FIGURE. Percentage of women aged 18--49 years pregnant at any time during October 2010--January 2011 (N = 1,457) who received influenza vaccination before, during, or after pregnancy for the 2010--11 influenza season, by month of delivery or expected month of delivery --- United States, Internet panel survey, April 2011
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Influenza Vaccination Coverage Among Pregnant Women --- United States, 2010--11 Influenza Season
Women are at increased risk for morbidity and mortality from influenza during pregnancy (1). Vaccinating pregnant women for influenza can protect both the women and their infants, especially infants aged <6 months who are not old enough to receive influenza vaccination (2--4). Since 2004, the Advisory Committee on Immunization Practices and the American College of Obstetricians and Gynecologists have recommended inactivated influenza vaccine for all women who are pregnant during influenza season, regardless of trimester (1,5). Before 2009, estimated influenza vaccination coverage among pregnant women had been consistently low (approximately 15%) (1,5). However, vaccination levels increased substantially in response to the 2009 influenza A (H1N1) pandemic to nearly 50% (6--7). To estimate influenza vaccination coverage among pregnant women for the 2010--11 season, CDC analyzed data from an Internet panel survey conducted in April 2011 among women who were pregnant any time during October 2010--January 2011. Among 1,457 survey respondents, 49% reported that they had received influenza vaccination: 12% were vaccinated before pregnancy, 32% during pregnancy, and 5% after pregnancy. Women offered influenza vaccination by a health-care provider (62%) were more likely to be vaccinated (71%) than other women (14%) and were more likely to have positive attitudes about vaccine effectiveness and safety. These results indicate that the higher vaccination level achieved the previous season (2009--10) was sustained and emphasize the critical role of health-care providers in promoting influenza vaccination. Continued efforts are needed to encourage health-care providers to strongly recommend and offer influenza vaccination to pregnant patients to protect both the mothers and their infants.
CDC conducted an Internet panel survey during April 4--25, 2011, to provide end-of-season estimates of influenza vaccination coverage and information on knowledge, attitudes, and behaviors related to influenza vaccination among pregnant women. Women aged 18--49 years who were pregnant at any time since August 1, 2010, were recruited from the SurveySpot panel operated by Survey Sampling International.* Of all panel members contacted in April 2011, a total of 2,126 were determined to be eligible for the survey, and 1,937 (91%) completed the online survey. The sample was weighted to reflect the age and race/ethnicity distribution based on census region estimates from the U.S. population of pregnant women (8). To be consistent with a previous study (6), the study population was limited to 1,457 women reporting pregnancy at any time during the peak influenza vaccination period (October 2010--January 2011).
Survey respondents were asked if they had an influenza vaccination since August 1, 2010, and if yes, in which month and whether it was before, during, or after pregnancy. Pregnancy status questions included whether respondents were currently pregnant or pregnant at any time since August 1, 2010, and if so, what were the actual months of pregnancy. Respondents who were pregnant at the time of the survey were asked their expected delivery date. All respondents were asked if their doctor or other health professional had offered them influenza vaccination during an office visit and their attitudes toward influenza and influenza vaccination. Weighted analyses were conducted using statistical software. Confidence intervals were calculated, and chi-square tests were used to assess statistical significance of differences in vaccination coverage levels between subgroups.
Of the 1,457 women pregnant at any time during October 2010--January 2011, 49% reported influenza vaccination for the 2010--11 season: 12% were vaccinated before pregnancy, 32% during pregnancy, and 5% after pregnancy. Vaccination after pregnancy was more prevalent for women delivering early in the vaccination period, and vaccination before pregnancy was more prevalent among women who were in earlier stages of pregnancy later in the vaccination period (Figure). Younger women (aged 18--24 years) were less likely to be vaccinated than older women (aged 25--49 years) (44% versus 52%) (Table 1). College graduates were more likely to be vaccinated than those with less education. Women with health insurance coverage also were more likely to report influenza vaccination compared with those who were not insured.
