, or pager) who has knowledge of the hazards of the material being shipped and emergency response and incident mitigation information in case a handler comes in contact with the released contents of the package. This number also can be that of a commercial service which can provide the appropriate mitigation information. If the telephone number of a commercial service is used, the contract number or service agreement number assigned by the commercial service must be entered on the document, as well as the name of the service provider.
13.6.3. Airbills
IATA carriers are required to prepare airbills to describe air cargo and accompany shipments in transit. Some dangerous goods shipments, such as Biological Substances Category B shipments, require preparation of this document but not a Shipper's Declaration. Specific preparation instructions are detailed in each IATA package instruction and in the "Documentation" section of the Dangerous Goods Regulations.
13.7. Refrigerants
Packaging must be leakproof when wet ice is used. Dry ice is a Class 9 dangerous good; it must be packaged according to PI 954, and its use requires completion of a Shipper's Declaration if it is used to ship a Category A substance.
Note: Dry ice is an explosion hazard and must never be placed into a tightly sealed container. Dry ice must be placed outside the secondary container, and the outer packaging must permit the release of CO.
13.8. Training and Certification
- Anyone involved in packing and shipping infectious substances must receive formal training in this activity; every 2 years by IATA, and every 3 years by DOT. The essential components of a training program must include
— general awareness and familiarity with packing and shipping infectious substances;
— importance, nature, and contents of IATA and DOT regulations;
— hands-on and/or demonstrations of packaging and packing techniques;
— marking and labeling;
— documentation of shipments of dangerous goods;
— safety training;
— pre- and posttraining testing; and
— issuance of a certificate after successful completion of the training. - Acceptable training materials and methods include manuals, training courses, and workshops, all of which are commercially available from professional organizations and commercial suppliers of packaging materials for dangerous goods. A training program or workshop that includes didactic, hands-on training and demonstrations can be developed by any hospital, laboratory, school, institution, or other facility.
- IATA and DOT require all aspects of training to be documented. The most important document used to prove appropriate and timely training is a certificate issued after training is complete.
— Employers are to keep a record for each employee who is trained.
— The record should include employee's name, location and date of training, name of the trainer, course content, documentation of testing, and a copy of the certificate of training. IATA and DOT certification is valid for 2 and 3 years, respectively. - DOT, through its Pipeline and Hazardous Materials Safety Administration (PHMSA), and the Federal Aviation Administration have authority to perform unannounced inspections of clinical laboratories whose employees pack and ship infectious substances, to inspect these facilities for compliance with the training regulations, and to inspect training records at these facilities. Facilities that do not comply with prescribed regulations are subject to substantial fines.
14. Emergency Procedures and Responsibilities
The risk of acquiring a laboratory-associated infection (LAI) after physically contacting a microorganism (an "exposure") in the workplace is real, always present, and an integral part of working in a diagnostic laboratory, and in particular the clinical microbiology laboratory. The potential for an exposure exists whenever a laboratorian manipulates and transports microorganisms, processes and stores patient specimens, and operates instruments used in the process. Diagnostic laboratories can be safe places to work if standard and appropriate safe work practices and procedures are easily accessible, understood by employees, enforced, and followed. These procedures are to be properly outlined in an exposure control plan and laboratory manuals. These plans are composed of essential elements related to preventing an exposure, and, equally important, they describe employer and employee involvement and responsibilities before and after an exposure. Appropriate actions taken after an exposure can greatly reduce or even eliminate the chance that an exposure will result in an LAI. Well-designed plans with the full support of the director and higher management can reduce workers' chances of exposures to microorganisms and can help ensure a culture of safety in diagnostic laboratories.
14.1. Responsibilities of Employers Before an Exposure
14.1.1. Exposure control plan
- Employers need to establish a formal plan outlining work practices and procedures to minimize the potential for an exposure.
- Employers are to have a formal plan to address actions to be taken after an employee is exposed. The plan is to incorporate at least the elements presented here (or the equivalent) and be easily accessible to employees.
- Both the employer and the employee must embrace the plan and rigorously participate in protecting themselves, their colleagues, the staff, and patients.
- Develop the plan before a first exposure with input from the director of the laboratory, laboratory workers, and representatives of employee health and infection control or a local site medical services provider to ensure the inclusion of appropriate immunization, postexposure prophylaxis, and infection control measures.
- Document annual review of safer medical devices and other advances in research technology that can reduce or eliminate potential risks of exposure.
- Develop a "safety culture" with appropriate buy-in from all levels of employees, including upper management and front-line workers. Develop a nonpunitive reporting structure for potential occupational exposures, and have provisions in place for employees who have self-reported changes in health status to be reassigned, if required, without negatively influencing their job performance or their performance review.
— Ensure that all potential incidents are reported regardless of whether the employee thinks an exposure occurred. The incident is to be evaluated by the employer and occupational physician to determine if postexposure prophylaxis is needed.
— Medical practices designated to perform these medical evaluations should be provided with the current U.S. Public Health Service recommendations for medical evaluations and postexposure prophylaxis for the infectious agents in use.
— Work collaboratively with occupational medical providers to ensure they are equipped to evaluate clinical laboratory workers who may have had occupational exposure to a variety of infectious agents.
14.1.2. Documentation of potential exposures
- Develop a record or form to include the following information:
— Date, time, and location of the exposure;
— Employee's name and employee number;
— Names of other employees related to the incident (e.g., witnesses);
— Details of the incident or exposure; brand names and manufacturer of any devices or instruments involved, including lot numbers of medical devices and personal protective equipment (PPE) used at the time of exposure;
— Relevant health information/status of the exposed employee at the time of the exposure;
— Immediate or remedial actions taken, including first aid;
— Actions recommended that the exposed employee take immediately or the same day, (e.g., visits to employee health or the emergency department, administration of chemoprophylaxis, consultations with physicians);
— Results of discussions with employee health clinicians;
— Monitoring and follow-up plans;
— Space to record results of monitoring and follow-up plans;
— Appropriate signatures (minimum: those of the employee and the employee's immediate supervisor).
14.1.3. Emergency response equipment and facilities
- Employees must receive training on the proper use of all emergency equipment.
- First aid kits must be visible and easily accessible within the laboratory. Eye wash stations and safety showers meeting American National Standards Institute (ANSI) specifications must be readily accessible and comply with ANSI standards and any Occupational Safety and Health Administration (OSHA) standards mandating their placement. Cleansing skin abrasions, flushing eyes, or showering after skin contact can greatly reduce or even eliminate the chance that an exposure will result in an LAI.
- Employees are to be made aware and frequently reminded of the importance and locations of emergency equipment and facilities. Ensure that procedures are in place to document that all equipment is functioning properly (e.g., ANSI standards for eyewashes and showers) and within acceptable expiration dates (e.g., first aid kit).
14.1.4. Immunizations
The Advisory Committee on Immunization Practices, in addition to recommending immunization of health-care personnel with vaccines recommended for all adults (influenza, measles/mumps/rubella, varicella, and tetanus/diphtheria/pertussis), recommends meningococcal or hepatitis B vaccination for those at risk for occupational exposure (211,217,218).
- Microbiologists who are routinely exposed to isolates of Neisseria meningitidis should be vaccinated (217). Laboratorians manipulating N. meningitidis isolates are among the groups at increased risk for meningococcal disease, and fatal laboratory-acquired meningococcal disease has been documented (218).
- Immunization of employees in general, and these special situations in particular, is most effectively addressed in institutional employee health or infection control policies. Consult CDC's Vaccines website for information and updated immunization recommendations (http://www.cdc.gov/vaccines). Employers are to make laboratorians aware of the existence and availability of all vaccines.
14.1.5. Education of employees
- Employees must receive initial training and refresher training at least annually, and this training is to be documented. Training will encompass all the necessary laboratory procedures employed in the facility to reduce the risk of exposure. Training includes the following information:
— Engineering controls in place (including safety equipment);
— Proper procedures used in the manipulation of samples;
— PPE and procedures to be followed in the event of an exposure. - Employees are to be made aware and frequently reminded that
— Many reasonable and helpful actions taken after an exposure can greatly reduce or even eliminate the chance that an exposure will result in an LAI.
— These actions are detailed in an exposure management plan.
— Employee health clinicians and infection control practitioners are available for consultation after an exposure.
— Immunizations appropriate for workers in clinical microbiology laboratories are available. - They are to also be made aware of symptoms of infections and follow-up procedures.
- Document that the employee has read and understood all procedures that have been adopted in support of the laboratory's formal exposure management plan.
14.2. Responsibilities of Employees Before an Exposure
It is the responsibility of laboratory employees to do the following:
- Follow Standard Precautions and other established institutional laboratory safety practices at all times.
