NMI Notes

April 2021
Overall Updates
NNDSS eSHARE Updates
- The next National Notifiable Diseases Surveillance System (NNDSS) eSHARE session is scheduled for Thursday, 4/29/2021, from 3:00 to 4:30 PM ET. Please note the date change and extended time frame.
- The April eSHARE webinar will provide important training for current and new users of the Message Validation, Processing, and Provisioning System (MVPS) Portal.
- At the beginning of May, MVPS will start processing data from all NNDSS case notification formats.
- New functionality in the MVPS Portal will allow users to view message and case count information from all message formats, verify cases for low-incidence conditions, and delete cases through the user interface.
- This webinar will be a valuable opportunity to learn how to use the new functionality and management of new user roles and permissions.
- Access past eSHARE presentations at the NNDSS eSHARE website.
COVID-19 Vaccination Information
- CDC encourages jurisdictions to send vaccine history information in coronavirus disease 2019 (COVID-19) case notifications using these options:
- Full COVID-19 message mapping guide (MMG),
- COVID Lite (generic v2 MMG plus vaccine history from COVID-19 MMG), or
- DCIPHER/HHS Protect CSV upload using the updated January 2021 CSV template.
- Find resources at the NNDSS Technical Resource Center.
COVID-19 Value Sets Updated
- CDC reminds jurisdictions to update their COVID-19 case notification value sets. CDC expects frequent updates for certain value sets.
- The most recently updated value sets include:
- Lab Test Type (COVID-19) v4 and
- Lab Test Interpretation (VPD) v6.
Core Requested Data Elements for COVID-19
- As resources allow, jurisdictions are encouraged to send additional case information through the CSV upload to DCIPHER or through the COVID-19 MMG.
NNDSS 2020 Annual Data Reconciliation
- Jurisdictions will reconcile data using MVPS.
- Jurisdictions will view and extract the line list and aggregate tables from MVPS for all NNDSS case notification methods:
- HL7 (including generic version 1 and varicella messages),
- National Electronic Telecommunications System for Surveillance (NETSS), and
- NEDSS Base System (NBS) master message.
- Jurisdictions can work at their own pace but must still meet the established deadline.
- The process for state epidemiologist sign-off will be in MVPS.
- Jurisdictions will view and extract the line list and aggregate tables from MVPS for all NNDSS case notification methods:
- Timeline: CDC recognizes the demands of the pandemic on jurisdictions and CDC resources; to accommodate this, the timeline for reconciliation of the 2020 data is delayed compared to a non-pandemic year.
- June 2021: Jurisdictions receive training and have access to line lists and aggregate tables in MVPS.
- September 30, 2021: Jurisdiction deadline for sign-off on final data.
Join a Cohort to Jumpstart Your MMG Implementations!
- The NNDSS Association of Public Health Laboratories Technical (APHL) assistance team provides technical assistance to help groups—also known as cohorts—of public health agencies build and implement NNDSS HL7 case notification messages.
- The APHL assistance team has established cohorts of four-six public health agencies focusing on the generic v2 and the COVID-19 MMGs.
- APHL and NBS subject matter experts guide participants through planning, gap analysis, data extraction, HL7 message creation, and validation. At completion, public health agencies will be ready to begin onboarding.
- For those interested in joining a cohort, please email the CDC Electronic Data Exchange mailbox at edx@cdc.gov.
Production HL7 Case Notification Messages
Arboviral v1.3
- CDC has posted the updated arboviral MMG v1.3.1 on the NNDSS Technical Resource Center.
