Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Hepatitis A Vaccination Coverage Among U.S. Children Aged 12--23 Months --- Immunization Information System Sentinel Sites, 2006--2009
Hepatitis A vaccine was first licensed as a 2-dose vaccine for children aged ≥24 months in 1995. In 1996 and 1999, the Advisory Committee on Immunization Practices (ACIP) recommended routine hepatitis A vaccination for children aged ≥24 months in communities with the highest rates of the disease (1,2). In August 2005, the minimum age for which the vaccine was licensed was lowered to 12 months, and in May 2006, ACIP recommended routine vaccination of all children aged 12--23 months, regardless of risk category or location (3). As a result, hepatitis A incidence in the United States reached a historic low in 2007, the most recent year for which data are available (4). To assess hepatitis A vaccine coverage among children aged 12--23 months from 2006 through 2009, CDC used data from eight Immunization Information System (IIS) sentinel sites. Average (unweighted) hepatitis A vaccination coverage with ≥1 dose at the sites increased from 17% in 2006 to 47% in 2009. Average full vaccination coverage with ≥2 vaccine doses through age 23 months increased from 1% in 2006 to 15% in 2009. Vaccination coverage with ≥1 dose increased the most during 2006 through the first quarter of 2007, after which the rate of increase slowed. The 2006 ACIP recommendations for routine hepatitis A vaccination of all children aged 12--23 months resulted in improved coverage, but coverage has plateaued. Immunization programs and vaccine providers should encourage hepatitis A vaccination of all children beginning at age 12 months.
IIS are confidential, population-based, computerized data systems that were designed primarily to consolidate vaccination records for all children within a geographic area from multiple vaccine providers. Data are collected from health-care providers, vital records, and billing systems. Although not designed to be representative of the U.S. population, IIS sentinel sites are population based and cover more than 1.8 million children aged <6 years residing in diverse regions of the United States. For the 2008--2012 sentinel site project period, CDC awarded supplemental funds to eight IIS* sites that met the following criteria: 1) ≥85% of child vaccine provider sites were enrolled in the IIS, 2) ≥85% of children aged <19 years who resided in the sentinel site region had ≥2 vaccinations recorded in the IIS, and 3) >70% of doses were reported to IIS within 30 days of vaccination. The six sentinel site areas in Arizona, Colorado, Michigan, Minnesota, Oregon, and Wisconsin consist of contiguous geographic counties, postal code areas, or census tracts. The other two sentinel sites consist of the entire state of North Dakota and all of New York City. The total enrollment of children aged 12--23 months at the eight sites was 331,054 for 2006 and 307,030 for 2009. Enrollment in 2009 varied from 143,533 in New York City to 2,223 in Colorado. Data from the IIS sentinel sites were used to calculate hepatitis A vaccination coverage among children aged 12--23 months. Hepatitis A vaccination coverage with ≥1 dose among children aged 12--23 months was calculated by dividing the number of children who received ≥1 dose before age 24 months by the total number of children aged 12--23 months residing in each sentinel site area and enrolled in the IIS, by year and quarter, from 2006--2009.
To be considered valid, the 2 hepatitis A vaccine doses must be administered at least 6 months apart; therefore, children aged 12--17 months are not eligible to be fully vaccinated. To calculate full vaccination coverage, the number of children who received ≥2 valid hepatitis A vaccine doses before age 24 months was divided by the total number of children aged 18--23 months residing in each sentinel site area and enrolled in the IIS, by year and quarter, from 2006--2009. The unweighted average site-specific coverage for the eight sites was calculated by summing the percentages of children vaccinated at each site and dividing by the total number of sites (eight).
In 2006, average hepatitis A vaccination coverage with ≥1 dose among children aged 12--23 months was 17% across the eight IIS sentinel sites (range: 11%--26%) (Table 1). In 2009, Michigan, North Dakota, Oregon, and Wisconsin reached ≥50% coverage among children aged 12--23 months. Coverage ranged from 29%--49% in the other sites. Among children recommended for routine hepatitis A vaccination and eligible to be fully vaccinated (i.e., children aged 18--23 months), full vaccination coverage with ≥2 doses increased from 1% in 2006 to 15% in 2009 (range: 6%--24%) (Table 2). Coverage increased 5--10 percentage points each quarter of 2006 through the first quarter of 2007. The change in coverage during these five quarters accounted for 73% of the total increase in hepatitis A vaccination coverage during 2006--2009. In the remaining 11 quarters (second quarter of 2007 through fourth quarter 2009), coverage increased less and declined slightly (between -2 and 2 percentage points per quarter) (Figure).
Reported by
MB Kurilo, MPH, Oregon Dept of Human Svcs. LJ Pabst, MPH, National Center for Immunization and Respiratory Diseases; C Weinbaum, MD, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC.
