Seasonality of Respiratory Syncytial Virus — United States, 2017–2023
Weekly / April 7, 2023 / 72(14);355–361
Sarah Hamid, PhD1,2; Amber Winn, MPH2; Rishika Parikh, MPH2,3; Jefferson M. Jones, MD2; Meredith McMorrow, MD2; Mila M. Prill, MSPH2; Benjamin J. Silk, PhD2; Heather M. Scobie, PhD2; Aron J. Hall, DVM2 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
In the United States, the timing of seasonal respiratory syncytial virus (RSV) epidemics (October–April) was disrupted during the COVID-19 pandemic.
What is added by this report?
RSV circulation was historically low during 2020–21 and began earlier and continued longer during 2021–22 than during prepandemic seasons. The 2022–23 season started later than the 2021–22 season but earlier than prepandemic seasons, suggesting a return toward prepandemic seasonality.
What are the implications for public health practice?
Ongoing monitoring of RSV seasonality can guide the timing of immunoprophylaxis and evaluation of new immunization products. Although an eventual return to prepandemic RSV seasonality is expected, clinicians should be aware that off-season RSV circulation might continue.
Altmetric:
In the United States, respiratory syncytial virus (RSV) infections cause an estimated 58,000–80,000 hospitalizations among children aged <5 years (1,2) and 60,000–160,000 hospitalizations among adults aged ≥65 years each year (3–5). U.S. RSV epidemics typically follow seasonal patterns, peaking in December or January (6,7), but the COVID-19 pandemic disrupted RSV seasonality during 2020–2022 (8). To describe U.S. RSV seasonality during prepandemic and pandemic periods, polymerase chain reaction (PCR) test results reported to the National Respiratory and Enteric Virus Surveillance System (NREVSS)* during July 2017–February 2023 were analyzed. Seasonal RSV epidemics were defined as the weeks during which the percentage of PCR test results that were positive for RSV was ≥3% (9). Nationally, prepandemic seasons (2017–2020) began in October, peaked in December, and ended in April. During 2020–21, the typical winter RSV epidemic did not occur. The 2021–22 season began in May, peaked in July, and ended in January. The 2022–23 season started (June) and peaked (November) later than the 2021–22 season, but earlier than prepandemic seasons. In both prepandemic and pandemic periods, epidemics began earlier in Florida and the Southeast and later in regions further north and west. With several RSV prevention products in development,† ongoing monitoring of RSV circulation can guide the timing of RSV immunoprophylaxis and of clinical trials and postlicensure effectiveness studies. Although the timing of the 2022–23 season suggests that seasonal patterns are returning toward those observed in prepandemic years, clinicians should be aware that off-season RSV circulation might continue.
Each week, participating clinical and public health laboratories voluntarily report to NREVSS aggregate numbers of RSV PCR tests performed and numbers of positive test results. Although antigen tests are sometimes performed, this analysis was restricted to PCR tests because they accounted for >90% of tests reported (9). Surveillance years were defined based on troughs in RSV circulation. During 2017–2020 (the prepandemic period), surveillance years began in early July (epidemiologic week 27) and ended the following year in late June (week 26). Because the typical winter RSV epidemic was absent during 2020–21, and the 2021–22 epidemic began in the spring, the 2021–22 and 2022–23 surveillance years (pandemic period) were defined as early March (week 9) to late February (week 8) of the following year.§ Several methods for characterizing RSV seasonality were explored (Supplementary Table 1, https://stacks.cdc.gov/view/cdc/126381) (Supplementary Table 2, https://stacks.cdc.gov/view/cdc/126380). A 3% test positivity threshold was chosen because it prospectively identified a high proportion of annual RSV detections during epidemic periods of moderate duration. The epidemic onset and offset (or end) weeks were defined, respectively, as the first and last of 2 consecutive weeks when the percentage of PCR tests positive for RSV was ≥3%. The epidemic duration was the inclusive number of weeks between onset and offset. The peak was defined as the week with the highest percentage of PCR tests positive for RSV.
