Key points
- Never delay or withhold treatment pending the receipt of laboratory test results, or an initially negative result. Early recognition and prompt treatment with doxycycline is critical.
- Polymerase chain reaction on whole blood is best for confirming anaplasmosis if testing is done in the first week of illness.
- Always take a thorough patient history, including information on recent tick bites, exposure to areas where ticks are found, and travel history.
- Anaplasmosis is a nationally notifiable condition.

Clinical diagnosis
Early recognition and treatment
- Prevent progression to severe illness through early recognition and prompt treatment.
- Anaplasmosis can be difficult to diagnose, particularly in the early stages of illness.
- Treatment should be started as soon as anaplasmosis is suspected without waiting for confirmatory laboratory testing.
- Maintain a high level of clinical suspicion for anaplasmosis or other tickborne diseases in cases of non-specific febrile illness. Be alert during spring and summer months when ticks are most active.
Patient history
Always thoroughly review patient history, including:
- History of recent tick bite. Many people do not remember being bitten. Do not rule out a tickborne infection if your patient does not remember a tick bite.
- Exposure to areas where ticks are commonly found, including wooded areas or brushy areas with high grasses and leaves. This includes around the home activities such as gardening.
- Travel history to areas where anaplasmosis is endemic.
Laboratory confirmation
Laboratory confirmation is helpful for disease surveillance and understanding burden of anaplasmosis infection in the United States.
Diagnostic methods video
The following video provides information on rickettsial disease diagnostic methods for healthcare providers, including what tests are available and when it is most appropriate to collect samples. It focuses on the use of polymerase chain reaction (or PCR) tests, and the indirect fluorescent antibody (IFA) test for rickettsial disease diagnosis.
Laboratory diagnosis
- Testing for anaplasmosis should be considered for any person with a compatible illness especially in endemic regions and for those with known risk factors, such as history of a tick bite or exposure to tick habitat.
- Anaplasmosis is a nationally notifiable condition. Testing for and reporting of cases of anaplasmosis is important to improve our understanding of disease prevalence, where it occurs, and how the incidence and geographic distribution change over time.
Recommended tests
- PCR is performed on whole blood specimens.
- This method is most sensitive in the first week of illness and decreases in sensitivity within 48 hours of administration of appropriate antibiotics.
- Although a positive PCR result should be treated as clear evidence of active infection, a negative result does not rule out the diagnosis. Treatment should not be withheld due to a negative result if anaplasmosis is suspected.
- PCR can be used to identify anaplasmosis from solid tissue and bone marrow specimens.
- The reference standard serologic test for diagnosis of anaplasmosis is the indirect fluorescent antibody (IFA) test for immunoglobulin G (IgG) using Anaplasma phagocytophilum antigen.
- IgG IFA tests should be performed on paired acute and convalescent serum samples, with the acute specimen collected during the first two weeks of illness and the convalescent collected 2-10 weeks later. Seroconversion is defined as a four-fold increase in titer from the convalescent compared to acute.
- Antibody titers determined using immunoglobulin G (IgG) IFA tests are frequently negative (titer of less than 1:64) in the first week of illness. Anaplasmosis cannot be confirmed using single acute antibody results. A single IgG titer of 1:128 or greater is classified as supportive laboratory evidence.
- Immunoglobulin M (IgM) is not a reliable method for the diagnosis of anaplasmosis and should not be used as an indicator of recent infection.
- Antibodies to A. phagocytophilum might remain elevated for many months after the disease has resolved. Persistently elevated IgG titers should not prompt additional treatment in the absence of new symptoms.
- In certain people, high titers of antibodies against A. phagocytophilum have been observed up to four years after the acute illness.
- Between 5–10% of healthy people in some areas might have elevated antibody titers due to past exposure to A. phagocytophilum or similar organisms.
- Seroconversion provides the best evidence of recent infection.
Cross Reactivity
- Closely related organisms, including those in the Ehrlichia and Anaplasma genera, share similar antigens such that antibodies directed to one of these antigens can cross-react.
- Most commercial labs are unable to differentiate between Anaplasma species.
- In areas endemic for ehrlichiosis and anaplasmosis, IFA using antigen from both Ehrlichia and Anaplasma species should be run side-by-side.
- Culture isolation and IHC assays of A. phagocytophilum are only available at specialized laboratories; routine hospital blood cultures cannot detect the organism.
- PCR, culture, and IHC assays can also be applied to autopsy tissue specimens.
- If a bone marrow biopsy is performed as part of the investigation of cytopenia, immunostaining of the bone marrow biopsy specimen can diagnose A. phagocytophilum infection.
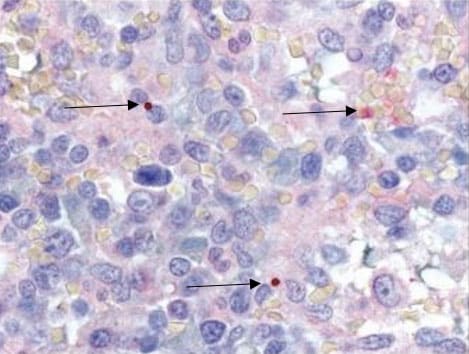
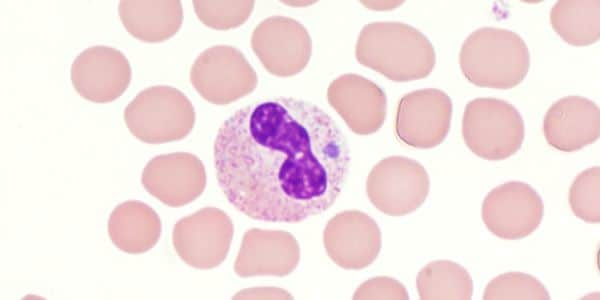
- During the first week of illness, a microscopic examination of a peripheral blood smear might reveal morulae (microcolonies of anaplasmae) in the cytoplasm of granulocytes. If these morulae are identified, anaplasmosis should be strongly suspected.
- Blood smear examination for morulae is insufficiently sensitive and should not be relied upon solely to diagnose anaplasmosis.
- Observing morulae in a particular type of white blood cell cannot conclusively differentiate between Anaplasma and Ehrlichia species.
Bobbi Pritt, MD, Mayo Clinic
