FIGURE 1. Classification of noroviruses into 5 genogroups (GI--V) and 35 genotypes based on sequence diversity in the complete capsid protein VP1 (human strains cluster within GI, GII, and GIV)
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Updated Norovirus Outbreak Management and Disease Prevention Guidelines
Please note: An erratum has been published for this article. To view the erratum, please click here.
The material in this report originated in the National Center for Immunization and Respiratory Diseases, Anne Schuchat, MD, Director; and the Division of Viral Diseases, Jane Seward, MBBS, Acting Director.
Corresponding preparer: Aron J. Hall, DVM, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, CDC, 1600 Clifton Road, MS A-47, Atlanta, GA 30333. Telephone: 404-639-1869; Fax: 404-639-8665; E-mail: ajhall@cdc.gov.
Summary
Noroviruses are the most common cause of epidemic gastroenteritis, responsible for at least 50% of all gastroenteritis outbreaks worldwide, and a major cause of foodborne illness. In the United States, approximately 21 million illnesses attributable to norovirus are estimated to occur annually. Since 2001, when the most recent norovirus recommendations were published (CDC. "Norwalk-like viruses." Public health consequences and outbreak management. MMWR 2001;50[No. RR-9]), substantial advances have been made in norovirus epidemiology, immunology, diagnostic methods, and infection control. As molecular diagnostic techniques have improved in performance and become more widely available, detection and reporting of norovirus outbreaks have increased. Although the inability to culture human noroviruses in vitro has hampered progress, assessment of the performance of disinfectants has been facilitated by the discovery of new, cultivable surrogates for human noroviruses. In addition, the periodic emergence of epidemic strains (from genogroup II type 4, GII.4) and outbreaks in specific populations (e.g., the elderly in nursing homes) have been characterized. This report reviews these recent advances and provides guidelines for outbreak management and disease prevention. These recommendations are intended for use by public health professionals investigating outbreaks of acute gastroenteritis, including state and local health authorities, as well as academic and research institutions.
Introduction
The prototype norovirus was first identified as the cause of a gastroenteritis outbreak in Norwalk, Ohio, in 1968 (1). However, the epidemiology of noroviruses remained poorly characterized until relatively recently because of the lack of widespread availability of sensitive diagnostic methods. Electron microscopy was the first tool used for identifying noroviruses but proved to be a time-consuming and insensitive method that was rarely available outside of research settings. Immunologic assays developed during the late 1970s and 1980s improved detection rates (2) but lacked broad reactivity to detect the full spectrum of noroviruses and were not widely available, limiting their utility for routine use in outbreak investigations. Consequently, the cause of the majority of gastroenteritis outbreaks could not be determined. Because of these limitations of laboratory diagnostics, clinical and epidemiologic criteria were developed to help attribute gastroenteritis outbreaks to norovirus (3). These criteria have subsequently been validated as highly sensitive (99%) and moderately specific (68%) (4). Since the 1990s, the development and subsequent widespread use of molecular diagnostic assays have led to substantial improvements in understanding the role of noroviruses in gastroenteritis outbreaks (5), particularly among those involving foodborne transmission and those occurring in long-term--care facilities (6,7). Nonetheless, progress in the characterization and control of norovirus has been hampered by the lack of a rapid and sensitive assay for use in clinical settings and the inability to cultivate human noroviruses in cell culture.
Since 2001, when CDC published the most recent norovirus recommendations (8), substantial advances have been made in norovirus epidemiology, immunology, diagnostic methods, and infection control. This report reviews these advances and provides guidelines for outbreak management and disease prevention. Specific recommendations include standardized collection of clinical specimens during norovirus outbreaks, use of two recently launched surveillance systems for reporting of norovirus outbreaks, and use of appropriate control measures focusing on hand hygiene, environmental disinfection, and exclusion of ill persons (Box). Their implementation by public health professionals is intended to guide efficient use of public health resources for effective prevention and control of norovirus disease.
Methods
These guidelines were developed by CDC subject matter experts based on a critical review of published literature and unpublished data from outbreak investigations. In June 2010, a preliminary version of these guidelines was presented at the Council of State and Territorial Epidemiologists annual conference to solicit input from state and local public health partners. A draft report was subsequently shared for comment with representatives from five state or local health departments and the U.S. Food and Drug Administration (FDA). These recommendations complement the CDC Healthcare Infection Control Practices Advisory Committee (HICPAC) guideline for the prevention and control of norovirus gastroenteritis outbreaks in health-care settings (9), the model FDA Food Code for retail food and institutional foodservice settings (10), and other applicable state or local regulatory guidance, by providing general principles advisable across all outbreak settings.
Biology and Epidemiology
The Virus
Noroviruses are a group of nonenveloped, single-stranded RNA viruses classified into the genus Norovirus (previously referred to as Norwalk-like viruses [NLVs] or small round-structured viruses [SRSVs]) of the family Caliciviridae. Other genera within the Caliciviridae family include Sapovirus (previously referred to as Sapporo-like viruses [SLVs]), which also cause acute gastroenteritis (AGE) in persons, as well as Lagovirus, Vesivirus, and Nebovirus, which are not pathogenic for humans (11). Noroviruses can be divided into at least five genogroups, designated GI--GV, based on amino acid identity in the major structural protein (VP1) (12). The strains that infect humans (referred to collectively as "human noroviruses") are found in GI, GII, and GIV, whereas the strains infecting cows and mice are found in GIII and GV, respectively (Figure 1). Although interspecies transmission of noroviruses has not been documented, strains that infect pigs are found in GII (13), and a GIV norovirus was discovered recently as a cause of diarrhea in dogs (14), suggesting the potential for zoonotic transmission. On the basis of >85% sequence similarity in the complete VP1 genome, noroviruses can be classified further into genotypes, with at least eight genotypes belonging to GI and 21 genotypes belonging to GII (12,13; CDC, unpublished data, 2011). Since 2001, GII.4 viruses have been associated with the majority of viral gastroenteritis outbreaks worldwide (15). Recent studies have demonstrated that these viruses evolve over time through serial changes in the VP1 sequence, which allow evasion of immunity in the human population (15,16).
Clinical Features
Noroviruses cause acute gastroenteritis in persons of all ages. The illness typically begins after an incubation period of 12--48 hours and is characterized by acute onset, nonbloody diarrhea, vomiting, nausea, and abdominal cramps. Some persons might experience only vomiting or diarrhea. Low-grade fever and body aches also might be associated with infection, and thus the term "stomach flu" often is used to describe the illness, although there is no biologic association with influenza. Although symptoms might be severe, they typically resolve without treatment after 1--3 days in otherwise healthy persons. However, more prolonged courses of illness lasting 4--6 days can occur, particularly among young children, elderly persons, and hospitalized patients (17,18). Approximately 10% of persons with norovirus gastroenteritis seek medical attention, which might include hospitalization and treatment for dehydration with oral or intravenous fluid therapy (7,19,20). Norovirus-associated deaths have been reported among elderly persons and in the context of outbreaks in long-term--care facilities (21,22). Necrotizing enterocolitis in neonates, chronic diarrhea in immunosuppressed patients, and postinfectious irritable bowel syndrome also have been reported in association with norovirus infection (23--25); however, more data from analytic studies are needed to confirm a causal link with these conditions.
Norovirus is shed primarily in the stool but also can be found in the vomitus of infected persons, although it is unclear if detection of virus alone indicates a risk for transmission. The virus can be detected in stool for an average of 4 weeks following infection, although peak viral shedding occurs 2--5 days after infection, with a viral load of approximately 100 billion viral copies per gram of feces (26). However, given the lack of a cell culture system or small animal model for human norovirus, whether these viruses represent infectious virus is unknown, and therefore the time after illness at which an infected person is no longer contagious also is unknown. Furthermore, up to 30% of norovirus infections are asymptomatic, and asymptomatic persons can shed virus, albeit at lower titers than symptomatic persons (26--28). The role of asymptomatic infection in transmission and outbreaks of norovirus remains unclear.
Immunity
Protective immunity to norovirus is complex and incompletely understood. In human challenge studies, infected volunteers were susceptible to reinfection with the same strain as well as to infection with heterologous strains (29--32). In addition, those with preexisting antibodies were not protected from infection unless repeated exposure to the same strain occurred within a short period. Two of these studies demonstrated that homologous antibody protection might last anywhere from 8 weeks to 6 months (30,31). However, the infectious dose of virus given to volunteers in these challenge studies was several-fold greater than the dose of virus capable of causing human illness, and thus immunity to a lower natural challenge dose might be greater and more cross-protective.
