* Available at http://www.searo.who.int/linkfiles/diarrhoea,_ari_and_hepatitis_hepatitis-wha63.pdf.
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Progress Toward Prevention and Control of Hepatitis C Virus Infection — Egypt, 2001–2012
Worldwide, 130–170 million persons are living with chronic hepatitis C virus (HCV) infection (1), which, if left untreated, can result in cirrhosis and liver cancer. Egypt has the largest burden of HCV infection in the world, with a 10% prevalence of chronic HCV infection among persons aged 15–59 years (2). HCV transmission in Egypt is associated primarily with inadequate infection control during medical and dental care procedures (3,4). In response, the Egyptian Ministry of Health and Population (MOHP) in 2001 implemented a program to reduce health-care–associated HCV transmission and in 2008 launched a program to provide care and treatment. This report describes the progress of these programs, identifies deficiencies, and recommends enhancements, including the establishment of a comprehensive national viral hepatitis control program. Infection control programs implemented in 2001 at MOHP facilities resulted in improvements in infection control practices and a decrease in the annual incidence of HCV infection among dialysis patients from 28% to 6%. Through June 2012, a total of 23 hepatitis treatment facilities had been established in Egypt, providing care and treatment to nearly 190,000 persons with chronic HCV infection. Despite these programs, Egypt continues to face an ongoing hepatitis C epidemic. A comprehensive plan is needed to prevent and control hepatitis C in Egypt. This plan should address increasing community awareness and education, preventing of HCV infection in health-care settings, ensuring a safe blood supply, establishing surveillance and monitoring to track the effectiveness of control programs, and providing care and treatment.
Epidemiology of HCV infection in Egypt
The hepatitis C epidemic in Egypt began during 1960–1980, when mass campaigns were conducted to control schistosomiasis through parenteral antischistosomal therapy (PAT) administered by health-care workers using improperly sterilized glass syringes (5). HCV transmission is ongoing in Egypt, and incidence rates have been estimated at 2.4 per 1,000 person-years (165,000 new infections annually) (6). In 2008, nearly 15% of the population aged 15–59 years had antibodies to HCV (anti-HCV), and 10% (approximately 5 million persons) had chronic HCV infection (2); overall, an estimated 6 million Egyptians had chronic HCV infection in 2008. Prevalence of chronic HCV infection in Egypt is higher among men than women (12% and 8%, respectively), increases with age (reaching >25% among persons aged >50 years), and is higher among persons residing in rural versus urban areas (12% versus 7%) (2). Primary modes of HCV transmission include unsafe injections, other inadequate infection control practices, and unsafe blood transfusions (4,6). HCV transmission also occurs among injection-drug users in Egypt (3).
National Infection Control Program
Approximately 280 million injections were administered in Egypt during 2001, of which an estimated 8% (23 million) might have been unsafe (7). In response, MOHP launched an infection control program that year to promote safe health care in hospitals and health facilities throughout Egypt (8). A baseline assessment of MOHP facilities revealed 1) a lack of health-care workers with specific training or expertise in infection control; 2) a lack of formal infection control programs in most facilities; 3) poor understanding among health-care workers regarding standard precautions for infection control; and 4) absent or inadequate equipment reprocessing, sterilization practices, and waste management. In response, in 2003, national infection control guidelines were developed, and infection control programs were established in 450 MOHP facilities. To ensure ongoing improvement, health-care workers received training, and supervision and monitoring were conducted on a monthly basis. After the implementation of the national infection control program, improvements were observed in health-care worker compliance with standard precautions (e.g., hand hygiene, use of personal protective equipment, safe injection practices, appropriate reprocessing of instruments, and waste management) (Table). Among 60 facilities with dialysis units, the annual incidence of HCV infection among previously uninfected recipients of renal dialysis decreased from 28% (before program implementation) to 6% (3 years after implementation).
In 2008, the Egyptian infection control guidelines were revised and adopted by the Eastern Mediterranean Regional Office of the World Health Organization for use in the region. In 2011, the program underwent an International Health Regulations assessment, which concluded that the program had substantially decreased iatrogenic transmission of HCV.
National Control Strategy for Viral Hepatitis
Given the high burden of viral hepatitis in Egypt, in 2006, MOHP established the National Committee for the Control of Viral Hepatitis (NCCVH). By April 2008, this committee had developed a National Control Strategy for Viral Hepatitis, which called for effective surveillance, enhancements in prevention to reduce the incidence of hepatitis B virus (HBV) and HCV infection, and expanded access to care and treatment for those with chronic infection. To date, implementation largely has been limited to the care and treatment component of the strategy; a national network of 23 viral hepatitis facilities has been established to provide viral hepatitis care and treatment at a substantially reduced cost. Facilities are located throughout Egypt and within 100 kilometers of every Egyptian city and village, allowing greater access to care and treatment. Each facility is directed by a trained hepatologist to ensure that care and treatment standards are met and provides a full spectrum of care. Persons 1) are tested to confirm HCV infection, 2) are screened for eligibility for subsidized treatment using uniform inclusion and exclusion criteria, 3) are given a baseline clinical assessment, and 4) receive care and treatment services, including medications and testing according to a standardized protocol. Standard therapy for HCV consists of 48 weeks of pegylated interferon and ribavirin. Oversight is provided by NCCVH, which collects electronic medical record data from the treatment sites, maintains a patient registry, regularly analyzes these data, and provides feedback to ensure provision of quality patient care. During 2008–2011, nearly 190,000 patients were provided with care and treatment (Figure). Preliminary results indicate that 51% of patients (most with HCV genotype 4, which causes approximately 90% of HCV infections in Egypt) achieved a sustained virologic response.