- Never perform laboratory procedures, manipulate microorganisms, process patient specimens, or operate microbiology identification instruments if doing so will increase the risk for an LAI.
- Know what to do immediately if an exposure or suspected exposure has occurred.
— Be familiar with laboratory infection control manuals and procedures and where they are located.
— Know the location of first aid kits, eye wash stations, and showers, and how to use them. - Keep the supervisor informed of their health status (e.g., immunocompetency, cuts, abrasions, pregnancy, breathing problems) to determine the possibility of an increased LAI risk while performing a particular job.
Note: Employees are advised to report changes in health status, but they cannot be compelled to do so. A provision should be developed and in place regarding how to handle this situation. - Know any unique signs and symptoms of any aerosol-transmitted infectious agent that is routinely isolated and manipulated in the laboratory. Report such symptoms to supervisors if experienced.
14.3. Responsibilities of Employers After an Exposure
14.3.1. Determination of the extent of exposure
- The likelihood that an exposure will result in an LAI is directly related to several variables, including
— concentration of the microorganism to which the employee was exposed;
— physical form of the microorganism (e.g., broth, colony, lyophilized, aerosol);
— innate virulence of the microorganism;
— length of time the worker was exposed to the microorganism;
— proper use of PPE;
— immunocompetency status of the exposed worker; and
— portal of entry (intact, inflamed, abraded, or cut skin; needle stick; mucous membrane; respiratory route; oral route). - Immediately after any exposure:
— Examine (together with the exposed employee, if possible) all details of the exposure incident.
— Determine the degree to which these variables (and others if applicable) were involved in the exposure.
— Make a decision regarding the degree of exposure.
— Agree to an acceptable course of action to prevent or reduce the chances of the employee's developing an LAI. - Communication or consultation with the employee's health-care provider may be necessary, depending upon the severity of exposure.
Note: Most exposures in laboratories are mitigated by thorough hand washing, and although some are significant enough to document and take additional actions, all are potentially risky. - Initiate a root cause analysis to determine all facts regarding the exposure, the root causes, and an action plan to correct identified causes. The action plan needs to be developed, implemented, and monitored.
- After an incident, retraining of personnel may be warranted, and procedure manuals may require review to ensure appropriate safety measures are listed.
14.3.2. Documentation of exposures
- Document on an exposure incident form all details of the exposure and all decisions related to the exposure. The form is to be signed by the employee and the employee's immediate supervisor.
- Gather information from personnel who might have witnessed the incident or assisted in the clean-up.
- Allow employees access to medical services for consultation.
14.3.3. Consultation with employee health clinicians
The employee and the supervisor of an employee who has experienced a potential exposure are to contact the employee health physician or nurse and discuss the exposure. These clinicians are the persons most likely to provide advice regarding timely chemoprophylaxis and to able to administer appropriate antimicrobial agents.
14.3.4. Counseling exposed employees
- Keep the exposed employee well informed during the postexposure period as information continues to be gathered and documented.
- Tell the employee all the findings of the exposure investigation, the supervisor's opinion regarding the extent of exposure, what and when actions are expected to happen, whom he/she should see for medical consultation, where additional information can be found, and which postexposure prophylaxis is advised and available.
- Counsel the employee to seek medical advice and treatment for any acute illnesses that occurs after the exposure and during follow-up. The employee should receive a copy of the initial and final exposure report forms.
14.3.5. Exposure to Mycobacterium tuberculosis
- Approaches to situations in which an employee has likely been exposed to Mycobacterium tuberculosis are best addressed in institutional employee health or infection control policies, many of which are based on CDC guidelines (219).
- If an employee is exposed to M. tuberculosis, e.g., by possible inhalation of aerosolized M. tuberculosis, the employer should arrange to have the employee skin- or blood-tested to determine if an exposure has occurred. The results of this test can be compared with the employee's baseline or previous annual test to detect a conversion.
14.3.6. Exposure to Neisseria meningitidis
- Employees who are exposed percutaneously to a N. meningitidis isolate from a sterile site should receive treatment with an appropriate antibiotic.
- Employees who have a mucosal exposure to a N. meningitidis isolate from a sterile site should also receive antimicrobial chemoprophylaxis.
14.3.7. Exposure to bloodborne pathogens
- Approaches to employee percutaneous or membrane exposures to bloodborne pathogens, e.g., human immunodeficiency virus and hepatitis viruses B and C, are most effectively addressed in institutional employee health or infection control policies, many of which are based on CDC guidelines (220,221). These policies address the availability of postexposure prophylaxis for exposure to hepatitis B and human immunodeficiency viruses.
14.4. Responsibilities of Employees After an Exposure
- Never assume a laboratory injury or exposure is insignificant or unimportant.
- Employees must be empowered to report all incidents, with the goal of protecting themselves, their colleagues, and their families without fear of reprisal. Report all exposures to the supervisor immediately, and discuss the exposure to determine what, if any, actions need to be taken. Actively participate in the documentation of the exposure, and provide pertinent information that will be used in the development of the corrective-action plan
- Cooperate fully with the laboratory's approved postexposure processes, and follow prudent medical advice.
- Follow the directions of the supervisor to the degree they are judged to be reasonable.
15. Biosafety Education
Biosafety education efforts begin even before an employee begins working in the laboratory. The employer must develop an accurate job description so that the employee understands the job responsibilities. Knowledge, skills, and abilities needed for the job are to be defined. Evaluate incoming employees to see if they meet these criteria. Develop a mentoring plan and fill any training gaps before employees are placed in a position that would put them at risk for exposure. Evaluate and document the employees' competency before they are allowed to work independently.
Educational opportunities to reinforce safe behaviors must be ongoing and supported by all levels of management and staff. In accordance with Occupational Safety and Health Administration (OSHA) requirements, education about the risks of exposure to infectious agents begins with a new employee's first orientation to the laboratory or assignment to technical work and is to be specific to the tasks the employee performs. Training must include an explanation of the use and limitations of methods that will reduce or prevent exposure to infectious materials. These include engineering controls, work practices, and personal protective equipment. Annual retraining for these employees must be provided within 1 year of their original training and should emphasize information on new engineering controls and practices. Annual safety training offers a chance to review key biosafety measures that may be forgotten during everyday work pressures.
The responsibility for overseeing the safety education of laboratory personnel must be clearly assigned. This responsibility may be delegated to the biosafety officer or other staff member who has been given additional training through specialized courses or work experience and whose competency to perform the training has been verified. Because laboratory tests might be performed outside a traditional laboratory setting (e.g., doctor's office, outpatient clinic, community setting), these recommendations for training and education must be adapted to suit the employees performing the tests and the person who is overseeing them.
15.1. Biosafety Training/Exercises
Employee training can be accomplished by any of several methods, and nearly all of these can be adapted or combined to fit the needs of employees in a particular laboratory.
- Live demonstrations are effective because they allow interaction between the laboratory worker and an expert who can ask and answer questions.
- Demonstrations of common problems or exposures that laboratory scientists might encounter in their normal work duties can be presented for individuals or groups. This might be done by rigging (or "pre-positioning") a laboratory area or work station with deliberate problems or safety errors and asking, "What's wrong here and how would you address it?" "What would you do if you encountered this problem?" For example:
— Set up a biological safety cabinet (BSC) with excess clutter inside, or demonstrate someone going in and out of the hood excessively.
— Practice what to do if you drop a liquid culture, possibly of Mycobacterium tuberculosis, in the mycobacteriology laboratory.
— Discuss what to do if a syringe is sent to the laboratory with a needle attached.
— How would you handle a broken tube with a specimen in the centrifuge?
— Demonstrate what you would do if you got a splash of a bacterial suspension in your face/eyes/mouth.
— Demonstrate ejecting pipette tips where they could bounce up and strike the laboratory scientist in the face, and demonstrate the correct procedure.
— Discuss how to deal with a leaking specimen in the pneumatic tube. - All proper behavior for avoiding or dealing with safety breaches such as those highlighted in these demonstration events should be described in a standard safety practices and procedures section in the laboratory safety manual.
- Monthly safety mini-presentations can be presented during regularly scheduled laboratory meetings. Employees who have had a "near-miss" or actual incident could share their experience with their coworkers, in accordance with institutional privacy considerations. Having this information coming from their coworkers would increase its impact. Elicit and discuss creative solutions to prevent biosafety problems.
- Alternatively, each laboratory employee could take a turn at presenting at a staff meeting what they perceive as a biosafety hazard. This would also help to keep everyone aware of daily safety issues so easily forgotten in a busy workplace.
