* Import-associated cases include 1) internationally imported cases, 2) import-linked cases (those related epidemiologically to imported cases), and 3) imported virus cases (for which no epidemiologic link has been identified but viral genetic evidence indicates an imported measles genotype). Additional information at http://www.cdc.gov/osels/ph_surveillance/nndss/casedef/measles_2010.htm.
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Measles Imported by Returning U.S. Travelers Aged 6--23 Months, 2001--2011
In the first 2 months of 2011, CDC received reports of seven imported measles cases among returning U.S. travelers aged 6--23 months; four required hospitalization. Young children are at greater risk for severe measles, death, or sequelae such as subacute sclerosing panencephalitis (1,2). Although all seven children had been eligible for vaccination before travel, none had received measles, mumps, and rubella (MMR) vaccine, the only measles-containing vaccine currently available in the United States. To characterize imported measles cases reported in the first 2 months of 2011 in U.S. travelers aged 6--23 months and compare them with cases in recent years, CDC analyzed data from the National Notifiable Diseases Surveillance System (NNDSS) for the period January 2001--February 2011. The results of that analysis indicated that, during January--February 2011, a total of 13 imported cases were reported in U.S. residents, including the seven children aged 6--23 months. During 2001--2010, a total of 159 imported cases were reported in U.S. residents, including 47 (range: 3--8 per year) in children aged 6--23 months (three of whom had been vaccinated before travel). Because measles remains endemic in much of the world, international travelers should be up-to-date on vaccinations. In accordance with the Advisory Committee for Immunization Practices (ACIP) recommendations, U.S. children who travel or live abroad should be vaccinated at an earlier age than those living in the United States because of the greater risk for exposure to measles outside the United States, and particularly outside the Americas (3).
In the United States, measles cases are reported to CDC by local and state health departments via NNDSS, using standard case definitions (4). Cases are classified epidemiologically according to the source of infection. An internationally imported measles case is defined as one resulting from exposure to measles virus outside the United States as evidenced by at least some of the exposure period (7--21 days before rash onset) occurring outside the United States and rash occurring within 21 days of entering the United States, with no known exposure to measles in the United States during that time. All other U.S. cases are considered U.S.-acquired. Laboratory confirmation of measles is made by detection in serum of measles-specific immunoglobulin M antibodies, isolation of measles virus, or detection of measles virus by nucleic acid amplification in an appropriate clinical specimen (e.g., nasopharyngeal/oropharyngeal swabs, nasal aspirates, throat washes, or urine). Monitoring of viral genotypes is an important component of measles surveillance and a tool to identify the likely source of imported viruses.
Imported Measles During January--February 2011
Of the 29 measles cases reported during January--February, 2011, a total of 28 were import-associated,* of which 16 (57%) were classified as imported cases; 13 of the imported cases (81%) were among U.S. residents. Of these 13 cases, seven (54%) occurred in children aged 6--23 months. All seven patients had recently traveled internationally; they were residents of Massachusetts (two patients), Texas (one), New York (one), Pennsylvania (one), Washington (one), and California (one). Median age was 10 months (range: 7--23 months) (Table).
Four of the seven patients were hospitalized for measles-related complications: two with diarrhea and dehydration, one with persistent fever, and one with pneumonia. The median duration of hospitalization was 3.5 days; all seven recovered. Diagnosis of measles was delayed in three of the seven patients. One was hospitalized for 3 days for pneumonia and "drug-induced rash," with a measles diagnosis made only after an unvaccinated sibling developed measles. A second patient visited a pediatrician three times before a diagnosis of measles was made in an emergency department and the child was hospitalized. A third patient did not receive an initial medical evaluation until 6 days after onset, and measles was not suspected until a follow-up visit 2 days later.
Measles was laboratory confirmed in patients 2--7 (Table). Although specimens were not obtained from patient 1, five siblings epidemiologically linked to this patient subsequently developed measles, and four of those cases were laboratory confirmed.
The seven patients had traveled to at least six different countries; two traveled to countries in the World Health Organization (WHO) Region of the Americas. Although patient 1 traveled to Haiti during the exposure period, an extensive investigation in Haiti found no evidence of measles transmission in that country. Further investigation identified another U.S. resident with onset of measles at about the same time as patient 1. This person had been in the same terminal of an international airport as patient 1 within a 2-hour interval suggesting a common exposure. For patient 3, an investigation conducted in the Dominican Republic identified as the likely source of this infection a European tourist with measles who stayed in a resort room adjacent to patient 3's family. The genotypes of virus isolated from patient 3 and patients linked epidemiologically to patient 1 all were D4 (Table), with genetic sequences closely related to those of viruses circulating in Europe. The genotypes detected in patients 2, 5, and 6 were the same as the genotypes associated with endemic transmission or recent outbreaks in the country of travel.