Because preexisting antibodies among challenged volunteers did not necessarily convey immunity, and some persons seemed to remain uninfected despite significant exposure, both innate host factors and acquired immunity have been hypothesized to contribute to the susceptibility to infection (31). Histo-blood group antigens (HBGAs), including H type, ABO blood group, and Lewis antigens have been proposed as candidate receptors for norovirus. Expression of HBGAs is associated with strain-specific susceptibility to norovirus infection (17,33--38). Resistance to norovirus infections has been associated with mutations in the 1,2-fucosyltransferase (FUT2) gene leading to a lack of expression of HBGAs on the surface of intestinal cells (33--35,39). Thus, persons who have the normal FUT2 gene and who express these antigens are termed "secretors" whereas mutations in the FUT2 gene leading to the absence of HBGA expression result in "nonsecretor" persons who are less susceptible to infection. However, secretor status does not completely explain the differences seen among infected and uninfected persons for all strains of norovirus. Thus, additional mechanisms of immunity are likely involved, and this remains an ongoing field of research. In addition, evidence suggests that new GII.4 variants evolve to escape the build-up of acquired immunity and innate resistance in the human population (16,40).
Transmission
Norovirus is extremely contagious, with an estimated infectious dose as low as 18 viral particles (41), suggesting that approximately 5 billion infectious doses might be contained in each gram of feces during peak shedding. Humans are the only known reservoir for human norovirus infections, and transmission occurs by three general routes: person-to-person, foodborne, and waterborne. Person-to-person transmission might occur directly through the fecal-oral route, by ingestion of aerosolized vomitus, or by indirect exposure via fomites or contaminated environmental surfaces. Foodborne transmission typically occurs by contamination from infected food handlers during preparation and service but might also occur further upstream in the food distribution system through contamination with human waste, which has been demonstrated most notably by outbreaks involving raspberries and oysters as vehicles (42--46). A recent outbreak involving consumption of delicatessen meat also demonstrated the potential for norovirus contamination during processing (47). Finally, recreational and drinking water can serve as vehicles of norovirus transmission and result in large community outbreaks (48,49). These outbreaks often involve well water that becomes contaminated from septic tank leakage or sewage (50) or from breakdowns in chlorination of municipal systems (51).
Sporadic Disease
As diagnostic methods have improved and become more widely available, the role of noroviruses as the leading cause of sporadic gastroenteritis in all age groups has become clear. Approximately 21 million illnesses caused by norovirus are estimated to occur each year in the United States, approximately one quarter of which can be attributed to foodborne transmission (52). A recent systematic review of 31 community, outpatient, and hospital-based studies in both developed and developing countries estimated that noroviruses accounted for 10%--15% of severe gastroenteritis cases in children aged <5 years and for 9%--15% of mild and moderate diarrhea cases among persons of all ages (53). Although laboratory-based data on endemic norovirus disease in the United States are sparse, recent studies suggest that norovirus is the leading cause of acute gastroenteritis in the community and among persons seeking care in outpatient clinics or emergency departments across all age groups (54,55; CDC, unpublished data, 2011).
Serosurveys have demonstrated that norovirus infections are prevalent throughout the world, with initial exposure typically occurring early in life (5). In population-based studies from Australia, England, Hong Kong, and the Netherlands, norovirus infection has accounted for 9%--24% of gastroenteritis cases (19,20,56--58). In these studies, infection was more frequent in certain age groups (e.g., children aged <5 years and adults aged >65 years). In studies that have used sensitive molecular assays, a relatively high prevalence of norovirus-positive samples in asymptomatic persons has been detected, ranging from 5% in the Netherlands to 16% in England (20,56). This background rate of asymptomatic infection, coupled with innate resistance attributable to secretor status and acquired immunity, helps explain why attack rates rarely exceed 50% in outbreaks.
Outbreaks
Noroviruses are the predominant cause of gastroenteritis outbreaks worldwide. Data from the United States and European countries have demonstrated that norovirus is responsible for approximately 50% of all reported gastroenteritis outbreaks (range: 36%--59%) (5). Outbreaks occur throughout the year although there is a seasonal pattern of increased activity during the winter months. In addition, periodic increases in norovirus outbreaks tend to occur in association with the emergence of new GII.4 strains that evade population immunity (Table) (40,59). These emergent GII.4 strains rapidly replace existing strains predominating in circulation and can sometimes cause seasons with unusually high norovirus activity, as in 2002--2003 and 2006--2007. Because the virus can be transmitted by food, water, and contaminated environmental surfaces as well as directly from person to person, and because there is no long lasting immunity to norovirus, outbreaks can occur in a variety of institutional settings (e.g., nursing homes, hospitals, and schools) and affect people of all ages. Whereas prior national estimates of outbreak attribution by mode of transmission were likely biased toward foodborne disease (60,61), more recent data from individual states indicate that the majority of norovirus outbreaks primarily involve person-to-person transmission (62,63). Multiple routes of transmission can occur within an outbreak; for example, point-source outbreaks from a food exposure often result in secondary person-to-person spread within an institution or community. Of the 660 norovirus outbreaks laboratory confirmed by CDC during 1994--2006 that indicated the setting, 234 (35.4%) were from long-term--care facilities (e.g., nursing homes); 205 (31.1%) were from restaurants, parties, and events; 135 (20.5%) were from vacation settings (including cruise ships); and 86 (13.0%) were from schools and communities (59). Although GII.4 variants predominate overall, the role of GI and other GII genotypes appears to be greater in settings that involve foodborne or waterborne transmission (Figure 2).
Long-Term Care and Other Health-Care Facilities
Health-care facilities including nursing homes and hospitals are the most commonly reported settings of norovirus outbreaks in the United States and other industrialized countries (59,60,63--65). Virus can be introduced from the community into health-care facilities by staff, visitors, and patients who might either be incubating or infected with norovirus upon admission or by contaminated food products. Outbreaks in these settings can be prolonged, sometimes lasting months (66). Illness can be more severe in hospitalized patients than for otherwise healthy persons (18), and associated deaths have been reported (21,22). Strict control measures (including isolation or cohorting of symptomatic patients, exclusion of affected staff, and restricting new admissions into affected units) are disruptive and costly but might be required to curtail outbreaks (9,67,68).
Restaurants and Catered Events
Norovirus is now recognized as the leading cause of foodborne disease outbreaks in the United States. Norovirus accounted for 822 (35%) of the 2,367 foodborne disease outbreaks reported to CDC during 2006--2007 (Figure 3) and half of the 1,641 foodborne disease outbreaks with a confirmed or suspected etiology (69,70). Food can become contaminated with norovirus at any point during production, processing, distribution, and preparation. Thus, a variety of products have been implicated in outbreak investigations; foods eaten raw (e.g., leafy vegetables, fruits, and shellfish) are identified most commonly (69,70). Handling of ready-to-eat foods by infected food employees is commonly identified as a contributing factor in outbreaks of foodborne norovirus associated with food-service establishments (71,72). Norovirus outbreaks also have resulted from fecal contamination of certain food products at the source. For example, oysters harvested from fecally contaminated growing waters and raspberries irrigated with sewage-contaminated water have been implicated in outbreaks (44,73). Because gross sewage contamination will contain a collection of viruses circulating in the community, multiple norovirus genotypes often are detected in such outbreaks. Contamination with norovirus also can occur during processing, as demonstrated by a recent outbreak involving delicatessen meat (47). Only a small dose of virus is needed to cause infection, and thus infected food handlers can contaminate large quantities of product. For example, approximately 500 cases of gastroenteritis were reported during a 2006 outbreak caused by a foodhandler who vomited at work (74). Drinking water or ice also might become contaminated with norovirus and result in outbreaks in food-service settings. Secondary person-to-person transmission is common following point-source food or water exposures.
Schools and Other Institutional Settings
Norovirus outbreaks occur in a range of other institutional settings including schools, child care centers, colleges, prisons, and military encampments. Outbreaks of gastroenteritis in child care centers also are caused by other pathogens, including rotavirus, sapovirus, and astrovirus, as a result of a lack of immunity to these viruses in young populations (75). Outbreaks have been reported recently from multiple universities in different states and have led to campus closures (76). An intervention study in elementary schools demonstrated that improved hand hygiene and surface disinfection can lead to lower rates of absenteeism caused by nonspecific gastroenteritis and reduced surface contamination with norovirus (77). Norovirus was also the most common cause of gastroenteritis in U.S. marines during Operation Iraqi Freedom (78) and a common cause of outbreaks among deployed British troops (79).
