HIV Pre-exposure Prophylaxis (PrEP) Care System
Pre-exposure Prophylaxis (PrEP) delivery in the United States includes a system of prevention and support services, a PrEP care system, that contributes to making PrEP available, accessible, and acceptable.
About PrEP
PrEP is a medication that users take daily to lower their chances of getting HIV.
The Centers for Disease Control and Prevention (CDC) recommends PrEP as an HIV prevention strategy. Taking PrEP medication as prescribed reduces the risk of getting HIV via sexual contact by about 99% and reduces the risk of getting HIV by at least 74% among persons who inject drugs.
Two oral medications, emtricitabine and tenofovir disoproxil fumarate (Truvada®) and emtricitabine and tenofovir alafenamide or (Descovy®) are currently approved for daily use as HIV PrEP. Multiple generic formulations of tenofovir disoproxil fumarate/emtricitabine—the equivalent of Truvada—are now available in the United States for HIV prevention with PrEP. In addition to the oral medications already approved, in December 2021, the FDA approved APRETUDE (cabotegravir extended-release injectable suspension), for intramuscular use as HIV PrEP.
PrEP Delivery System: Clinical Care and Public Health Collaborative Care Models
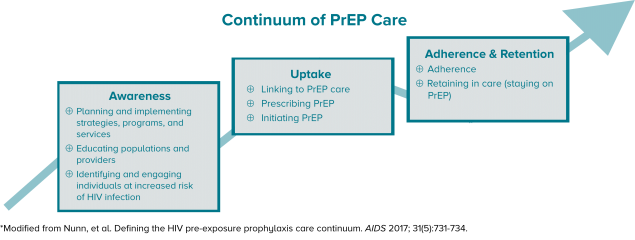
Health departments, community-based organizations (CBOs), and clinics can form partnerships as part of a comprehensive PrEP Care System to make PrEP available, accessible, and acceptable. A continuum of PrEP care, like the one below*, provides a framework for the steps needed for population health and prevention with PrEP.
⇓ Click image to enlarge ⇓
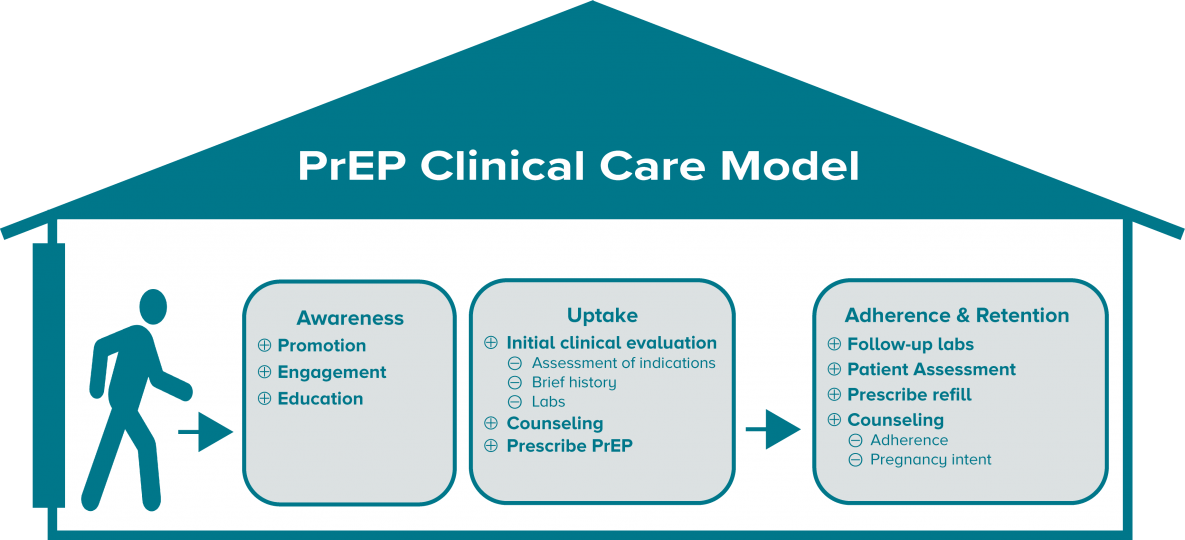
I. PrEP Clinical Care Model
The following graphic shows the essential elements and the basic flow of PrEP care in a clinic. PrEP clinical care includes identifying and engaging patients in need of PrEP, conducting necessary exams and lab tests and prescribing PrEP for the patients, as well as ongoing patient monitoring with follow-up visits and prescriptions—for as long as the patient needs PrEP.