15.2. Educational Reinforcement
- Educational reinforcement through communications and quizzes is to be ongoing and random. Unpredictable quizzes or challenges might be most effective because they force quick, deliberate thought about how to respond. A specific scenario could be handed to a randomly selected technologist, and some laboratories may have the technological capacity to send safety challenges to individual workers when they sign on for the day. Alternatively, educational reinforcement might be as simple as having a question of the week posted in the laboratory. These approaches would enable safety exercises to be carried out for all shifts.
- If meeting presentations are not possible, short one-page written "safety puzzles" can be shared in the break room over coffee, or posted on the lockers or on the laboratory bulletin board. An example is a "what's wrong with this picture?" format.
— Show a photo of a person working with moulds outside the biosafety cabinet.
— Show someone working in the biosafety cabinet with objects blocking the vent of the biosafety hood.|
— Show a person vortexing bacterial suspensions for antimicrobial susceptibility testing without a lid on the tube.
— Post the correct answers with the next safety quiz. - Signs reminding personnel to comply with safety policies might increase awareness and compliance. OSHA, CDC, and other organizations provide job aids that can be downloaded and printed. However, because signs that remain unchanged quickly lose their effectiveness, rotate these signs with new messages that are designed to keep staff alert to the hazards at the work stations.
- Consult the American Biological Safety Association website (http://www.absa.org/resbslinks.html) for links to numerous government agencies and other professional organizations, rules/regulations/guidelines, information on biosecurity and bioterrorism, technical links and papers, and information on their week-long biosafety review course.
15.3. Annual Checklist of Critical Safety Items and Procedures
There is no one "official" set of questions for an annual safety checklist. Although many common activities might be performed by all personnel, customize the list to reflect the actual job duties. Analyze each work station for the type of biosafety risks associated with it, and target the checklist to each of these risks. If practical, ask individual laboratorians to draft their own checklists for the duties they perform, and have their list reviewed by their supervisor and safety officer.
15.4. Assessment and Documentation
Employee training and competency assessment should be documented for the following:
- Constructing and properly labeling containers for medical waste;
- Following all federal, state, and local regulations regarding waste management, including handling, disposal, and storage of medical waste;
- Transportation of medical waste, including any required Department of Transportation (DOT) labeling of transport containers (e.g., with a "known infectious substance" label);
- Transportation of specimens;
- Use of appropriate supplies — e.g., containers, plastic bags, labeling — for transport of all laboratory specimens both within and outside of the hospital facility;
- Following all federal, state, and local regulations regarding transport of laboratory specimens; and
- Transportation of specimens outside the facility (93).
15.5. Monitoring Compliance with Safety Procedures
- To reinforce the importance of safety training and education, make it a part of the annual performance review. During this review, ensure that the employee participates in annual training, follows policies, and actively participates in laboratory-wide safety quizzes. Those who have experienced a series of incidents during the evaluation period are to be counseled and receive more intense retraining in those areas.
- Ensure that employees understand the importance of reporting incidents to Employee Health and their supervisor or manager. Explain that this is important for the employee's protection and that linking an injury or infection to an incident that may have occurred in the workplace will help to compensate the employee appropriately and improve practices to protect others.
- Develop an expedited exposure control plan whereby a laboratory employee is "streamlined" through the employee health process and is, therefore, more likely to report an incident.
- The supervisor or manager must follow up on every employee who has incurred a problem to protect the employee and to make sure these incidents are monitored and systematic risks are eliminated.
- Conduct an annual review of engineering controls because changes in practices and technology may put the laboratory staff at new risks. New engineering controls should be reviewed by appropriate persons, as warranted, e.g., the safety officer, risk management, purchasing, or employees who use needles and other sharps in the course of their duties.
- Laboratory sections can appoint a "safety captain of the month," rotating the position among employees. The safety captain is to reinforce good safety behaviors and encourage coworkers to comply with safety policies.
- Reserve punitive measures for egregious or repeat violations, and create a positive attitude about safety rather than a negative one with administrative consequences. In this regard, the laboratory staff could become a cadre of safety "shoppers," who could be encouraged to report on any new safety ideas implemented by the staff as well as examples of unsafe practices or situations they see occurring in the laboratory. The purpose is not to report people, but to promote a safe work environment and describe situations they see where safe practices are not being followed so that safety can be reinforced and recognized at the next staff meeting.
16. Continuous Quality Improvement
Integrate continuous quality improvement for biosafety with the continuous quality improvement for the entire laboratory. The 12 quality system essentials, as defined by the Clinical Laboratory Standards Institute, provide a comprehensive basis and reference for continuous quality improvement (222). More detailed and specific biosafety considerations have been listed for each of these elements (Table 17).
Acknowledgments
We acknowledge the assistance of Tanya Graham, DVM, South Dakota State University, Brookings, SD; Larry Thompson, DVM, PhD, Nestle Purina Pet Care, St. Louis, MO; R. Ross Graham, DVM, PhD, Merrick and Company; Corrine Fantz, PhD, Emory University, Atlanta, GA; Thomas Burgess, PhD, and Quest Diagnostics, Tucker, GA.
We appreciate the review and input into the document provided by the Office of Health and Safety, CDC; American Association of Veterinary Laboratory Diagnosticians; American Biological Safety Association; College of American Pathologists; American Society for Microbiology; Association of Public Health Laboratories; and subject matter experts at CDC.
References
- CDC/National Institutes of Health. Biosafety in microbiological and biomedical laboratories. 5th ed. Available at http://www.cdc.gov/biosafety/publications/bmbl5/BMBL.pdf.
- Clinical and Laboratory Standards Institute. Protection of laboratory workers from occupationally acquired infections; approved guideline—fourth edition (CLSI document M29-A4). Wayne, PA: Clinical and Laboratory Standards Institute; 2011. In press.
- CDC. Laboratory-acquired vaccinia exposures and infections—United States, 2005–2007. MMWR 2008;57:401–4.
- CDC. Laboratory-acquired brucellosis—Indiana and Minnesota, 2006. MMWR 2008;57:39–42.
- CDC. Update: potential exposures to attenuated vaccine strain Brucella abortus RB51 during a laboratory proficiency test—United States and Canada, 2007. MMWR 2008;57:36–9.
- CDC. Laboratory exposure to Burkholderia pseudomallei—Los Angeles, California, 2003. MMWR 2004;53:988–90.
- CDC. Laboratory-acquired meningococcal disease—United States, 2000. MMWR 2002;51:141–4.
- CDC. Suspected cutaneous anthrax in a laboratory worker—Texas, 2002. MMWR 2002;51:279–81.
- CDC. Laboratory-acquired West Nile virus infections—United States, 2002. MMWR 2002;51:1133–5.
- CDC. Laboratory-acquired human glanders—Maryland, May 2000. MMWR 2000;49:532–5.
- CDC. Epidemiologic notes and reports. Laboratory-acquired meningococcemia—California and Massachusetts. MMWR 1991;40:46–47,55.
- Jacobson JT, Orlob RB, Clayton JL. Infections acquired in clinical laboratories in Utah. J Clin Microbiol 1985;21:486–9.
- Harrington JM, Shannon HS. Incidence of tuberculosis, hepatitis, brucellosis, and shigellosis in British medical laboratory workers. Br Med J 1976;1:759–62.
- Reid DD. Incidence of tuberculosis among workers in laboratories. Br Med J 1957;2:10–14.
- Vesley D, Hartmann HM. Laboratory-acquired infections and injuries in clinical laboratories: a 1986 survey. Am J Public Health 1988;78:1213–15.
- Walker D, Campbell D. A survey of infections in United Kingdom laboratories, 1994–1995. J Clin Pathol 1999;52:415–8.
- Sejvar JJ, Johnson D, Popovic T, et al. Assessing the risk of laboratory-acquired meningococcal disease. J Clin Microbiol 2005;43:4811–14.
- Sewell DL. Laboratory-associated infections and biosafety. Clin Microbiol Rev 1995;8:389-405.
- Pike RM. Laboratory-associated infections: incidence, fatalities, causes, and prevention. Annu Rev Microbiol 1979;33:41–66.
- Herwaldt, BL. Laboratory-acquired parasitic infections from accidental exposures. Clin Microbiol Rev 2001;14:659–88.
- Pragay DA, Howard SF, Gill ES. Clinical laboratory accidents, and some recommended remedies. Clin Chem 1980;26:1107–8.
- Kyes K. Blood safety. Clinical Laboratory Products. June 2007. Available at http://www.clpmag.com/issues/articles/2007-06_02.asp.
- Collins CH. Laboratory-acquired infections: history, incidence, causes, and prevention. 3rd ed. London: Butterworth-Heinemann, Ltd; 1993.
- Pike RM. Laboratory-associated infections: incidence, fatalities, causes, and prevention. Annu Rev Microbiol 1979;33:41–66.
- Pike RM. Laboratory-associated infections: summary and analysis of 3921 cases. Health Lab Sci 1976;13:105–14.