Cruise Ships and Other Transportation Settings
Passengers and crew aboard cruise ships are affected frequently by norovirus outbreaks (80). Virus generally is introduced on board by passengers or crew infected before embarkation but might also result from food items contaminated before loading or persistently contaminated environmental surfaces from previous cruises. Virus also might be acquired when ships dock in countries where sanitation might be inadequate, either through contaminated food or water or passengers becoming infected while docked. Repeated outbreaks can occur in subsequent sailings as a result of environmental persistence or infected crew, particularly if control measures have not been implemented consistently and thoroughly. Genotyping of outbreak strains from repeated outbreaks has demonstrated that this can occur through the introduction of new virus or recrudescence of virus from one sailing to the next (81,82). CDC's Vessel Sanitation Program assists the cruise ship industry in preventing and controling the introduction and transmission of gastrointestinal illness by inspecting cruise ships, monitoring gastrointestinal illnesses, and responding to outbreaks (http://www.cdc.gov/nceh/vsp). Outbreaks also have been reported in association with bus tours and air travel, in which environmental contamination and proximity to ill passengers facilitated norovirus transmission (83,84).
Diagnostic Methods
The majority of clinical virology laboratories perform real-time reverse transcription-polymerase chain reaction (RT-PCR) assays (specifically, TaqMan-based real-time assays) for norovirus detection. These assays have not been cleared by FDA although commercial kits are available in the United States. Positive samples can be typed subsequently by DNA sequencing of conventional RT-PCR products. FDA has recently cleared the RIDASCREEN Norovirus 3rd Generation (r-Biopharm, AG, Darmstadt, Germany) enzyme immunoassay (EIA) for preliminary identification of norovirus as the cause of an AGE outbreak. However, negative EIA results should be confirmed by RT-PCR reference methods.
Real-Time RT-PCR Assays
The ORF1-ORF2 junction region is the most conserved region of the norovirus genome, with a high level of nucleotide sequence identity across strains within a genogroup (85,86). This feature makes this region ideal for designing broadly reactive primers and probes for TaqMan-based real-time RT-PCR (RT-qPCR) assays (Figure 4). The majority of published assays are derived from the original publication (86), which has been modified into one-step and duplex assays (87,88) for high throughput screening in clinical diagnostic laboratories. In addition to detecting norovirus RNA in clinical samples, relatively small optimizations of the primers and probes have resulted in sensitive assays to detect norovirus RNA in environmental samples (e.g., food and water) (64,89--91). The extreme analytic sensitivity of RT-qPCR permits the detection of very low titers of virus that might be present in samples from persons without disease caused by norovirus (i.e., asymptomatic infection) (28). For this reason, low titer results (i.e., high cycle threshold [CT] values) should be interpreted with caution.
Conventional RT-PCR Assays for Genotyping
Four different regions (designated A--D) of the genome have been used successfully for genotyping of noroviruses (92,93). However, typing based on the capsid gene (region C and region D) allows for better differentiation between strains (Figure 4). Although region C sequences have been used widely for genotyping strains by clinical diagnostic laboratories in the United States, Europe, and Japan, the resolution of this region is not sufficient to distinguish differences between certain GII.4 variants, which are the predominant strains associated with outbreaks (59). The gold standard for genotyping norovirus strains is full capsid sequencing. However, for clinical samples with high copy numbers, amplifying partial capsid sequences is more practical and is only slightly less discriminatory than full capsid sequencing (92). For this reason, laboratories participating in CaliciNet, CDC's electronic norovirus outbreak surveillance network (see Reporting and Assistance), use region D genotyping.
Enzyme Immunoassays
For detection of norovirus antigen in clinical samples, rapid assays (e.g., EIA) offer an attractive alternative to expensive and technically demanding molecular detection assays. However, the development of a broadly reactive EIA for noroviruses has been challenging because of the number of antigenically distinct norovirus strains and the high viral load required for a positive signal by these assays. Commercial kits, including IDEIA Norovirus EIA (Oxoid, Ely, United Kingdom), SRSV (II)-AD (Denka Seiken Co. Ltd., Tokyo, Japan), and RIDASCREEN include pools of cross-reactive monoclonal and polyclonal antibodies (94--101). In evaluations, the sensitivity of these kits when compared with RT-PCR has ranged from 36% to 80%, and specificity has ranged from 47% to 100% (94--101). Because of the modest performance of these commercial kits, particularly their poor sensitivity, they are not recommended for clinical diagnosis of norovirus infection in sporadic cases of gastroenteritis. However, given their ease of use and rapid results, EIA kits with high specificity (>85%) and at least moderate sensitivity (>50%) might be useful for preliminary screening of multiple fecal samples associated with an outbreak of acute gastroenteritis. These kits are not widely available in the United States, although the RIDASCREEN Norovirus 3rd Generation EIA has been recently cleared by FDA for preliminary identification of norovirus as the cause of AGE outbreaks. Nonetheless, negative samples will have to be confirmed by a second technique such as RT-qPCR, and therefore EIA kits should not be regarded as a replacement for molecular methods during outbreak investigations.
Specimen Collection
Clinical Specimens
Whole stool specimens should be collected for laboratory diagnosis of norovirus and are preferred over rectal swabs because of the higher quantity of virus present in whole stool. Vomitus also may be collected and tested using molecular assays, although unlike for stool, the sensitivity of these assays for vomitus is unknown. To aid in interpretation, each specimen should be accompanied with dates of symptom onset and specimen collection. An outbreak of acute gastroenteritis is considered to be laboratory-confirmed as attributable to norovirus if stool or vomitus specimens from two or more ill persons are positive for norovirus by RT-PCR, EIA, or electron microscopy (71).
Stool
Timing. Specimen collection for norovirus testing should begin as early as possible in an epidemiologic investigation. Unnecessary delays while awaiting results from testing for bacterial or parasitic agents could preclude establishment of a viral diagnosis. Ideally, whole stool specimens should be obtained during the acute phase of illness (i.e., within 48--72 hours after onset) while the stools are still liquid or semisolid and viral excretion is at its peak. When this is not possible, specimens collected later in illness or after resolution (i.e., up to 7--10 days after onset) might still provide a diagnosis and an opportunity to confirm norovirus infection in a suspected source case-patient (e.g., a food handler with a recent history of diarrhea). If specimens are collected late in the illness, the utility of viral diagnosis and interpretation of the results should be discussed with laboratory personnel before tests are conducted (see Reporting and Assistance).
Number and quantity. Obtaining a collection of high quality (i.e., sufficient volume collected during acute illness) diagnostic specimens is crucial for laboratory confirmation. Whole stool specimens from at least five ill persons are recommended. This number should be increased if specimens are taken after the acute phase of illness, in large or protracted outbreaks, or if less sensitive EIA diagnostics are employed. On the basis of test characteristics of commercially available EIAs, six specimens are required to achieve >90% sensitivity in outbreak diagnosis (101,102). Bulk samples (i.e., 10--50 mL of whole stool placed in a stool cup or urine container) are preferred, because rectal swabs are generally insufficient (CDC, unpublished data, 2011).
Storage and transport. Whole stool specimens should be kept refrigerated at 39°F (4°C) if testing occurs within 2--3 weeks. At this temperature, specimens can be stored without compromising diagnostic yield. If the specimens have to be transported to a laboratory for testing, they should be individually bagged and sealed and kept on ice or frozen refrigerant packs in an insulated, waterproof container. If testing is expected to occur >3 weeks after collection or for archiving purposes, stool samples should be frozen at -4°F (-20°C) or -94°F (-70°C). Norovirus RNA can be detected by using molecular assays after at least 5 years when samples are stored under these conditions (59). Although it was previously recommended to avoid freezing stool specimens because of degradation of viral morphology and consequent hampering of diagnosis by electron microscopy (8), this is no longer a concern because of the widespread use of molecular assays.
Vomitus
Specimens of vomitus can be collected to supplement the diagnostic yield from stool specimens during an investigation. Recommendations for collection, storage, and shipment of vomitus specimens are the same as those for stool specimens.
Serum
Serum specimens might be useful in unique circumstances or for special studies but are not recommended for routine diagnostics. If feasible and warranted, acute- and convalescent-phase serum specimens may be obtained to test for a greater-than-fourfold rise in IgG titer to noroviruses. Acute-phase specimens should be obtained during the first 5 days after symptom onset, and the convalescent-phase specimens should be collected during the third to fourth week after resolution of symptoms. Because these assays are not available commercially, CDC may be contacted for further guidance on collection, processing, and storage of serum specimens (see Reporting and Assistance).
