Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Expanding Poliomyelitis and Measles Surveillance Networks to Establish Surveillance for Acute Meningitis and Encephalitis Syndromes — Bangladesh, China, and India, 2006–2008
Introduction
Quality surveillance is critical to the control and elimination of vaccine-preventable diseases (VPDs). A key strategy for enhancing VPD surveillance, outlined in the World Health Organization (WHO) Global Framework for Immunization Monitoring and Surveillance (GFIMS) (1), is to expand and link existing VPD surveillance systems (particularly those developed for polio eradication and measles elimination) to include other priority VPDs. Since the launch of the Global Polio Eradication Initiative in 1988, the incidence of polio has decrease by 99% worldwide (2). A cornerstone of this success is a sensitive surveillance system based on the rapid and timely reporting of all acute flaccid paralysis (AFP) cases in children aged <15 years, with confirmatory diagnostic testing performed by laboratories that are part of a global network. As countries achieve polio-free status, many have expanded syndromic surveillance to include persons with rash and fever, and have built measles diagnostic capacity in existing polio reference laboratories. Acute meningitis/encephalitis syndrome (AMES)* and acute encephalitis syndrome (AES)† are candidates for expanded surveillance because they are most often caused by VPDs of public health importance for which confirmatory laboratory tests exist. Vaccine-preventable cases of encephalitis include approximately 68,000 Japanese encephalitis (JE) cases, resulting in 13,000–20,000 deaths each year in Asia (3). Moreover, although bacterial meningitis incidence in Asia is not as well-documented, pneumococcal and meningococcal meningitis outbreaks have been reported in Bangladesh (4) and China (5), and the incidence of Haemophilus influenzae type b (Hib) meningitis in children aged <5 years in India has been estimated to be 7.1 per 100,000 population, similar to that in European countries before the introduction of vaccine (6). This report describes a prototype for expanding existing polio and measles surveillance networks in Bangladesh, China, and India to include surveillance for viral and bacterial vaccine-preventable causes of AMES and AES and presents data from 2006–2008.
Background
AMES and AES surveillance rely on identification of persons presenting with a clinically compatible syndrome, collection and testing of specimens, and laboratory confirmation (7,8). During 2006–2008, Bangladesh and China introduced AMES surveillance, and India introduced AES surveillance. In all three countries, surveillance was initiated in areas with well-established AFP and rash/fever surveillance systems, high AFP performance indicators, no endemic polio transmission,§ and expressed interest by their ministries of health (MoHs) to introduce AMES/AES surveillance.
Implementation
Active AMES/AES surveillance was established at sentinel hospitals in three districts of Bangladesh (three sites), four prefectures in four provinces of China (24 sites), and four states of India (four sites). Case investigations were conducted by polio and hospital surveillance medical officers (Bangladesh and India) and VPD surveillance staff (China). Blood and cerebrospinal fluid (CSF) were collected from patients at sentinel sites who had an illness that met the clinical case definition. Case investigation data were entered into standardized electronic data management systems that were developed separately for each implementing country. Every month, summary results were reported by the respective national program office to the MoH, which provided feedback to sentinel sites.
The laboratory methods, staff, and equipment needed for JE diagnosis were similar to those used for measles testing, and JE testing was conducted by the global polio/measles network laboratories and staff members. JE was diagnosed by detecting anti-JE virus immunoglobulin M (IgM) in CSF or serum by IgM-capture enzyme-linked immunosorbent assay at the National Institute of Public Health (Bangladesh), the Chinese Center for Disease Control and Prevention (CCDC) prefectural laboratories (China), and sentinel hospital laboratories (India).
Because considerable personnel, procedural, and specimen-processing differences exist between indirect viral assays and bacterial cultures, bacterial testing was difficult to establish in the polio/measles viral laboratories. Bacterial culture, Gram stain, and latex agglutination (LA) were performed at sentinel hospital laboratories in Bangladesh and China for bacterial (i.e., Neisseria meningitidis, Streptococcus pneumoniae, and Hib) meningitis etiologies (Figure). When specimens were adequate, real-time polymerase chain reaction (rt-PCR) for Hib, S. pneumoniae, and N. meningitidis was performed on CSF or serum (or both) at CDC (Bangladesh) and on CSF at the CCDC provincial laboratories (China). CDC provided training, reference strains, and proficiency panel testing for quality assurance and quality control (QA/QC).
During 2006–2008, a total of 4,197 AMES/AES patients were reported from Bangladesh (n = 632), China (n = 2,815) and India (n = 750). For ≥90% of these, specimens were tested in the AMES/AES laboratory network (Table 1).
Comment
Field surveillance, including case investigations, was well integrated into existing polio and measles surveillance activities, which historically have been conducted through the immunization program in Bangladesh, China, and India. Other MoH departments had responsibility for JE surveillance (Bangladesh and India) and bacterial meningitis surveillance (Bangladesh, China, and India).
