Ocular Monkeypox — United States, July–September 2022
Weekly / October 21, 2022 / 71(42)
On October 17, 2022, this report was posted online as an MMWR Early Release.
Please note: This report has been corrected. An erratum has been published.
Shama Cash-Goldwasser, MD1,2,3; Sarah M. Labuda, MD1; David W. McCormick, MD1; Agam K. Rao, MD1; Andrea M. McCollum, PhD1; Brett W. Petersen, MD1; James Chodosh, MD4; Catherine M. Brown, DVM5; Suk Yin Chan-Colenbrander, MD6; Caitlin M. Dugdale, MD7; Michael Fischer, MD8; Amy Forrester9; Jayne Griffith, MPH3; Rachel Harold, MD10; Bruce W. Furness, MD1,10; Vivian Huang, MD11; Aaron R. Kaufman, MD12; Ellen Kitchell, MD13; Rachel Lee, DO14; Nicholas Lehnertz, MD3; Ruth Lynfield, MD3; Ketzela Jacobowitz Marsh, MD6; Lawrence C. Madoff, MD5; Nelson Nicolasora, MD15; Dharmendra Patel, MD16; Roberto Pineda II, MD12; Trey Powrzanas, FNP17; Afsoon Roberts, MD14; Maria Teresa Seville, MD16; Ami Shah, MD16; Joshua M. Wong, MD1; Jana M. Ritter, DVM1; Caroline A. Schrodt, MD1; Elliot Raizes, MD1; Sapna Bamrah Morris, MD1; Jeremy A. W. Gold, MD1; CDC Monkeypox Clinical Escalations Team (View author affiliations)
View suggested citationSummary
What is already known about this topic?
Patients with monkeypox can experience serious ocular complications, which are not well described during the current outbreak.
What is added by this report?
This report describes five cases of ocular monkeypox identified in the United States during July–September 2022. Patients with ocular monkeypox, including those with HIV-associated immunocompromise, have experienced delays in treatment initiation, prolonged illness, hospitalization, and vision impairment.
What are the implications for public health practice?
Health care providers and public health practitioners should be aware that ocular monkeypox, although rare, is a sight-threatening condition. Patients with signs and symptoms compatible with ocular monkeypox should be considered for urgent ophthalmologic evaluation and treatment. Prompt notification of public health officials can help support these efforts.
As of October 11, 2022, a total of 26,577 monkeypox cases had been reported in the United States.* Although most cases of monkeypox are self-limited, lesions that involve anatomically vulnerable sites can cause complications. Ocular monkeypox can occur when Monkeypox virus (MPXV) is introduced into the eye (e.g., from autoinoculation), potentially causing conjunctivitis, blepharitis, keratitis, and loss of vision (1,start highlight2end highlight). This report describes five patients who acquired ocular monkeypox during July–September 2022. All patients received treatment with tecovirimat (Tpoxx)†; four also received topical trifluridine (Viroptic).§ Two patients had HIV-associated immunocompromise and experienced delays between clinical presentation with monkeypox and initiation of monkeypox-directed treatment. Four patients were hospitalized, and one experienced marked vision impairment. To decrease the risk for autoinoculation, persons with monkeypox should be advised to practice hand hygiene and to avoid touching their eyes, which includes refraining from using contact lenses (start highlight3end highlight). Health care providers and public health practitioners should be aware that ocular monkeypox, although rare, is a sight-threatening condition. Patients with signs and symptoms compatible with ocular monkeypox should be considered for urgent ophthalmologic evaluation and initiation of monkeypox-directed treatment. Public health officials should be promptly notified of cases of ocular monkeypox. Increased clinician awareness of ocular monkeypox and of approaches to prevention, diagnosis, and treatment might reduce associated morbidity.
