March 2022
April 19, 2022, 3:00–4:00 PM ET: eSHARE webinar. CDC will announce the topic of the April 2022 eSHARE webinar in the April issue of Case Surveillance News.
June 19–23, 2022: Council of State and Territorial Epidemiologists (CSTE) Annual Conference, Louisville, Kentucky. Learn more

New format for 2022 weekly data on Data.CDC.gov
On February 22, 2022, the National Notifiable Diseases Surveillance System (NNDSS) transitioned weekly data publication for 2022 data on Data.CDC.gov to a single-table format to make it easier for users to extract NNDSS data. This change consolidates 42 weekly NNDSS tables on Data.CDC.gov into one table that includes all conditions and data published in the 2022 NNDSS Weekly WONDER tables. If you need technical support using the new table, visit the Socrata helpdesk.
NNDSS to begin reconciliation of 2021 annual data in June 2022
The CDC NNDSS team plans to begin annual reconciliation of 2021 data in June 2022. The process will take advantage of new reconciliation functionality in the Message Validation, Processing, and Provisioning System (MVPS).
COVID-19 data will not go through a reconciliation process. Instead, aggregate 2021 NNDSS COVID-19 data will be collected using an Epi Info web form similar to the one used for the 2020 COVID-19 data.

CDC reminds jurisdictions to:
- begin clean-up of your 2021 data, if you haven’t already started;
- work with CDC programs to finalize your case counts;
- use the MVPS portal to review your 2021 data for low-incidence conditions that require special verification (see the information below in “News You Can Use”); and
- send case notification updates to correct data as appropriate.
Stay tuned for more information about 2021 annual reconciliation coming soon!
Redirects ended for old NNDSS website
In April 2021, CDC launched a redesigned NNDSS website at cdc.gov/nndss. To help faciliate a smooth transition to the new website, CDC used URL redirects to automatically send all users to the new website.

CDC has now fully retired the previous NNDSS website, including ending the automatic URL redirects. Users who navigate to the old NNDSS website URL—such as users who have bookmarked old webpages or have clicked links in documents that still have the old URL—will now get an error message instead of being redirected to the new website.
The January 2022 Notice to Data Users at NNDSS Weekly Tables, Notice To Data Users (cdc.gov) summarizes old and new NNDSS website locations to help you find information on the new NNDSS website.
Remember to update your bookmarks and visit the new website to learn about case surveillance, access case definitions, and find other great surveillance resources!
Email surveillancepractice@cdc.gov if you have questions or comments about the new website.
New CDC grant provides opportunity to strengthen U.S. public health infrastructure, workforce, and data systems

