Reported Tuberculosis in the United States, 2022
TB Treatment and Case Outcomes

Successful completion of TB treatment for persons with TB disease is important to cure the patient of TB disease, prevent the development of drug resistance to TB treatments, and minimize the risk of spreading TB to others.
CDC collects information on the date and reason the patient stopped TB treatment (e.g., the patient completed treatment or the patient died). CDC uses this information to calculate completion-of-treatment percentages. CDC stratified cases of TB disease by the indicated length of treatment, based on American Thoracic Society, CDC, and Infectious Diseases Society of America treatment guidelines in effect during the period covered, and the patient’s initial drug-susceptibility test results, age, and disease site.
TB treatment regimens vary in the duration of the regimen, the types of anti-TB drugs prescribed, and the dose and frequency of the drugs.
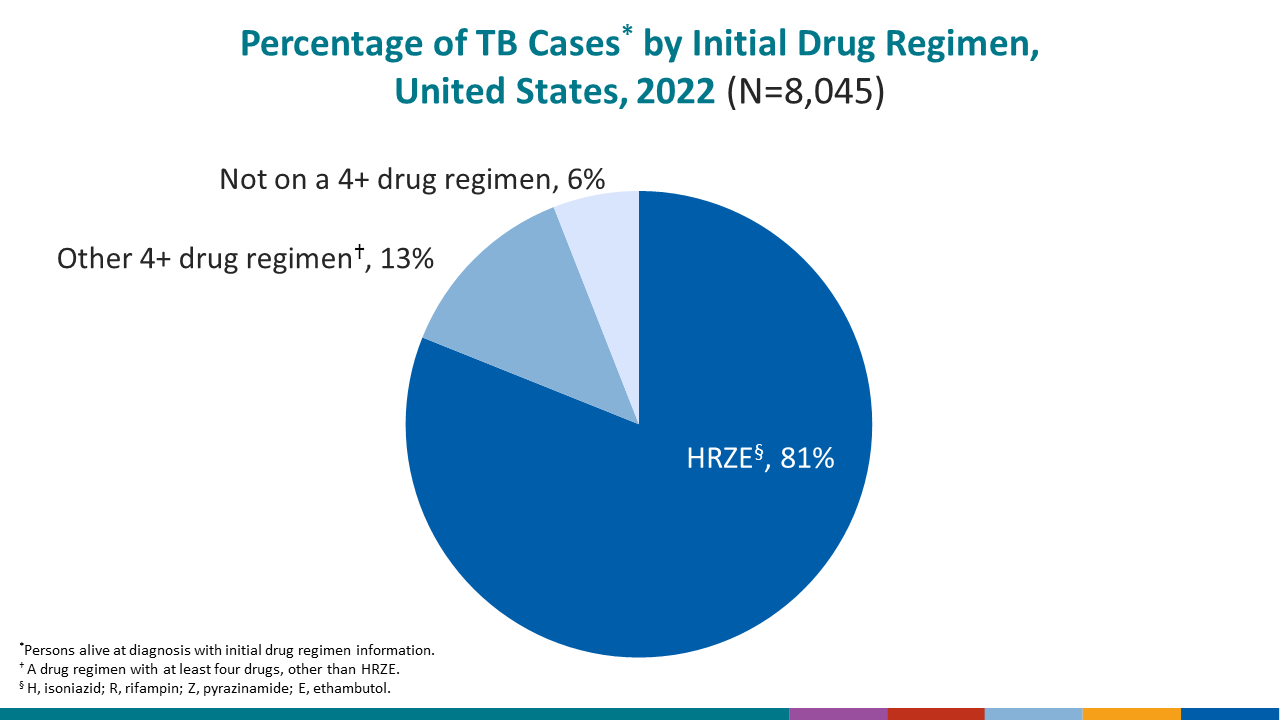
The most common initial treatment regimen for drug-susceptible TB disease and for TB disease when drug-susceptibility is unknown, includes isoniazid, H; rifampin, R; pyrazinamide, Z; and ethambutol, E (HRZE), also known as RIPE.
