NIOSH Respiratory Protective Device Information
Updated June 9, 2021
NIOSH CA 2021-1033R1
June 2021
Since August 24, 2020, the National Institute for Occupational Safety and Health (NIOSH) has accepted and prioritized approval applications for surgical N95 filtering facepiece respirators (surgical N95s), a subset of single-use dispoasable N95 filtering facepiece respirators (FFRs). NIOSH-approved N95 FFRs are also regulated by the Food and Drug Administration (FDA) as class II devices under 21 CFR § 878.4040, classified as product code MSH, and commonly referred to as surgical N95s. This notice is intended to inform users about a subset of surgical N95s, exempted by regulation from the FDA’s premarket notification (510(k)) requirements. This subset of NIOSH Approved surgical N95s are easily identified by the NIOSH approval labels, see Figures 1 and 2.
NIOSH-approved surgical N95s offer the same level of respiratory protection as a NIOSH-approved N95 FFR. Additionally, the surgical N95 has demonstrated conformance to FDA specified flammability, fluid resistance, and biocompatibility requirements and is intended for use in all healthcare settings and for all medical purposes in which an N95 level of respiratory protection is needed to protect the wearer.
Users can recognize a NIOSH-approved surgical N95 by the abbreviated label, which is printed on each respirator. The surgical N95 abbreviated label includes the name of the approval holder (i.e., name of the company that was issued NIOSH approval), the model or part number, the approval number assigned by NIOSH (e.g., TC-84A-XXXX), “Surgical N95 Respirator,” “NIOSH,” and the lot number. An example of an abbreviated label on a NIOSH-approved surgical N95 is shown in Figure 1 below.
The full NIOSH approval label, which is found on or within the respirator packaging, will indicate the protection type as being “Surgical/N95.” An example of a NIOSH surgical N95 approval label is shown in Figure 2 below. The approval label will have the caution and limitation “S – Special or Critical User Instructions and/or specific use limitations apply. Refer to User Instructions before donning.” The “S” caution and limitation will correspond to a section in the User Instructions that indicates, this respirator has been approved as a NIOSH N95 filtering facepiece respirator, for use in healthcare settings, as a Surgical N95 Respirator conforming to recognized standards for biocompatibility, flammability, and fluid resistance.
NIOSH-approved surgical N95s can be identified on the NIOSH Certified Equipment List (CEL) and can be searched using the term “surgical.”
In addition to surgical N95s approved under the process described above, users can select and use a surgical N95 FFR that previously received NIOSH approval and FDA 510(k) clearance. The User Instructions and packaging will include the caution and limitation “P – NIOSH does not evaluate respirators for use as surgical masks.” The abbreviated label, which is printed on the respirator, will also not include “surgical.” However, NIOSH can approve an approval holder’s updated User Instructions and packaging to remove the caution and limitation “P” and include caution and limitation “S.” The abbreviated label can also be updated to include “surgical.” It is expected that eventually all Surgical N95s will have consistent labeling and packaging as approval holders submit updates to NIOSH.
Surgical N95s exempt from the 510(k) premarket notification requirements must comply with applicable FDA labeling requirements and all other statutory and regulatory requirements. When the labeling and packaging is updated, these surgical N95 FFRs can also be found on the CEL by using the term “surgical.”
Figure 1: Example of an abbreviated label on a NIOSH Approved surgical N95
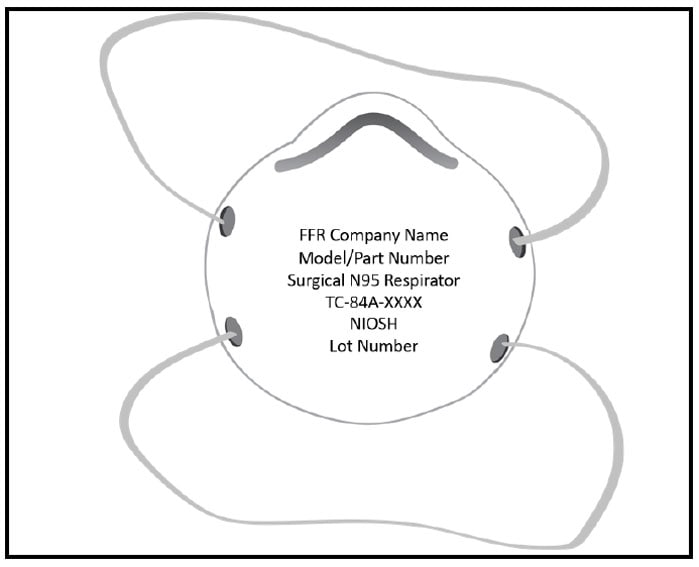
Figure 2: Example of a NIOSH surgical N95 full respirator approval label, often located on or within the packaging.

REFERENCES:
FDA Final Order
FDA Postmarket Requirements (Devices)
Approval of Respiratory Protective Devices, 42 CFR Part 84
MOU 225-18-006
NIOSH Conformity Assessment Letter to Manufacturers (CA 2018-1010R1) | NPPTL | NIOSH | CDC
NIOSH Conformity Assessment Letter to Manufacturers (CA 2020-1031) | NPPTL | NIOSH | CDC
| Revision (R) | Date | Reason for Revision |
|---|---|---|
| 1.0 | 12 May 2021 | NIOSH revised this notice based on HHS review and comments. |