Overall, 62% of women reported that they were offered influenza vaccination by their health-care providers; among those offered vaccination, 71% received influenza vaccination, substantially higher than the 14% vaccination level among women whose health-care providers did not offer vaccination (Table 1). Forty-five percent of women reported influenza vaccination in a previous influenza season, and these women were four times as likely to report 2010--11 vaccination as women without previous vaccination (84% versus 21%).
Compared with women whose health-care provider did not offer vaccination, women who received a health-care provider offer were more likely to have positive attitudes about the effectiveness of influenza vaccination (82% versus 54%), safety of influenza vaccination for pregnant women (78% versus 53%), and safety of vaccination for their infants (75% versus 47%) (Table 2). In addition, women who received a health-care provider offer consistently had higher vaccination levels than those who did not receive a health-care provider offer, regardless of their perceptions of vaccination safety or effectiveness expressed in April 2011. Moreover, women with a negative attitude toward vaccination who had received a health-care provider offer of vaccination were more likely to be vaccinated than women who had a positive attitude without a health-care provider offer.
The top five "main" reasons for not receiving influenza vaccination were "I am concerned about possible safety risks to my baby if I got vaccinated" (20%), "I am concerned that the vaccination would give me the flu" (17%), "I don't think the vaccination is effective in preventing flu" (14%), "I am concerned about possible safety risk to myself if I got vaccinated" (11%), and either "I don't think I would get very sick if I got the flu" or "I think if I get the flu, I will just get some medication to treat it" (14%).
The majority of women who were vaccinated during pregnancy received vaccination at their obstetrician/gynecologist or midwife's office (61%), followed by another doctor's office or another medical-related place (22%), a pharmacy or grocery store (8%), health department (5%), and their workplace or school (5%). Among women vaccinated either before or after pregnancy, 18% were vaccinated in an obstetrician/gynecologist or midwife's office, and 61% in another doctor's office or another medical-related place.
Reported by
Deborah K Walker, EdD, Sarah Ball, ScD, Robert Black, MPH, David Izrael, MS, Abt Associates, Inc., Atlanta, Georgia. Helen Ding, MD, Gary L. Euler, DrPH, James A. Singleton, MS, Carolyn B. Bridges, MD, Immunization Svc Div, Lisa A. Grohskopf, MD, Influenza Div, National Center for Immunization and Respiratory Diseases; Denise J. Jamieson, MD, Div of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, CDC. Corresponding contributor: Helen Ding, hding@cdc.gov, 404-639-8513.
Editorial Note
Results from this survey indicate that the record high influenza vaccination levels among pregnant women reported for the previous influenza season (2009--10) were sustained during the 2010--11 season. During 2009--10, pregnant women were included in the initial target groups to receive the inactivated 2009 H1N1 pandemic vaccine, and CDC worked closely with key partners, especially the American College of Obstetricians and Gynecologists, to increase awareness that pregnant women were at increased risk for severe illness from influenza and were recommended for influenza vaccination to protect themselves and their infants (9). However, vaccination levels are still below the Healthy People 2020 target of 80% influenza vaccination coverage for pregnant women.†
This study found that women who received a health-care provider offer were more likely to believe influenza vaccination was effective, protective, and safe for themselves and their infants, and were nearly five times more likely to report receipt of vaccination compared with those who visited a doctor but did not receive an offer of vaccination. Pregnant women who had previously received influenza vaccination were four times more likely to receive influenza vaccination compared with those without a prior history of influenza vaccination. Because influenza vaccination is now recommended for all persons aged ≥6 months (1), further implementation of the universal vaccination recommendation among women of childbearing age might help to increase the likelihood of influenza vaccination before and during pregnancy.
Pregnant women who receive regular prenatal care have many more opportunities for a health-care provider offer of influenza vaccination than nonpregnant women. However, nearly four out of 10 women in this survey did not receive an offer of vaccination even though they visited a health-care provider at least one time. Barriers to providing influenza vaccination in health-care providers' offices identified by previous studies include lack of infrastructure for vaccine storage, lack of training for nurses to administer vaccines, and concern about safety and related lawsuits for vaccinating first trimester women (10). Another finding of this study was that women still reported safety risk to their infant as the most common main reason for refusing influenza vaccination, even though influenza vaccination during pregnancy can protect women and their infants (2--4). This study also indicated that a substantial proportion of women who delivered early in the influenza season received their vaccination after delivery. Vaccination of members of households with an infant aged <6 months is important for minimizing influenza risk for the upcoming influenza season.