CDC’s OE22-2203: Strengthening U.S. Public Health Infrastructure, Workforce, and Data Systems grant will provide funding to improve critical public health infrastructure needs. This investment, a key component of the American Rescue Plan funding, will help ensure that U.S. public health systems are ready to respond to public health emergencies like COVID-19 and meet the evolving and complex needs of the communities and populations they serve. Learn more at Workforce Resources for Organizations.
CDC will not hold an eSHARE webinar in March 2022. CDC will hold the next eSHARE training webinar on April 19, 2022, 3:00–4:00 PM ET.
To join an eSHARE webinar, please see your calendar invitation or contact the CDC Electronic Data Exchange mailbox at edx@cdc.gov for login information with the subject line “eSHARE invitation.”
You can find links to the latest event code list and value set, Morbidity and Mortality Weekly Report weeks calendars, and other helpful resources for case notifications at the “Event Codes and other surveillance resources” webpage in the NNDSS Technical Resource Center.
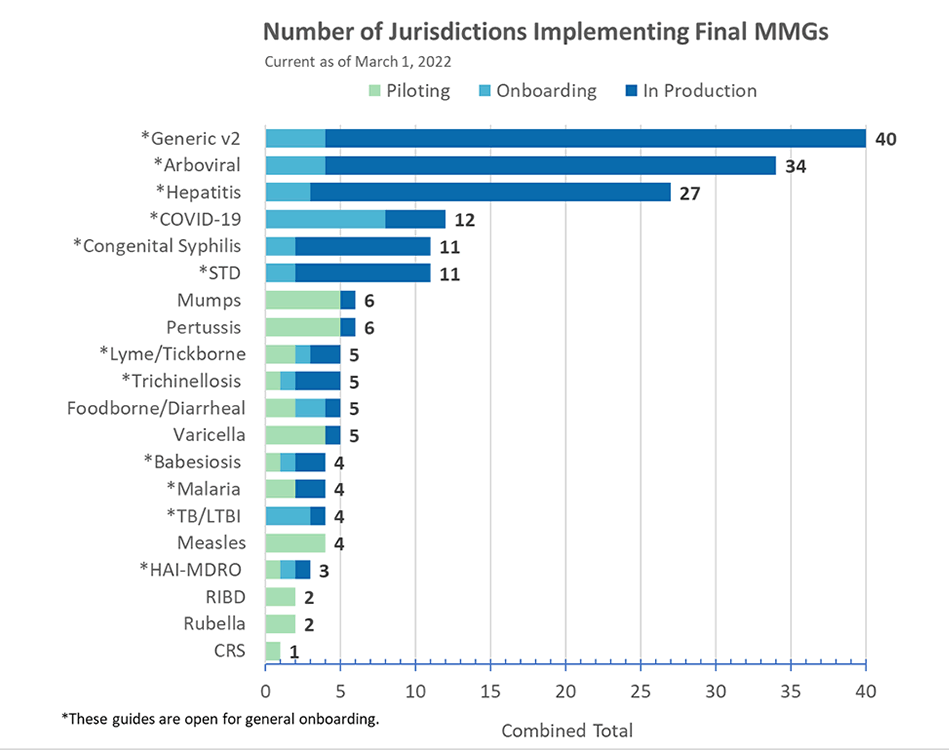
COVID-19 Message Mapping Guide (MMG)
The PHIN Vocabulary Access and Distribution System (VADS) COVID-19 Case Notification View version 12 is now available for COVID-19 MMG version 1.0 and version 1.1. The changes in version 12 include:
- three additional values for Lab Test Type (COVID-19),
- one additional value for Vaccine Type (NND), and
- three additional values and changes to preferred concept names for Lab Test Interpretation (VPD).
Healthcare-Associated Infections, Multi-Drug Resistant Organisms (HAI-MDRO) MMG
HAI-MDRO is now open for general onboarding. HAI-MDRO MMG version 1.0.2 contains formatting updates and the most up-to-date value sets for International Destination(s) of Recent Travel (82764-2) and Specimen Type (66746-9). All value sets associated with this guide are in the PHIN VADS HAI MDRO Case Notification View version 2.
If you are interested in joining a cohort or onboarding an MMG, please email the CDC Electronic Data Exchange mailbox at edx@cdc.gov.
Spotlight: Job aid for verifying low-incidence conditions
Data for low-incidence, national notifiable conditions require verification before being published in the NNDSS tables on the CDC Wide-ranging Online Data for Epidemiologic Research (WONDER) platform.
In August 2021, CDC released new features in MVPS to facilitate the verification of cases of low-incidence conditions. The CDC NNDSS team developed a job aid for jurisdictions with instructions for how to use the MVPS portal to verify low-incidence cases. Learn how to get access to job aids on MVPS.
Conditions that need low-incidence verification
Some low-incidence conditions need verification by both the jurisdiction and CDC program to determine whether to publish the case, while other low-incidence conditions require only verification from the jurisdiction.
These conditions require only jurisdiction approval to publish the case:
- Diphtheria;
- Hantavirus infection, non-hantavirus pulmonary syndrome;
- Hantavirus pulmonary syndrome;
- Rabies, human; and
- Rubella, congenital syndrome.
These conditions require jurisdiction and CDC program approval to publish the case:
- Anthrax;
- Cholera (toxigenic Vibrio cholerae O1 or O139);
- Novel influenza A virus infections;
- Plague;
- Poliomyelitis, paralytic;
- Poliovirus infection, nonparalytic;
- Severe acute respiratory syndrome (SARS)-associated coronavirus disease (SARS-CoV);
- Smallpox;
- Vancomycin-resistant Staphylococcus aureus (VRSA);
- Viral hemorrhagic fevers resulting from infection with Ebola virus, Chapare virus, Crimean-Congo hemorrhagic fever virus, Guanarito virus, Junín virus, Lassa virus, Lujo virus, Machupo virus, Marburg virus, and Sabia virus; and
- Yellow fever.
Cases that meet three criteria will be displayed in the MVPS “Low Incidence” section:
- the condition needs low-incidence case verification,
- the condition is reportable by the jurisdiction in the year it was reported, and
- the case meets the case classification status criteria for publication in the NNDSS tables.
If a case meets the criteria above, MVPS will send automated emails and periodic reminders to jurisdictions and CDC programs letting them know they need to verify the case.
How MVPS determines whether to publish a case
Once both the jurisdiction and the CDC program respond to a low-incidence verification request, MVPS uses an algorithm to determine whether to publish a case in the WONDER tables. The full algorithm is available on page 7 of the low-incidence verification job aid (registration required for access).
If CDC and a jurisdiction disagree about whether to publish a case, MVPS will use the jurisdiction’s response to decide whether to publish. In some situations, jurisdictions or CDC programs may indicate that they need more time to investigate a case or to discuss a case before providing a final response.
For conditions where program response is not required, MVPS will apply the algorithm once the jurisdiction provides a verification response.