- Pike RM. Past and present hazards of working with infectious agents. Arch Pathol Lab Med 1978;102:333–6.
- Harding AL, Byers KB. Epidemiology of laboratory-associated infections. In: Fleming DO, Hunt DL, eds. Biological safety: principles and practices. 3rd ed. Washington, DC: ASM Press; 2000:35–54.
- Vesley D, Hartmann HM. Laboratory-acquired infections and injuries in clinical laboratories: a 1986 survey. Am J Public Health. 1988;78:1213–15.
- National Fire Protection Association. Section 5.1.3.3.2 (7). In: NFPA 99 Standard for health care facilities, 2005 Edition., Quincy, MA: National Fire Protection Association; 2005:37.
- Zohar D. The effects of leadership dimensions, safety climate, and assigned priorities on minor injuries in work groups. Journal of Organizational Behavior 2002;23:75–92.
- Zohar D, Luria G. A multilevel model of safety climate: Cross-level relationships between organization and group-level climates. J. Appl Psychol 2005;90:616–28.
- Clinical and Laboratory Standards Institute. Laboratory design: approved guideline—second edition (CLSI document GP18-A2). Wayne, PA: Clinical and Laboratory Standards Institute; 2007.
- Occupational Safety and Health Administration. Occupational safety and health standards. Z. Toxic and hazardous substances. Bloodborne pathogens. Standard no.1910.1030. Available at http://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=standards&p_id=10051.
- International Federation of Biosafety Associations. Biosafety compendium on regulations and information sources. Available at http://www.internationalbiosafety.org/english/internlCompendium.asp.
- American Biological Safety Organization. Available at http://www.absa.org.
- World Health Organization. Laboratory biosafety manual, 3rd edition. Geneva: World Health Organization, 2004. Available at http://www.who.int/csr/resources/publications/biosafety/Biosafety7.pdf.
- Occupational Safety and Health Administration. Occupational safety and health standards. I. Personal protective equipment. Standard no. 1910.132. Available at http://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=9777.
- Rutala W, Weber D. Use of inorganic hypochlorite (bleach) in health-care facilities. Clin Microbiol Rev 1997;597–610.
- Clinical and Laboratory Standards Institute. Clinical laboratory waste management; approved guideline—third edition (CLSI document GP05-A3). Wayne, PA: Clinical and Laboratory Standards Institute; 2011.
- CDC, US Department of Agriculture, Animal and Plant Health Inspection Service. National select agent registry. Available at http://www.selectagents.gov.
- Miller JM. The Select Agent Rule and its impact on clinical laboratories. Clin Microbiol Newsletter 2006;28(8):57–63.
- Johansen BV. Hazards related to EM-laboratory instrumentation. Ultrastruct Pathol 1984;7:219–25.
- Bance GN, Barber VC, Sholdice JA. Safety in the SEM laboratory—1981 update. Scan Electron Microsc 1981;II:87–94.
- Fleming DO, Richardson JH, Tulis JJ, Vesley D. Laboratory safety: principles and practices. 2nd ed. Washington DC: American Society for Microbiology; 1995.
- Clinical and Laboratory Standards Institute. Implementing a needlestick and sharps injury prevention program in the clinical laboratory (CLSI document CLSI X03RE). Wayne, PA: Clinical and Laboratory Standards Institute; 2002.
- CDC. Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. MMWR 2005;54(No. RR-17).
- Collins CH. Laboratory acquired infections. Med Lab Sci 1980;37:291–8.
- Chatigny MA, Hatch MT, Wolochow H, et al. Studies on release and survival of biological substances used in recombinant DNA laboratory procedures. Recombinant DNA technical bulletin. Bethesda, MD: US Department of Health and Human Services, National Institutes of Health. 1979.
- Wiedbrauk DL, Stoerker J. Quality assurance in the molecular virology laboratory. In: Wiedbrauk DL, Farkas DH, eds. Molecular methods for virus detection. San Diego, CA: Academic Press; 1995: 25–37.
- Darlow HM. Safety in the microbiology laboratory: an introduction. In: Shapton DA, Board RG, eds. Safety in microbiology. London: Academic Press; 1972:1–19.
- Schmid I, Lambert C, Ambrozak D, Marti GE, Moss DM, Perfetto SP. International Society for Analytical Cytology biosafety standard for sorting of unfixed cells. Cytometry Part A. 2007;6:414–37.
- Kubica GP. Your tuberculosis laboratory: are you really safe from infection? Clinical Microbiology Newsletter 1990;12:85–7.
- Murray PR, Baron EJ, Horgensen JH, Pfaller MA, Yolken RH. Manual of clinical microbiology, Eighth edition. Washington, DC: ASM Press; 2003.
- Fleming DO, Hunt DL. Biological safety: principles and practices. Washington, DC: ASM Press; 2006.
- Occupational Safety and Health Administration. Occupational safety and health standards. I. Personal protective equipment: Respiratory protection. Standard no. 1910.134. Section 4. Available at http://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=12716.
- Nolte K, Taylor D, Richmond J. Biosafety considerations for autopsy. Am J Forensic Med Pathol 2002;23(2):107–22.
- Collins C, Grange J. Tuberculosis acquired in laboratories and necropsy rooms. Communicable Disease and Public Health 1999;2:161–7.
- Burnett L, Lunn G, Coico R. Biosafety: guidelines for working with pathogenic and infectious microorganisms. In: Curr Protoc Microbiol.[serial online] May 2009; DOI: 10.1002/9780471729259.mc01a01s13.
- Nolte KB, Foucar K, Richmond JY. Hanta viral biosafety issues in the autopsy room and laboratory: concerns and recommendations. Hum Pathol 1996;27:1253–4.
- De Carli G, Puro V, Scognamiglio P, Ippolito G. Infection with hepatitis C virus transmitted by accidental needlesticks. Clin Infect Dis 2003;37:1718–9.
- Nyberg M, Suni J, Haltia M. Isolation of human immunodeficiency virus (HIV) at autopsy one to six days postmortem. Am J Clin Pathol 1990;94:422–5.
- Petrosillo N, Puro V, De Carli G, Ippolito G, SIROH Group. Risks faced by laboratory workers in the AIDs era. J Biol Regul Homeost Agents 2001;15:243–8.
- CDC. Updated US Public Health Service guidelines for the management of occupational exposures to HBV, HCV, and HIV and recommendations for postexposure prophylaxis. MMWR 2001;50(No. RR-11).
- Brown P, Gibbs CJ, Gajdusek D, Cathala F, LaBauge R. Transmission of Creutzfeldt-Jakob disease from formalin-fixed, paraffin-embedded human brain tissue. N Engl J Med 1986;315:1614–5.
- Sitwell L LB, Attack E, Izukawa, D. Creutzfeldt-Jakob disease in histopathology technicians N Engl J Med 1988;318:854.
- Wilkins D WA, Cossar YE, Miller DC. Creutzfeldt-Jakob disease in histopathology technicians. N Engl J Med 1988;318:853–4.
- Miller D. Creutzfeldt-Jakob disease in histopathology technicians. N Engl J Med 1988;318:853–4.
- Canadian Food Inspection Agency. Biosafety guidelines developed for National TSE Veterinary Diagnostic Laboratory Network. Version 1.0, March 8, 2002.
- Newsom S, Rowlands C, Mathews J, Elliot CJ. Aerosols in the mortuary. J Clin Pathol 1983;36:127–32.
- Occupational Safety and Health Administration. Occupational safety and health standards. Z. Toxic and hazardous substances. 1910.1048, Formaldehyde. Available at http://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=10075.
- Code of Federal Regulations. Title 40, Protection of Environment. Available at http://ecfr.gpoaccess.gov/cgi/t/text/text-idx?c=ecfr;sid=4990e762d7b81851bef18f82dc851826;rgn0=div5;view=text;node=40%3A25.0.1.1.2;idno=40;cc=ecfr#40:25.0.1.1.2.3.1.5.
- Occupational Safety and Health Administration. Occupational safety and health standards. I. Personal protective equipment. 1910.133, Eye and face protection. Available at http://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=9778.
- National Tuberculosis Advisory Committee. Guidelines for Australian Mycobacteriology Laboratories. Communicable Diseases Intelligence 2006;30:116–28.
- Hedwell E. Incidence of tuberculosis among medical students at Lund University. American Review of Tuberculosis 1940;41:770–80.
- Weston J, Locker G. Frequency of glove puncture in the postmortem room. J Clin Pathol 1992;45:177–8.
- Templeton GL, Illing LA, Young L, Cave D, Stead WW, Bates JH. The risk for transmission of Mycobacterium tuberculosis at the bedside and during autopsy. Ann Intern Med 1995;122:922–5.