During the 2022 multinational outbreak, CDC has provided consultation to clinicians treating patients with monkeypox.¶ This report describes demographic characteristics, clinical features, and outcomes as of October 11 for five patients who received a diagnosis of ocular monkeypox during July–September 2022. Ocular monkeypox was defined as the presence of new ocular disease compatible with Orthopoxvirus (OPXV) infection in a patient with probable or confirmed monkeypox** and no alternative explanation for the ocular disease. CDC obtained data during clinical consultation and worked with treating clinicians and jurisdictional health departments to follow patient progress. Patient permission for the use of the clinical image was obtained. This activity was reviewed by CDC and was conducted consistent with applicable federal law and CDC policy.††
Patient A
In August 2022, a man aged 20–29 years with HIV disease (Table) (Figure 1) (CD4 = 25 cells/mm3, not receiving antiretroviral therapy [ART]) was evaluated in an outpatient clinic for a rash on his buttocks, chest, arms, and hands that was compatible with monkeypox.§§ Swabs collected from lesions on his chest were sent for polymerase chain reaction (PCR) testing for OPXV, and results were negative. Ten days later, the patient presented for care again, this time with progressive rash as well as left eye symptoms, including pain, itching, swelling, discharge, foreign body sensation, photosensitivity, and vision changes. The rash was swabbed again to test for OPXV, and he was provided a referral to ophthalmology. Seven days later, PCR testing returned positive results for OPXV, and he commenced treatment with oral tecovirimat. Two days later, he was admitted to a hospital because of worsening ocular symptoms. Ophthalmologic exam was notable for left eye conjunctivitis. Visual acuity in the left eye was 20/40. The patient started intravenous tecovirimat and topical trifluridine drops to his left eye on admission, and he was initiated on ART. His ocular symptoms improved, and he was discharged after 5 days with prescriptions for oral tecovirimat, topical trifluridine, and ART. During the next 4 weeks he developed new facial lesions and decreasing left eye vision, for which he was readmitted to the hospital; health care providers suspected nonadherence with prescribed medications. Ophthalmologic examination revealed left eye conjunctivitis, keratitis, and a conjunctival ulcer (Figure 2). Left eye visual acuity was measured at 20/300. A swab of the conjunctival lesion yielded a positive PCR test result for OPXV, and biopsy showed necroulcerative conjunctivitis with extensive intralesional orthopoxviral antigen detected by immunohistochemistry. The patient was restarted on intravenous tecovirimat, and his left eye was treated with topical trifluridine for 1 week as well as topical povidone-iodine. Currently, the patient is on day 14 of intravenous tecovirimat and remains hospitalized for treatment of ocular monkeypox. He has experienced waxing and waning of left eye pain, irritation, and photosensitivity. Left eye visual acuity was most recently measured at 20/800 (profound visual impairment), although bedside visual acuity assessments have been challenging. His prognosis for vision recovery is currently unknown.
Patient B
In July 2022, a man aged 30–39 years with HIV disease (CD4 = 78 cells/mm3, not on ART) was evaluated at an emergency department with a rash on his chest, legs, perianal area, and face, including on the bridge of his nose near his right eye (Table) (Figure 1). Swabs of lesions from his face and scalp were taken to test for OPXV, but because tecovirimat was not available in the emergency department, he was referred to an outpatient clinic to receive tecovirimat. The swabs tested PCR-positive for OPXV. The patient was evaluated at an outpatient clinic 9 days after testing and was prescribed ART and 14 days of oral tecovirimat. His rash began to resolve during treatment. Two weeks after completion of tecovirimat, he developed new and worsening facial lesions. The lesion on his nose expanded onto the right medial canthus and over the conjunctiva, and he experienced right eye redness, pain, itching, and photosensitivity, for which he was hospitalized. He did not experience vision changes. Ophthalmologic exam results were notable for right eye conjunctivitis, several small conjunctival nodular lesions, and corneal ulcers. He was treated again with intravenous tecovirimat for 10 days and with topical trifluridine drops for 5 days and antibacterial eye drops to the right eye. He was discharged upon regression of the eye lesion and improvement in conjunctivitis 10 days after admission, without further treatment for monkeypox.
Patient C
In August 2022, a previously healthy man aged 30–39 years developed rectal pain and perianal lesions. He went to an emergency department 3 days later and swabs of those lesions were taken for OPXV testing (Table) (Figure 1). Three days later, when the swabs yielded positive PCR test results for OPXV, the patient was prescribed oral tecovirimat for rectal pain. Two days later, he was evaluated again in the emergency department with right eye pain, redness and discharge. He did not experience vision changes. Ophthalmologic exam was notable for right eye conjunctivitis. He subsequently developed bilateral conjunctivitis; the treating physicians suspected the patient had autoinoculated both eyes with MPXV by rubbing them. The patient’s bilateral conjunctivitis persisted for 3 weeks after resolution of his perianal lesions. The course of tecovirimat was extended until all ocular symptoms resolved, which occurred after 1 month of treatment.
Patient D
In August 2022, a previously healthy man aged 30–39 years developed a groin rash (Table) (Figure 1). One week later, he was examined at an emergency department for right eye redness, pain, and eyelid swelling. He reported rubbing his right eye. Lesions were noted on his penis, abdomen, and one wrist. Samples were collected from the body lesions for OPXV testing; the patient received empiric treatment for gonorrhea and chlamydia. Providers attributed the eye symptoms to bacterial preseptal cellulitis and he was discharged on oral antibiotics. Two days later, the patient returned with multiple right eyelid lesions, periorbital swelling, and eye pain, for which he was admitted to a hospital. He did not experience vision changes. Ophthalmologic exam was notable for right eye conjunctivitis as well as four ulcers on the eyelid margin and three lesions on the palpebral conjunctiva, which were swabbed for OPXV testing. He was started on oral tecovirimat empirically, after which all test results from swabs of skin and eye lesions returned PCR-positive for OPXV. The patient also received topical trifluridine for 5 days and antibacterial drops to the right eye, as well as intravenous antibiotics for preseptal cellulitis. He was discharged upon clinical improvement 5 days after admission, to complete a 14-day course of oral tecovirimat.
