Purpose
Overview
GISP sentinel surveillance collected N. gonorrhoeae isolates monthly from up to the first 25 men with symptomatic gonococcal urethritis seeking care at participating STI clinics. Clinical and demographic data were abstracted from medical records. Isolates were shipped from participating clinics to the regional laboratories participating in the Antibiotic Resistance Laboratory Network (ARLN) for agar dilution antimicrobial susceptibility testing. In 2023 and 2024, isolates were tested to determine minimum inhibitory concentrations (MICs) of penicillin, tetracycline, ceftriaxone, cefixime, ciprofloxacin, azithromycin, and gentamicin. Cefixime susceptibility testing was discontinued in 2007 and re-started in 2009. Gentamicin susceptibility testing was first started in 2015. Clinical, demographic, and laboratory data are compiled and analyzed at CDC for public use.
In 2017, GISP expanded in a subset of clinical sites to conduct N. gonorrhoeae surveillance in non-urethral isolates (i.e., pharyngeal, rectal, and endocervical isolates). The Enhanced Gonococcal Isolate Surveillance Project (eGISP) was established to help understand if the pharynx and/or rectum may be anatomic niches that select for or foster resistance and to evaluate if gonococcal susceptibility patterns vary between men and women.
N. gonorrhoeae isolates were collected monthly in eGISP from up to the first 25 men or women with gonococcal infections at the pharynx or rectum and from up to the first 25 women with cervical or vaginal gonococcal infections who sought care at participating STI clinics. The submission of isolates, susceptibility testing by agar dilution, and the transmission of clinical, demographic, and laboratory data were the same as in GISP.
Findings from GISP directly contributed to the CDC's STI Treatment Guidelines released in 1993, 1998, 2002, 2006, 2010, 2015, and 2021 and the Gonorrhea Treatment recommendation updates released in 2000, 2004, 2007, 2012, and 2020. Findings from GISP also directly contributed to CDC's AR Threats Reports in 2013 and 2019. GISP and CARGOS support the ability to detect changes in susceptibility patterns across different populations and to detect resistant infections sooner for improved surveillance and public health response.
Findings
Antimicrobial susceptibility criteria used in the 2023 and 2024 GISP and eGISP profiles:
Antimicrobial
MIC Value
MIC Interpretation
Ceftriaxone
MIC ≥0.125 µg/ml
Elevated MIC
Cefixime
MIC ≥0.25 µg/ml
Elevated MIC
Azithromycin
MIC ≥2.0 µg/ml
Resistance
Ciprofloxacin
MIC ≥1.0 µg/ml
Resistance
Penicillin
MIC ≥2.0 µg/ml or Beta lactamase positive
Resistance
Tetracycline
MIC ≥2.0 µg/ml
Resistance
Gentamicin
MIC values correlated with susceptibility and resistance have not been established
The majority of these criteria are consistent with Clinical and Laboratory Standards Institute (CLSI) criteria. Criteria for azithromycin are consistent with the FDA-Recognized Antimicrobial Susceptibility Test Interpretive Criteria (FDA-STIC). As of September 2025, CLSI and FDA-STIC criteria for resistance to ceftriaxone and cefixime, and criteria for resistance and susceptibility to gentamicin have not been established for N. gonorrhoeae.
GISP and eGISP Site Profiles
2023 Profiles:
- GISP Profiles National Data
- GISP Profiles Site Specific Data
- eGISP Profiles National Data
- eGISP Profiles Site Specific Data
2024 Profiles:
2023 GISP Clinical Sites and Regional Laboratories
STI clinics affiliated with 30 state or city health departments contributed 3,502 gonococcal isolates to GISP from January-December 2023 and 1,820 gonococcal isolates from January-July 2024. Of these 30 sites, 10 current sites have participated continuously since 1987.