Environmental Specimens
Because human noroviruses cannot be grown in cell culture, current detection methods (e.g., RT-qPCR) allow detection of norovirus RNA in water, food, and environmental specimens; however, validated methods are available only for water (at CDC) and shellfish (at FDA's Gulf Coast Seafood Laboratory). If a food or a water source is strongly suspected as the source of an outbreak, a sample should be obtained as early as possible with respect to the time of exposure and preferably stored frozen at -4°F (-20°C), and CDC or FDA should be contacted for further guidance on testing. Detection of norovirus in food requires appropriate elution and concentration techniques which need to be adapted for each food vehicle. At present, these methods are not available routinely, except for shellfish. Water can be tested for noroviruses after concentration of large volumes (e.g., up to 100 L of water) through specially designed filters (103). Environmental surface swabs have also been demonstrated to detect norovirus RNA in specific outbreak settings (104,105). Depending on the specific situation, epidemiologic findings, and expected level of contamination, collection of environmental specimens might be warranted and should be pursued in consultation with CDC. (See Reporting and Assistance).
Prevention and Control
Hand Hygiene
Appropriate hand hygiene is likely the single most important method to prevent norovirus infection and control transmission. Reducing any norovirus present on hands is best accomplished by thorough handwashing with running water and plain or antiseptic soap. Washing with plain soap and water reduces the number of microbes on hands via mechanical removal of loosely adherent microorganisms (106). The efficacy of alcohol-based and other hand sanitizers against norovirus remains controversial with mixed evidence depending on the product formulation and evaluation methodology. In finger pad studies, soap and water used for 20 seconds have been demonstrated to reduce norovirus by 0.7--1.2 log10 by RT-PCR assay, whereas alcohol-based hand sanitizers did not demonstrate any appreciable reduction of viral RNA (107). However, such studies cannot determine whether the residual virus remains viable given the inability to cultivate human norovirus in vitro.
Studies using cultivable surrogate viruses such as murine norovirus (MNV) or feline calicivirus (FCV) have demonstrated that ethanol has superior efficacy against FCV compared with other alcohols (e.g., propanol-1 and propanol-2), and formulations containing 70% ethanol were able to reduce infectious MNV by 2.5 log10 after 30 seconds (108--110). The sensitivity of FCV to low pH and the relatively high susceptibility of MNV to alcohols suggest that sanitizers that are effective against both surrogate viruses might be more likely to be effective against human norovirus, than those effective against only one of the surrogates (111). In addition, reduction of viral RNA (as opposed to reduced infectivity of cultivable surrogates) does not appear to be a reliable means of estimating the effectiveness of hand sanitizers against human norovirus (111).
Overall, studies suggest that proper hand washing with soap and running water for at least 20 seconds is the most effective way to reduce norovirus contamination on the hands, whereas hand sanitizers might serve as an effective adjunct in between proper handwashings but should not be considered a substitute for soap and water handwashing (106--111). As an additional preventive strategy, no bare-hand contact with ready-to-eat foods (foods edible without washing, cooking, or additional preparation to achieve food safety) is recommended (112; FDA, unpublished data, 2011).
Exclusion and Isolation
Considering the highly infectious nature of norovirus, exclusion and isolation of infected persons are often the most practical means of interrupting transmission of virus and limiting contamination of the environment. This is particularly important in settings where people reside or congregate such as long-term--care facilities, acute-care hospitals, cruise ships, and college dormitories. Empiric evidence for the effectiveness of exclusion and isolation strategies is limited (113); rather, strategies are based on infection-control principles. The principle underpinning isolation is to minimize contact with persons during the most infectious periods of their illness. This includes the acute phase of illness, a period following recovery while the person is still shedding virus at high levels (usually 24--72 hours), and, in some situations in health-care facilities, exclusion of exposed and potentially incubating persons. Isolation of both exposed and unexposed well persons might be useful during outbreaks in long-term--care facilities to help break the cycle of transmission and prevent additional cases. In health-care facilities, ill patients may be cohorted together in a unit or part thereof, with dedicated nursing staff providing care for infected persons (9). Analogously, passengers with gastroenteritis on cruise ships may be asked to remain isolated voluntarily in their cabins during their illness and for a period of 24--48 hours after their symptoms have resolved. Ill patients generally should not be transferred to unaffected units in the facility or to other facilities, except in the case of medical necessity and after consultation with infection-control staff. To minimize the risk of spread from incubating or asymptomatically infected patients and staff in health-care facilities, such persons should not be transferred to or work on (respectively) unaffected areas, typically for 48 hours after exposure. Ill staff members in health-care facilities and food handlers should be excluded during their illness and for 48--72 hours following resolution of symptoms. Asymptommatic food-service workers who have tested positive for norovirus, which might occur during an outbreak investigation, also should be excluded or restricted per the FDA Food Code (10). Regulatory authority approval might be required for excluded food-service workers to return to work (10), although requiring negative stool results prior to returning to work is not recommended. Sick pay and sick leave policies that do not penalize ill workers might help to facilitate such staff exclusion. Nonessential personnel, including visitors, may be screened for symptoms and excluded or, at the minimum, should be cautioned about the risks and made aware of the heightened importance of hand hygiene through washing with soap and water. In certain situations, units in a health-care facility may be closed to new admissions to prevent the introduction of new susceptible patients (113).
Environmental Disinfection
The use of chemical disinfectants is one of the key approaches to interrupt norovirus spread from contaminated environmental surfaces. Particular attention should be given to the likely areas of greatest environmental contamination such as bathrooms and high-touch surfaces (e.g., door knobs and hand rails). Sodium hypochlorite (chlorine bleach) has been widely recommended to disinfect human norovirus from surfaces, and its efficacy has been well documented (114--117). Environmental surfaces potentially contaminated with norovirus should be disinfected using a sodium hypochlorite solution or other commercial product registered with EPA as effective against norovirus. A list of EPA-approved products is available at http://www.epa.gov/oppad001/list_g_norovirus.pdf. Products containing phenolic compounds (including triclosan and quarternary ammonium compounds) are less effective against nonenveloped viruses such as human norovirus (114,118--120). Furthermore, this list should be interpreted with caution because the efficacy of these products is determined by using the surrogate FCV, which exhibits different physiochemical properties than human norovirus and therefore might not reflect a similar disinfection efficacy profile (121). In addition, several recent reports have demonstrated that FCV is not the most resistant surrogate virus to predict inactivation of human norovirus (111,122,123). Therefore, the use of multiple surrogate viruses rather than a single surrogate has been suggested to assess the efficacy of disinfectants (111).
Because of this uncertainty, whenever possible, chlorine bleach solution should be applied to hard, nonporous, environmental surfaces at a concentration of 1,000--5,000 ppm (5--25 tablespoons household bleach [5.25%] per gallon of water). Given the potential for evaporative dilution, bleach solutions should be freshly prepared for use within 24 hours, or the target concentration should be doubled (e.g., 2,000--10,000 ppm) for storage and used within 30 days. Although the upper end of this range has been recommended previously when soiling is present (124), few data are available to support the effectiveness of sodium hypochlorite on fecally soiled surfaces (125). For example, 5,000 ppm of sodium hypochlorite was not able to completely eliminate norovirus dried in 20% fecal stool suspension on surfaces (126). A recent study indicated that longer exposure times (approximately 4 minutes) of 5,000 ppm sodium hypochlorite on fecally soiled surfaces were needed to inactivate both human norovirus surrogates (MNV and FCV) by 4 log10 (122),which has been proposed as a satisfactory reduction level for norovirus contamination (127). In conclusion, initial cleaning of contaminated surfaces to remove organic loads such as fecal material should be performed before sodium hypochlorite disinfection.
In health-care settings, cleaning products and disinfectants used should be EPA-registered and have label claims for use in health-care settings; personnel performing environmental services should adhere to the manufacturer's instructions for dilution, application, and contact time. Heat disinfection (i.e., pasteurization to 140°F [60°C]) has been suggested and used successfully under laboratory conditions, for items that cannot be subjected to chemical disinfectants such as chlorine bleach (128--131). Other disinfection approaches such as ozone, hydrogen peroxide, or coating surfaces with antimicrobial materials (e.g., titanium dioxide [TiO2] film) also have been proposed for routine environmental control of noroviruses (132--134).
Reporting and Assistance
Individual cases of norovirus are not nationally notifiable; however, all outbreaks of acute gastroenteritis should be reported to the appropriate local or state health departments, in accordance with local regulations. Additional outbreak assistance, including coordination of multistate outbreak investigations, epidemiologic consultation, and laboratory support can be provided by the CDC Norovirus Program located within the Division of Viral Diseases, National Center for Immunization and Respiratory Diseases. After telephone consultation with the National Calicivirus Laboratory (404-639-1923), state or local public health laboratories can ship outbreak specimens to CDC at National Calicivirus Laboratory, Dash Unit 75, CDC, 1600 Clifton Road, N.E., Atlanta, GA 30333.
