Type II Necrotizing Fasciitis
Necrotizing fasciitis is a rapidly progressive infection that destroys deep soft tissues including muscle fascia and overlying subcutaneous fat. Multiple species of bacteria can cause necrotizing fasciitis. This page focuses on type II necrotizing fasciitis where Streptococcus pyogenes is isolated alone or in combination with another species.
Etiology
Type II necrotizing fasciitis, known as hemolytic streptococcal gangrene, is characterized by the isolation of S. pyogenes with or without other bacterial species. Staphylococcus aureus is the most common species found in co-infection cases.1
S. pyogenes are gram-positive cocci that grow in chains (see Figure 1). They exhibit β-hemolysis (complete hemolysis) when grown on blood agar plates. They belong to group A in the Lancefield classification system for β-hemolytic Streptococcus. Thus, these bacteria are also called group A Streptococcus or group A strep.
Streptococcal necrotizing fasciitis is a rare infection that typically occurs after trauma, sometimes minor or unapparent trauma, and surgery.2 Necrotizing fasciitis may also occur as a superinfection complicating varicella lesions.3
Clinical features
Necrotizing fasciitis can affect any part of the body, but most commonly affects extremities, particularly the legs. Necrotizing fasciitis begins with pain in the affected area accompanied by the following signs:
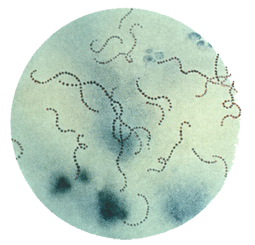
Figure 1. Streptococcus pyogenes (group A Streptococcus) on Gram stain. Source: Public Health Image Library, CDC
- Swelling
- Erythema
- Tenderness
- Heat
The pain experience by the patient is usually out of proportion to the signs of the local skin infection.3 Treating clinicians often underestimate the extent of tissue infected due to:3
- Spread within subcutaneous tissue with relative sparing of the overlying skin
- Absence of lymphadenitis and lymphangitis
Swelling progresses to brawny edema and then to dark-red induration. Within 24 to 48 hours, the overlying skin may turn dusky, indicating small vessels in the dermal papilla have thrombosed.3 Cutaneous ischemia also develops.3 Bullae form and are filled with straw colored fluid that progressively turns to hemorrhagic fluid. Affected tissues progressively darken from red to purple to blue to black. As gangrene sets in, skin becomes anesthetized due to destruction of superficial nerves.1
Indications of advanced disease include:3
- Bullae
- Ecchymosis
- Crepitus
- Anesthesia
- Necrosis
The lesions become sharply demarcated and, as skin sloughs, a necrotic eschar forms, resembling a third-degree burn.1 The patient may quickly progress to sepsis, shock, organ failure, and death. In the extremities, infection may progress to compartment syndrome, requiring emergent fasciotomy.3
With superinfection secondary to varicella in young children, obvious cutaneous signs of streptococcal infection may not be present.3 Three or four days after symptom onset, children may have return or persistence of high fever and appear toxic.3
Transmission
Necrotizing fasciitis can occur after both penetrating and non-penetrating traumas.
In necrotizing fasciitis, the initial entry of group A strep into the body can occur by several routes. Most patients do not remember preceding streptococcal infections. In non-penetrating trauma (e.g., bruises, muscle strain), the most likely mechanism is seeding from transient bacteremia.4 When penetrating trauma breaks the skin, bacteria may pass directly to the site of infection from the environment.4
Risk factors
Healthy people can get streptococcal necrotizing fasciitis. However, it is most commonly found in the setting of systemically or locally immunocompromised individuals.1,3 Classically, these are adults with underlying disease, such as diabetes mellitus, peripheral vascular disease, or neoplasm. In the past, retrospective reports linked nonsteroidal anti-inflammatory drugs to necrotizing fasciitis; however, prospective studies have not confirmed this linkage.1 Cirrhosis and corticosteroid therapy have also been shown to be predisposing factors.1 Furthermore, the streptococcal strain may influence the development of necrotizing fasciitis. The most commonly involved strains contain M protein types 1, 3, 12, and 28 (usually elaborating pyrogenic exotoxin A).1 Necrotizing fasciitis may also occur as a superinfection complicating varicella lesions.3
Diagnosis and testing
Differentiating cellulitis and necrotizing fasciitis can be difficult when presenting symptoms are non-specific (e.g., unexplained fever, pain, edema, erythema).1,4 Where there is low suspicion for necrotizing fasciitis, imaging may be helpful, but can delay diagnosis.1,5 Suspicion based upon clinical findings (e.g., profound pain, areas of decreased sensation, crepitation, bullae, skin necrosis) should lead to:1,5
- Prompt surgical exploration
- Gram stain
- Culture
Likewise, in patients whose systemic findings are out of proportion to local findings, surgical exploration or biopsy should be expeditious.5
Computed tomography (CT) scanning or magnetic resonance imaging (MRI) can detect subcutaneous and fascial edema or tissue gas.1 This information can be helpful early in the process when pain and swelling are present without cutaneous changes. However, these are not definitive studies and may delay the diagnosis. In the case of streptococcal necrotizing fasciitis, abscess formation or gas in the tissues are usually not seen on imaging.2 Muscle tear, hematoma, or prior surgery can complicate interpretation of imaging studies; therefore, these studies should never delay surgical exploration.2
Labs such as leukocytosis, thrombocytopenia, and azotemia are common. Gram stain can be highly informative to determine whether the etiology is group A strep.1,3
Treatment
Primary treatment of necrotizing fasciitis is prompt surgery and appropriate antibiotic therapy. Imaging studies should never delay surgical exploration.