Imported Measles During 2001--2010
Of the 692 measles cases reported during 2001--2010, a total of 604 (87%) were import-associated. Of these, 292 cases (48%) were imported; 159 (54%) of the imported cases were in U.S. residents. Among the imports in U.S. residents, 47 (30%) were among children aged 6--23 months (range: 3--8 cases per year) (Figure). Among these 47 children, 23 were aged 6--11 months, 18 were aged 12--15 months, and six were aged 16--23 months. Although all 47 children had been eligible for MMR vaccination, only three (6%) had been vaccinated for measles before their departure. Fourteen (30%) of the 47 children were hospitalized; no deaths were reported.
Reported by
NM Cocoros, MPH, Massachusetts Dept of Public Health. J Zipprich, PhD, California Dept of Public Health. D Kuhles, MD, Nassau County Dept of Health; E Rausch-Phung, MD, CR Schulte, DS Blog, MD, New York State Dept of Health. P Lurie, MD, Pennsylvania Dept of Health. R Wiseman, MPH, Texas Dept of State Health Svcs. C Kroll MPH, Clark County Public Health; C DeBolt, MPH, Washington State Dept of Health. PK Kutty, MD,* SB Redd, AE Barskey, MPH, JS Rota, MPH, PA Rota, PhD, GL Armstrong, MD, WJ Bellini, PhD, KM Gallagher, DSc, Div of Viral Diseases, National Center for Immunization and Respiratory Diseases; AS Mahamud, MD, EIS Officer, CDC. *Corresponding contributor: Preeta Kutty, Div of Viral Diseases, National Center for Immunization and Respiratory Diseases, CDC, pkutty@cdc.gov.
Editorial Note
Since measles was declared eliminated in the United States in 2000, elimination status has been maintained through high MMR vaccination coverage, and most measles cases have been associated with importation (5). MMR vaccine is recommended by ACIP and the American Academy of Pediatrics for routine use in all U.S. children at age 12--15 months, with a booster at age 4--6 years (3,6). Children aged ≥12 months who are traveling internationally should receive 2 doses of MMR vaccine, separated by at least 28 days. Children aged 6--11 months should receive 1 dose of MMR vaccine. Because serologic response to the measles component of the vaccine varies among infants aged 6--11 months, infants vaccinated before age 12 months should be revaccinated on or after the first birthday with 1 dose of MMR vaccine followed by a second dose at least 28 days later (3,6).
In this report, none of the seven children aged 6--23 months with imported measles in the first 2 months of 2011 had received MMR vaccine, and only three of the 47 with imported measles during 2001--2010 had received MMR vaccine. The reasons for nonvaccination of children often are unknown, but contributing to these might be a lack of perceived risk for severe measles (7), which resulted in the hospitalization of four of these seven children aged 6--23 months with measles reported in 2011.
Measles often is not considered in the initial differential diagnosis of children returning from international travel with a rash illness; as a result, diagnosis of measles frequently is delayed, as in three of the seven children with reported measles in 2011. One child was hospitalized for 3 days, yet a measles diagnosis was only made retrospectively after a sibling developed measles. Another visited a pediatrician three times before a diagnosis of measles was made in an emergency department, and a third did not have measles suspected until 8 days after onset. All of these infections and the associated sequelae were potentially preventable through adherence to recommendations for vaccination of children traveling outside the United States.
The frequency of imported measles among children aged 6--23 months also suggests that parents and clinicians might not be aware of recommendations to administer MMR vaccine to children as young as age 6 months when they are living or traveling abroad (7). The parents of one of these 2011 patients asked their pediatrician about vaccination for their child before traveling and were advised that it was unnecessary. Travelers to the WHO European Region should be aware that measles is endemic in several countries of that region, which was the source of 39% of U.S. measles imports during 2005--2008 (8).