National Outbreak Reporting System
In response to a position statement by the Council of State and Territorial Epidemiologists, CDC developed the National Outbreak Reporting System (NORS) as an integrated national surveillance system for enteric disease outbreaks that combined and expanded on the longstanding national surveillance systems for foodborne and waterborne outbreaks. Launched in February 2009, NORS provides a framework through which all outbreaks of enteric disease, regardless of transmission mode, may be reported from state and local health departments to CDC. This web-based platform includes modules for outbreaks attributable to foodborne, waterborne, and person-to-person outbreaks, thereby providing comprehensive national surveillance for all gastroenteritis outbreaks, including those caused by norovirus. Data collected through NORS will help determine the burden and trends of epidemic norovirus and attribution to settings, transmission routes, and food vehicles. Such information is critical to develop appropriate prevention and control strategies that target points of intervention most likely to yield the greatest impact. States are encouraged to contribute to this national surveillance effort by reporting all suspected and confirmed norovirus outbreaks through NORS.
CaliciNet
Genotyping of norovirus strains has been demonstrated to be useful in confirming or identifying links between clusters of norovirus illness, and direct sequencing of RT-PCR products has become the method of choice. To increase the value of national norovirus surveillance, CDC has developed and implemented an electronic norovirus outbreak surveillance network (CaliciNet) with state and local public health and food regulatory agency laboratories. CaliciNet is intended to link norovirus outbreaks to aid in identifying common (e.g., food) vehicles, identify trends, and identify emerging norovirus strains. Using standardized genotyping methods, CaliciNet-certified laboratories upload norovirus sequences and conduct rapid comparison with sequences already in the database. CaliciNet was implemented in March 2009, and as of January 2011, a total of 21 states were certified for participation. With continued development and expansion of the network, CaliciNet might help elucidate the role of food contamination throughout the continuum from production to preparation and the evolution of specific norovirus strains in causing outbreaks, which ultimately can help better target interventions.
References
-
Kapikian AZ, Wyatt RG, Dolin R, Thornhill TS, Kalica AR, Chanock RM. Visualization by immune electron microscopy of a 27-nm particle associated with acute infectious nonbacterial gastroenteritis. J Virol 1972;10:1075--81.
-
Kaplan JE, Gary GW, Baron RC, et al. Epidemiology of Norwalk gastroenteritis and the role of Norwalk virus in outbreaks of acute nonbacterial gastroenteritis. Ann Intern Med 1982;96(6 Pt 1):756--61.
-
Kaplan JE, Feldman R, Campbell DS, Lookabaugh C, Gary GW. The frequency of a Norwalk-like pattern of illness in outbreaks of acute gastroenteritis. Am J Public Health 1982;72:1329--32.
-
Turcios RM, Widdowson MA, Sulka AC, Mead PS, Glass RI. Reevaluation of epidemiological criteria for identifying outbreaks of acute gastroenteritis due to norovirus: United States, 1998--2000. Clin Infect Dis 2006;42:964--9.
-
Patel MM, Hall AJ, Vinje J, Parashar UD. Noroviruses: a comprehensive review. J Clin Virol 2009;44:1--8.
-
Rosenthal NA, Lee LE, Vermeulen BA, et al. Epidemiological and genetic characteristics of norovirus outbreaks in long-term care facilities, 2003--2006. Epidemiol Infect 2011; 139:286--94.
-
Widdowson MA, Sulka A, Bulens SN, et al. Norovirus and foodborne disease, United States, 1991--2000. Emerg Infect Dis 2005;11:95--102.
-
MacCannell T, Umsheid CA, Agarwal RK, et al. Guideline for the prevention and control of norovirus gastroenteritis outbreaks in healthcare settings. Atlanta, GA: US Department of Health and Human Services, CDC; 2010.
-
Food and Drug Administration. Food code 2009. College Park, MD: Food and Drug Administration; 2010. Available at http://www.fda.gov/Food/FoodSafety/RetailFoodProtection/FoodCode/FoodCode2009/default.htm. Accessed February 2, 2011.
-
Green KY, Ando T, Balayan MS, et al. Taxonomy of the caliciviruses. J Infect Dis 2000;181(Suppl 2):S322--30.
-
Zheng DP, Ando T, Fankhauser RL, Beard RS, Glass RI, Monroe SS. Norovirus classification and proposed strain nomenclature. Virology 2006;346:312--23.
-
Wang QH, Han MG, Cheetham S, Souza M, Funk JA, Saif LJ. Porcine noroviruses related to human noroviruses. Emerg Infect Dis 2005;11:1874--81.
-
Martella V, Lorusso E, Decaro N, et al. Detection and molecular characterization of a canine norovirus. Emerg Infect Dis 2008;14:1306--8.
-
Siebenga JJ, Vennema H, Zheng DP, et al. Norovirus illness is a global problem: emergence and spread of Norovirus GII.4 Variants, 2001--2007. J Infect Dis 2009;200:802--12.
-
Lindesmith LC, Donaldson EF, Lobue AD, et al. Mechanisms of GII.4 norovirus persistence in human populations. PLoS Med 2008;5:e31.
-
Rockx B, De Wit M, Vennema H, et al. Natural history of human calicivirus infection: a prospective cohort study. Clin Infect Dis 2002;35:246--53.
-
Lopman BA, Reacher MH, Vipond IB, Sarangi J, Brown DW. Clinical manifestation of norovirus gastroenteritis in health care settings. Clin Infect Dis 2004;39:318--24.
-
Phillips G, Tam CC, Conti S, et al. Community incidence of norovirus-associated infectious intestinal disease in England: improved estimates using viral load for norovirus diagnosis. Am J Epidemiol 2010;171:1014--22.
-
de Wit MA, Koopmans MP, Kortbeek LM, et al. Sensor, a population-based cohort study on gastroenteritis in the Netherlands: incidence and etiology. Am J Epidemiol 2001;154:666--74.
-
Harris JP, Edmunds WJ, Pebody R, Brown DW, Lopman BA. Deaths from norovirus among the elderly, England and Wales. Emerg Infect Dis 2008;14:1546--52.
-
CDC. Norovirus activity---United States, 2006--2007. MMWR 2007;56:842--6.
-
Turcios-Ruiz RM, Axelrod P, St John K, et al. Outbreak of necrotizing enterocolitis caused by norovirus in a neonatal intensive care unit. J Pediatr 2008;153:339--44.
-
Westhoff TH, Vergoulidou M, Loddenkemper C, et al. Chronic norovirus infection in renal transplant recipients. Nephrol Dial Transplant 2009;24:1051--3.
-
Marshall JK, Thabane M, Borgaonkar MR, James C. Postinfectious irritable bowel syndrome after a food-borne outbreak of acute gastroenteritis attributed to a viral pathogen. Clin Gastroenterol Hepatol 2007;5:457--60.
-
Atmar RL, Opekun AR, Gilger MA, et al. Norwalk virus shedding after experimental human infection. Emerg Infect Dis 2008;14:1553--7.
-
Graham DY, Jiang X, Tanaka T, Opekun AR, Madore HP, Estes MK. Norwalk virus infection of volunteers: new insights based on improved assays. J Infect Dis 1994;170:34--43.
-
Phillips G, Lopman B, Tam CC, Iturriza-Gomara M, Brown D, Gray J. Diagnosing norovirus-associated infectious intestinal disease using viral load. BMC Infect Dis 2009;9:63.
-
Baron RC, Greenberg HB, Cukor G, Blacklow NR. Serological responses among teenagers after natural exposure to Norwalk virus. J Infect Dis 1984;150:531--4.
-
Johnson PC, Mathewson JJ, DuPont HL, Greenberg HB. Multiple-challenge study of host susceptibility to Norwalk gastroenteritis in US adults. J Infect Dis 1990;161:18--21.
-
Parrino TA, Schreiber DS, Trier JS, Kapikian AZ, Blacklow NR. Clinical immunity in acute gastroenteritis caused by Norwalk agent. N Engl J Med 1977;297:86--9.
-
Wyatt RG, Dolin R, Blacklow NR, et al. Comparison of three agents of acute infectious nonbacterial gastroenteritis by cross-challenge in volunteers. J Infect Dis 1974;129:709--14.
-
Hutson AM, Atmar RL, Graham DY, Estes MK. Norwalk virus infection and disease is associated with ABO histo-blood group type. J Infect Dis 2002;185:1335--7.
-
Lindesmith L, Moe C, Marionneau S, et al. Human susceptibility and resistance to Norwalk virus infection. Nat Med 2003;9:548--53.
