Acute Rheumatic Fever
Acute rheumatic fever is a delayed sequela of pharyngitis and skin infections due to Streptococcus pyogenes, which are also called group A Streptococcus (group A strep). The etiology, clinical features, diagnosis and treatment options, prognosis and complications, and prevention are described below.
Etiology
Acute rheumatic fever is a nonsuppurative, delayed sequela of pharyngitis and skin infections, such as impetigo, due to S. pyogenes. The exact disease process is not fully known. However, the disease is in part due to an autoimmune response to S. pyogenes infection involving multiple organ systems. Organ systems involved typically include the heart, joints, and central nervous system. Streptococcal pharyngitis typically precedes the onset of acute rheumatic fever by 1 to 5 weeks.1
S. pyogenes are gram-positive cocci that grow in chains (see Figure 1). They exhibit β-hemolysis (complete hemolysis) when grown on blood agar plates. They belong to group A in the Lancefield classification system for β-hemolytic Streptococcus, and thus are also called group A streptococci.
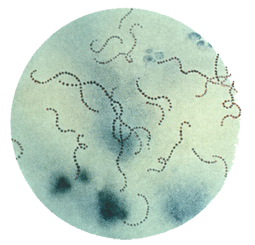
Figure 1. Streptococcus pyogenes (group A Streptococcus) on Gram stain. Source: Public Health Image Library, CDC
Clinical features
Acute rheumatic fever is usually characterized by fever and affects multiple organ systems.
Cardiovascular system
Carditis is the major cardiac manifestation of acute rheumatic fever, occurring in 50% to 70% of first episodes, and is associated with valvulitis. Clinical signs of carditis include cardiomegaly, new onset heart murmur (usually with mitral or aortic valvular disease), pericardial friction rub, pericardial effusion, and congestive heart failure. Additionally, a prolonged PR interval can be seen on electrocardiography.
Subclinical carditis may also be present. In these cases, classic murmurs may not be appreciated on physical exam, and valve disease is found solely by echocardiography/Doppler studies.2,3 Carditis is the only manifestation that may result in long-term disability or death.
Musculoskeletal system
Polyarthritis is the major musculoskeletal manifestation. The arthritis is typically migratory and involves the following large joints: elbows, wrists, knees, and ankles. Joint involvement may range from general arthralgia to a painful, inflammatory arthritis.
Integumentary system
Subcutaneous nodules and erythema marginatum are the two major skin manifestations. Subcutaneous nodules are firm, painless, variable in size (typically between a few millimeters and 2 centimeters in diameter), and usually found over joint extensor surfaces. Nodules are most commonly present in patients with carditis.
Erythema marginatum is an erythematous, non-pruritic, non-painful macular lesion on the trunk or proximal extremities. Lesions are transient and tend to extend outward with central clearing and are often described as serpiginous.
Central nervous system
Chorea, also called Sydenham’s chorea or St. Vitus dance, is the major central nervous system manifestation. Chorea is a neurological disorder characterized by abrupt, purposeless, non-rhythmic, involuntary movements that is often associated with muscle weakness and emotional lability. Chorea often appears after the other manifestations of acute rheumatic fever. It also can appear as the only manifestation of acute rheumatic fever.1,4
Risk factors
Inadequate or lack of antibiotic treatment of streptococcal pharyngitis increases the risk of someone developing acute rheumatic fever. In approximately one-third of patients, acute rheumatic fever follows subclinical streptococcal infections or infections for which medical attention was not sought.1
Individuals with a history of acute rheumatic fever have an increased risk of recurrence with subsequent streptococcal pharyngeal infections.1,5 The risk of recurrence after streptococcal infection is highest within the first few years after the initial attack and then declines.
The incidence of acute rheumatic fever is highest in children between the ages of 5 and 15 years. Acute rheumatic fever is very rare in children 3 years of age and younger in the United States. First-onset acute rheumatic fever is rare in adults, although recurrence may occur through adulthood.5, 6
Crowding, such as found in schools, military barracks, and daycare centers, increases the risk of spreading group A strep and thus increases the risk of developing acute rheumatic fever.