The findings in this report are subject to at least two limitations. First, selection bias might remain after weighting adjustments, given the exclusion of women with no Internet access and the self-selection processes for entry into the panel and participation in the survey. However, influenza vaccination coverage estimated from this study, restricted to women who were pregnant at any time during December 2010 (48%), was similar to the coverage estimates based on December 2010 Behavioral Risk Factor Surveillance System (BRFSS) interviews of women who were pregnant at that time (51%) (CDC, unpublished data, 2011). BRFSS is a telephone survey and also might be subject to selection bias because of exclusion of households without landline telephone service. Pregnant women account for only 1% of the general population, and conducting a random-digit--dialing survey or a mail survey large enough to obtain an adequate sample size would be costly and time-consuming. The similar estimate from BRFSS provides more evidence to support the use of Internet panels as useful surveillance data sources for timely midseason and postseason evaluation of influenza vaccination among pregnant women. Second, the survey was self-administered, and because pregnancy and vaccination status were not validated by medical record review, all responses are subject to recall and reporting error.
This study found that the higher vaccination level achieved during the 2009--10 influenza season (the fall wave of 2009 H1N1 virus activity) among pregnant women was repeated the following season, and identified key elements highly associated with pregnant women's acceptance of influenza vaccination, such as the health-care provider offer of vaccination and past receipt of influenza vaccination. Continued efforts are needed to encourage health-care providers to strongly recommend and offer influenza vaccination to their pregnant patients. Additional efforts are needed to remove barriers for health-care providers to administer influenza vaccination as part of routine practice. Messages to pregnant women from health-care providers and others should emphasize the safety and effectiveness of maternal influenza vaccination to maximize protection of pregnant patients and their infants.
Acknowledgments
John Boyle, PhD, Chuck Shuttles, Abt SRBI, Inc., Washington, DC. Peng-jun Lu, MD, Leah N Bryan, MPH, Immunization Svc Div, National Center for Immunization and Respiratory Diseases, CDC.
References
- CDC. Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR 2010;59(No. RR-8).
- Eick AA, Uyeki TM, Klimov A, et al. Maternal influenza vaccination and effect on influenza virus infection in young infants. Arch Pediatr Adolesc Med 2011;165:104--11.
- Zaman K, Roy E, Arifeen SE, et al. Effectiveness of maternal influenza immunization in mothers and infants. N Engl J Med 2008;359:1555--64.
- Poehling KA, Szilagyi PG, Staat MA, et al., Impact of maternal immunization on influenza hospitalizations in infants. Am J Obstet Gynecol 2011;204(6 Suppl 1):S141--8.
- American College of Obstetricians and Gynecologists Committee on Obstetric Practice. Influenza vaccination and treatment during pregnancy. Obstet Gynecol 2004;104(5 pt 1):1125--6.
- Ding H, Santibanez TA, Jamieson DJ, et al. Influenza vaccination coverage among pregnant women---National 2009 H1N1 Flu Survey (NHFS). Am J Obstet Gynecol 2011;204(6 Suppl 1):S96--106.
- CDC. Seasonal influenza and 2009 H1N1 influenza vaccination coverage among pregnant women---10 states, 2009--10 influenza season. MMWR 2010;59:1541--5.
- CDC. Estimated pregnancy rates for the United States, 1990--2005: an update. Natl Vital Stat Rep 2009;58(4).
- Rasmussen SA, Kissin DM, Yeung LF, et al; Pandemic Influenza and Pregnancy Working Group. Preparing for influenza after 2009 H1N1: special considerations for pregnant women and newborns. Am J Obstet Gynecol 2011;204(6 Suppl 1):S13--20.