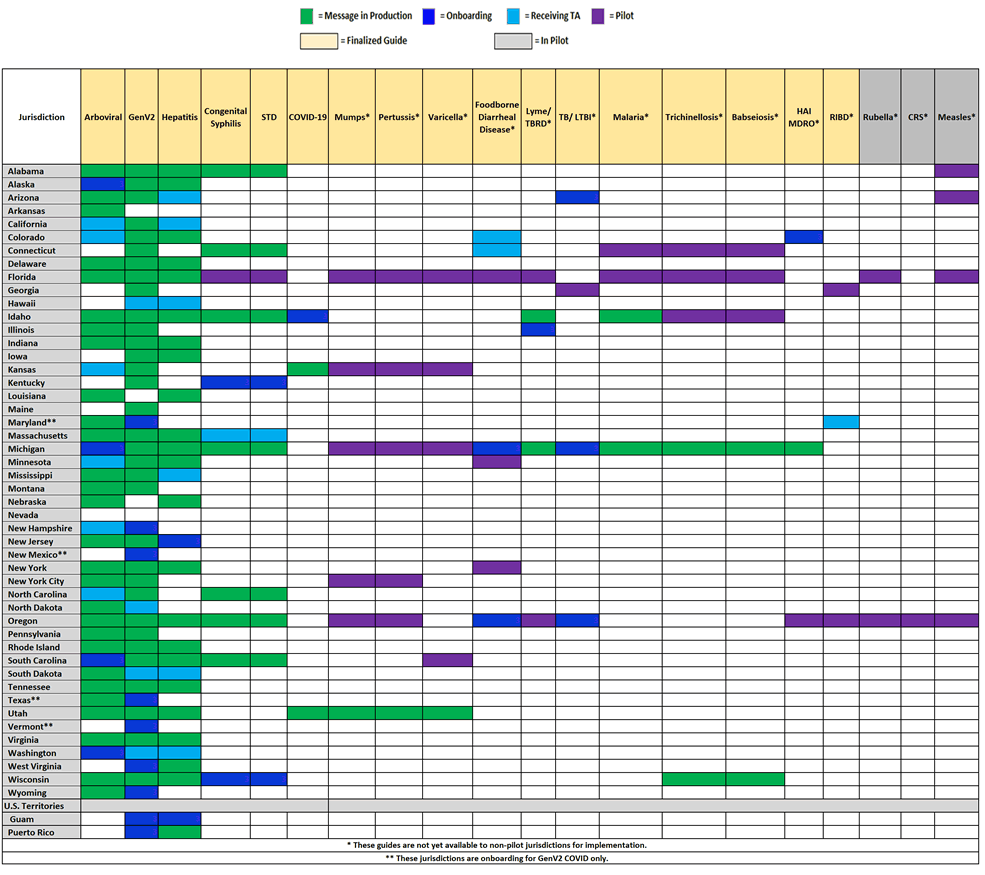
- There are 28 jurisdictions in production for arboviral.
- Alaska, Michigan, South Carolina, and Washington are engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Babesiosis
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- Please use this guide to begin implementing your case notification messages in preparation for onboarding.
- There are two jurisdictions in production for babesiosis. Wisconsin is the latest jurisdiction in production for babesiosis. Congratulations, Wisconsin!
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Congenital Syphilis and Sexually Transmitted Diseases
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- There are seven jurisdictions in production for congenital syphilis and sexually transmitted diseases.
- Kentucky and Wisconsin are engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Coronavirus Disease 2019
- MMG and Data Elements
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- Due to the evolving nature of COVID-19 vaccines and lab testing, the value sets that are affected will be updated regularly.
- The COVID-19 case notification vocabulary view in the PHIN Vocabulary Access and Distribution System (PHIN VADS) will be updated routinely to reflect these changes.
- Onboarding
- The COVID-19 MMG is open for onboarding, and the first cohort has begun onboarding. For those interested in implementing the COVID-19 MMG, please email edx@cdc.gov.
-
- There are two jurisdictions in production for COVID-19.
- Idaho is engaged in onboarding the COVID-19 MMG.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Foodborne and Diarrheal Diseases
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- Pilot jurisdictions Oregon and Michigan are engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Generic v2
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- There are 33 jurisdictions in production for generic v2.
- Guam, New Hampshire, Puerto Rico, West Virginia, and Wyoming are engaged in onboarding.