Diagnosis and testing
The differential diagnosis of acute rheumatic fever is broad due to the various symptoms of the disease. The differential diagnosis may include but is not limited to: rheumatoid arthritis, juvenile idiopathic arthritis, septic arthritis, systemic lupus erythematosus, serum sickness, Lyme disease, infective endocarditis, viral myocarditis, Henoch-Schonlein purpura, gout, sarcoidosis, leukemia, and Hodgkin’s disease.1
There is no definitive diagnostic test for acute rheumatic fever. A clinical diagnosis of acute rheumatic fever should be made using the Jones Criteria. A 2015 revised version of the Jones Criteria endorsed by the American Heart Association now includes the addition of subclinical carditis as a major criteria and stratification of the major and minor criteria based upon epidemiologic risk (e.g., low, moderate, or high risk populations).2
Diagnosis of initial acute rheumatic fever illness
The presence of 2 major manifestations, or 1 major and 2 minor manifestations (see below), indicates a high probability of an initial acute rheumatic fever illness in any risk population. More than one joint and more than one cardiac manifestation can only be classified as either one major or one minor criteria, not both. For example, if there is evidence of carditis (a major criteria), a prolonged PR interval should not also be counted as a minor criteria. Similarly, if there is evidence of arthritis (a major criteria), then arthralgia should not also be counted as a minor criteria.
In most cases, there should also be evidence of preceding group A streptococcal infection.2 Evidence to support an antecedent group A strep infection include:
- Positive throat culture or rapid streptococcal antigen test
- Elevated or rising streptococcal antibody titer
Table: Revised Jones Criteria for diagnosing acute rheumatic fever
| Part A. For all patient populations with evidence of preceding group A strep infection | ||
|---|---|---|
| Diagnosis: Initial ARF |
2 major manifestations or 1 major plus 2 minor manifestations
|
|
| Diagnosis: Recurrent ARF |
2 major manifestations or 1 major plus 2 minor manifestations or 3 minor manifestations
|
|
| Low-risk populations* | Moderate- and high-risk populations* | |
| Part B. Major manifestations | Carditis
Arthritis
Chorea Erythema marginatum Subcutaneous nodules |
Carditis
Arthritis
Chorea Erythema marginatum Subcutaneous nodules |
| Part C. Minor manifestations | Polyarthralgia
Fever (≥38.5oC) Elevated acute phase reactants (ESR ≥60 mm in the first hour and/or CRP ≥3.0 mg/dl) Prolonged PR interval on electrocardiography, after accounting for age variability (unless carditis is a major criterion) |
Monoarthralgia
Fever (≥38oC) Elevated acute phase reactants (ESR ≥30 mm/hr and/or CRP >3.0 mg/dl) Prolonged PR interval on electrocardiography, after accounting for age variability (unless carditis is a major criterion) |
ARF = acute rheumatic fever; ESR = erythrocyte sedimentation rate; CRP = C-reactive protein; mm = millimeters; mg/dl = milligrams per deciliter
*Low-risk population is defined as an acute rheumatic fever incidence of <2 per 100,000 school-aged children or all age rheumatic heart disease prevalence of ≤1 per 1000 population per year. Those not included in the low-risk population are defined as moderate or high risk depending upon their reference population.
In addition, routine echocardiography/Doppler is now recommended for all confirmed or suspected acute rheumatic fever cases regardless of the presence or absence of murmur on physical exam.2 A heart murmur without echocardiographic evidence of carditis and other clinical signs or symptoms can exclude the presence of rheumatic carditis.2
In some instances, a presumptive diagnosis of acute rheumatic fever can be made without fulfilling the Jones Criteria. For example, when clinical evidence is lacking in areas of high acute rheumatic fever incidence, clinical judgment must be used regarding the appropriate diagnosis and use of antibiotic prophylaxis.2 In addition, acute rheumatic fever can be considered in cases of chorea and indolent, chronic carditis despite the lack of group A streptococcal laboratory confirmation or fulfillment of Jones Criteria.2
Diagnosis of recurrent disease
Individuals with a history of rheumatic heart disease or prior episode of acute rheumatic fever are at increased risk for recurrences of acute rheumatic fever. In these individuals, a presumptive diagnosis of a recurrence can be made in the presence of a documented group A streptococcal infection with any of the following:2
- 2 major manifestations
- 1 major and 2 minor manifestations
- 3 minor manifestations.