- Meyer J. TB Plagues Office of the LA Coroner. Los Angeles Times April 25, 1997;A1–27. Available at http://articles.latimes.com/1997-04-25/news/mn-52228_1_coroner-s-office.
- Kappel T, Reinarts J, Schmid J, Holter J, Azar M. The viability of Mycobacterium tuberculosis in formalin-fixed pulmonary autopsy tissue: review of the literature and brief report. Hum Pathol 1996;27:1361–4.
- Ford A. Taking stock of biosafety perils in surg path. CAP Today 2009;23:1.
- Gärtner H-V, Seidl C, Luckenbach C, et al. Genetic analysis of a sarcoma accidentally transplanted from a patient to a surgeon. N Engl J Med 1996;335:1494–7.
- Goette D, Jacobson, KW, Doty RD. Primary inoculation tuberculosis of the skin; prosector's paronychia. Arch Dermatol 1978;114:567–9.
- Vetter J. Microbial aerosols from a freezing microtome. American Society for Clinical Microbiology Summary Report; 1977.
- CDC. Tuberculosis infection associated with tissue processing—California. MMWR 1981;30(6):73–4.
- Smith J. The hazard to pathologists from tuberculosis specimens. Hum Pathol 1996;27:1251–2.
- Gerston K, Blumberg L, Tshabalala V, Murray J. Viability of mycobacteria in formalin-fixed lungs. Hum Pathol 2004;35:571–5.
- Richard M, Biacabe A, Perret-Liaudet A, McCardle L, Ironside J, Kopp N. Protection of personnel and environment against Creutzfeldt-Jakob disease in pathology laboratories. Clin Expl Pathol 1999;47:192–200.
- Clinical and Laboratory Standards Institute. Procedures for the recovery and identification of parasites from the intestinal tract; approved guideline—second edition (CLSI document M28-A2). Wayne, PA: Clinical and Laboratory Standards Institute; 2005.
- Wheatley W. A rapid staining procedure for intestinal amoebae and flagellates. Am J Clin Pathol 1951;21:990–1.
- Garcia LS. Diagnostic medical parasitology, 5th ed. Washington, DC: ASM Press; 2007.
- International Air Transport Association. 2005[a]. Dangerous Goods Regulations, 46th edition. Montreal, Canada: International Air Transport Association; 2005.
- International Air Transport Association. 2005[b]. Dangerous Goods Regulations, 46th ed. Addendum II. March 22. Montreal, Canada: International Air Transport Association; 2005.
- International Air Transport Association. 2005[c]. Dangerous Goods Regulations, 46th ed. Addendum III. July 5. Montreal, Canada: International Air Transport Association; 2005.
- US Department of Transportation, Pipeline and Hazardous Materials Safety Administration. Hazardous materials: infectious substances; harmonization with the United Nations recommendations; final rule (49 CFR Parts 171 et al.). Federal Register 2006;71:32244–63. Available at https://hazmatonline.phmsa.dot.gov/services/publication_documents/Transporting%20Infectious%20Substances%20Safely.pdf.
- US Department of Transportation, Research and Special Programs Administration. Harmonization with the United Nations Recommendations, International Maritime Dangerous Goods Code, and International Civil Aviation Organization's Technical Instructions; final rule (CFR 42, Parts 171, 172, et al.). Federal Register 2004;69:76044–76187 (http://www.labsafety.com/refinfo/fedreg/FRPDF/122004.pdf).
- US Department of Transportation, Research and Special Programs Administration. Hazardous materials: revision to standards for infectious substances and genetically modified organisms; final rule (CFR 42, Parts 171 et al.). Federal Register 2002;67:53118–53144.
- US Postal Service. Domestic mail manual. Hazardous materials (601.10.1). 2011. Available at http://pe.usps.com/cpim/ftp/manuals/dmm300/full/mailingStandards.pdf.
- World Health Organization. Transport of infectious substances. Background to the 17 amendments adopted in the 13th revision of the United Nations Model Regulations guiding the transport of infectious substances. Geneva: World Health Organization; 2004. Available at http://www.who.int/csr/resources/publications/WHO_CDS_CSR_LYO_2004_9/en.
- World Health Organization. 2005. Guidance on Regulations for the Transport of Infectious Substances 2009–2010. Geneva: World Health Organization; 2009. Available at http://www.who.int/csr/resources/publications/biosafety/WHO_HSE_EPR_2008_10/en/index.html.
- Garcia LS, Shimizu RY, Shum A, Bruckner DA. Evaluation of intestinal protozoan morphology in polyvinyl alcohol preservative: comparison of zinc sulfate and mercuric chloride based compounds for use in Schaudinn's fixative. J Clin Microbiol 1993;31:307–10.
- Jungbauer FH, van der Harst JJ, Groothoff JW, Coenraads PJ. Skin protection in nursing work: promoting the use of gloves and hand alcohol. Contact Dermatitis 2004;51:135–40.
- Kampf G, Kramer A. Epidemiologic background of hand hygiene and evaluation of the most important agents for scrubs and rubs. Clin Microbiol Rev 2004;17:863–93.
- CDC. A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States. Recommendations of the Advisory Committee on Immunization Practices. Part II: Immunization of adults. MMWR 2006;55(No. RR-16).
- Calcofluor white reagent dropper (material safety data sheet). Franklin Lakes, NJ: Becton Dickinson; 2011. Available at http://www.bdregdocs.com/msds/view/msds-usa-261195.pdf.
- Potassium hydroxide 10% w/v (material safety data sheet). Austin, TX: Science Stuff, Inc.; 2006. Available at http://www.sciencestuff.com/msds/C2360.html.
- Calcobrite fungi staining kit (material safety data sheet). Guildford, UK: Clin-Tech, LTD; 2009. Available at http://www.clin-tech.co.uk/msds/63115X.pdf.
- Potassium hydroxide (10–45%) solutions and concentrates (material safety data sheet). Phillipsburg, NJ: Mallinckrodt Baker, Inc.; 2008. Available at http://www.jtbaker.com/msds/englishhtml/P5887.htm.
- Lactophenol cotton blue stain droppers (material safety data sheet). Franklin Lakes, NJ: Becton Dickinson; 2003. Available at http://www.bd.com/ds/productCenter/261188.asp.
- BactiDrop lactophenol aniline blue (material safety data sheet). Hants, UK: Oxoid; 2007. Available at http://www.oxoid.com/pdf/msds/EN/R21526.pdf.
- Lactophenol–cotton blue mounting fluid (material safety data sheet). Gibbstown, NJ: EMD Chemicals; 2010. Available at http://www.emdchemicals.com/lactophenol/EMD_CHEM-R03465/p_uuid?attachments=MSDS.
- Higgins waterproof black drawing India ink (material safety data sheet). Bellwood, IL: Sanford Corporation; 2001. Available at http://web.grcc.cc.mi.us/Pr/msds/visualarts-photography/HIGGINS%20Waterproof%20Black%20Drawing%20Indian%20Ink.pdf.
- Methenamine silver stain (package insert). Carpinteria, CA: Dako North America; 2008. Available at http://www.dako.com/us/searchresultlist?search=methenamine+silver+stain.
- Giemsa stain (material safety data sheet). Torrance, CA: Medical Chemical Corp.; 2006. Available at http://www.med-chem.com/msds/591A.pdf.
- Larone DH. Medically important fungi, a guide to identification, 4th ed., Washington, DC: ASM Press; 2002.
- Stevens DA, Clemons KV, Levine HB, et al. Expert opinion: what to do when there is Coccidioides exposure in a laboratory. Clin Infect Dis 2009; 49:919–23.
- Indoor allergens: assessing and controlling adverse health effects. In: Pope AM, Patterson R, eds. Washington, DC: National Academy Press; 1993.
- Occupational allergy to antibiotics. Med Pr 1986;37:383–7.
- Coutts II, Dally MB, Taylor AJ, Pickering CA, Horsfield N. Asthma in workers manufacturing cephalosporins. Br Med J (Clin Res Ed) 1981;283:950.
- Davies RJ, Hendrick DJ, Pepys J. Asthma due to inhaled chemical agents: ampicillin, benzyl penicillin, 6 amino penicillanic acid and related substances. Clin Allergy 1974;4:227-47.
- Malo J-L, Cartier A. Occupational asthma in workers of a pharmaceutical company processing spiramycin. Thorax 1988;45:371–7.
- Paggiaro PL, Loi AM, Toma A. Bronchial asthma and dermatitis due to spiramycin in a chick breeder. Clin Allergy 1979;9:571–4.
- Bance GN, Barber VC, Sholdice JA. Safety in the SEM laboratory—1981 update. Scan Electron Microsc 1981;II:87–94.