Evidence to date supports the finding that measles has been eliminated throughout the Region of the Americas over the last decade. The region expects to document elimination of measles and rubella in every country by 2012. Nonetheless, in the United States, measles importations and transmission from imported cases continue to pose a threat to U.S. residents (8,9). Travelers can be exposed to measles in the country of travel or while en route to and from that country, in airports or on airplanes. An estimated 1.9 million U.S. children travel overseas each year and often are at risk for acquiring infectious diseases that might not be common in the United States (10). These findings highlight the importance of reviewing the vaccination history of anyone planning international travel. Clinicians also should maintain a high level of suspicion for measles in patients with febrile rash illnesses and recent travel outside the United States. Finally, physicians should report suspected measles cases immediately to their local health department and obtain specimens for measles testing, including viral specimens for confirmation and genotyping.
References
- Bellini WJ, Rota JS, Lowe LE, et al. Subacute sclerosing panencephalitis: more cases of this fatal disease are prevented by measles immunization than was previously recognized. J Infect Dis 2005;192:1686--93.
- Miller DL. Frequency of complications of measles, 1963. Report on a national inquiry by the Public Health Laboratory Service in collaboration with the Society of Medical Officers of Health. Br Med J 1964;2:75--8.
- CDC. Measles, mumps, and rubella---vaccine use and strategies for elimination of measles, rubella, and congenital rubella syndrome and control of mumps: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1998;47(No. RR-8).
- CDC. Manual for the surveillance of vaccine-preventable diseases. 4th ed., 2008--2009. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/vaccines/pubs/surv-manual/default.htm. Accessed April 4, 2011.
- Orenstein WA, Papania MJ, Wharton ME. Measles elimination in the United States. J Infect Dis 2004;189(Suppl 1):S1--3.
- American Academy of Pediatrics. Measles. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. Red book: 2009 report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009:444--55. Available at http://aapredbook.aappublications.org/cgi/content/full/2009/1/3.77. Accessed April 4, 2011.
- CDC. Preventable measles among U.S. residents, 2001--2004. MMWR 2005;54:817--20.
- Parker Fiebelkorn A, Redd SB, Gallagher K, et al. Measles in the United States during the postelimination era. J Infect Dis 2010;202:1520--8.
- CDC. Measles outbreak---Hennepin County, Minnesota, February--March 2011. MMWR 2011;60:421.
- CDC. The pre-travel consultation: routine vaccine-preventable diseases. In: Traveler's health: yellow book [Chapter 2]. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://wwwnc.cdc.gov/travel/yellowbook/2010/chapter-2/measles.aspx. Accessed April 4, 2011.
What is already known on this topic?
Although measles was declared eliminated in the United States in 2000, imported cases continue to occur among U.S. travelers returning from areas of the world where measles is endemic.
What is added by this report?
In the first 2 months of 2011, a total of 13 imported cases were reported in U.S. residents, including seven cases in unvaccinated children aged 6--23 months. These 2-month totals were comparable to the number reported each year during 2001--2010, when a total of 159 imported cases were reported in U.S. residents, including 47 (range: 3--8 per year) in children aged 6--23 months, only three of whom had been vaccinated.
What are the implications for public health practice?
Before any international travel, children aged 6--11 months traveling outside the United States should receive 1 dose of measles, mumps, and rubella (MMR) vaccine, and children aged ≥12 months should receive 2 doses of MMR vaccine at least 28 days apart.
FIGURE. Number of imported measles cases in U.S. residents (N = 172), by age group --- January 2001--February 2011
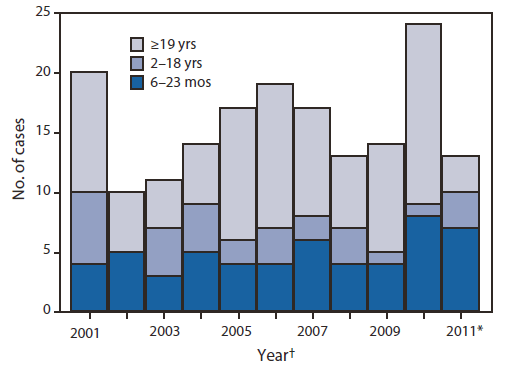
* January--February only.
† Based on date of rash onset.
Alternate Text: The figure above shows the number of imported measles cases in U.S. residents (N = 172) by age group during January 2001-February 2011. Among the imports in U.S. residents during 2001-2010, 47 (30%) were among children aged 6-23 months (range: 3-8 cases per year).
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