See Table above for additional details.
If relying on the presence of 3 minor manifestations, the diagnosis of recurrent acute rheumatic fever should only be made if other more likely causes have been excluded.
Treatment
Patients with acute rheumatic fever should start on therapy for the symptomatic management of acute rheumatic fever, including salicylates and anti-inflammatory medicines to relieve inflammation and decrease fever, as well as management of cardiac failure. These patients should also be started on antibiotics for treatment of group A strep, regardless of the presence or absence of pharyngitis at the time of diagnosis, in order to eliminate any residual group A strep Streptococcus.5
Prognosis and complications
Rheumatic heart disease is the most important long-term sequela of acute rheumatic fever due to its ability to cause disability or death.1 Untreated rheumatic fever increases a person’s risk of recurrent attacks and worsens prognosis. Prognosis is related to the prevention of recurrent attacks, degree of cardiac valvular damage, and degree of overall cardiac involvement. Cardiac complications may vary in severity and include, but are not limited to, pericarditis, endocarditis, arrhythmias, valvular damage, and congestive heart failure.
Prevention
Diagnosis and adequate antibiotic treatment of group A strep pharyngitis is the primary means of preventing acute rheumatic fever. However, in about one-third of patients, acute rheumatic fever follows subclinical streptococcal infections or infections for which medical attention was not sought.1
Secondary prevention of rheumatic fever requires antibiotic prophylaxis to reduce the likelihood of recurrent attacks in persons with a history of acute rheumatic fever. Because acute rheumatic fever frequently recurs with subsequent group A strep pharyngitis infections, long-term prophylaxis duration should be individually tailored but is usually indicated at least until age 21. Prophylaxis typically involves an intramuscular injection of benzathine penicillin every 4 weeks or oral penicillin V twice daily. Sulfadiazine or oral macrolides can be taken daily by individuals who are allergic to penicillin.5,7 Current American Heart Association guidelines no longer recommend bacterial endocarditis prophylaxis for patients with rheumatic heart disease, unless the patient has a prosthetic valve.8
The spread of group A strep infection can be reduced by good hand hygiene, especially after coughing and sneezing and before preparing foods or eating, and respiratory etiquette (e.g., covering your cough or sneeze). Treating an infected person with an antibiotic for 12 hours or longer limits their ability to transmit the bacteria. Thus, people with group A strep pharyngitis or scarlet fever should stay home from work, school, or daycare until:
- They are afebrile
AND - At least 12-24 hours after starting appropriate antibiotic therapy*
Epidemiology and surveillance
The incidence of acute rheumatic fever has declined significantly in the developed world over the last century and now is significantly less common in the United States compared to less developed countries.9 Worldwide, however, rheumatic heart disease remains a significant cause of cardiovascular disease.
Global estimates of the burden of disease:
- 470,000 new cases of acute rheumatic fever each year10
- 282,000 new cases of rheumatic heart disease each year10
- 33.4 million cases of rheumatic heart disease11
- 305,000 people die each year from rheumatic heart disease or its complications12
The annual national incidence of acute rheumatic fever in the United States is largely unknown as it is no longer a nationally notifiable disease; however, there is likely significant regional variation. For instance, Hawaii and American Samoa have higher annual incidence rates than the continental United States, and may be as high as 1.1 to 1.5 cases per 1,000 population.13,14 The rate in Tennessee was estimated to be 0.6 cases per 100,000 population in 1977–1981.15 The rate in Utah, a state with a history of resurgences of acute rheumatic fever in the 1980s, has been estimated to be 3.7 cases per 100,000 population.16 Hospitalization data suggests that Asian/Pacific Islanders, Hispanics, blacks, and Native Americans were more likely to be hospitalized for acute rheumatic fever compared to white persons.17
Footnote
Per the American Academy of Pediatrics Red Book 2021–2024, children with group A strep pharyngitis should not return to school or a childcare setting until well appearing and at least 12 hours after beginning appropriate antibiotic therapy. In certain scenarios, such as an infection in a healthcare worker or in a group A strep outbreak setting, staying home for at least 24 hours after beginning appropriate antibiotics should be considered.