Editorial Note
Before the 2006 changes in ACIP recommendations for routine vaccination, all eight sentinel sites had low hepatitis A vaccination coverage (17%) among children aged 12--23 months. By 2009, coverage with ≥1 dose and ≥2 doses of hepatitis A vaccine had increased across all sites, ranging from 29% to 58% and 6% to 24%, respectively. The 2008 National Immunization Survey (NIS) reported vaccination coverage with ≥2 doses of approximately 40% (5). However, results from NIS are not comparable to results from IIS because 1) NIS includes children aged 19--35 months, while these IIS data include only children aged 12--23 months; 2) NIS counts all doses administered, while IIS counts only those doses that are valid according to ACIP recommendations for the minimum age at administration and the minimum interval between doses; and 3) NIS includes any doses administered by the date of the household interview (may include doses administered through age 35 months), whereas these IIS results include only doses administered from ages 12--23 months. IIS results also might differ from NIS because of underreporting to IIS.
The variation in vaccination coverage across the sentinel sites in 2009 highlights state-specific differences in vaccine administration that likely are influenced by many factors, including provider practices, parental attitudes, access to care, reimbursement and insurance policies, and education and outreach regarding vaccine recommendations. Hepatitis A vaccination coverage at all eight sentinel sites increased substantially during 2006 through the first quarter of 2007, and then showed little increase through 2009. The slight decrease in ≥1-dose coverage in the third and fourth quarters of 2009 might reflect an actual decrease in vaccination coverage, a decrease or delay in reporting to IIS, or some other factor.
A single dose of hepatitis A vaccine is highly immunogenic; however, a second dose is indicated for long-term protection (2), and extending the interval between doses has no adverse effect on immunogenicity. Vaccine providers can use reminder/recall messages to help ensure that all children in their practice receive 2 hepatitis A vaccine doses. School or child-care entry vaccination requirements also might contribute to improved vaccination coverage (6). Two of the eight sentinel sites, North Dakota and Oregon, require that children entering child care, preschool, or Head Start programs be vaccinated against hepatitis A.† Both reminders/recalls and vaccination requirements for child care and school attendance are among a number of interventions recommended by the Task Force on Community Preventive Services to improve vaccination coverage (7,8).
The findings in this report are subject to at least three limitations. First, the analysis in this report is based on data from only eight IIS sentinel sites. Although these sites represent diverse regions of the United States, data from these sites might not be representative of immunization practices nationwide. Second, underestimates of coverage might have occurred. IIS are dependent on participation by providers to report vaccination information. Although at least 85% of provider sites must be enrolled in an IIS for it to be included in this coverage analysis, not all providers and health systems in each IIS sentinel site are enrolled, and enrolled providers might not submit all vaccine data to IIS. Finally, data assessment was limited to vaccinations received through age 23 months; 2-dose vaccination coverage at older ages was not assessed.
Even with modest levels of vaccination coverage, vaccination of U.S. children against hepatitis A has resulted in significant communitywide decreases in disease incidence (9). As of 2007, the incidence of acute hepatitis A had declined to the lowest level recorded (4). The 2005 licensure of the hepatitis A vaccine for use in children aged ≥12 months and the 2006 ACIP recommendations for routine vaccination of all U.S. children resulted in improved vaccination coverage and are expected to reduce hepatitis A rates further. However, the findings of this report suggest that increases in hepatitis A vaccination coverage among children aged 12--23 months are slowing. Immunization programs and vaccine providers should continue to encourage all children to receive hepatitis A vaccine, beginning at age 12 months. Because IIS vaccination data are especially timely (data on >70% of vaccine doses administered are entered into IIS sentinel sites within 30 days of vaccine administration), these data are particularly useful for monitoring hepatitis A vaccination coverage and evaluating the impact of interventions to increase coverage.
Acknowledgments
This report is based, in part, on contributions by the IIS sentinel site project contributors: P Gast, MS, L Rasmussen, Arizona Dept of Health Svcs; D Herrero, MS, K Dugger, MPH, Colorado Dept of Public Health and Environment Immunization Program; R Potter, DVM, B Salada, Michigan Dept of Community Health; KE White, MPH, EJ Peterson, Minnesota Dept of Health; M Sander, MPH, K LoMurray, North Dakota Dept of Health; V Papadouka, PhD, MA Hansen, MPH, New York City Dept of Health and Mental Hygiene; J Gaudino, MD, Oregon Immunization Program, Public Health Div, Dept of Human Svcs; and T Maerz, and S Schauer, PhD, Wisconsin Immunization Program.
References
- CDC. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1996;45(No. RR-15).