AMES/AES viral laboratory testing for JE was integrated successfully into the polio and measles networks in all three countries. However, development of capacity for bacterial meningitis diagnosis proved to be more challenging (Table 2). Because hospitals have primary responsibility for conducting bacterial testing to determine appropriate treatment, hospital laboratories routinely process blood and CSF for bacterial culture, whereas specimens for viral testing are sent to a surveillance reference laboratory (Figure). In many settings, CSF specimens were not collected or were collected after initiation of antibiotic therapy, and delays in processing, storage, and shipping affected culture results. In addition, although standard operating and QA/QC procedures exist for the polio and measles laboratory network, at the time of this activity, there were no established WHO-sponsored networks with standard operating and QA/QC procedures or an accreditation process for laboratories diagnosing bacterial diseases. Because hospital and public health bacteriology laboratories usually fall under a different jurisdiction than polio and measles surveillance reference viral laboratories, developing capacity in these laboratories requires building new relationships at national, provincial, and local levels. National bacteriology laboratories have not received the same level of attention and resources as have global surveillance networks for polio and measles.
WHO's global invasive bacterial vaccine-preventable disease (IB-VPD) surveillance and laboratory network was established in 2008 and is an important step towards providing the needed support, standardization, and quality assurance for bacterial testing in participating countries.
To enhance detection of bacterial meningitis, efforts were made to standardize laboratory quality at the sentinel hospital laboratories and to establish standard operating procedures, QA/QC procedures, and reference testing for laboratory diagnosis of bacterial diseases. To compensate for the limitations of bacterial culture, where resources are available, rt-PCR can be used to enhance the sensitivity of laboratory-supported bacterial meningitis surveillance (9).
Funding for surveillance often is disease-specific and time-limited, and can result in multiple, parallel surveillance systems that compete for resources, are not adequately funded, and are not sustainable. As the number of diseases targeted by immunization increases, the need for integrated surveillance systems also will increase (10). This effort to introduce AMES/AES surveillance was funded by CDC's Division of Global Disease Detection and Emergency Response.
This surveillance project represents the first effort to integrate surveillance for encephalitis and meningitis at the field and laboratory levels, capitalizes on the existing infrastructure and international investment in polio and measles surveillance, and should be considered one approach to implementing GFIMS. In China, the MoH assumed full funding of the project in 2010 and has been sustaining AMES surveillance in four provinces since then. Since 2010, Bangladesh, using local and external resources, has expanded AMES surveillance to include an additional sentinel site, for a total of four sentinel sites. All of India's AES surveillance sites have been sustained with local resources. Additionally, these sentinel sites are being integrated into the IB-VPD network as feasible. Lessons learned from this effort to integrate AMES/AES surveillance into existing VPD surveillance can inform planned integration programs in other areas. Successful implementation of GIFMS depends upon development of best practices, which can be applied to other integrated VPD surveillance projects.
Reported by
National Immunization Program, Institute for Viral Disease Control, Institute for Communicable Disease Control, Chinese Center for Disease Control and Prevention; Expanded Programme on Immunization, World Health Organization, China. National Vector Borne Disease Control Programme, Ministry of Health and Family Welfare; National Institute of Mental Health and Neurosciences; National Polio Surveillance Project, World Health Organization, New Delhi; Expanded Programme on Immunization, Immunization and Vaccine Development, Family and Health Research Dept, World Health Organization Regional Office for South-East Asia, New Delhi, India. Institute for Epidemiology, Disease Control, and Research, Institute of Public Health, Ministry of Health and Family Welfare, Bangladesh. International Centre for Diarrheal Diseases Research; Immunization and Vaccine Development, World Health Organization, Bangladesh. Expanded Programme on Immunization, World Health Organization Regional Office for the Western Pacific, Philippines. Expanded Programme on Immunization, World Health Organization, Switzerland. Div of Vector-Borne Diseases, National Center for Emerging and Zoonotic Diseases; Div of Bacterial Diseases, Div of Viral Diseases, National Center for Immunization and Respiratory Diseases; Global Immunization Division, Centre for Global Health, CDC. Corresponding contributor: Hardeep S. Sandhu, hsandhu@cdc.gov, 404-639-8976.
References
- World Health Organization. Global Framework for Immunization Monitoring and Surveillance. Geneva, Switzerland: World Health Organization; 2007. Available at http://whqlibdoc.who.int/hq/2007/who_ivb_07.06_eng.pdf. Accessed December 5, 2012.
- World Health Assembly. Global eradication of poliomyelitis by the year 2000. Resolution WHA41.28. Geneva, Switzerland: World Health Organization; 1988. Available at http://www.who.int/ihr/polioresolution4128en.pdf. Accessed December 12, 2012.