- Shulman ST, Bisno AL. Nonsupprative poststreptococcal sequelae: Rheumatic fever and glomerulonephritis. In Bennett J, Dolin R, Blaser M, editors. 8th Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. Philadelphia (PA): Elsevier. 2015;2:2300–9.
- Gewitz MH, Baltimore RS, Tani LY, et al. Revision of the Jones Criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography: A scientific statement from the American Heart Association. Circulation. 2015;131:1806.
- Reményi B, Wilson N, Steer A, et al. World Heart Federation criteria for echocardiographic diagnosis of rheumatic heart disease—An evidence-based guideline. Nat Rev Cardiol. 2012;9(5):297–309.
- Guidelines for the diagnosis of rheumatic fever. Jones Criteria, 1992 update. Special Writing Group of the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young of the American Heart Association. JAMA. 1992;268(15):2069–73. Erratum in: JAMA. 1993;269(4):476.
- Gerber MA, Baltimore RS, Eaton CB, et al. Prevention of rheumatic fever and diagnosis and treatment of acute streptococcal pharyngitis: A scientific statement from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, the Interdisciplinary Council on Functional Genomics and Translational Biology, and the Interdisciplinary Council on Quality of Care and Outcomes Research: Endorsed by the American Academy of Pediatrics. Circulation. 2009;119(11):1541–51.
- Shulman ST, Bisno AL, Clegg HW, Gerber MA, Kaplan EL, Lee G, et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis. 2012;55(10):1279–82.
- Committee on Infectious Diseases. Group A streptococcal infections. In Kimberlin DW, Barnett ED, Lynfield R, Sawyer MH, editors. 32nd ed. Red Book: 2021 Report of the Committee on Infectious Diseases. Elk Grove Village (IL): American Academy of Pediatrics; 2021:633–46.
- Wilson W, Taubert K, Gewitz M, et al. Prevention of Infective Endocarditis: Guidelines from the American Heart Association: A Guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007;116:1736–54.
- Carapetis JR, McDonald M, Wilson NJ. Acute rheumatic fever. Lancet. 2005;366(9480):155–68.
- Carapetis JR. The current evidence for the burden of group A streptococcal diseases. World Health Organization. Geneva. 2005.
- Watkins DA, Johnson CO, Colquhoun SM, et al. Global, regional, and national burden of rheumatic heart disease, 1990-2015. N Engl J Med. 2017;377(8):713-722.
- Global Burden of Disease Collaborative Network. Global Burden of Disease Study 2016 (GBD 2016) Results. Seattle, United States: Institute for Health Metrics and Evaluation (IHME), 2017, available at https://vizhub.healthdata.org/gbd-results/ (accessed 20 March 2018).
- Erdem G, Mizumoto C, Esaki D, et al. Group A streptococcal isolates temporally associated with acute rheumatic fever in Hawaii: Differences from the continental United States. Clin Infect Dis. 2007;45:e20–4.
- Beaudoin A, Edison L, Introcaso CE, Goh L, Marrone J, Mejia A, Van Beneden C. Acute rheumatic fever and rheumatic heart disease among children—American Samoa, 2011–2012. MMWR Morb Mortal Wkly Rep. 2015; 64(20);555–8.
- Land MA, Bisno AL. Acute rheumatic fever: A vanishing disease in suburbia. JAMA. 1983;249(7):895–98.
- Stockmann C, Ampofo K, Hersh AL, et al. Evolving epidemiologic characteristics of invasive group A streptococcal disease in Utah, 2002–2010. Clin Infect Dis. 2012;55(4):479–87.
- Miyake CY, Gauvreau K, Tani LY, Sundel RP, Newburger JW. Characteristics of children discharged from hospitals in the United States in 2000 with the diagnosis of acute rheumatic fever. Pediatrics. 2007;120(3):503–8.