- Naleway AL, Smith WJ, Mullooly JP. Delivering influenza vaccine to pregnant women. Epidemiol Rev 2006;28:47--53.
* Additional information available at http://www.surveysampling.com. The SurveySpot panelists generally were recruited from Internet sites that host large and frequent numbers of visitors and diverse Internet traffic. Multiple methods of recruitment were used, including banner ads, direct invitations, pop-ups, and web intercepts. The panel represents approximately 1 million households, and new panelists are continually being recruited; existing panelists are removed from the panel if they have opted-out or have not responded to an invitation within a specified period. A minimum incentive is routinely used to maintain the panel but not for an inducement to participate in a particular survey. Pregnant women panelists in this report were recruited from the SurveySpot panel using two methods. First, a message advertising the survey was placed on the main panel website (http://www.surveyspot.com), inviting panelists to view the survey eligibility questions on the panel's requirements page. A total of 18,789 respondents were invited, and 1,705 (9.1%) viewed the first eligibility question. Second, an e-mail invitation was sent to a sample of 11,688 panelists whose panel profiles indicated that they were women aged 18--49 years living in the United States. Of these, 1,370 (11.7%) replied. As a result of the two methods, a total of 3,075 panelists recruited went to the survey website and answered the first eligibility question.
† Additional information about Healthy People 2020 objectives for influenza vaccination is available at http://www.healthypeople.gov/2020/topicsobjectives2020/objectiveslist.aspx?topicId=23.
What is already known on this topic?
Pregnant women are recommended by the American College of Obstetricians and Gynecologists and the Advisory Committee on Immunization Practices to receive influenza vaccination regardless of trimester. Vaccination coverage among pregnant women was approximately 50% for the 2009--10 season, much higher than coverage reported for previous influenza seasons. Health-care provider recommendation is strongly associated with vaccination among pregnant women.
What is added by this report?
Approximately 49% of pregnant women in an Internet panel survey were vaccinated for influenza for the 2010--11 influenza season; 32% were vaccinated during pregnancy, and 17% before pregnancy or after delivery. Among the 62% of pregnant women who received a health-care provider offer for influenza vaccination, nearly three quarters were vaccinated, which was five times the coverage among those who didn't receive a health-care provider offer.
What are the implications for public health practice?
Continued efforts are needed to 1) encourage health-care providers to strongly recommend and offer inactivated influenza vaccination to their pregnant patients and 2) remove barriers for health-care providers to administer influenza vaccination as part of routine practice. Messages to pregnant women from health-care providers and others should emphasize the safety and effectiveness of maternal influenza vaccination to maximize protection of pregnant patients and their infants.
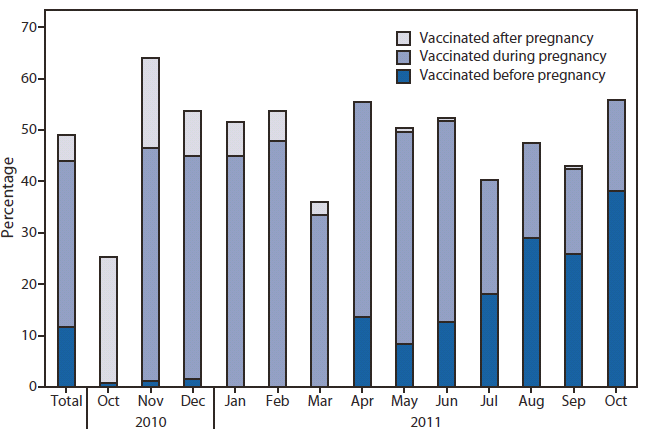
Alternate Text: The figure above shows the percentage of women aged 18-49 years pregnant at any time during October 2010-January 2011 (N = 1,457) who received influenza vaccination before, during, or after pregnancy for the 2010-11 influenza season, by month of delivery or expected month of delivery, in the United States, based on results of an Internet panel survey conducted in April 2011. Vaccination after pregnancy was more prevalent for women delivering early in the vaccination period, and vaccination before pregnancy was more prevalent among women who were in earlier stages of pregnancy later in the vaccination period.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