The estimated cost of the care and treatment program for the Egyptian government is $80 million annually, which covers 40% of total costs of the program; the remaining 60% is paid by insurance companies and patients. Market competition has driven down the price of medications for a standard 48-week course of treatment; since program inception, medication costs have decreased from approximately $12,000 to <$2,000 (9). Nevertheless, treatment costs remain a barrier, hampering efforts to reach a greater proportion of HCV-infected persons.
Reported by
Nasr El Sayed, MD, Amr Kandeel, MD, Mohamed Genedy, MD, Ministry of Health and Population; Gamal Esmat, MD, Wahid Doss, MD, Manal El Sayed, MD, National Committee for the Control of Viral Hepatitis; Naeema Al Gasseer, PhD, Nasr Eltantawy, MD, World Health Organization, Cairo; Hassan El Bushra, MD, Eastern Mediterranean Regional Office, World Health Organization, Cairo; Maha Talaat, DrPH, Infection Control Unit, Global Disease Detection and Response, US Naval Medical Research Unit No. 3, Cairo, Egypt. Hande Harmanci, MD, Global Hepatitis Program, World Health Organization, Geneva, Switzerland. Arnaud Fontanet, MD, Adeline Bernier, MPH, Emerging Disease Epidemiology Unit, Institut Pasteur, Paris, France. Rania A. Tohme, MD, Francisco Averhoff, MD, Div of Viral Hepatitis, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC. Corresponding contributor: Francisco Averhoff, faverhoff@cdc.gov, 404-718-8501.
Editorial Note
This report describes Egypt's efforts to control HCV infection by addressing infection control in health-care facilities and providing care and treatment to persons living with chronic HCV infection. Findings indicate that the infection control programs have been widely introduced and successful in reducing HCV infections in MOHP facilities. In addition, Egypt's care and treatment program is one of the largest national programs in the world, providing subsidized care and treatment services and medications to nearly 190,000 Egyptians with chronic HCV infection during 2008–2011. Egypt's market-based approach to reduce the cost of treatment for HCV infection has been effective and can serve as a model for other resource-constrained countries with a large burden of viral hepatitis seeking to provide affordable care and treatment services.
The primary focus for hepatitis control in Egypt has been on care and treatment; these activities consume up to 20% of the MOHP budget. However, Egypt's success in reducing the cost of treatment (9) has improved access for persons with chronic HCV infection. Nonetheless, continued monitoring of treatment responses and collection of cost-effectiveness data are needed to identify best practices and areas for improvement.
Despite Egypt's implementation of the infection control program in MOHP facilities, which provide care to 30% of the population, the remainder of the health-care system (e.g., university and private facilities) lacks a comprehensive, standardized approach to infection control. Decreasing the incidence and transmission of HCV infection in Egypt necessitates wider application of infection control standards to all providers of health and dental care. Best practices and guidelines should be disseminated to health-care and dental-care institutions and providers throughout Egypt, with an emphasis on expanding the number of trained experts capable of supporting and overseeing this effort. Beginning in 2003, the Egyptian government increased annual expenditures to support infection control programs, and by 2011, approximately $800,000 was dedicated to infection control activities in MOHP facilities, which amounts to only 1% of government expenditures for viral hepatitis care and treatment; increasing investments in health-care–facility infection control are necessary to limit the spread of hepatitis C and other health-care–associated infections in the country.
Given the current substantial burden of HCV infection in Egypt, hepatitis C–related morbidity and mortality are predicted to at least double in the coming 20 years (10). Aware of the need to effectively control the hepatitis C epidemic, in 2011, MOHP requested the assistance of the World Health Organization, Institut Pasteur, and CDC in the formation of a technical advisory group consisting of local and international experts in viral hepatitis and public health. This group, which first met in July 2011, was tasked with examining the burden of viral hepatitis, reviewing current control efforts, and providing MOHP with independent guidance on addressing viral hepatitis in Egypt. Consistent with the recommendations of resolution 63.18* adopted by the World Health Assembly in 2010, the group recognized the need for a comprehensive approach to address all elements of viral hepatitis prevention and control, including increasing community awareness and education, preventing HCV and HBV infection in health-care settings, ensuring a safe blood supply, enhancing access to hepatitis B vaccination, establishing surveillance and monitoring to track the effectiveness of control programs, and providing care and treatment to those already infected. With the technical assistance and support from the World Health Organization, the Institut Pasteur, and CDC, MOHP is working to develop and implement an enhanced prevention and control strategy.