- Drury P. Safety in electron microscopy and protective glove hazards. Can J Med Technol 1980;42:80–2.
- Pegum JS, Medhurst FA. Dermatitis from penetration of rubber gloves by acrylic monomer. Br Med J 1971;276:141–3.
- Tobler M, Wüthrich B, Freiburghaus AU. Contact dermatitis from acrylate and methacrylate compounds in Lowicryl embedding media for electron microscopy. Contact Dermatitis 1990;23:96–102.
- Lunn G, Sansone EB. Ethidium bromide: destruction and decontamination of solutions. Anal Biochem 1987;162:453–8. PMID 3605608.
- Gärtner HV, Seidl C, Luckenbach C, et al. Genetic analysis of a sarcoma accidentally transplanted from a patient to a surgeon. N Engl J Med 1996;335:1494–6.
- Nadler SH, Moore GE. Immunotherapy of malignant disease. Arch Surg 1969;99:376–81.
- Scanlon EF, Hawkins RA, Fox WW, Smith WS. Fatal homotransplanted melanoma: a case report. Cancer 1965;18:782–89.
- Southam CM. Homotransplantation of human cell lines. Bull NY Acad Med 1958;34:416–23.
- Blohme I, Nyberg G, Jeansson S, Svalander C. Adenovirus infection in a renal transplant patient. Transplant Proc 1992;24:295.
- Myerowitz RL, Stalder H, Oxman MN, et al. Fatal disseminated adenovirus infection in a renal transplant recipient. Am J Med 1975;59:591–8.
- Asim M, Chong-Lopez A, Nickeleit V. Adenovirus infection of a renal allograft. Am J Kidney Dis 2003;41:696–701.
- Hsuing GD. Latent virus infections in primate tissues with special reference to simian viruses. Bacteriol Rev 1968;32:185–205.
- Swack NS, Hsuing GD. Endogenous agents in primary cell cultures with special reference to latent viruses. In Vitro 1974;10:260–7.
- Hopps HE. Origin of endogenous and exogenous agents in cell cultures. In Vitro 1974;10:243–6.
- Patel R, Paya CV. Infections in solid-organ transplant recipients. Microbiol Rev 1997;10:86–124.
- van Gelder T, Kroes LC, Mulder A, Gratama JW, Weimar W. A living-related kidney donor as the source of a nearly fatal primary Epstein-Barr virus infection following transplantation. Transplantation 1994;58:852–4.
- Davis C, Gretch DR, Carithers RL. Hepatitis B and transplantation. Infect Dis Clin North Am 1995;9:925–41.
- Dummer JS, Armstrong J, Somers J, et al. Transmission of infection with herpes simplex virus by renal transplantation. J Infect Dis 1987;155:202–6.
- Goodman JL. Possible transmission of herpes simplex virus by organ transplantation. Transplantation 1989;47:609–13.
- Schwarz A, Hoffmann F, L'age-Stehr, J, Tegzess AM, Offermann G. Human immunodeficiency virus transmission by organ donation. Outcome in cornea and kidney recipients. Transplantation 1987;1:21–4.
- Sotir M, Switzer W, Schable C, Schmitt J, Vitek C, Khabbaz RF. Risk of occupational exposure to potentially infectious nonhuman primate materials and to simian immunodeficiency virus. J Med Primatol 1997;26:233–40.
- Khabbaz RF, Rowe T, Murphey-Corb M, et al. Simian immunodeficiency virus needlestick accident in a laboratory worker. Lancet 1992;340:271–3.
- Gonzalez-Perez MP, Munoz-Juarez L, Cardenas FC, Zarranz Imirizaldu JJ, Carranceja JC, Garcia-Saiz A. Human T-cell leukemia virus type I infection in various recipients of transplants from the same donor. Transplantation 2009;75:1006–11.
- Nakatsuji Y, Sugai YF, Watanabe S, et al. HTLV-I-associated myelopathy manifested after renal transplantation. J Neurol Sci 2000;177:154–6.
- Remesar MC, del Pozo AE, Pittis MG, Mangano AM, Sen L, Briones L. Transmission of HTLV-I by kidney transplant. Transfusion 2000;40:1421–2.
- Toro C, Rodes B, Poveda E, Soriano V. Rapid development of subacute myelopathy in three organ transplant recipients after transmission of human T-cell lymphotropic virus type I from a single donor. Transplantation 2003;75:102–4.
- CDC. Lymphocytic choriomeningitis virus infection in organ transplant recipients—Massachusetts, Rhode Island, 2005. MMWR 2005;54:537–9.
- Dykewicz CA, Dato VM, Fisher-Hoch SP, Ostroff SM, Gary H Jr, McCormick JB. Lymphocytic choriomeningitis outbreak associated with nude mice in a research institute. JAMA 2010;267:1349–53.
- Mahy BW, Dykewicz C, Fisher-Hoch S, Ostroff S, Sanchez A. Virus zoonoses and their potential for contamination of cell cultures. Dev Biol Stand 1991;75:183–9.
- Schmitt M, Pawlita M. High-throughput detection and multiplex identification of cell contaminations. Nucleic Acids Res Advance Access 2009;37(18):e119. Available at http://nar.oxfordjournals.org/content/37/18/e119.full.
- Emery JB, York CJ. Occurrence of a hemadsorption virus in normal monkey tissue culture. Virology 1960;11:313–5.
- Mengling WL. Porcine parvovirus: frequency of naturally occurring transplacental infection and viral contamination of fetal porcine kidney cell cultures. Am J Vet Res 1975;36:41–4.
- Srinivasan A, Burton EC, Kuehnert MJ, et al. Transmission of rabies virus from an organ donor to four transplant recipients. N Engl J Med 2005;352:1103–11.
- Robertson I. Corneal transplants and rabies. Med J Aust 1979;2:697.
- Martín-Dávila P, Fortún J, López-Vélez R, et al. Transmission of tropical and geographically restricted infections during solid-organ transplantation. Clin Microbiol Rev 2008;21:60–96.
- Javadi MA, Fayaz A, Mirdehghan SA, Ainollahi B. Transmission of rabies by corneal graft. Cornea 2009;15:431–3.
- Houff SA, Burton RC, Wilson RW, et al. Human-to-human transmission of rabies virus by corneal transplant. N Engl J Med 1979;300:603–4.
- Hellenbrand W, Meyer C, Rasch G, Steffens T, Amon A. Cases of rabies in Germany following organ transplantation. Eur Surveill 2005;10:E050224.
- Gode GR, Bhide NK. Two rabies deaths after corneal grafts from one donor. Lancet 1988;2:791.
- CDC. Human-to-human transmission of rabies via corneal transplant—Thailand. MMWR. 1981;30:473–4.
- Baer GM, Shaddock JH, Houff SA, Harrison AK, Gardner JJ. Human rabies transmitted by corneal transplant. Arch Neurol 2009;39:103–7.
- Patient received cornea: rabies case linked to transplant. American Medical News 1978;21:3.
- Hull RN, Minner JR, Smith JW. New viral agents recovered from tissue cultures of monkey kidney cells. I. Origin and properties of cytopathic agents SV1, SV2, SV4, SV5, SV6, SV11, SV12, and SV15. Am J Hyg 1956;63:204–15.
- Rustigian R, Johnston T, Reihart H. Infection of monkey kidney tissue cultures with virus-like agents. Proc Soc Exp Biol Med 1955;88:8–16.
- Peden K, Sheng L, Yacobucci M, et al. Recovery of strains of the polyomavirus SV40 from rhesus monkey kidney cells dating from the 1950s to the early 1960s. Virology 2008;370:63–76.
- Sweet BH, Hilleman MR. The vacuolating virus, SV40. Proc Soc Exp Biol Med 1960;105:420–7.
- Kekarainen T, Martínez-Guinó L, Segalés J. Swine torque teno virus detection in pig commercial vaccines, enzymes for laboratory use and human drugs containing components of porcine origin. J Gen Virol 2000;90:648–53.
- Middleton PG, Miller S, Ross JA, Steel CM, Guy K. Insertion of SMRV-H viral DNA at the c-myc gene locus of a BL cell line and presence in established cell lines. Int J Cancer 1992;54:451–4.
- Sun R., Grogan E, Shedd D, et al. Transmissible retrovirus in Epstein-Barr virus-producer B95-8 cells. Virology 1995;209:374–83.
- Antony S. Severe meningo-encephalitis and death in a renal transplant recipient resulting from West Nile virus infection. J Natl Med Assoc 2004;96:1646–7.
- Armali Z, Ramadan R, Chlebowski A, Azzam ZS. West Nile meningo-encephalitis infection in a kidney transplant recipient. Transplant Proc 2003;35:2935–6.