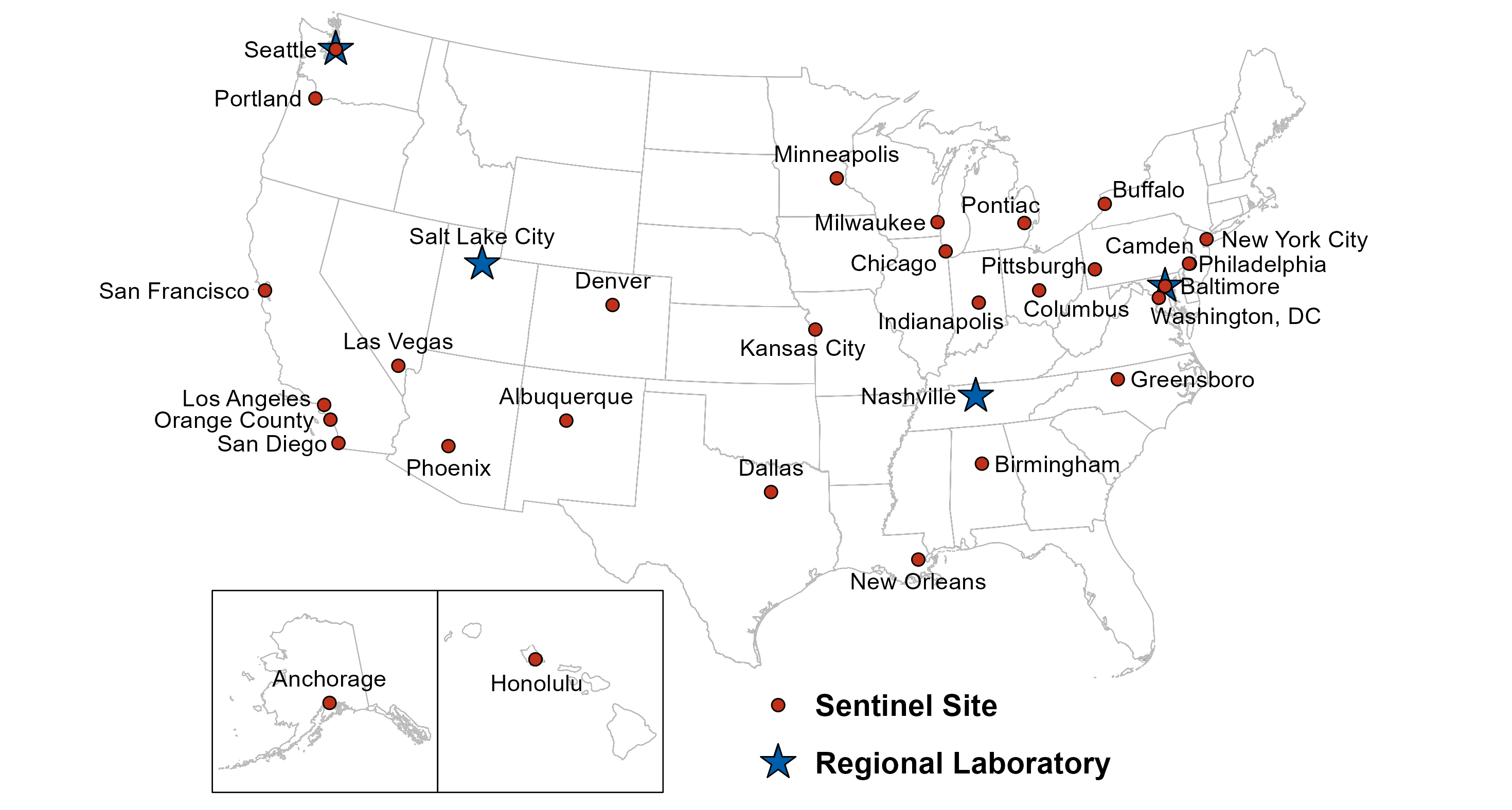
2023-2024 GISP Clinical Sites (years participated)
|
Albuquerque, New Mexico |
Greensboro, North Carolina |
Philadelphia, Pennsylvania |
|
Anchorage, Alaska |
Honolulu, Hawaii |
Phoenix, Arizona |
|
Baltimore, Maryland |
Indianapolis, Indiana |
Orange County, California |
|
Birmingham, Alabama |
Kansas City, Missouri |
Pittsburgh, Pennsylvania |
|
Buffalo, New York |
Las Vegas, Nevada |
Pontiac, Michigan |
|
Camden, New Jersey |
Los Angeles, California |
Portland, Oregon |
|
Chicago, Illinois |
Milwaukee, Wisconsin |
San Diego, California |
|
Columbus, Ohio |
Minneapolis, Minnesota |
San Francisco, California |
|
Dallas, Texas |
New Orleans, Louisiana |
Seattle, Washington |
|
Denver, Colorado |
New York, New York |
Washington, District of Columbia |
A subgroup of clinical sites began contributing gonococcal isolates to eGISP beginning in 2018. A total of 10 sites participated in eGISP in 2023 contributing 1,495 gonococcal isolates during the year. A total of 9 sites participated in eGISP in 2024, contributing 854 gonococcal isolates from January 1, 2024-July 31, 2024.