Epidemic onset, offset, peak, and duration were identified for each season at the national level and by U.S. Department of Health and Human Services (HHS) region.¶ Because patterns of weekly RSV circulation in Alaska, Florida, and Hawaii are different from those in other states within their assigned regions (HHS Regions 10, 4, and 9, respectively), these states were excluded from regional analyses. State-level seasonality for Florida is reported; however, an insufficient number of laboratories in Alaska and Hawaii consistently reported PCR data to present state-level seasonality in those states. The analysis included data from laboratories that consistently conducted PCR testing.** This activity was conducted consistent with applicable federal law and CDC policy.††
During the period with weeks ending July 8, 2017–February 25, 2023, five distinct RSV epidemics occurred: three before the COVID-19 pandemic (2017–18, 2018–19, and 2019–20) and two during the pandemic (2021–22 and 2022–23). Using the 3% epidemic threshold, no seasonal RSV epidemic was observed to occur during the 2020–21 surveillance year (Figure 1). The number of tests performed increased substantially during the pandemic (Table).
Nationally, RSV epidemics during the 3 surveillance years preceding the COVID-19 pandemic (2017–2020) began in October, peaked in December, and lasted a median of 27 weeks before the offset during March–April (Table). In contrast, the 2021–22 epidemic began 21 weeks earlier (May), peaked in July, and lasted 33 weeks until January 2022, although the peak percentage of RSV-positive PCR results (15%) was comparable with that during prepandemic seasons (Figure 1). During the 2022–23 surveillance year, onset occurred in June, the proportion of positive PCR results peaked in November, and the peak was higher (19%) than that during prepandemic seasons (range = 13%–16%). The epidemic lasted 32 weeks until the offset occurred in January.
In both the prepandemic and pandemic periods, RSV epidemics began earliest in Florida and the Southeast and later in regions further north and west (Figure 2). During the Florida prepandemic seasons, the median onset occurred in August, the peak occurred in November, and the epidemic continued until March (median duration = 30 weeks) (Table) (Supplementary Figure, https://stacks.cdc.gov/view/cdc/126382). In the 10 HHS regions (excluding Alaska, Florida, and Hawaii), the median onset ranged from September in Region 4 to December in Region 8. The median epidemic peaks ranged from November in Region 6 to February in Regions 8 and 9. Median offsets ranged from March in Region 5 to May in Region 7; offsets occurred 2–6 weeks earlier during the 2019–20 surveillance year (i.e., when the COVID-19 pandemic began) compared with the preceding 2 surveillance years. The shortest epidemic periods occurred in Region 10 (median = 21 weeks), and the longest occurred in Region 4 (median = 27 weeks).
During the 2021–22 (pandemic) surveillance year, epidemic onsets across the 10 HHS regions and Florida occurred a median of 20 weeks earlier (range = 13–25 weeks) than the median onsets during the prepandemic period (range = March [Florida] to August [Region 10]). Epidemic peaks also occurred earlier than they did during the prepandemic years, ranging from July in Region 6 to December in Region 10. Offsets ranged from November (Region 4) to February (Region 9), which is when prepandemic peaks typically occurred. During the 2021–22 surveillance year, the epidemic durations were a median of 6 weeks longer than the median durations of prepandemic RSV epidemics (range = 21 weeks [Region 2] to 38 weeks [Florida]).
During the 2022–23 season, early epidemic onsets (April–June) were observed in Florida and HHS Regions 3, 4, and 6, but the percentage of RSV-positive PCR test results levelled off before increasing again in September (Figure 1) (Table). In other regions, epidemics began between August and October. Seasons peaked from October in Region 4 to November in regions further north and west (Regions 2, 8, 9, and 10). Epidemics ended between December and February.
Discussion
In the United States, disruption of the seasonal circulation of RSV was observed during the COVID-19 pandemic as nonpharmaceutical interventions (e.g., school closures and masking) reduced respiratory virus transmission and led to an accumulation of susceptible persons resulting in large epidemics with atypical seasonality (10). After the implementation of nonpharmaceutical interventions in March 2020, the 2019–20 RSV epidemic ended earlier than the previous two epidemics. During 2020, RSV circulated at historically low levels. In 2021, RSV circulation began earlier (in late spring), when nonpharmaceutical interventions eased, and continued longer than it did during prepandemic years, although the percentage of RSV-positive PCR tests at the peak was comparable to those during prepandemic years. The 2022–23 epidemic began later than the 2021–22 epidemic but earlier than prepandemic epidemics, suggesting a reversion toward prepandemic seasonality with winter peaks. The peak percentage of positive RSV test results was higher than those in previous years, suggesting higher intensity of circulation. Across both prepandemic and pandemic years, RSV circulation began in Florida and the Southeast and later in regions to the north and west. The consistency of this pattern could help predict the timing of future epidemics in specific regions.
