Information for Health Professionals
Clinical Features
If you suspect your patient may have botulism, call your state public health department immediately. If there is no answer, contact CDC 24/7 at 770-488-7100.
- For non-infant cases: State public health officials can reach the CDC clinical emergency botulism service for consultation and antitoxin 24/7 at 770-488-7100.
- For infant botulism: The Infant Botulism Treatment and Prevention Program (IBTPP) at the California Department of Public Health provides consultation 24/7 and can be reached at 510-231-7600.
If clinical consultation supports botulism, request antitoxin immediately and begin treatment as soon as it is available. Do not wait for laboratory confirmation. If administered early in the course of illness, antitoxin can prevent progression of illness and shorten its duration.
See step-by-step guidance for clinicians of patients who may have infant botulism.

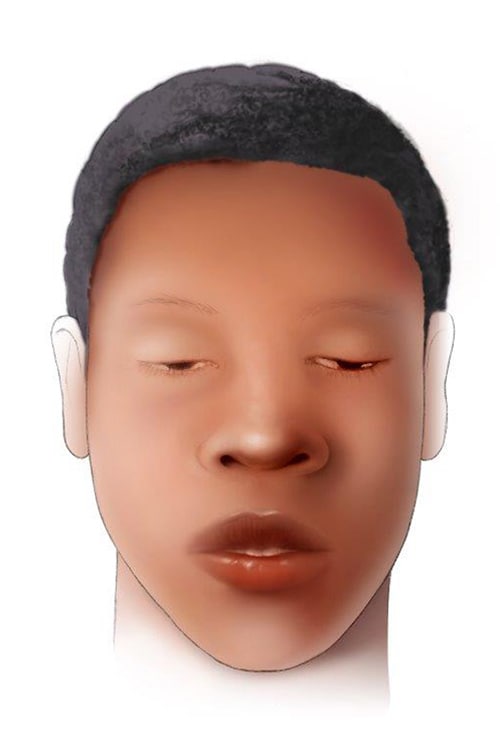
Botulism is a neuroparalytic illness characterized by symmetric, descending flaccid paralysis of motor and autonomic nerves, always beginning with the cranial nerves.
Signs and symptoms may include:
- Dysphagia
- Muscle weakness
- Diplopia
- Ptosis
- Blurry vision
- Slurred speech
- Respiratory distress or failure
- Ocular palsy
Possible signs and symptoms in foodborne botulism may also include:
- Vomiting
- Nausea
- Abdominal pain
- Diarrhea
Signs and symptoms in an infant may include:
- Constipation
- Poor feeding
- Ptosis
- Sluggish pupils
- Flattened facial expression
- Diminished suck and gag reflexes
- Weak and altered cry
- Respiratory distress or failure
If untreated, illness from any type of botulism can progress to descending paralysis of respiratory muscles, arms, and legs.
Diagnosis
Initial diagnosis is based on clinical symptoms. Do not wait for laboratory confirmation to begin treatment.
Botulism differs from other flaccid paralyses in that it typically manifests initially with prominent cranial nerve palsies. It also differs in its invariable descending progression, in its symmetry, and in its absence of sensory nerve dysfunction.
Botulism is frequently misdiagnosed, most often as a polyradiculoneuropathy (Guillain-Barré or Miller-Fisher syndrome), myasthenia gravis, or other diseases of the central nervous system.
Differential Diagnoses
- Bacterial or chemical food poisoning
- Cerebrovascular accident (CVA)
- Chemical intoxication
(e.g., carbon monoxide, opioid intoxication) - Congenital myopathy
- Electrolyte-mineral imbalance
- Guillain-Barré syndrome
- Lambert-Eaton myasthenic syndrome
- Leigh syndrome
- Meningitis
- Miller-Fisher syndrome
- Mushroom poisoning
- Myasthenia gravis
- Poliomyelitis
- Reye’s syndrome
- Sepsis
- Tick paralysis
- Werdnig-Hoffman disease
- West Nile Virus
Routine laboratory test results are usually unremarkable for people with botulism. A normal Tensilon test helps to differentiate botulism from myasthenia gravis; borderline positive tests can occur in botulism. A normal CT or MRI helps to rule out cerebrovascular accident.
Treatment
If consultation supports botulism, request treatment and administer it as soon as possible. Botulism Antitoxin Heptavalent and BabyBIG do not reverse paralysis but arrest its progression. Recovery follows the regeneration of new neuromuscular connections.
Exercise meticulous intensive care, including monitoring of respiratory function and, when required, mechanical ventilation. In more severe cases, ventilator support may be required for weeks to months.
Treatment for wound botulism may also include wound debridement to remove the source of toxin‑producing bacteria and antibiotic therapy.
Diagnostic Testing
Laboratory confirmation is done by demonstrating the presence of botulinum toxin in serum, stool, or food, or by culturing botulinum neurotoxin-producing species of Clostridium (C. botulinum, C. butyricum, or C. baratii) from stool or a wound.
Diagnostic testing is done through the state public health department’s laboratory. CDC provides testing services for some state public health departments.
This specialized testing often takes days to complete. Follow up with your state health department if you do not receive results within 5 days.
See information on submitting a specimen to CDC >
Sequelae
Death can result from respiratory failure or the consequences of extended paralysis. About 5% of patients die. Recovery takes weeks to months. Those who survive may have fatigue and shortness of breath for years.
Notification and Reporting
Botulism is a notifiable disease in the United States. Physicians must promptly notify the state health department of suspected cases, and laboratories must notify the state health department of all confirmed cases.
State health departments report confirmed cases to CDC through the National Notifiable Diseases Surveillance System.
Clinical Guidelines for Diagnosis and Treatment of Botulism, 2021
These are the first comprehensive clinical care guidelines for botulism. They include
- Best practices for diagnosing, monitoring, and treating botulism, including during outbreaks
- Special considerations for infants, children, and people who are pregnant or breastfeeding
- Recommendations for supportive care, including psychosocial support for patients and family members

“Botulism,” a CDC-sponsored supplement to Clinical Infectious Diseases
The supplement includes six systematic reviews and other articles that provided the evidence base for the clinical guidelines. Contents include reviews and articles on
- Clinical presentation
- Effectiveness of antitoxin
- Antitoxin adverse reactions
- Outbreaks
- And more