-
Tan M, Jin M, Xie H, Duan Z, Jiang X, Fang Z. Outbreak studies of a GII-3 and a GII-4 norovirus revealed an association between HBGA phenotypes and viral infection. J Med Virol 2008;80:1296--301.
-
Hutson AM, Atmar RL, Estes MK. Norovirus disease: changing epidemiology and host susceptibility factors. Trends Microbiol 2004;12:279--87.
-
Larsson MM, Rydell GE, Grahn A, et al. Antibody prevalence and titer to norovirus (genogroup II) correlate with secretor (FUT2) but not with ABO phenotype or Lewis (FUT3) genotype. J Infect Dis 2006;194:1422--7.
-
Lindesmith L, Moe C, Lependu J, Frelinger JA, Treanor J, Baric RS. Cellular and humoral immunity following Snow Mountain virus challenge. J Virol 2005;79:2900--9.
-
Marionneau S, Ruvoen N, Le Moullac-Vaidye B, et al. Norwalk virus binds to histo-blood group antigens present on gastroduodenal epithelial cells of secretor individuals. Gastroenterology 2002;122:1967--77.
-
Cannon JL, Lindesmith LC, Donaldson EF, Saxe L, Baric RS, Vinje J. Herd immunity to GII.4 noroviruses is supported by outbreak patient sera. J Virol 2009;83:5363--74.
-
Teunis PF, Moe CL, Liu P, et al. Norwalk virus: how infectious is it? J Med Virol 2008;80:1468--76.
-
Dowell SF, Groves C, Kirkland KB, et al. A multistate outbreak of oyster-associated gastroenteritis: implications for interstate tracing of contaminated shellfish. J Infect Dis 1995;171:1497--503.
-
Morse DL, Guzewich JJ, Hanrahan JP, et al. Widespread outbreaks of clam- and oyster-associated gastroenteritis. Role of Norwalk virus. N Engl J Med 1986;314:678--81.
-
Falkenhorst G, Krusell L, Lisby M, Madsen SB, Bottiger B, Molbak K. Imported frozen raspberries cause a series of norovirus outbreaks in Denmark, 2005. Euro Surveill 2005;10:E050922.2.
-
Gaulin CD, Ramsay D, Cardinal P, D'Halevyn MA. [Epidemic of gastroenteritis of viral origin associated with eating imported raspberries.] Can J Public Health 1999;90:37--40.
-
Le Guyader FS, Mittelholzer C, Haugarreau L, et al. Detection of noroviruses in raspberries associated with a gastroenteritis outbreak. Int J Food Microbiol 2004;97:179--86.
-
Malek M, Barzilay E, Kramer A, et al. Outbreak of norovirus infection among river rafters associated with packaged delicatessen meat, Grand Canyon, 2005. Clin Infect Dis 2009;48:31--7.
-
Beller M, Ellis A, Lee SH, Drebot MA, et al. Outbreak of viral gastroenteritis due to a contaminated well. International consequences. JAMA 1997;278:563--8.
-
Kukkula M, Maunula L, Silvennoinen E, von Bonsdorff CH. Outbreak of viral gastroenteritis due to drinking water contaminated by Norwalk-like viruses. J Infect Dis 1999;180:1771--6.
-
Scallan E, Hoekstra RM, Angulo FJ, et al. Foodborne illness acquired in the United States---major pathogens. Emerg Infect Dis 2011;;17:7--15.
-
Patel MM, Widdowson MA, Glass RI, Akazawa K, Vinje J, Parashar UD. Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg Infect Dis 2008;14:1224--31.
-
Nataro JP, Mai V, Johnson J, et al. Diarrheagenic Escherichia coli infection in Baltimore, Maryland, and New Haven, Connecticut. Clin Infect Dis 2006;43:402--7.
-
Hall AJ, Rosenthal M, Gregoricus N, et al. Burden of noroviruses and other enteric viruses in community and outpatient settings, Georgia, United States [Presentation]. 4th International Conference on Caliciviruses, Santa Cruz, Chile; October 16--19, 2010.
-
Amar CF, East CL, Gray J, Iturriza-Gomara M, Maclure EA, McLauchlin J. Detection by PCR of eight groups of enteric pathogens in 4,627 faecal samples: re-examination of the English case-control Infectious Intestinal Disease Study (1993--1996). Eur J Clin Microbiol Infect Dis 2007;26:311--23.
-
Lau CS, Wong DA, Tong LK, et al. High rate and changing molecular epidemiology pattern of norovirus infections in sporadic cases and outbreaks of gastroenteritis in Hong Kong. J Med Virol 2004;73:113--7.
-
Marshall JA, Hellard ME, Sinclair MI, et al. Incidence and characteristics of endemic Norwalk-like virus-associated gastroenteritis. J Med Virol 2003;69:568--78.
-
Zheng DP, Widdowson MA, Glass RI, Vinje J. Molecular epidemiology of genogroup II-genotype 4 noroviruses in the United States between 1994 and 2006. J Clin Microbiol 2010;48:168--77.
-
Blanton LH, Adams SM, Beard RS, et al. Molecular and epidemiologic trends of caliciviruses associated with outbreaks of acute gastroenteritis in the United States, 2000--2004. J Infect Dis 2006;193:413--21.
-
Fankhauser RL, Monroe SS, Noel JS, et al. Epidemiologic and molecular trends of "Norwalk-like viruses" associated with outbreaks of gastroenteritis in the United States. J Infect Dis 2002;186:1--7.
-
Doyle TJ, Stark L, Hammond R, Hopkins RS. Outbreaks of noroviral gastroenteritis in Florida, 2006--2007. Epidemiol Infect 2009;137:617--25.
-
Yen C, Wikswo M, Lopman B, Hall A. Temporal trends in norovirus activity--- United States, January 2007--April 2010 [Presentation]. 4th International Conference on Caliciviruses, Santa Cruz, Chile; October 16--19, 2010.
-
Svraka S, Duizer E, Vennema H. Etiological role of viruses in outbreaks of acute gastroenteritis in The Netherlands from 1994 through 2005. J Clin Microbiol 2007;45:1389--94.
-
Lopman BA, Reacher MH, Van Duijnhoven Y, Hanon FX, Brown D, Koopmans M. Viral gastroenteritis outbreaks in Europe, 1995--2000. Emerg Infect Dis 2003;9:90--6.
-
Johnston CP, Qiu H, Ticehurst JR, et al. Outbreak management and implications of a nosocomial norovirus outbreak. Clin Infect Dis 2007;45:534--40.
-
Lopman BA, Reacher MH, Vipond IB, et al. Epidemiology and cost of nosocomial gastroenteritis, Avon, England, 2002--2003. Emerg Infect Dis 2004;10:1827--34.
-
CDC. Surveillance for foodborne disease outbreaks---United States, 2006. MMWR 2009;58:609--15.
-
CDC. Surveillance for foodborne disease outbreaks---United States, 2007. MMWR 2010;59:973--9.
-
Hall A. Foodborne norovirus outbreaks in the United States: burden and attribution [Presentation]. 4th International Conference on Caliciviruses, Santa Cruz, Chile; October 16--19, 2010.
-
Westrell T, Dusch V, Ethelberg S, et al. Norovirus outbreaks linked to oyster consumption in the United Kingdom, Norway, France, Sweden and Denmark, 2010. Euro Surveill 2010;15(12).
-
Lyman WH, Walsh JF, Kotch JB, Weber DJ, Gunn E, Vinje J. Prospective study of etiologic agents of acute gastroenteritis outbreaks in child care centers. J Pediatr 2009;154:253--7.
-
Sandora TJ, Shih MC, Goldmann DA. Reducing absenteeism from gastrointestinal and respiratory illness in elementary school students: a randomized, controlled trial of an infection-control intervention. Pediatrics 2008;121:e1555--62.
-
Thornton SA, Sherman SS, Farkas T, Zhong W, Torres P, Jiang X. Gastroenteritis in US Marines during Operation Iraqi Freedom. Clin Infect Dis 2005;40:519--25.
-
Bailey MS, Gallimore CI, Lines LD, et al. Viral gastroenteritis outbreaks in deployed British troops during 2002--7. J R Army Med Corps 2008;154:156--9.
-
Cramer EH, Blanton CJ, Blanton LH, Vaughan GH Jr, Bopp CA, Forney DL. Epidemiology of gastroenteritis on cruise ships, 2001--2004. Am J Prev Med 2006;30:252--7.
-
sakbaeva ET, Widdowson MA, Beard RS, et al. Norovirus transmission on cruise ship. Emerg Infect Dis 2005;11:154--8.
