Latent Tuberculosis Infection Resources
Updated February 22, 2024

The online latent TB infection (LTBI) resource hub is a one-stop shop for resources related to:
- Education and training
- Guidelines
- Testing and diagnosis
- Infection control and prevention
- Treatment
To order free latent TB infection resources, visit CDC-INFO on Demand.
- Advisory Council for the Elimination of Tuberculosis (ACET): Roadmap for Advancing TB Elimination in the United States through Scale up of Testing and Treatment of Latent TB Infection [PDF – 1 MB] (published in ACET Minutes of the Meeting June 16, 2020, Addendum 1, pages 40-51 [PDF – 612KB])
This statement from ACET updates the strategy to eliminate TB in the United States. The report provides new recommendations to prevent TB, specifically through focus on identifying individuals at risk and testing and treating latent TB infection.

- USPSTF Latent Tuberculosis Infection: Screening Recommendation
The US Preventive Services Task Force (USPSTF) makes recommendations about the effectiveness of specific preventive care services for patients without obvious related signs or symptoms.
- Interferon-Gamma Release Assays (IGRAs) – Blood Tests for TB Infection
Interferon-Gamma Release Assays (IGRAs) are whole-blood tests that can aid in diagnosing Mycobacterium tuberculosis infection. - Targeted Tuberculosis Testing and Interpreting Tuberculin Skin Test Results
CDC and USPSTF recommend testing populations that are at increased risk for TB infection. Health care providers should consider several criteria when classifying positive TST reactions.
- Guidelines for the Treatment of Latent Tuberculosis Infection: Recommendations from the National Tuberculosis Controllers Association and CDC, 2020
These updated 2020 LTBI treatment guidelines include the NTCA- and CDC-recommended treatment regimens that comprise three preferred rifamycin-based regimens and two alternative monotherapy regimens with daily isoniazid.
- Update on Recommendations for Use of Once-weekly Isoniazid-Rifapentine Regimen to Treat Latent Mycobacterium Tuberculosis Infection
CDC continues to recommend 3HP for treatment of LTBI in adults and now recommends use of 3HP 1) in persons with LTBI aged 2–17 years; 2) in persons with LTBI who have HIV infection, including acquired immunodeficiency syndrome (AIDS), and are taking antiretroviral medications with acceptable drug-drug interactions with rifapentine; and 3) by DOT or self-administered therapy (SAT) in persons aged ≥2 years.
-
- The 12-Dose Regimen for Latent TB Infection Treatment: Fact Sheet for Clinicians
- Frequently Asked Questions on the 12-dose regimen for Latent TB Infection for Providers
- Frequently Asked Questions on the 12-dose regimen for Latent TB Infection for Pharmacists
- CDC Medscape Commentary: Latent TB Infection Updated Treatment Recommendations
- Treatment Options of Latent Tuberculosis Infection
Available fact sheets on TB treatment include:
- Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV
These guidelines are intended for clinicians, other health care providers, patients with HIV, and policy makers in the United States.

- Latent TB Infection Testing and Treatment: Summary of U.S. Recommendations
This summary includes information on testing for TB infection, recommended test for TB infection, and recommended treatment regimens for latent TB infection. - Latent Tuberculosis Infection: A Guide for Primary Health Care Providers
The guide includes latent TB infection diagnosis and treatment information, sample documentation forms, and additional resources. - Targeted Tuberculosis (TB) Testing and Treatment of Latent TB Infection (slide set)
The slide set provides an overview of the current recommendations for the testing and treatment of latent tuberculosis. The target audience for this slide set is primary health care providers. - CDC Messages and Resources
This information may be helpful in communication activities to inform and educate partners, stakeholders, and media about the importance of expanded latent TB infection testing and treatment in eliminating TB in the United States.
- Testing for Tuberculosis
This fact sheet provides information about the two kinds of tests that are used to determine if a person has been infected with TB bacteria: the tuberculin skin test and TB blood tests. - Tuberculin Skin Testing
The Mantoux tuberculin skin test is one method of determining whether a person is infected with Mycobacterium tuberculosis.
Information and tools for patients on latent TB treatment.
- 12-Dose Regimen for Latent TB Infection
- 12-Dose Regimen for Latent TB Infection-Medication Tracker and Symptom Checklist
- 4 Months Daily Rifampin Schedule for Latent Tuberculosis (TB) Infection
- 3 Months of Daily Isoniazid plus Rifampin (3HR)
These fact sheets will give an overview of regimens for latent TB infection and provide resources for patients.