Primary treatment of necrotizing fasciitis is early and aggressive surgical exploration and debridement of necrotic tissue.5 Surgery is coupled with appropriate broad-spectrum parenteral antibiotic therapy.3 Upon first exploration, extensive incisions that go beyond the area of apparent involvement are usually necessary. The wound should be left open and re-inspected 24 hours later to ensure adequacy of the initial debridement.1 Antibiotic therapy is based upon Gram stain findings. Once group A strep is confirmed to be the etiology, high dose penicillin or ampicillin and clindamycin (which interferes with toxin production) should be given.1 Intravenous immunoglobulin may be considered in cases of severe necrotizing fasciitis, although efficacy has not been proven.5
Prognosis and complications
In the most recent five years, the mortality rate of necrotizing fasciitis ranges from 11% to 22%.1 Coincident necrotizing fasciitis and streptococcal toxic shock syndrome (STSS) have a mortality rate ranging from 16% – 33%.3 Extensive surgical debridement and amputations are not uncommon.
Prevention
CDC does not recommend chemoprophylaxis or routine screening of household contacts of individuals with confirmed invasive group A streptococcal infections.6 Certain groups are at increased risk of sporadic invasive group A strep disease. In addition, people 65 years or older are at increased risk of death if they get an invasive infection. For this reason, healthcare providers may choose to offer chemoprophylaxis to all members of households of individuals with invasive group A strep infection with elderly household contacts or otherwise high-risk household contacts.6
Epidemiology and surveillance
CDC tracks necrotizing fasciitis caused by group A strep with a special system called Active Bacterial Core surveillance (ABCs). Since 2010, approximately 700 to 1,150 cases occur each year in the United States. This is likely an underestimate. According to ABCs data, the number of annual group A strep necrotizing fasciitis infections reported to ABCs does not appear to be rising.
- Pasternack MS, Swartz MN. Cellulitis, necrotizing fasciitis, and subcutaneous tissue infections. In Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. Philadelphia (PA): Churchill Livingstone Elsevier; 2015:1:1195–216.
- Bryant AE, Stevens DL. Streptococcus pyogenes. In Bennett J, Dolin R, Blaser M, editors. 8th Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. Philadelphia (PA): Elsevier; 2015:2:2285–300.
- Morelli JG. Subcutaneous tissue infections. In Kliegman RM, Stanton B, Geme JWS, Schor NF, Behrman RE, editors. Nelson Textbook of Pediatrics. Philadelphia (PA): Elsevier Saunders; 2011:2299–308.
- Stevens DL. Infections of the skin, muscles, and soft tissues. In Kasper D, Fauci A, Hauser S, et al, editors. 19th Harrison’s Principles of Internal Medicine. New York (NY): McGraw-Hill; 2015. Accessed December 02, 2015.
- Committee on Infectious Diseases. Group A streptococcal infections. In Kimberlin DW, Barnett ED, Lynfield R, Sawyer MH, editors. 32nd ed. Red Book: 2021 Report of the Committee on Infectious Diseases. Elk Grove Village (IL): American Academy of Pediatrics. 2021:633–46.
- Prevention of Invasive Group A Streptococcal Infections Workshop Participants. Prevention of invasive group A streptococcal disease among household contacts of case patients and among postpartum and postsurgical patients: Recommendations from the Centers for Disease Control and Prevention. Clin Infect Dis. 2002;35(8):950–9. Erratum in: Clin Infect Dis. 2003;36(2):243.