- CDC. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1999;48(No. RR-12).
- CDC. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2006;55(No. RR-7).
- CDC. Surveillance for acute viral hepatitis---United States, 2007. Surveillance Summaries, May 22, 2009. MMWR 2009;58(No. SS-3).
- CDC. National, state, and local area vaccination coverage among children aged 19--35 months---United States, 2008. MMWR 2009;58:921--6.
- Orenstein WA, Hinman AR. The immunization system in the United States---the role of school immunization laws. Vaccine 1999;17(Suppl 3):S19--24.
- Task Force on Community Preventive Services. Recommendations regarding interventions to improve vaccination coverage in children, adolescents, and adults. Am J Prev Med 2000;18:92--6.
- Briss PA, Rodewald LE, Hinman AR, et al. Reviews of evidence regarding interventions to improve vaccination coverage in children, adolescents, and adults. Am J Prev Med 2000;18:97--140.
- Armstrong GL, Billah K, Rein DB, Hicks KA, Wirth KE, Bell BP. The economics of routine childhood hepatitis A immunization in the United States: the impact of herd immunity. Pediatrics 2007;119:e22--9.
* Additional information available at http://www.cdc.gov/vaccines/programs/iis/default.htm.
† North Dakota's immunization requirements are available at http://www.legis.nd.gov/information/acdata/pdf/33-06-05.pdf; Oregon's requirements are available at http://www.oregon.gov/dhs/ph/imm/school.
What is already known on this topic?
In August 2005, the youngest age for which hepatitis A vaccine was licensed was lowered from 24 months to 12 months, and in May 2006, the Advisory Committee for Immunization Practices (ACIP) recommended routine vaccination of all children aged 12--23 months, regardless of risk category or location.
What is added by this report?
After the 2006 ACIP recommendations for routine hepatitis A vaccination of all children aged 12--23 months, unweighted average coverage with ≥1 dose at eight Immunization Information System (IIS) sentinel sites increased rapidly through the first quarter of 2007 to approximately 40%, but then plateaued.
What are the implications for public health practice?
Immunization programs and vaccine providers should encourage all children to receive hepatitis A vaccine, beginning at age 12 months; interventions, including reminder/recall notification and school or child-care entry vaccination requirements, can contribute to increased vaccination coverage.
FIGURE. Mean hepatitis A vaccination coverage with ≥1 dose and full coverage* among children aged 12--23 months, by year and quarter --- Immunization Information Systems (IIS) sentinel sites,† 2006--2009
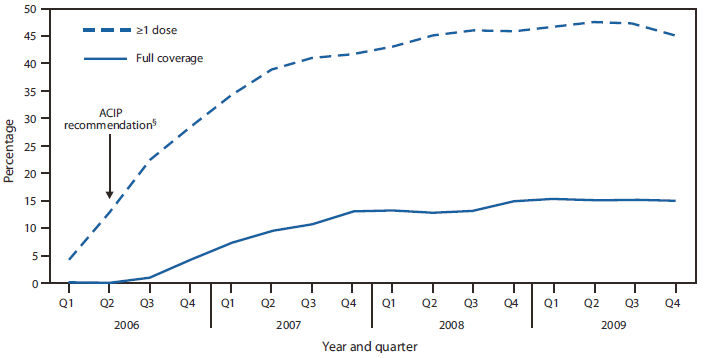
* ≥2 doses separated by ≥6 months.
† The sentinel sites for this analysis included areas in Arizona, Colorado, Michigan, Minnesota, Oregon, and Wisconsin, all of North Dakota, and New York City. Additional information is available at http://www.cdc.gov/vaccines/programs/iis/default.htm. Enrollment of children aged 12--23 months in all eight sites was 331,054 in 2006 and 307,030 in 2009. Enrollment in 2009 was as follows: Arizona, 12,656; Colorado, 2,223; Michigan, 85,430; Minnesota, 18,741; North Dakota, 10,393; New York City, 143,533; Oregon, 18,710; and Wisconsin, 15,344.
§ In May 2006, the Advisory Committee on Immunization Practices (ACIP) recommended routine vaccination against hepatitis A of all children aged 12--23 months, regardless of risk category or location.
Alternate Text: The figure above shows mean hepatitis A vaccination coverage with ≥1 dose and full coverage among children aged 12-23 months, by year and quarter for 2006-2009. Coverage increased 5-10 percentage points each quarter of 2006 through the first quarter of 2007. The change in coverage during these five quarters accounted for 73% of the total increase in hepatitis A vaccination coverage during 2006-2009. In the remaining 11 quarters (second quarter of 2007 through fourth quarter 2009), coverage increased less and declined slightly (from -2 to 2 percentage points per quarter).
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