2023-2024 eGISP Clinical Sites (years participated)
|
Columbus, Ohio |
New Orleans, Louisiana |
Pontiac, Michigan |
|
Honolulu, Hawaii |
Orange County, California |
San Diego, California |
|
Las Vegas, Nevada |
Philadelphia, Pennsylvania |
|
|
Minneapolis, Minnesota |
Phoenix, Arizona |
|
Starting in 2017, four of the seven ARLN state public health laboratories began functioning as GISP Regional Laboratories. In 2023-2024, antimicrobial susceptibility testing of all GISP and eGISP isolates was conducted by these four laboratories:
- Maryland Department of Health and Mental Hygiene (Baltimore, Maryland)
- Tennessee Department of Health (Nashville, Tennessee)
- Utah Department of Health (Salt Lake City, Utah)
- Washington State Department of Health (Seattle, Washington)
Summary
Susceptibility to Antimicrobial Agents
Susceptibility to cefixime
Susceptibility testing for cefixime in GISP began in 1992, was discontinued in 2007, and was re-started in 2009. The distribution of cefixime MICs each year for the previous 5-year period is displayed in Figure 1 of the GISP National Profiles. Each year, approximately 80-90% of isolates exhibited cefixime MICs ≤0.03 µg/ml. The percentage of isolates with elevated cefixime MICs (≥0.25 µg/ml) fluctuated between approximately 0.1% and 0.5% during 2019 through 2024. Site-specific data are presented in the Site Profiles section of this report.
Susceptibility to ceftriaxone
Susceptibility testing for ceftriaxone began in 1987. The distribution of ceftriaxone MICs each year for the previous 5-year period is displayed in Figure 2 of the GISP National Profiles. Each year, approximately 90% of isolates exhibited ceftriaxone MICs ≤0.015 µg/ml. The percentage of GISP isolates that exhibited elevated ceftriaxone MICs, defined as ≥0.125 µg/ml, never exceeded 0.1% between 2019 and 2024. Site-specific data are presented in the Site Profiles section of this report.
Susceptibility to azithromycin
Susceptibility testing for azithromycin began in 1992. The distribution of azithromycin MICs each year for the previous 5-year period is displayed in Figure 3 of the GISP National Profiles. Most isolates had azithromycin MICs of 0.125–1.0 µg/ml. The proportion of GISP isolates with azithromycin MICs of ≥2.0 µg/ml peaked in 2020 at 5.8%. Site-specific data are presented in the Site Profiles section of this report.
Susceptibility to ciprofloxacin
Susceptibility testing for ciprofloxacin began in 1990. The distribution of ciprofloxacin MICs each year for the previous 5-year period is displayed in Figure 4 of the GISP National Profiles. Each year, approximately 35% of isolates exhibited ciprofloxacin MICs ≥1.0 µg/ml. Site-specific data on resistance to ciprofloxacin are presented in the Site Profiles section of this report.