- Bragin-Sanchez D, Chang PP. West Nile virus encephalitis infection in a heart transplant recipient: a case report. J Heart Lung Transplant 2005;24:621–3.
- CDC. West Nile virus activity–United States, September 26–October 2, 2002, and investigations of West Nile virus infections in recipients of blood transfusion and organ transplantation. JAMA 2002;288:1975–6.
- CDC. West Nile virus infection in organ donor and transplant recipients–Georgia and Florida, 2002. JAMA 2002;288:1465–6.
- CDC. Update: West Nile virus screening of blood donations and transfusion-associated transmission—United States. MMWR 2004;53:281–4.
- CDC. Transfusion-associated transmission of West Nile virus—Arizona, 2004. MMWR. 2004;53:842–4.
- CDC. West Nile virus infections in organ transplant recipients—New York and Pennsylvania, August–September, 2005. MMWR 2005;54:1021–3.
- DeSalvo D, Roy-Chaudhury P, Peddi R, et al. West Nile virus encephalitis in organ transplant recipients: another high-risk group for meningoencephalitis and death. Transplantation 2004;77.
- Hardinger KL, Miller B, Storch GA, Desai NM, Brennan DC. West Nile virus-associated meningoencephalitis in two chronically immunosuppressed renal transplant recipients. Am J Transplant 2003;3:1312–5.
- Harrington T, Kuehnert MJ, Kamel H, et al. West Nile virus infection transmitted by blood transfusion. Transfusion 2003;43:1018–22.
- Iwamoto M, Jernigan DB, Guasch A, et al. Transmission of West Nile virus from an organ donor to four transplant recipients. N Engl J Med 2003;348:2196–203.
- Kleinschmidt-DeMasters BK, Marder BA, Levi ME, et al. Naturally acquired West Nile virus encephalomyelitis in transplant recipients: clinical, laboratory, diagnostic, and neuropathological features. Arch Neurol 2004;61:1210–20.
- Kumar D, Prasad GVR, Zaltzman J, Levy GA, Humar A. Community-acquired West Nile virus infection in solid-organ transplant recipients. Transplantation 2004;77.
- Kusne S, Smilack J. Transmission of West Nile virus by organ transplantation. Liver Transplant 2005;11:239–41.
- Montgomery SP, Brown JA, Kuehnert MJ, et al. Transfusion-associated transmission of West Nile virus. United States 2003 through 2005. Transfusion 2006;46:2038–46.
- Murtagh B, Wadia Y, Messner G, Allison P, Harati Y, Delgado R. West Nile virus infection after cardiac transplantation. J Heart Lung Transplant 2005;24:774–6.
- Pealer LN, Marfin AA, Petersen LR, et al. Transmission of West Nile virus through blood transfusion in the United States in 2002. N Engl J Med 2003;349:1236–45.
- Ravindra K, Freifeld A, Kalil A, et al. West Nile virus associated encephalitis in recipients of renal and pancreas transplants: case series and literature review. Clin Infect Dis 2004;38:1257–60.
- Shephert JC, Subramanian A, Montgomery RA, et al. West Nile virus encephalitis in a kidney transplant recipient. Am J Transplant 2004;4:830–3.
- Trijzelaar B. Regulatory affairs and biotechnology in Europe: III. Introduction into good regulatory practice—validation of virus removal and inactivation. Biotherapy 1993;62:93–102.
- Hazelton PR, Gelderblom HR. Electron microscopy for rapid diagnosis of infectious agents in emergent situations. Emerg Infect Dis 2003;9:294–303.
- Johansen BV. Hazards related to EM-laboratory instrumentation. Ultrastruct Pathol 1984;7:219–25.
- CDC. Interim biosafety guidance for all individuals handling clinical specimens or isolates containing 2009-H1N1 influenza A virus (Novel H1N1), including vaccine strains. August 15, 2009. Available at: http://cdc.gov/h1n1flu/guidelines_labworkers.htm. 2-16-2010.
- Clinical and Laboratory Standards Institute. Performance of the bleeding time test; approved guideline—fourth edition (CLSI document H45-A2).Wayne, PA: Clinical and Laboratory Standards Institute; 2005.
- CDC. Workbook for designing, implementing, and evaluating a sharps injury prevention program. Atlanta, GA: US Department of Health and Human Services, CDC. Available at http://www.cdc.gov/sharpssafety/pdf/sharpsworkbook_2008.pdf.
- Clinical and Laboratory Standards Institute. Clinical flow cytometric analysis of neoplastic hematolymphoid cells; approved guideline—second edition (CLSI document H43-A2). Wayne, PA: Clinical and Laboratory Standards Institute; 2007.
- Clinical and Laboratory Standards Institute. Collection, transport, and processing of blood Specimens for testing plasma-based coagulation assays and molecular hemostasis assays; approved guideline—fifth edition (CLSI document H21-A5). Wayne, PA: Clinical and Laboratory Standards Institute; 2008.
- Clinical and Laboratory Standards Institute. Nucleic acid amplification assays for molecular hematopathology; approved guideline (CLSI document MM05-A). Wayne, PA: Clinical and Laboratory Standards Institute; 2003.
- Clinical and Laboratory Standards Institute. Collection, Transport, Preparation, and storage of specimens for molecular methods; approved guideline (CLSI document MM13-A). Wayne, PA: Clinical and Laboratory Standards Institute; 2005.
- Clinical and Laboratory Standards Institute. Procedures for the collection of diagnostic blood specimens by venipuncture; approved standard—sixth edition (CLSI document H03-A6). Wayne, PA: Clinical and Laboratory Standards Institute; 2007.
- Clinical and Laboratory Standards Institute. Procedures and devices for the collection of diagnostic capillary blood specimens; approved standard—sixth edition (CLSI document H04-A6). Wayne, PA: Clinical and Laboratory Standards Institute; 2008.
- Clinical and Laboratory Standards Institute. Procedures for the handling and processing of blood specimens for common laboratory tests; approved guideline—fourth edition (CLSI document H18-A4). Wayne, PA: Clinical and Laboratory Standards Institute; 2010.
- Selecting, evaluating, and using sharps disposal containers. Cincinnati, OH: US Department of Health and Human Services, CDC, National Institute for Occupational Safety and Health; 1998. Available at http://www.cdc.gov/niosh/pdfs/97-111.pdf.
- US Environmental Protection Agency. Selected EPA-registered disinfectants. Washington DC: US Environmental Protection Agency; 2009. Available at http://www.epa.gov/oppad001/chemregindex.htm.
- American Veterinary Medical Association. One health: a new professional imperative. Schaumburg, IL: American Veterinary Medical Association; 2008. Available at http://www.avma.org/onehealth.
- Torrey EF, Yolken RH. Beasts of the earth. New Brunswick, NJ: Rutgers University Press; 2005.
- Taylor LH, Latham SM, Woolhouse ME. Risk factors for human disease emergence. Philos Trans R Soc Lond B Biol Sci 2001;356:983–9.
- Biosafety and biosecurity in the veterinary microbiology laboratory and animal facilities. In: OIE manual of diagnostic tests and vaccines for terrestrial animals. Paris, France: World Organization of Animal Health (OIE), Office International Des Epizooties; 2008.
- The National Research Council, Committee on Hazardous Biological Substances in the Laboratory. Biosafety in the laboratory. Prudent practices for the handling and disposal of infectious materials. Washington, DC: National Academy Press; 1989. Available at http://www.nap.edu/openbook.php?isbn=0309039754.
- Immunization Action Coalition. Healthcare personnel vaccination recommendations. St. Paul, MN: Immunization Action Coalition; 2011. Available at http://www.immunize.org/catg.d/p2017.pdf.
- CDC. Human rabies prevention—United States, 2008: Recommendations of the Advisory Committee on Immunization Practices. MMWR 2008;57(No. RR-3).
- American Society for Microbiology. Sentinel level clinical microbiology laboratory guidelines. Washington, DC: American Society for Microbiology; 2010. Available at http://www.asm.org/?option=com_content&view=article&id=6342&Itemid=639.
- Gray LD, Snyder JW. Sentinel laboratory guidelines for suspected agents of bioterrorism and emerging infectious diseases. Packing and shipping infectious substances. Washington, DC: American Society for Microbiology; 2010. Available at http://www.asm.org/images/pdf/Clinical/ps11-15-10final.pdf.
- Gray LD, Snyder JW. Packing and shipping biological materials. In: Fleming DO, Hunt DL, eds. Biological safety: principles and practices, 4th edition. Washington, DC: ASM Press; 2006.
- International Air Transport Association. Dangerous goods regulations, 52nd. edition. Montreal, Canada: International Air Transport Association; 2011.
- CDC. Prevention and control of meningococcal disease. Recommendations of the Advisory Committee on Immunization Practices. MMWR 2005;54(No. RR-7).