- Maryland, New Mexico, Texas, and Vermont are engaged in onboarding the COVID-19 condition for GenV2. Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Healthcare-Associated Infections, Multi-Drug Resistant Organisms (HAI MDRO)
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- Please use this guide to begin implementing your case notification messages in preparation for onboarding.
- There is one jurisdiction in production for HAI MDRO.
- Pilot jurisdiction Colorado is engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Hepatitis
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- There are 23 jurisdictions in production for hepatitis.
- Guam and New Jersey are engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Lyme and Tickborne Rickettsial Diseases (TBRD)
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- There are two jurisdictions in production for Lyme and TBRD.
- Pilot jurisdiction Illinois is engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Malaria
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- CDC expects to post minor updates to the MMG and artifacts and an FAQ document by late spring 2021.
- There are two jurisdictions in production for malaria.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Mumps and Pertussis
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- There is one jurisdiction in production for mumps and pertussis.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Respiratory Invasive Bacterial Disease
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
Trichinellosis
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- Please use this guide to begin implementing your case notification messages in preparation for onboarding.
- There are two jurisdictions in production for trichinellosis. Wisconsin is the latest jurisdiction in production for trichinellosis. Congratulations, Wisconsin!
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Tuberculosis and Latent Tuberculosis Infection
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- Pilot jurisdictions Arizona, Michigan, and Oregon are engaged in onboarding.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Varicella
- The MMG and artifacts are available on the NNDSS Technical Resource Center.
- There is one jurisdiction in production for varicella.
- Please see the chart below, Jurisdiction Implementation of NNDSS HL7 Case Notification Messages, for more details.
Developing HL7 Case Notification Messages
Bacterial Special Pathogens (Stage I—Draft)
- Prioritization of response activities has delayed the external review comment period to gather jurisdiction feedback.
- CDC expects to post the four MMGs (anthrax, brucellosis, Hansen’s disease, and leptospirosis) for external review in mid-2021.
Carbon Monoxide Poisoning (Stage II—Pilot Test-ready)
- CDC has postponed the pilot start date.
Lead Poisoning (Requirements Analysis)
- No new updates at this time.
Listeriosis (Stage I—Draft)
- No new updates at this time.
Measles (Stage III—Final MMG)
- CDC anticipates posting the final guide and artifacts on the NNDSS Technical Resource Center in 2021.
Rubella and Congenital Rubella Syndrome (Stage III—Final MMG)
- CDC anticipates posting the final guides and artifacts on the NNDSS Technical Resource Center in 2021.
Resources
- NNDSS Technical Resource Center:
https://www.cdc.gov/nndss/trc/ - NNDSS Message Mapping Guides:
https://ndc.services.cdc.gov/message-mapping-guides/ - NNDSS eSHARE Monthly Webinar:
https://www.cdc.gov/nndss/trc/onboarding/eshare.html - NNDSS News:
https://www.cdc.gov/nndss/trc/news/
Jurisdiction Implementation of NNDSS HL7 Case Notification Messages
This chart shows the status of state public health departments who are piloting, receiving technical assistance, onboarding, or in production for HL7 case notification messages developed as part of the NNDSS Modernization Initiative.
Upcoming Events
- NMI eSHARE; 4/29/21, 3:00PM ET;
https://www.cdc.gov/nndss/trc/onboarding/eshare.html - CSTE 2021 Annual Conference, June 13-17, 2021;
https://www.csteconference.org/index.php/registration2021/
NMI Notes provides monthly news updates about the National Notifiable Diseases Surveillance System (NNDSS) Modernization Initiative (NMI). It is a collaboration by the Centers for Disease Control and Prevention (CDC), Council of State and Territorial Epidemiologists (CSTE), and Association of Public Health Laboratories (APHL).
If a colleague forwarded this issue to you, we encourage you to subscribe at this link to ensure that you receive future issues of NMI Notes.
Write: edx@cdc.gov Visit: www.cdc.gov/nndss/
www.cste.org
Ready to request technical assistance or onboarding?
Please contact edx@cdc.gov for more information.
Have questions or feedback on NMI Notes?
Email edx@cdc.gov.