Spike Gene Target Amplification in a Diagnostic Assay as a Marker for Public Health Monitoring of Emerging SARS-CoV-2 Variants — United States, November 2021–January 2023
Weekly / February 3, 2023 / 72(5);125–127
On January 25, 2023, this report was posted online as an MMWR Early Release.
Heather M. Scobie, PhD1; Akilah R. Ali, MPH1; Philip Shirk, PhD1; Zachary R. Smith, MA2; Prabasaj Paul, PhD3; Clinton R. Paden, PhD1; Norman Hassell, MS1; Xiao-yu Zheng, PhD1; Anastasia S. Lambrou, PhD1,4; Rebecca Kondor, PhD1; Duncan MacCannell, PhD3; Natalie J. Thornburg, PhD1; Joseph Miller, PhD2; Dave Wentworth, PhD1; Benjamin J. Silk, PhD1 (View author affiliations)
View suggested citationAltmetric:
Monitoring emerging SARS-CoV-2 lineages and their epidemiologic characteristics helps to inform public health decisions regarding vaccine policy, the use of therapeutics, and health care capacity. When the SARS-CoV-2 Alpha variant emerged in late 2020, a spike gene (S-gene) deletion (Δ69–70) in the N-terminal region, which might compensate for immune escape mutations that impair infectivity (1), resulted in reduced or failed S-gene target amplification in certain multitarget reverse transcription–polymerase chain reaction (RT-PCR) assays, a pattern referred to as S-gene target failure (SGTF) (2). The predominant U.S. SARS-CoV-2 lineages have generally alternated between SGTF and S-gene target presence (SGTP), which alongside genomic sequencing, has facilitated early monitoring of emerging variants. During a period when Omicron BA.5–related sublineages (which exhibit SGTF) predominated, an XBB.1.5 sublineage with SGTP has rapidly expanded in the northeastern United States and other regions.
As part of the Increasing Community Access to Testing (ICATT) program,* specimens collected at a national pharmacy chain were tested at a commercial laboratory that exclusively used the TaqPath COVID-19 Combo Kit (ThermoFisher Scientific) (3). Real-time RT-PCR cycle threshold (Ct) results for three gene targets (S, N, and ORF1ab) were reported to U.S. Department of Health and Human Services (HHS) Protect† during November 1, 2021–January 14, 2023. The proportion of SGTF or SGTP§ (2) results was calculated weekly at the national and HHS regional levels¶; SGTF data were reported on a public dashboard.** CDC also collects genomic sequencing data from the National SARS-CoV-2 Strain Surveillance program,†† contracted commercial laboratories, and partners that label sequencing results in public repositories as baseline surveillance (4). Sequencing data are used to calculate variant proportions, which are published weekly on CDC’s COVID Data Tracker.§§ Genomic sequencing results lag 2–3 weeks behind specimen collection, which necessitates nowcasting estimates (4) for the most recent 3 weeks (December 25, 2022–January 14, 2023). Geographic representativeness and median interval from specimen collection to result were calculated for both data sources. Trends were assessed in SGTP proportions, variant proportions, and nowcast estimates; all were weighted to represent RT-PCR–positive specimens by state (4). Genomic sequencing results, including for a random sample of ICATT specimens, were assessed by SGTF/SGTP status. This activity was conducted consistent with applicable federal law and CDC policy.¶¶
During November 1, 2021–December 24, 2022, national weekly SGTF and SGTP results ranged from 3,104 to 83,805 (median = 102; IQR = 327 per jurisdiction***) and genomic sequencing results ranged from 6,313 to 69,280 (median = 195; IQR = 460 per jurisdiction†††). During December 25, 2022–January 14, 2023, the national weekly average number of SGTF/SGTP and sequencing results were 5,005 and 847, respectively. After specimen collection, SGTF/SGTP results were available sooner (median = 2 days; IQR = 1) than were genomic sequencing results (median = 16 days; IQR = 10).