The findings in this report are subject to at least four limitations. First, reporting to NREVSS is voluntary, and analysis is limited to laboratories that consistently report, which might not represent local and state circulation. Second, differences in testing across regions and changes in testing practices and diagnostics over time, including increased panel testing during the COVID-19 pandemic, could have affected the baseline percentage of positive test results and trends, and thus the onset, offset, and duration of epidemics. Third, there is no standard method for characterizing seasonality; seasonal attributes vary depending on the method used. An earlier description of RSV seasonality in the United States used a more sensitive method (retrospective slope 10§§) that can only be applied retrospectively and results in longer epidemic durations (6,9). However, the 3% RSV-positive PCR threshold used in the current analysis can be applied in near real time and identified epidemic periods that included a high concentration of detections (9). Finally, this analysis describes regional and national trends; locally available data and region-specific thresholds might better reflect circulation patterns within specific jurisdictions.
Although the peak in RSV circulation during November 2022 suggests that seasonal patterns are returning to those observed in prepandemic years, it is uncertain whether this reversion will continue in the upcoming surveillance year. To monitor RSV circulation, CDC has conducted year-round surveillance using a variety of approaches including active, population-based surveillance for RSV-associated hospitalizations and outpatient visits.¶¶ Clinicians should be aware that atypical RSV epidemics might continue and consider testing patients for multiple respiratory pathogens when indicated. With new prevention products nearing licensure, including vaccines for older adults, maternal vaccines, and long-acting RSV immunoprophylaxis for infants and children, policy makers should consider RSV seasonality when making recommendations about the timing of studies and administration of new immunization and other RSV prevention products.
Corresponding author: Sarah Hamid, tsl4@cdc.gov.
1Epidemic Intelligence Service, CDC; 2Coronavirus and Other Respiratory Viruses Division, National Center for Immunizations and Respiratory Diseases, CDC; 3Goldbelt C6, Chesapeake, Virginia.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
* https://www.cdc.gov/surveillance/nrevss/index.html
† https://www.path.org/resources/rsv-vaccine-and-mab-snapshot
§ Defining surveillance year start and end dates based on troughs in RSV activity ensures that seasonal epidemics are encompassed in a 12-month period and that calculated proportions of annual detections that occur in the epidemic period are comparable over time (i.e., that the denominator for the proportion is the 12-month period between troughs in RSV circulation).
¶ https://www.hhs.gov/about/agencies/iea/regional-offices/index.html
** Consistent reporting was defined by the following criteria: 1) reported RSV PCR testing results for ≥30 weeks during the 12-month surveillance year and 2) reported an average of 10 or more RSV-positive PCR tests per week during the surveillance year.
†† 45 C.F.R. part 46.102(l)(2), 21 C.F.R. part 56; 42 U.S.C. Sect. 241(d); 5 U.S.C. Sect. 552a; 44 U.S.C. Sect. 3501 et seq.
§§ Retrospective slope 10 is a method for retrospectively characterizing RSV seasons that captures a high percentage of PCR detections. It uses a centered 5-week moving average of RSV detections normalized to a season peak of 1,000 detections. The season onset is defined as the second of 2 consecutive weeks when the slope, or normalized 5-week moving average of RSV detections between subsequent weeks, exceeds 10. The season offset is the last week when the standardized (normalized) detections exceed the standardized detections at onset.