- CDC. Laboratory-acquired meningococcal disease—United States, 2000. MMWR 2000;51:141–4.
- CDC. Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. MMWR 2005;54(No. RR-17).
- CDC. Updated US Public Health Service guidelines for the management of occupational exposure to HIV and recommendations for postexposure prophylaxis. MMWR 2005;54(No, RR-9).
- CDC. Updated US Public Health Service guidelines for the management of occupational exposure to HBV, HCV, and HIV and recommendations for postexposure prophylaxis. MMWR 2001;50(No. RR-11).
- Clinical and Laboratory Standards Institute. Application of a quality management system model for laboratory services; approved guideline—third edition (CLSI document GP26-A3). Wayne, PA: Clinical and Laboratory Standards Institute; 2004.
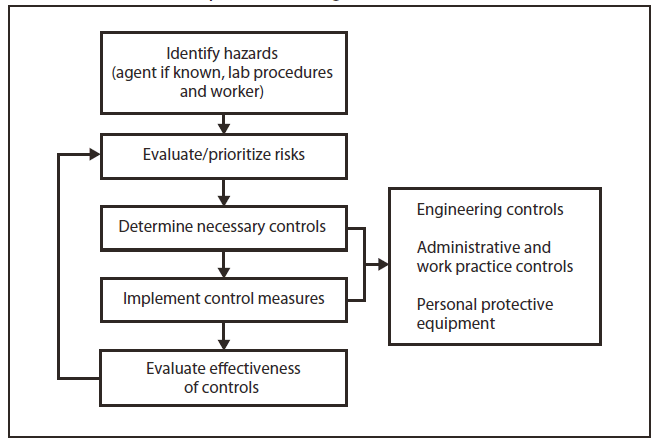
Alternate Text: The figure is a flow chart that presents the risk assessment process for a biologic hazard
|
BSL |
Practices |
Safety equipment and facilities |
|---|---|---|
|
1 |
Standard microbiological practices |
None required |
|
2 |
• BSL-1 practices • Limited access • Display biohazard signs • Sharps precautions • Staff trained with pathogens • Safety manual available |
• BSC used for specimen processing and work producing aerosols or splashes • PPE (coats, gloves, face shields) as needed • Autoclave available |
|
3 |
• BSL-2 practices • Controlled access • Collect baseline serum from personnel |
• BSL-2 equipment/facilities • BSC used for work with all specimens and cultures • PPE (gowns and masks) as needed • Negative pressure airflow • Self-closing double doors • Exhaust air not recirculated |
|
4 |
• BSL-3 practices • Change clothing before entering • Shower on exit • Decontaminate all waste on exit |
• BSL-3 equipment/facilities • Separate building or facility • BSC and full-body, air-supplied positive pressure suit for all procedures • Specialized ventilation and decontamination system |
|
Abbreviations: NIH = National Institutes of Health; BSL = biosafety level; BSC = biological safety cabinet; PPE = personal protective equipment. Source: Adapted from CDC/National Institutes of Health. Biosafety in microbiological and biomedical laboratories. 5th ed. 2007 (1). |
||
|
BOX 3. High-consequence livestock pathogens and select agents |
|
Livestock African horse sickness virus African swine fever virus Akabane virus Avian influenza virus (highly pathogenic) Bluetongue virus (exotic) Bovine spongiform encephalopathy Camel pox virus Classic swine fever virus Foot-and-mouth disease virus Goat pox virus Japanese encephalitis virus Lumpy skin disease virus Menangle virus Mycoplasma capricolum subspecies Mycoids small colony (MmmSC) (contagious bovine pleuropneumonia) Peste des petits ruminants virus Rinderpest virus Sheep pox virus Swine vesicular disease virus Vesicular stomatitis virus (exotic)—Indiana subtypes VSV-IN2, VSV-IN3 Virulent Newcastle disease virus US Department of Agriculture/Department of Health and Human Services overlap agents Bacillus anthracis Brucella abortus Brucella melitensis Brucella suis Burkholderia mallei Burkholderia pseudomallei Hendra virus Nipah virus Rift Valley fever virus Venezuelan equine encephalitis virus Source: Adapted from USDA/APHIS select agent and toxin list, 2010. (http://www.aphis.usda.gov/programs/ag_selectagent/ag_bioterr_toxinlist). |
|
TABLE 14. Rabies pre-exposure prophylaxis guide — United States, 2008 |
|||
|---|---|---|---|
|
Risk category |
Nature of risk |
Typical populations |
Pre-exposure recommendations |
|
Continuous |
Virus present continuously, often in high concentrations. Specific exposures likely to go unrecognized. Bite, nonbite, or aerosol exposure. |
Rabies research laboratory workers; rabies biologics production workers. |
Primary course. Serologic testing every 6 months; booster vaccination if antibody titer is below acceptable level.* |
|
Frequent |
Exposure usually episodic, with source recognized, but exposure also might be unrecognized. Bite, nonbite, or aerosol exposure. |
Rabies diagnostic laboratory workers, cavers, veterinarians and staff, and animal-control and wildlife workers in areas where rabies is enzootic. All persons who frequently handle bats. |
Primary course. Serologic testing every 2 years; booster vaccination if antibody titer is below acceptable level.* |
|
Infrequent (greater than population at large) |
Exposure nearly always episodic with source recognized. Bite or nonbite exposure. |
Veterinarians and animal-control staff working with terrestrial animals in areas where rabies is uncommon to rare. Veterinary students. Travelers visiting areas where rabies is enzootic and immediate access to appropriate medical care including biologics is limited. |
Primary course. No serologic testing or booster vaccination. |
|
Rare (population at large)* |
Exposure always episodic with source recognized. Bite or nonbite exposure. Minimum acceptable antibody level is complete virus neutralization at a 1:5 serum dilution by the rapid fluorescent focus inhibition test. A booster dose should be administered if the titer falls below this level. |
U.S. population at large, including persons in areas where rabies is epizootic. |
No vaccination necessary. |
|
Source: CDC. Human rabies prevention — United States, 2008. MMWR 2008 (212). * Minimum acceptable antibody level is complete virus neutralization at a 1:5 serum dilution by the rapid fluorescent focus inhibition test. A booster dose should be administered if the titer falls below this level. |
|||
Alternate Text: The figure is a flow chart that presents the process for classifying an infectious substance for shipment.
FIGURE 3. A completely labeled outer package. The primary container inside the package contains a Biological Substance, Category B infectious substance and is packed according to PI 650
Abbreviation: PI = packing instructions.
Alternate Text: The figure is a diagram of a shipping package with the appropriate labeling for a Category B infectious substance.
FIGURE 4. A completely labeled outer package. The primary container inside contains a liquid Category A infectious substance and is packed according to PI 620
Abbreviation: PI = packing instructions.
Alternate Text: The figure is a diagram of a shipping package with the appropriate labeling for a liquid Category A infectious substance.
Blue Ribbon Panel for Issues of Clinical Laboratory Safety
Kathleen G. Beavis, MD, College of American Pathologists, Chicago, Illinois; Ellen Jo Baron, PhD, Stanford, California; William R. Dunn, MS, Greater New York Hospital Association Regional Laboratory Task Force, New York, New York; Larry Gray, PhD, American Society for Microbiology, Cincinnati, Ohio; Bill Homovec, MPH, American Clinical Laboratory Association, Burlington, North Carolina; Michael Pentella, PhD, Association of Public Health Laboratories, Iowa City, Iowa; Bruce Ribner, MD, Atlanta, Georgia; William A. Rutala, PhD, Chapel Hill, North Carolina; Daniel S. Shapiro, MD, Burlington, Massachusetts; Lisa A. Skodack-Jones, MT, Salt Lake City, Utah; Christine Snyder, American Society for Clinical Laboratory Science, Helena, Montana; Robert L. Sunheimer, MS, American Society for Clinical Pathology, Syracuse, New York; Christina Z. Thompson, MS, American Biological Safety Association, Greenfield, Indiana.
CDC Staff: Nancy L. Anderson, MMSc; Rex Astles, PhD; D. Joe Boone, PhD; David S. Bressler, MS; Roberta Carey, PhD; Casey Chosewood, MD; Mitchell L. Cohen, MD; Judy Delaney, MS; Thomas L. Hearn, DrPH; Kathleen F. Keyes, MS; Davis Lupo, PhD; Robert Martin, DrPH; Alison C. Mawle, PhD; Terra McConnel; J. Michael Miller, PhD; Shana Nesby, DVM; Janet K. Nicholson, PhD; John P. O'Connor, MS; Anne Pollock; John C. Ridderhof, DrPH; Pamela Robinson; Elizabeth G. Weirich, MS; Ae S. Youngpairoj.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services. |
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