-
Widdowson MA, Cramer EH, Hadley L, et al. Outbreaks of acute gastroenteritis on cruise ships and on land: identification of a predominant circulating strain of norovirus---United States, 2002. J Infect Dis 2004;190:27--36.
-
Kirking HL, Cortes J, Burrer S, et al. Likely transmission of norovirus on an airplane, October 2008. Clin Infect Dis 2010;50:1216--21.
-
Kornylo K, Kim DK, Widdowson MA, Turabelidze G, Averhoff FM. Risk of norovirus transmission during air travel. J Travel Med 2009;16:349--51.
-
Katayama K, Shirato-Horikoshi H, Kojima S, et al. Phylogenetic analysis of the complete genome of 18 Norwalk-like viruses. Virology 2002;299:225--39.
-
Kageyama T, Kojima S, Shinohara M, et al. Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. J Clin Microbiol 2003;41:1548--57.
-
Trujillo AA, McCaustland KA, Zheng DP, et al. Use of TaqMan real-time reverse transcription-PCR for rapid detection, quantification, and typing of norovirus. J Clin Microbiol 2006;44:1405--12.
-
Pang XL, Preiksaitis JK, Lee B. Multiplex real time RT-PCR for the detection and quantitation of norovirus genogroups I and II in patients with acute gastroenteritis. J Clin Virol 2005;33:168--71.
-
Jothikumar N, Cromeans TL, Hill VR, Lu X, Sobsey MD, Erdman DD. Quantitative real-time PCR assays for detection of human adenoviruses and identification of serotypes 40 and 41. Appl Environ Microbiol 2005;71:3131--6.
-
Loisy F, Atmar RL, Guillon P, Le Cann P, Pommepuy M, Le Guyader FS. Real-time RT-PCR for norovirus screening in shellfish. J Virol Methods 2005;123:1--7.
-
da Silva AK, Le Saux JC, Parnaudeau S, Pommepuy M, Elimelech M, Le Guyader FS. Evaluation of removal of noroviruses during wastewater treatment, using real-time reverse transcription-PCR: different behaviors of genogroups I and II. Appl Environ Microbiol 2007;73:7891--7.
-
Vinje J, Hamidjaja RA, Sobsey MD. Development and application of a capsid VP1 (region D) based reverse transcription PCR assay for genotyping of genogroup I and II noroviruses. J Virol Methods 2004;116:109--17.
-
Kojima S, Kageyama T, Fukushi S, et al. Genogroup-specific PCR primers for detection of Norwalk-like viruses. J Virol Methods 2002;100:107--14.
-
Okitsu-Negishi S, Okame M, Shimizu Y, et al. Detection of norovirus antigens from recombinant virus-like particles and stool samples by a commercial norovirus enzyme-linked immunosorbent assay kit. J Clin Microbiol 2006;44:3784--6.
-
Burton-MacLeod JA, Kane EM, Beard RS, Hadley LA, Glass RI, Ando T. Evaluation and comparison of two commercial enzyme-linked immunosorbent assay kits for detection of antigenically diverse human noroviruses in stool samples. J Clin Microbiol 2004;42:2587--95.
-
de Bruin E, Duizer E, Vennema H, Koopmans MP. Diagnosis of Norovirus outbreaks by commercial ELISA or RT-PCR. J Virol Methods 2006;137:259--64.
-
Dimitriadis A, Marshall JA. Evaluation of a commercial enzyme immunoassay for detection of norovirus in outbreak specimens. Eur J Clin Microbiol Infect Dis 2005;24:615--8.
-
Richards AF, Lopman B, Gunn A, et al. Evaluation of a commercial ELISA for detecting Norwalk-like virus antigen in faeces. J Clin Virol 2003;26:109--15.
-
Morillo SG, Luchs A, Cilli A, Ribeiro CD, Calux SJ, Carmona RD, Timenetsky MD. Norovirus 3rd generation kit: an improvement for rapid diagnosis of sporadic gastroenteritis cases and valuable for outbreak detection. J Virol Methods 2010 Dec 28 [Epub ahead of print].
-
Wilhelmi de Cal I, Revilla A, del Alamo JM, Roman E, Moreno S, Sanchez-Fauquier A. Evaluation of two commercial enzyme immunoassays for the detection of norovirus in faecal samples from hospitalised children with sporadic acute gastroenteritis. Clin Microbiol Infect 2007;13:341--3.
-
Costantini V, Grenz L, Fritzinger A, et al. Diagnostic accuracy and analytical sensitivity of IDEIA Norovirus assay for routine screening of human norovirus. J Clin Microbiol 2010;48:2770--8.
-
Duizer E, Pielaat A, Vennema H, Kroneman A, Koopmans M. Probabilities in norovirus outbreak diagnosis. J Clin Virol 2007;40:38--42.
-
Smith CM, Hill VR. Dead-end hollow-fiber ultrafiltration for recovery of diverse microbes from water. Appl Environ Microbiol 2009;75:5284--9.
-
Boxman IL, Dijkman R, te Loeke NA, et al. Environmental swabs as a tool in norovirus outbreak investigation, including outbreaks on cruise ships. J Food Prot 2009;72:111--9.
-
Sickbert-Bennett EE, Weber DJ, Gergen-Teague MF, Sobsey MD, Samsa GP, Rutala WA. Comparative efficacy of hand hygiene agents in the reduction of bacteria and viruses. Am J Infect Control 2005;33:67--77.
-
Liu P, Yuen Y, Hsiao HM, Jaykus LA, Moe C. Effectiveness of liquid soap and hand sanitizer against Norwalk virus on contaminated hands. Appl Environ Microbiol 2010;76:394--9.
-
Kampf G, Grotheer D, Steinmann J. Efficacy of three ethanol-based hand rubs against feline calicivirus, a surrogate virus for norovirus. J Hosp Infect 2005;60:144--9.
-
Macinga DR, Sattar SA, Jaykus LA, Arbogast JW. Improved inactivation of nonenveloped enteric viruses and their surrogates by a novel alcohol-based hand sanitizer. Appl Environ Microbiol 2008;74:5047--52.
-
Lages SLS, Ramakrishnan MA, Goyal SM. In-vivo efficacy of hand sanitisers against feline calicirvirus: a surrogate for norovirus. J. Hospital Inf 208;68:159--63.
-
Park GW, Barclay L, Macinga D, Charbonneau D, Pettigrew CA, Vinje J. Comparative efficacy of seven hand sanitizers against murine norovirus, feline calicivirus, and GII.4 norovirus. J Food Prot 2010;73:2232--8.
-
National Advisory Committee on Microbiological Criteria for Foods. Recommendations on bare hand contact with ready-to-eat foods. Washington, DC: Food Safety Inspection Service. Available at http://www.fsis.usda.gov/Frame/FrameRedirect.asp?main=http://www.fsis.usda.gov/OPHS/NACMCF/1999/report_barehand.htm. Accessed February 2, 2011.
-
Harris JP, Lopman BA, O'Brien SJ. Infection control measures for norovirus: a systematic review of outbreaks in semi-enclosed settings. J Hosp Infect 2010;74:1--9.
-
Doultree JC, Druce JD, Birch CJ, Bowden DS, Marshall JA. Inactivation of feline calicivirus, a Norwalk virus surrogate. J Hosp Infect 1999;41:51--7.
-
Duizer, E, Bijkerk P, Rockx B, De Groot A, Twisk F, Koopmans M. Inactivation of caliciviruses. Appl Environ Microbiol 2004;70:4538--43.
-
Girard M, Ngazoa S, Mattison K, Jean J. Attachment of noroviruses to stainless steel and their inactivation using household disinfectants. J Food Prot 2010;73:400--4.
-
Park GW, Boston DM, Kase JA, Sampson MN, Sobsey MD. Evaluation of liquid- and fog-based application of Sterilox hypochlorous acid solution for surface inactivation of human norovirus. Appl Environ Microbiol 2007;73:4463--8.
-
Eleraky NZ, Potgieter LN, Kennedy MA. Virucidal efficacy of four new disinfectants. J Am Anim Hosp Assoc 2002;38:231--4.
-
Gulati BR, Allwood PB, Hedberg CW, Goyal SM. Efficacy of commonly used disinfectants for the inactivation of calicivirus on strawberry, lettuce, and a food-contact surface. J Food Prot 2001;64:1430--4.
-
Jimenez L, Chiang M. Virucidal activity of a quaternary ammonium compound disinfectant against feline calicivirus: a surrogate for norovirus. Am J Infect Control 2006;34:269--73.
-
Cannon JL, Papafragkou E, Park GW, Osborne J, Jaykus LA, Vinje J. Surrogates for the study of norovirus stability and inactivation in the environment: a comparison of murine norovirus and feline calicivirus. J Food Prot 2006;69:2761--5.