¶¶ The Respiratory Syncytial Virus Hospitalization Surveillance Network conducts active, population-based surveillance for laboratory-confirmed RSV-associated hospitalizations (https://www.cdc.gov/rsv/research/rsv-net/dashboard.html). The New Vaccine Surveillance Network conducts active, population-based surveillance for RSV-associated acute respiratory illness among children in outpatient, emergency department, and hospital settings at seven U.S. medical centers. https://www.cdc.gov/surveillance/nvsn/index.html
References
- Hall CB, Weinberg GA, Iwane MK, et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med 2009;360:588–98. https://doi.org/10.1056/NEJMoa0804877 PMID:19196675
- McLaughlin JM, Khan F, Schmitt HJ, et al. Respiratory syncytial virus-associated hospitalization rates among US infants: a systematic review and meta-analysis. J Infect Dis 2022;225:1100–11. https://doi.org/10.1093/infdis/jiaa752 PMID:33346360
- McLaughlin JM, Khan F, Begier E, Swerdlow DL, Jodar L, Falsey AR. Rates of medically attended RSV among US adults: a systematic review and meta-analysis. Open Forum Infect Dis 2022;9:ofac300. https://doi.org/10.1093/ofid/ofac300 PMID:35873302
- Zheng Z, Warren JL, Shapiro ED, Pitzer VE, Weinberger DM. Estimated incidence of respiratory hospitalizations attributable to RSV infections across age and socioeconomic groups. Pneumonia 2022;14:6. https://doi.org/10.1186/s41479-022-00098-x PMID:36280891
- Havers FP, Whitaker M, Pham H, et al. 2209. RSV-associated hospitalizations in adults aged ≥18 years and the impact of the COVID-19 pandemic in the United States, October 2018–February 2022. Open Forum Infect Dis 2022;9(Suppl 2):ofac492.1828. https://doi.org/10.1093/ofid/ofac492.1828
- Rose EB, Wheatley A, Langley G, Gerber S, Haynes A. Respiratory syncytial virus seasonality—United States, 2014–2017. MMWR Morb Mortal Wkly Rep 2018;67:71–6. https://doi.org/10.15585/mmwr.mm6702a4 PMID:29346336
- Haynes AK, Prill MM, Iwane MK, Gerber SI; CDC. Respiratory syncytial virus—United States, July 2012–June 2014. MMWR Morb Mortal Wkly Rep 2014;63:1133–6. PMID:25474034
- Olsen SJ, Winn AK, Budd AP, et al. Changes in influenza and other respiratory virus activity during the COVID-19 pandemic—United States, 2020–2021. MMWR Morb Mortal Wkly Rep 2021;70:1013–9. https://doi.org/10.15585/mmwr.mm7029a1 PMID:34292924
- Midgley CM, Haynes AK, Baumgardner JL, et al. Determining the seasonality of respiratory syncytial virus in the United States: the impact of increased molecular testing. J Infect Dis 2017;216:345–55. https://doi.org/10.1093/infdis/jix275 PMID:28859428
- Baker RE, Park SW, Yang W, Vecchi GA, Metcalf CJE, Grenfell BT. The impact of COVID-19 nonpharmaceutical interventions on the future dynamics of endemic infections. Proc Natl Acad Sci U S A 2020;117:30547–53. https://doi.org/10.1073/pnas.2013182117 PMID:33168723
 FIGURE 1. Percentage* of polymerase chain reaction test results positive for respiratory syncytial virus, by epidemiologic week — National Respiratory and Enteric Virus Surveillance System, United States, July 2017–February 2023
FIGURE 1. Percentage* of polymerase chain reaction test results positive for respiratory syncytial virus, by epidemiologic week — National Respiratory and Enteric Virus Surveillance System, United States, July 2017–February 2023
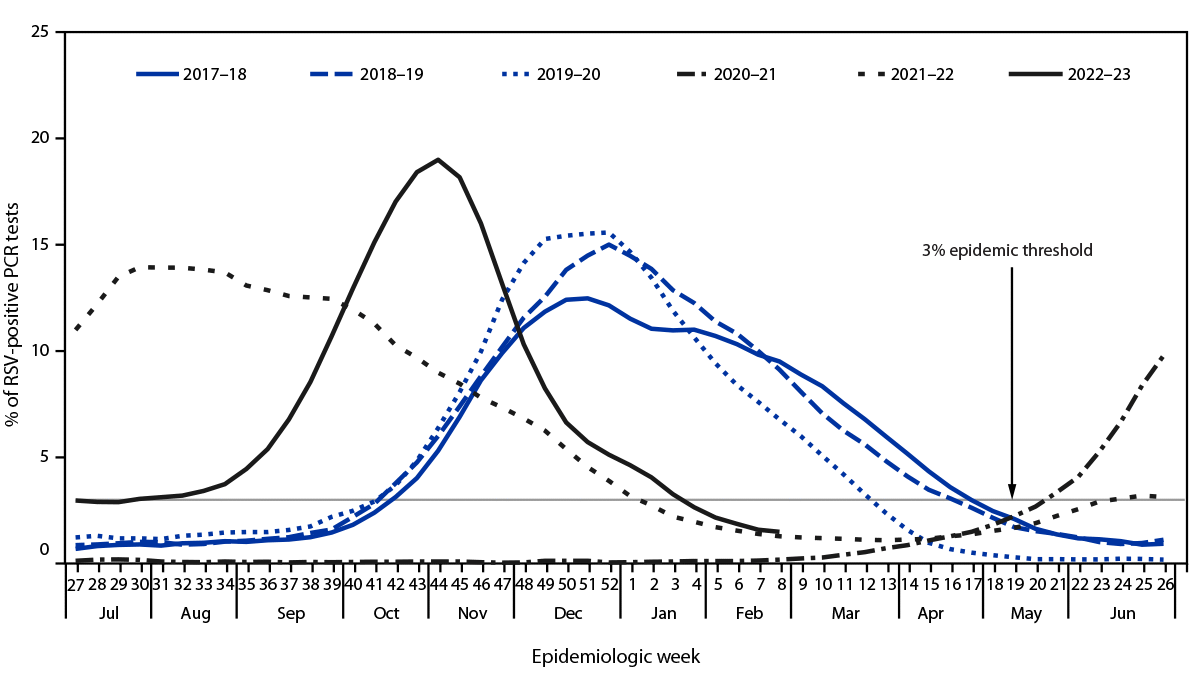
Abbreviations: PCR = polymerase chain reaction; RSV = respiratory syncytial virus.