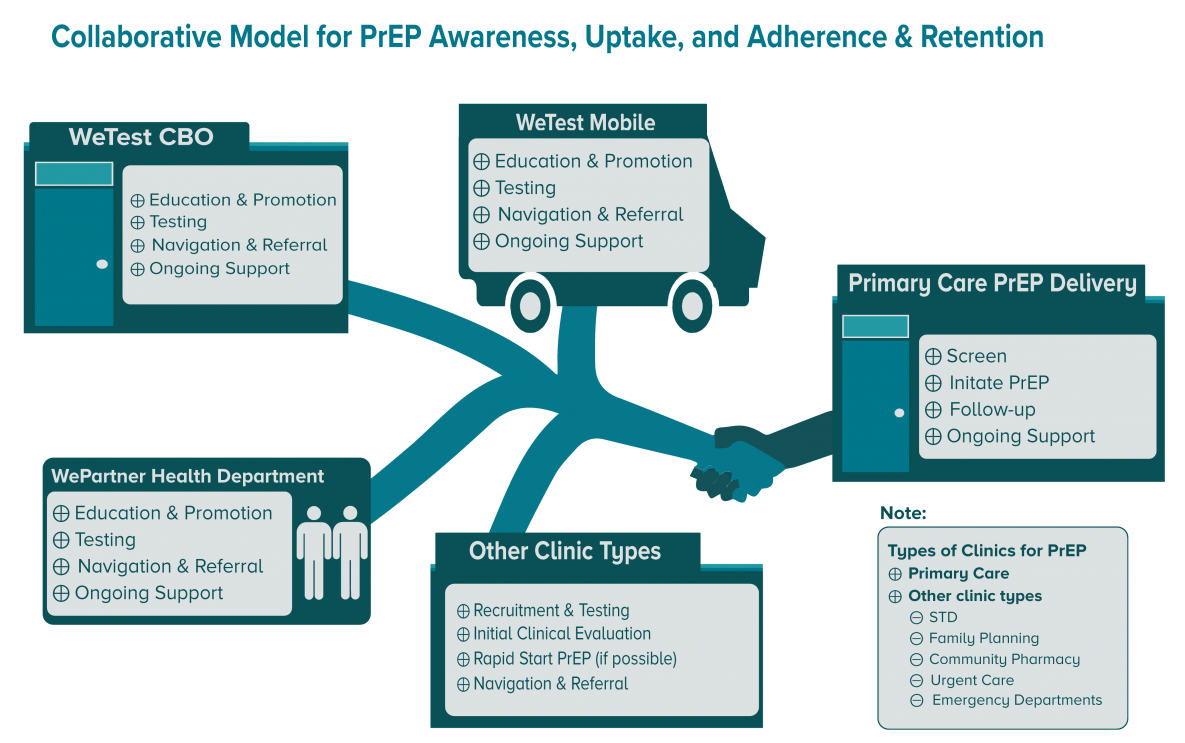
II. Collaborative Model
Health departments fund, guide, and lead collaborative planning and delivery of health protective measures—like PrEP. A public health model for prevention with PrEP includes national, state, and local levels of public health collaborating with and supporting clinics and nonclinical CBOs to ensure that PrEP is available, accessible, and acceptable.
III. Public Health Resources for PrEP: Awareness, Uptake, and Adherence & Retention
Increase Consumer and Provider Knowledge
Community-wide education on PrEP happens in a variety of complementary ways, including:
- Visuals, brochures, and pamphlets;
- Media campaigns, including the use of social media;
- Radio and television public service announcements; and
- Partnering with community members, key stakeholders, and peers to build trust and credibility.
Some examples include:
- Let’s Stop HIV Together-PrEP
Includes a PrEP brochure, graphic button with link, and an interactive widget to locate HIV Prevention Services that can be embedded in your website. - Building Healthy Online Communities (BHOC)
BHOC is intended to help providers conduct HIV prevention online. The site contains useful resources like a clearinghouse of PrEP publicity campaigns.
There are several community-level interventions that use effective strategies for promoting PrEP.
- PROMISE for HIP promotes PrEP by creating and distributing community- and population-specific role model stories to spread educational messages and modeling community member experiences and endorsements.
- d-up: Defend Yourself! is based on a cultural adaptation of the Popular Opinion (POL) intervention and is intended to reach black gay, bisexual, and other men who have sex with men (MSM). d-up: Defend Yourself! includes skills building for coping with community- and individual-level stigma.
Enhance Provider Knowledge and Support of PrEP
Health departments can develop their own PrEP educational materials and provide training for clinicians.
- HIV NEXUS- CDC Resources for Clinicians are appropriate for clinical detailing or for use in clinician education.
Identification, Recruitment and Navigation of People in Need of PrEP
Persons at risk for HIV can be identified in clinics, through outreach, community testing, and other community program services and interventions. Integrating PrEP education and referrals into program services is an effective way to promote PrEP to likely candidates.
Testing and risk assessment resources
- HIV Testing in Nonclinical Settings- Program guidance for HIV testing providers
- Discussing Sexual Health with Patients (brochure)
Uptake
Linkage and Navigation
Those at risk of HIV often encounter barriers, such as stigma, medical mistrust, and perceived payment barriers, that prevent them from receiving health services. PrEP navigation includes identifying and linking persons in need of PrEP-to-PrEP care, assisting with health insurance, identifying and reducing barriers to care, and tailoring education to the client to influence his or her health-related attitudes and behaviors. This may also include retention—supporting the person to stay on PrEP.