- Questions and Answers about Tuberculosis
Questions and Answers About Tuberculosis (TB) provides information on the diagnosis and treatment of TB infection and TB disease.
- Slide Set—Introduction to Tuberculosis
This slide set provides a basic overview of TB using plain language and visual aids.
- Kelcie’s Story of LTBI Testing and Treatment
Like many people, Kelcie wasn’t familiar with latent TB infection and wasn’t sure what she should do about it.
- 5 Things to Know about TB
This video highlights the continuing problem of tuberculosis (TB) in the United States.
All 50 states and the District of Columbia require that cases of TB disease be reported to local or state health authorities and the CDC; however, reporting latent TB infections to CDC is optional. Some states and localities have developed legal reporting requirements for latent TB infection as a tool to prevent TB disease.
CDC’s Office of Public Health Law Services completed a legal epidemiology assessment of statutes and regulations associated with latent TB infection reporting across 50 states and the District of Columbia. The map and table below provide a summary of findings as of April, 2021.
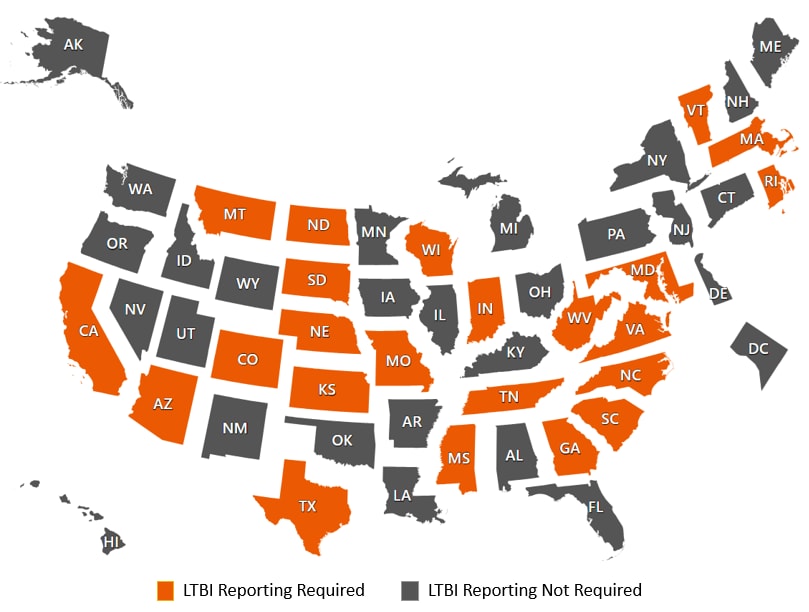
This map is an Equal-Area Cartogram. The area of each state has been modified so that geographic patterns in the data are easier to identify. In traditional maps of the United States, small states are visually overpowered by large states. An Equal-Area Cartogram may appear distorted because states must be separated to accommodate their new sizes.
1State websites included in this table were last visited 9/15/2022 and may differ from current webpages or PDFs. The current (2023) Georgia Notifiable Disease General Reporting Requirements can be found at NDCondition_5.12.23
2At the time of data collection, there was a discrepancy between New Hampshire’s Reportable Infectious Disease List , which listed “Tuberculosis infection, latent (lab reporting only)” as a reportable disease and New Hampshire regulations, which did not. See N.H. Code Admin R. He-P 301.02 (listing only “Tuberculosis Disease” and “Mycobacterium tuberculosis”).
3Current (2023) South Carolina List of Reportable Conditions is found at South Carolina List of Reportable Conditions | SCDHEC. But see supra note 1.
4Current (2023) Tennessee Reportable Disease List for Healthcare Providers is found at 2023-Provider-list.pdf (tn.gov). But see supra note 1.

Copy the code below to embed this image.(200×200)