* Three-week centered moving averages of percentage of RSV-positive PCR test results nationally. The threshold for a seasonal epidemic was set at 3% RSV-positive PCR test results (not based on a moving average).
Abbreviations: HHS = U.S. Department of Health and Human Services; PCR = polymerase chain reaction; RSV = respiratory syncytial virus.
* https://www.hhs.gov/about/agencies/iea/regional-offices/index.html. Patterns of weekly RSV circulation in Alaska, Florida, and Hawaii are distinct from other states within their assigned regions (HHS regions 10, 4, and 9, respectively); therefore, these states were excluded from regional analyses. State-level seasonality for Florida is reported; however, there are an insufficient number of laboratories consistently reporting RSV PCR data to present state-level seasonality in Alaska and Hawaii.
† Because the typical seasonal RSV epidemic was notably absent during the 2020–21 surveillance year, data from this surveillance year are not shown. Surveillance years were defined based on troughs in RSV circulation. During 2017–2020, surveillance years began in epidemiologic week 27 (early July) and ended the following year in epidemiologic week 26 (late June). During the COVID-19 pandemic (2021–22 and 2022–23), surveillance years began in epidemiologic week 9 (early March) and ended the following year in epidemiologic week 8 (late February).
§ The epidemic onset was defined as the first of 2 consecutive weeks when the percentage of PCR tests positive for RSV was ≥3%.
¶ The epidemic peak was defined as the week with the highest percentage of PCR tests positive for RSV.
** The epidemic offset was defined as the last of 2 consecutive weeks when the percentage of PCR tests positive for RSV was ≥3%.
†† The epidemic duration was the inclusive number of weeks between onset and offset.
§§ Annual percentage of detections in the epidemic period was defined as the proportion of all detections during a surveillance year that occurred during the epidemic period.
 FIGURE 2. Respiratory syncytial virus epidemic onsets* in U.S. Department of Health and Human Services Regions 1–10† and in Florida — National Respiratory and Enteric Virus Surveillance System, United States, July 2017–February 2023§
FIGURE 2. Respiratory syncytial virus epidemic onsets* in U.S. Department of Health and Human Services Regions 1–10† and in Florida — National Respiratory and Enteric Virus Surveillance System, United States, July 2017–February 2023§

Abbreviations: FL = Florida; RSV = respiratory syncytial virus.
* The epidemic onset was defined as the first of 2 consecutive weeks of a surveillance year when the percentage of PCR tests positive for RSV was ≥3%. Median epidemic onset weeks were calculated for the three RSV epidemics that occurred before the COVID-19 pandemic (2017–18, 2018–19, and 2019–20).
† https://www.hhs.gov/about/agencies/iea/regional-offices/index.html. Patterns of weekly RSV circulation in Alaska, Florida, and Hawaii are distinct from other states within their assigned regions; therefore, these states were excluded from regional analyses. State-level seasonality for Florida is reported; however, there are an insufficient number of laboratories consistently reporting polymerase chain reaction testing data to present state-level seasonality in Alaska and Hawaii.
§ Surveillance years were defined based on troughs in RSV circulation. During 2017–2020, surveillance years began in epidemiologic week 27 (early July) and ended the following year in epidemiologic week 26 (late June). The aberrant 2020–21 surveillance year was defined as week 27 through week 8 (late February) inclusive. During the COVID-19 pandemic (2021–22 and 2022–23), surveillance years began in epidemiologic week 9 (early March) and ended the following year in epidemiologic week 8.
Suggested citation for this article: Hamid S, Winn A, Parikh R, et al. Seasonality of Respiratory Syncytial Virus — United States, 2017–2023. MMWR Morb Mortal Wkly Rep 2023;72:355–361. DOI: http://dx.doi.org/10.15585/mmwr.mm7214a1.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.