CBOs that do not provide clinical services may develop working relationships with PrEP clinicians to facilitate linkage of community members to PrEP services. Clinical programs may also involve staff from CBOs to help to support PrEP patients. Working relationships with behavioral health clinics, substance abuse treatment clinics, and other clinical service providers may be beneficial for providing services to persons in need of PrEP.
- PrEP Clinic Locator Find PrEP providers whether insured or uninsured.
- Find a Community Health Center Use this HRSA locator to find local health centers.
- The Toolkit for Technology-based Partner Services The toolkit serves as a general resource for health departments, community-based organizations, and others on how to use the internet and other technologies to trace and contact persons potentially exposed to STIs including HIV.
Clinical/Prescribing PrEP
- The U.S. Public Health Service’s Clinical Practice Guideline [PDF – 2 MB], updated in 2021, provides the framework for clinical prevention with PrEP. Also see: Clinical Provider’s Supplement, 2021 [PDF – 809 KB].
- National Clinician Consultation Center for PrEP. National Clinician Consultation Center provides direct guidance on all aspects of PrEP management. This service includes guidance on assessing indications for PrEP, prescribing practices, PrEP-related laboratory protocols, and assistance with adherence issues. Via telephone: The PrEP Warmline: (855) 448-7737 or (855) HIV-PrEP, Monday – Friday, 9 a.m.- 8 p.m. EST.
- Materials for Your Practice and Patients (HIV NEXUS- CDC Resources for Clinicians)
- Screening for HIV
- Telehealth for HIV Prevention and Care Services
Financing PrEP
Jurisdictions may have local funds and leverage existing patient assistance program resources to establish their own PrEP patient assistance program. Providers may be able to leverage other public health resources such as:
- 340B Drug Pricing Program
- Diversifying PrEP Financing: Strategies to Leverage Funding across the PrEP Care Continuum
PrEP candidates may need assistance with transportation, communication with clinicians, and other services, such as navigating payment coverage. Here are some resources to help with the cost of PrEP:
Continuing Education (CE)
elearning modules on CDC TRAIN
- HIV PrEP: Engaging Patients in U.S. Clinics (TRAIN Course ID 1085296)
- HIV PrEP: Prescribing PrEP (TRAIN Course ID 1087417)
- HIV PrEP: PrEP and Pregnancy (TRAIN Course ID 1086182)
Classroom (or virtual, synchronous learning) on CDC TRAIN
- Implementing PrEP in Your Clinic: A Course for Decision Makers(TRAIN Course ID 1095262)
Adherence & Persistence
PrEP users should be provided all the information and support needed to ensure they take PrEP daily as directed. Health departments can support clinics and CBOs with adherence-related activities by providing PrEP education to clinical providers and providing resources for patients.
Compendium of Evidence-Based Interventions and Best Practices for HIV Prevention- PrEP Chapter
Continuing Education (CE)
eLearning modules on CDC TRAIN
- Strengthening Strategies that Support PrEP Adherence (TRAIN Course ID 1105465)
Training and Continuing Education Online (TCEO)
A comprehensive directory of HIV and other health services should support navigation and guidance of clients. The health department, local CBOs, or jurisdictional planning groups may have created such a directory. If none are available in your jurisdiction, you may want to create one that includes online resources, programs, and services. Compiling, using, and maintaining a directory for PrEP providers and PrEP-related services is a great way to provide information to the community and collaborate with others on prevention with PrEP.
PrEP Training
To view and register for scheduled virtual, instructor-led trainings:
- Access the National HIV Classroom Learning Center training calendar hosted on Cicatelli Associates, Inc.’s (CAI’s) website.
- Pre-register for your selected course via the link provided in the training calendar.
- Complete your registration on CDC TRAIN as directed when you receive an email from the National HIV Classroom Learning Center. You must join the HIV CBA Learning Group and locate the HIV CBA Training Plan in order to complete your CDC TRAIN registration for a specific classroom session (step-by-step instructions are available).
To access eLearning modules, including training prerequisite courses:
- Log-in to CDC TRAIN and access the HIV CBA Training Plan (step-by-step instructions are available).
- Select the module you wish to take.
- Launch the module or save the module for later.
To request technical assistance or that a training session be scheduled:
- CDC’s directly funded health department and CBO partners may request delivery of technical assistance or a CDC-supported training by submitting a request in the CBA Tracking System.
- Organizations not directly funded by CDC may contact their local health department for assistance in submitting a technical assistance or training request.
If you have questions or need additional assistance, please contact HIVCBA@cdc.gov.
- Centers for Disease Control and Prevention. U.S. Public Health Service: Preexposure prophylaxis for the prevention of HIV infection in the United States—2021 Update: A Clinical Practice Guideline. Published 2021.