Among persons with TB who were alive at diagnosis and had initial drug regimen information available in 2022,
- 81.1% began treatment with HRZE.
- 13.0% began treatment with a four-drug regimen other than HRZE.
- 6.0% started on a regimen of less than four drugs, including persons who were not prescribed any drugs.
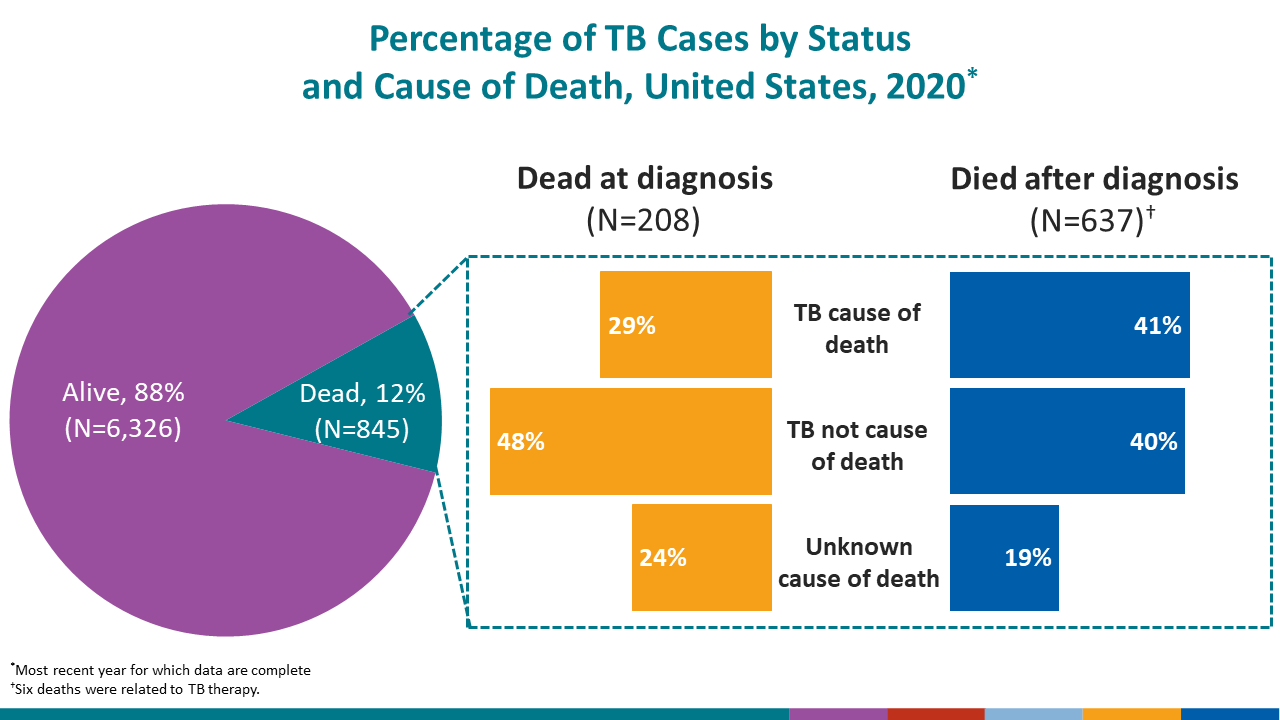
Among persons diagnosed with TB disease during 2020, a total of 845 (11.8%) persons died.
- Of the 845 deaths, 208 (24.6%) persons were dead at the time of TB diagnosis, and 637 (75.4%) persons died after diagnosis.
- Of the 208 deaths that occurred in persons who were dead at diagnosis, 28.8% of the deaths were attributed to TB.
- The percentage who died among persons with TB disease has remained consistent with previous years.
Learn more in the Executive Commentary.