Trends in SGTP proportions aligned with genomic sequencing results classified by SGTF and SGTP (Figure). For the week ending December 24, 2022, the latest week that weighted variant proportions were available from genomic sequencing, SGTP lineages accounted for 21.5% (XBB.1.5 = 11.8%; XBB = 4.4%; other BA.2-related sequences = 5.3%) of genomic sequences, while the weighted SGTP estimate from ICATT was 20% (95% CI = 18%–23%). For the week ending January 14, 2023, the SGTP estimate from ICATT was 40% (95% CI = 36%–45%); SGTP lineages from sequencing comprised 50.6% (XBB.1.5 = 43.0%; XBB = 3.9%; other BA.2-related sequences=3.7%) of lineages in the nowcast projections reported on January 13, 2023 and 45.5% (XBB.1.5 = 37.2%; XBB = 4.0%; other BA.2-related sequences=4.3%) of lineages in revised nowcast projections for the same week, subsequently reported on January 20, 2023%.§§§ SGTP accounted for >50% of specimens in HHS regions 1–3 and >20% in all other regions, except Region 10, where the estimated precision was low (Supplementary Figure; https://stacks.cdc.gov/view/cdc/123810). Among genomic sequences from ICATT specimens collected through January 2, 2023, 412 (99%) of 415 XBB-related sequences exhibited SGTP; among those collected during December 1, 2022–January 2, 2023, 294 (59%) of 495 SGTP specimens were XBB-related lineages.
SGTF/SGTP monitoring relies on diagnostic RT-PCR, which is less expensive, permits higher throughput, and faster turnaround of results than sequencing. Using SGTF/SGTP for early studies of emerging variants obviates the need to wait for sequencing results or >50% variant predominance. Limitations are that SGTF/SGTP monitoring is assay-dependent; presumes SARS-CoV-2 lineage classification, requiring further validation by genomic surveillance; cannot discriminate mutations beyond Δ69–70 (i.e., BA.2-related sequences or even between BA.4 and BA.5); and relies on continued changing SGTF/SGTP patterns compared with predominant lineages. SARS-CoV-2 sequencing remains the standard for genomic surveillance because it allows definitive classification of viral lineages and identification of emerging strains for further characterization.
When early nowcast estimates of rapidly emerging variants lacked precision and geographic resolution because of lags in genomic sequencing results, SGTF/SGTP estimates were used as complementary data by CDC and the SARS-CoV-2 Interagency Group to support guidance on the use of monoclonal antibody therapies.¶¶¶ SGTF/SGTP data were also used as proxy markers in several early studies of vaccine effectiveness and severity of emerging variants (3,5,6). Continued monitoring of SGTF/SGTP patterns will likely serve as a useful complement to genomic surveillance of SARS-CoV-2 lineages.
Acknowledgments
Summer Galloway, CDC; Strain Surveillance and Emerging Variants Bioinformatics and NS3 Working Groups, CDC; teams at Aegis Sciences Lab and Walgreens Co.
Corresponding author: Heather M. Scobie, hscobie@cdc.gov.
1National Center for Immunization and Respiratory Diseases, CDC; 2Center for Preparedness and Response, CDC; 3National Center for Emerging and Zoonotic Infectious Diseases, CDC; 4Epidemic Intelligence Service, CDC.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No other potential conflicts of interest were disclosed.
* https://www.cdc.gov/icatt/AboutICATT.html
† CDC’s HHS Protect is a secure decision-making and operations platform for the whole-of-government response to the COVID-19 pandemic and serves to collect, integrate, and share COVID-19 data across federal agencies and with state, local, territorial, and tribal partners. https://www.cdc.gov/ncezid/hhs-protect/index.html
§ Specimens with missing Ct values for N or ORF1ab were excluded. SGTF was defined as a SARS-CoV-2-positive specimen with amplification of the N and ORF1ab genes along with either a failure or reduced amplification of the S gene (S Ct value >4 cycles from the average of N and ORF1ab Ct values). SARS-CoV-2–positive specimens that were not SGTF were considered SGTP.
¶ https://www.hhs.gov/about/agencies/iea/regional-offices/index.html
** At the time of publication, the dashboard is not showing the SGTF proportions, but this functionality is expected to return in the near future. https://www.walgreens.com/businesssolutions/covid-19-index.jsp
†† https://www.cdc.gov/coronavirus/2019-ncov/variants/cdc-role-surveillance.html
§§ https://covid.cdc.gov/covid-data-tracker/#variant-proportions
¶¶ 45 C.F.R. part 46.102(l)(2), 21 C.F.R. part 56; 42 U.S.C. Sect.241(d); 5 U.S.C.0 Sect.552a; 44 U.S.C. Sect. 3501 et seq.