-
Park GW, Vinjé J, Sobsey MD. The Comparison of UV and Chlorine Disinfection Profiles of Murine Norovirus (MNV-1), Feline Calicivirus (FCV), and Coliphage MS2 [Presentation]. American Society for Microbiology (ASM), Orlando, Florida; May 21--25, 2006.
-
Park GW, Linden KG, Sobsey MD Inactivation of murine norovirus, feline calicivirus, and echovirus 12 as surrogates for human norovirus (NoV) and coliphage (F+) MS2 by ultraviolet light (254 nm) and the effect of cell association on UV inactivation. Lett Appl Microbiol 2011;56:162--7.
-
CDC. Norovirus in healthcare facilities fact sheet. Atlanta, GA: US Department of Health and Human Services, CDC; 2006. Available at http://www.cdc.gov/ncidod/dvrd/revb/gastro/downloads/noro-hc-facilities-fs-508.pdf.
-
Sattar SA, Springthorpe VS, Adegbunrin O, Zafer AA, Busa M. A disc-based quantitative carrier test method to assess the virucidal activity of chemical germicides. J Virol Methods 2003;112:3--12.
-
Barker, J, Vipond IB, Bloomfield SF. Effects of cleaning and disinfection in reducing the spread of Norovirus contamination via environmental surfaces. J Hosp Infect 2004;58:42--9.
-
Environmental Protection Agency. Initial virucidal effectiveness test: using feline calicivirus as surrogate for norovirus. Arlington, VA: Environmental Protection Agency. Available at http://www.epa.gov/oppad001/pdf_files/initial_virucidal_test.pdf.
-
Baert L, Uyttendaele M, Van Coillie E, Debevere J. The reduction of murine norovirus 1, B. fragilis HSP40 infecting phage B40-8 and E. coli after a mild thermal pasteurization process of raspberry puree. Food Microbiol 2008;25:871--4.
-
Butot S, Putallaz T, Amoroso R, Sanchez G. Inactivation of enteric viruses in minimally processed berries and herbs. Appl Environ Microbiol 2009;75:4155--61.
-
Hewitt J, Rivera-Aban M, Greening GE. Evaluation of murine norovirus as a surrogate for human norovirus and hepatitis A virus in heat inactivation studies. J Appl Microbiol 2009;107:65--71.
-
Topping JR, Schnerr H, Haines J, et al. Temperature inactivation of Feline calicivirus vaccine strain FCV F-9 in comparison with human noroviruses using an RNA exposure assay and reverse transcribed quantitative real-time polymerase chain reaction-A novel method for predicting virus infectivity. J Virol Methods 2009;156:89--95.
-
Hudson JB, Sharma M, Petric M. Inactivation of norovirus by ozone gas in conditions relevant to healthcare. J Hosp Infect 2007;66:40--5.
-
Cho M, Kim JH, VinjéJ, Park GW. Development of fluorinated TiO2 film and evaluation of its potential application to control human norovirus on the environmental surfaces [Presentation]. Meeting of the Association for Professionals in Infection Control and Epidemiology, New Orleans, Louisiana; July 11--15, 2010.
-
Pottage T, Richardson, Parks S, Walker JT, Bennett AM. Evaluation of hydrogen peroxide gaseous disinfection systems to decontaminate viruses. J Hosp Infect 2010;74:55--61.
|
BOX. Key points for investigation and response to norovirus outbreaks |
|---|
|
|
* Agents registered as effective against norovirus by EPA are available at http://www.epa.gov/oppad001/list_g_norovirus.pdf. Evidence for efficacy against norovirus usually is based on studies using feline calicivirus (FCV) as a surrogate. However, FCV and norovirus exhibit different physiochemical properties, and whether inactivation of FCV reflects efficacy against norovirus is unclear. |
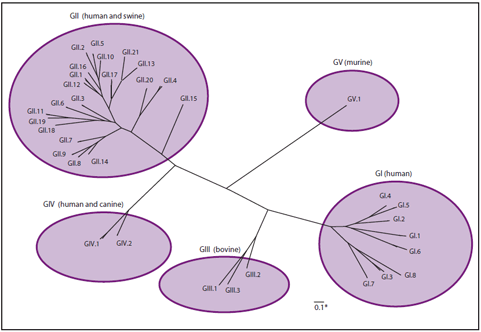
Sources: Data from Zheng DP, Ando T, Fankhauser RL, Beard RS, Glass RI, Monroe SS. Norovirus classification and proposed strain nomenclature. Virology 2006;346:312--23; Wang QH, Han MG, Cheetham S, Souza M, Funk JA, Saif LJ. Porcine noroviruses related to human noroviruses. Emerg Infect Dis 2005;11:1874--81; CDC, unpublished data, 2011; graphic developed by Everardo Vega, PhD, CDC.
* The scale bar of 0.1 reflects the number of amino acid substitutions per site.
Alternate Text: The figure illustrates the classification of noroviruses into 5 genogroups (GI-V) and 35 genotypes.
FIGURE 2. Number* of norovirus outbreaks laboratory-confirmed by CDC, by setting and genotype --- United States, 1994--2006
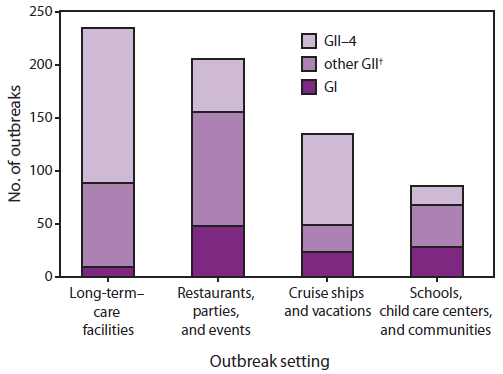
Source: Zheng DP, Widdowson MA, Glass RI, Vinje J. Molecular epidemiology of genogroup II-genotype 4 noroviruses in the United States between 1994 and 2006. J Clin Microbiol 2010;48:168--77.
* N = 660.
† Includes all other genoytpes within genogroup II except genogroup II type 4 (GII.4).
Alternate Text: The figure illustrates the number of norovirus outbreaks laboratory-confirmed by CDC for the United States during 1994-2006, by setting and genotype. The total number of outbreaks shown was 660.Four categories of outbreak settings are shown: 1) long-term-care facilities; 2) restaurants, parties, and events; 3) cruise ships and vacations; and 4) schools, child care centers, and communities.
FIGURE 3. Number* of foodborne disease outbreaks reported to CDC, by etiology --- United States, 2006--2007
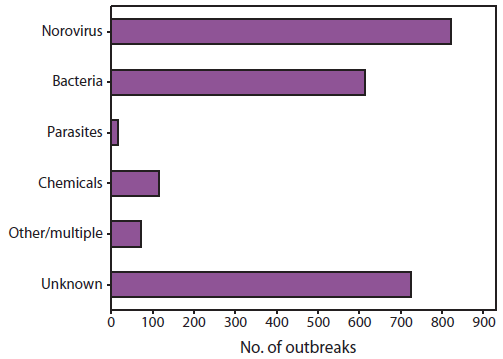
Sources: CDC. Surveillance for foodborne disease outbreaks ---United States, 2006. MMWR 2009;58:609--5; CDC. Surveillance for foodborne disease outbreaks---United States, 2007. MMWR 2010;59:973--9.
* No. = 2,367.
Atlernate Text: The figure illustrates the number of foodborne disease outbreaks reported to CDC for the United States during 2006-2007, by etiology. The total number of outbreaks shown was 2,367. Six etiologic categories are shown: norovirus, bacteria, parasites, chemical, other/multiple, and unknown.
FIGURE 4. Genomic regions targeted by reverse transcription-polymerase chain reaction (RT-PCR) assays used for norovirus detection and genotyping
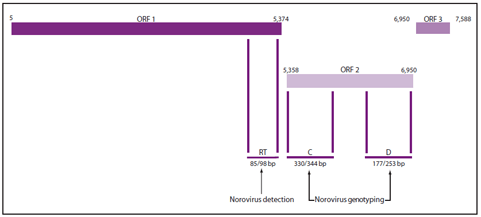
Abbreviations: ORF = open reading frame; RT = TaqMan real-time RT-PCR region for norovirus detection (87); C = region C for norovirus genotyping (93); D = region D for norovirus genotyping (92); and bp = base pairs.
Alternate Text: The figure illustrates the genomic regions targeted by reverse transcription-polymerase chain reaction (RT-PCR) assays used for norovirus detection and typing.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