- Campbell GL, Hills SL, Fischer M, et al. Estimated global incidence of Japanese encephalitis: a systematic review. Bull World Health Organ 2011;89:766–74.
- Gurley ES, Hossain MJ, Montgomery SP, et al. Etiologies of bacterial meningitis in Bangladesh: results from a hospital-based study. Am J Trop Med Hyg 2009;81:475–83.
- Shao Z, Li W, Ren J, et al. Identification of a new Neisseria meningitidis serogroup C clone from Anhui province, China. Lancet 2006;367:419–23.
- Minz S, Balraj V, Lalitha MK, et al. Incidence of Haemophilus influenzae type b meningitis in India. Indian J Med Res 2008;128:57–64.
- World Health Organization. Polio laboratory network. Geneva, Switzerland: World Health Organization; 2012. Available at http://www.who.int/immunization_monitoring/laboratory_polio/en/index.html. Accessed December 5, 2012.
- World Health Organization. Measles and rubella laboratory network. Geneva, Switzerland: World Health Organization; 2012. Available at http://www.who.int/immunization_monitoring/laboratory_measles/en. Accessed December 5, 2012.
- World Health Organization. Global invasive bacterial vaccine preventable diseases (IB-VPD) information and surveillance bulletin, January–June 2011. Vol 5. Geneva, Switzerland: World Health Organization; 2012. Available at http://www.who.int/nuvi/surveillance/IB_VPD_bulletin_Jan_June_2011_Final.pdf. Accessed December 5, 2012.
- Dabbagh A, Eggers R, Cochi S, Dietz V, Strebel P, Cherian T. A new global framework for immunization monitoring and surveillance. Bull World Health Organ 2007;85:904–5.
* An acute febrile illness with at least one of the following: altered mental status, new-onset seizures, or signs of meningeal irritation in a person of any age at any time of year.
† An acute febrile illness with at least one of the following: altered mental status or new-onset seizures in a person of any age at any time of year.
§ Endemic polio transmission was occurring in India; however, the states selected for AES surveillance had no ongoing transmission.
FIGURE. Functions of various components of AMES/AES surveillance and laboratory networks — China, Bangladesh, and India, 2006–2008
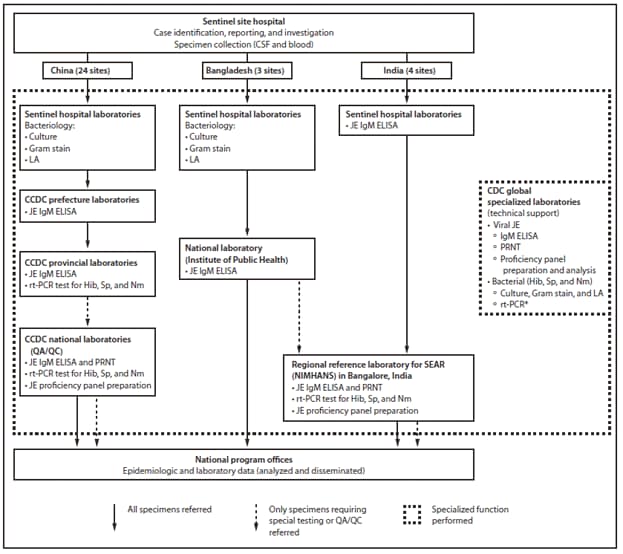
Abbreviations: AMES = acute meningitis/encephalitis syndrome; AES = acute encephalitis syndrome; CSF = cerebrospinal fluid; LA = latex agglutination; JE = Japanese encephalitis; IgM = immunoglobulin M; ELISA = enzyme-linked immunosorbent assay; CCDC = Chinese Center for Disease Control; rt-PCR = real-time polymerase chain reaction; Hib = Haemophilus influenzae type b; Sp = Streptococcus pneumoniae; Nm = Neisseria meningitidis; PRNT = plaque reduction neutralization test; QA/QC = quality assurance/quality control; WHO SEAR = World Health Organization South-East Asia Region; NIMHANS = National Institute of Mental Health and Neurosciences.
* CDC performed rt-PCR on CSF/serum for detection of Hib, Sp, and Nm for Bangladesh.
Alternate Text: The figure above shows functions of various components of acute meningitis/encephalitis syndrome (AMES/AES) surveillance and laboratory networks in China, Bangladesh, and India during 2006-2008. Because considerable personnel, procedural, and specimen-processing differences exist between indirect viral assays and bacterial cultures, bacterial testing was difficult to establish in the polio/measles viral laboratories. Bacterial culture, Gram stain, and latex agglutination (LA) were performed at sentinel hospital laboratories in Bangladesh and China for bacterial (i.e., Neisseria meningitidis, Streptococcus pneumoniae, and Hib) meningitis etiologies.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