*** Forty-seven states, the District of Columbia, and Puerto Rico.
††† Fifty states, the District of Columbia, and five territories (American Samoa, Guam, Northern Mariana Islands, Puerto Rico, and the U.S. Virgin Islands).
§§§ For data on variant proportions for the week ending January 14, 2023, nowcast estimates and 95% prediction intervals (95% PI) reported on the January 13, 2023, were 43.0% (26.4%–61.1%) for XBB.1.5 and 3.9% (95% PI = 3.0%–5.1%) for XBB. For the same week, nowcast estimates reported on January 20, 2023, were 37.2% (95% PI = 26.8%–49.0%) for XBB.1.5 and 4.0% (95% PI = 3.3%–4.7%) for XBB. Composite prediction intervals for other BA.2-related lineages were unavailable.
¶¶¶ https://www.cdc.gov/coronavirus/2019-ncov/variants/variant-classifications.html
References
- Meng B, Kemp SA, Papa G, et al.; COVID-19 Genomics UK (COG-UK) Consortium. Recurrent emergence of SARS-CoV-2 spike deletion H69/V70 and its role in the Alpha variant B.1.1.7. Cell Rep 2021;35:109292. https://doi.org/10.1016/j.celrep.2021.109292 PMID:34166617
- Clark C, Schrecker J, Hardison M, Taitel MS. Validation of reduced S-gene target performance and failure for rapid surveillance of SARS-CoV-2 variants. PLoS One 2022;17:e0275150. https://doi.org/10.1371/journal.pone.0275150 PMID:36190984
- Accorsi EK, Britton A, Fleming-Dutra KE, et al. Association between 3 doses of mRNA COVID-19 vaccine and symptomatic infection caused by the SARS-CoV-2 Omicron and Delta variants. JAMA 2022;327:639–51. https://doi.org/10.1001/jama.2022.0470 PMID:35060999
- Lambrou AS, Shirk P, Steele MK, et al.; Strain Surveillance and Emerging Variants Bioinformatic Working Group; Strain Surveillance and Emerging Variants NS3 Working Group. Genomic surveillance for SARS-CoV-2 variants: predominance of the Delta (B.1.617.2) and Omicron (B.1.1.529) variants—United States, June 2021–January 2022. MMWR Morb Mortal Wkly Rep 2022;71:206–11. https://doi.org/10.15585/mmwr.mm7106a4 PMID:35143464
- Link-Gelles R, Ciesla AA, Roper LE, et al. Early estimates of bivalent mRNA booster dose vaccine effectiveness in preventing symptomatic SARS-CoV-2 infection attributable to Omicron BA.5- and XBB/XBB.1.5-related sublineages among immunocompetent adults—Increasing Community Access to Testing Program, United States, December 2022–January 2023. MMWR Morb Mortal Wkly Rep 2023;72. https://www.cdc.gov/mmwr/volumes/72/wr/mm7205e1.htm?s_cid=mm7205e1_w