In the past decade, Egypt has made considerable progress in viral hepatitis control by implementing a national infection control program for MOHP facilities and establishing a national hepatitis C treatment program. To contain the HCV epidemic, Egypt must overcome major challenges, including the high cost of treatment and the large and growing number of persons living with chronic HCV infection. These challenges could be addressed by implementing a comprehensive viral hepatitis control program, including the expansion of the current infection control program beyond MOHP facilities, raising community awareness, and ensuring a safe blood supply. Establishment of a viral hepatitis surveillance system also is needed to guide implementation of prevention interventions and monitor their effectiveness. Upon implementation of a comprehensive viral hepatitis control program, Egypt can serve as a model for other highly endemic countries.
Acknowledgments
Rachel J. Wilson, Div of Viral Hepatitis, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC.
References
- The Global Burden of Hepatitis C Working Group. Global burden of disease (GBD) for hepatitis C. J Clin Pharmacol 2004;44:20–9.
- El-Zanaty F, Way A. Egypt demographic and health survey 2008. Cairo, Egypt: Ministry of Health, El-Zanaty and Associates, and Macro International; 2009. Available at http://www.measuredhs.com/pubs/pdf/fr220/fr220.pdf. Accessed July 18, 2012.
- Paez Jimenez A, Mohamed MK, Sharaf Eldin N, et al. Injection drug use is a risk factor for HCV infection in urban Egypt. PloS One 2009;4:e7193.
- Paez Jimenez A, Sharaf Eldin N, Rimlinger F, et al. HCV iatrogenic and intrafamilial transmission in greater Cairo, Egypt. Gut 2010;59:1554–60.
- Strickland G. Liver disease in Egypt: hepatitis C superseded schistosomiasis as a result of iatrogenic and biological factors. Hepatology 2006;43:915–22.
- Mostafa A, Taylor S, El-Daly M, et al. Is the hepatitis C virus epidemic over in Egypt? Incidence and risk factors of new hepatitis C virus infections. Liver Int 2010;31:560–6.
- Talaat M, El-Oun S, Kandeel A, et al. Overview of injection practices in two governorates in Egypt. Trop Med Int Health 2003;8:234–41.
- Talaat M, Kandeel A, Rasslan O, et al. Evolution of infection control in Egypt: achievements and challenges. Am J Infect Control 2006;34:193–200.
- Ford N, Singh K, Cooke GS, et al. Expanding access to treatment for hepatitis C in resource-limited settings: lessons from HIV/AIDS. Clin Infect Dis 2012;54:1465–72.
- Deuffic-Burban S, Mohamed M, Larouze B, Carrat F, Valleron A. Expected increase in hepatitis C-related mortality in Egypt due to pre-2000 infections. J Hepatol 2006;44:455–61.
What is already known on this topic?
Egypt has the largest burden of hepatitis C infection in the world, with a 10% prevalence of chronic hepatitis C virus (HCV) infection among persons aged 15–59 years. Health-care–associated transmission is a major contributor to HCV transmission in Egypt.
What is added by this report?
Implementation of an infection control program at Egyptian Ministry of Health and Population (MOPH) facilities resulted in improvements in infection control practices among health-care workers and a decrease in the annual incidence of HCV infection among dialysis patients (from 28% to 6%). In addition, Egypt's viral hepatitis care and treatment program, one of the largest national programs in the world, has provided subsidized care and treatment services to nearly 190,000 Egyptians with chronic HCV infection.
What are the implications for public health practice?
Despite the Egyptian government's efforts to control HCV transmission in health-care settings and to provide care and treatment to persons living with HCV infection in Egypt, Egypt continues to have a large and growing hepatitis C epidemic. A comprehensive viral hepatitis control program, including the expansion of the infection control program beyond MOHP facilities, raising community awareness, ensuring a safe blood supply, and establishing a viral hepatitis surveillance system, is needed to control the hepatitis C epidemic in Egypt.
FIGURE. Estimated number of chronic hepatitis C patients receiving care and treatment, by year — Egypt, 2008–2011
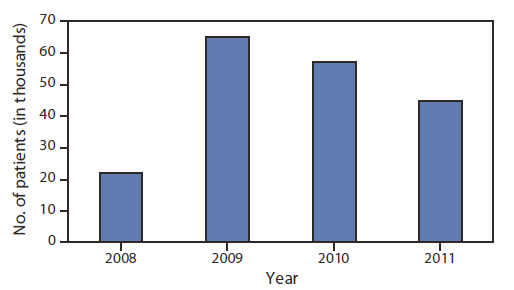
Source: National Committee for the Control of Viral Hepatitis, Egypt.
Alternate Text: The figure above shows the estimated number of chronic hepatitis C patients receiving care and treatment, by year, in Egypt, during 2008-2011. During this period, nearly 190,000 patients were provided with care and treatment.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