- Lewnard JA, Hong VX, Patel MM, Kahn R, Lipsitch M, Tartof SY. Clinical outcomes associated with SARS-CoV-2 Omicron (B.1.1.529) variant and BA.1/BA.1.1 or BA.2 subvariant infection in Southern California. Nat Med 2022;28:1933–43. https://doi.org/10.1038/s41591-022-01887-z PMID:35675841
 FIGURE. Trends in estimated proportions of SARS-CoV-2 reverse transcription–polymerase chain reaction test results with S-gene target presence and variant proportions and nowcast projections from genomic surveillance classified by S-gene target presence or S-gene target failure* — United States, November 1, 2021–January 14, 2023
FIGURE. Trends in estimated proportions of SARS-CoV-2 reverse transcription–polymerase chain reaction test results with S-gene target presence and variant proportions and nowcast projections from genomic surveillance classified by S-gene target presence or S-gene target failure* — United States, November 1, 2021–January 14, 2023
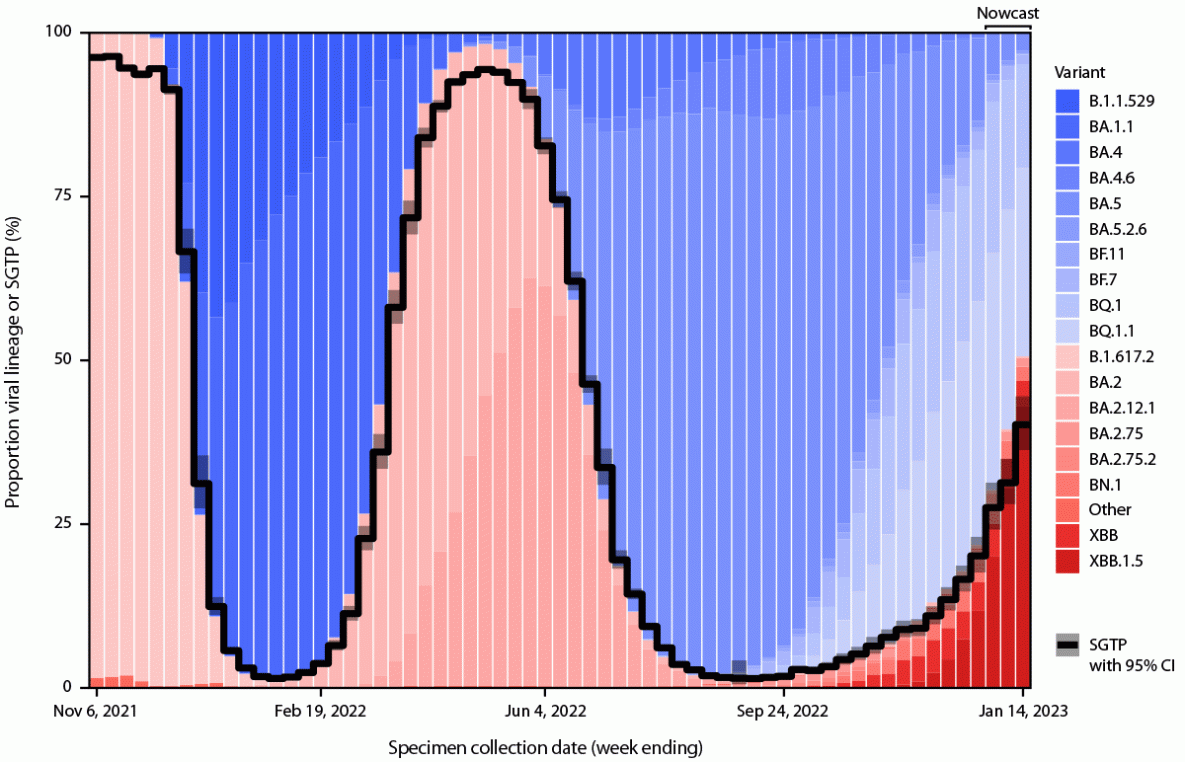
Abbreviations: S-gene = spike gene; SGTF = S-gene target failure; SGTP = S-gene target presence.
* Estimates of variant proportions and nowcast projections (for the most recent 3 weeks) are shown. The Delta (B.1.617.2) variant exhibited SGTP; the Omicron (B.1.1.529) variant and BA.1.1 sublineage exhibited SGTF; the Omicron BA.2 and BA.2.12.1 sublineages exhibited SGTP; Omicron BA.4 and BA.5 sublineages (which have the same spike sequence), BA.4-related (BA.4.6) and BA.5-related sublineages (BA.5.2.6, BQ.1, BQ.1.1, BF.7, and BF.11) exhibited SGTF; and BA.2-related sublineages (BA.2.75, BA.2.75.2, BN.1, XBB, and XBB.1.5) exhibited SGTP. The spike deletion (Δ69-70) that results in SGTF is not 100% penetrant in a lineage; SGTF/SGTP classification was made based on a 50% threshold. Most BA.2-related sublineages exhibit >99% SGTP.
Suggested citation for this article: Scobie HM, Ali AR, Shirk P, et al. Spike Gene Target Amplification in a Diagnostic Assay as a Marker for Public Health Monitoring of Emerging SARS-CoV-2 Variants — United States, November 2021–January 2023. MMWR Morb Mortal Wkly Rep 2023;72:125–127. DOI: http://dx.doi.org/10.15585/mmwr.mm7205e2.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.