Patient E
In July 2022, a previously healthy woman aged 30–39 years was evaluated for pustular lesions on her vaginal labia (Table) (Figure 1). A swab of those lesions tested PCR-positive for OPXV. During the week after symptom onset, lesions spread to her back, buttocks, chin, forehead, and left lower eyelid. She began experiencing left eye pain and redness. She sought medical care after noticing a lesion on the globe of her left eye, for which she was admitted to a hospital. Ophthalmologic exam was notable for left eye conjunctivitis, a bulbar conjunctival lesion, and a subconjunctival nodule. She did not experience vision changes. start highlightNeither tecovirimat nor trifluridine was immediately available; the patient was treated with naproxen.end highlight Her ocular symptoms improved, and she was discharged after 3 days with a 14-day course of oral tecovirimat and start highlighta 5-day course of topical trifluridine (2).end highlight
Discussion
This report highlights the varying clinical manifestations of ocular monkeypox and the importance of prompt evaluation and treatment to prevent sight-threatening complications. All five patients with ocular monkeypox described in this report suffered prolonged illness, four were hospitalized, and one experienced significant vision impairment. Two patients had HIV-associated immunocompromise and experienced delays in initiation of treatment for monkeypox. One of these patients experienced vision loss; he remains in treatment and his prognosis for vision recovery is currently unknown. Urgent referral for ophthalmologic evaluation and prompt antiviral therapy should be considered for patients with monkeypox and ocular signs or symptoms (e.g., vision changes or eye pain, itching, redness, swelling, or foreign body sensation) or lesions near the eye. Clinicians should consider initiation of prompt systemic antiviral therapy as well as topical trifluridine for patients with ocular monkeypox.¶¶
Several strategies might help prevent ocular monkeypox and associated complications. To decrease the risk for autoinoculation, persons with monkeypox should be advised to practice hand hygiene and to avoid touching their eyes, which includes refraining from using contact lenses start highlight(3).end highlight Short turnaround times for OPXV/MPXV PCR test results might help prevent delays in treatment initiation. For persons with suspected ocular monkeypox, or for persons with suspected monkeypox who are at risk for severe manifestations of the disease (e.g., those with HIV-associated immunocompromise), clinicians might consider initiating empiric treatment for monkeypox while test results are pending. Health care providers can contact their public health jurisdictions and CDC for support on the use of medical countermeasures to treat patients with monkeypox*** start highlight(4).end highlight
The findings in this report are subject to at least two limitations. First, the cases described might not be representative of patients with ocular monkeypox in the United States, and conclusions cannot be drawn about the frequency of reported events. Although the frequency of ocular monkeypox during the current outbreak is unknown, national surveillance data from the United States suggest that 5% of patients with monkeypox report ocular symptoms††† start highlight(5).end highlight Second, not every patient underwent testing of ocular lesions for OPXV/MPXV or exhaustive testing for other ocular infections. However, the clinical findings in these patients were compatible with descriptions of ocular monkeypox from other studies start highlight(6,7).end highlight
Ocular monkeypox is a potentially sight-threatening infection. Urgent ophthalmologic evaluation and the provision of timely medical countermeasures for patients with suspected or confirmed ocular monkeypox might help prevent poor outcomes.
Acknowledgments
Isaac Bleicher, Harvard Medical School; Shade Brady, Arizona Department of Health Sciences; Steven Yeh, University of Nebraska Medical Center; Melissa Yuan, Massachusetts Eye and Ear Infirmary.
CDC Monkeypox Clinical Escalations Team
Amimah Asif, CDC; Amy Beeson, CDC; Ramon Bhatia, CDC; Brian F. Borah, CDC; Kevin Chatham-Stevens, CDC; Rewa Choudhary, CDC; Eleanor Click, CDC; Thomas D. Filardo, CDC; Romeo R. Galang, CDC; Julia Haston, CDC; Sophia Hsu, CDC; Gurpreet Kaur, CDC; Anne Kimball, CDC; James T. Lee, CDC; Grace Marx, CDC; Janet McNicholl, CDC; Maureen J. Miller, CDC; Rebecca Noe, CDC; Siobhan O’Connor, CDC; Kevin O’Laughlin, CDC; Kia Padgett, CDC; Gail Thompson, CDC; Farrell Tobolowsky, CDC; Isaac Zulu, CDC
Corresponding author: Shama Cash-Goldwasser, tqx7@cdc.gov.
1CDC Monkeypox Emergency Response Team; 2Epidemic Intelligence Service, CDC; 3Minnesota Department of Health; 4University of New Mexico School of Medicine, Albuquerque, New Mexico; 5Massachusetts Department of Public Health; 6University of Minnesota Medical Center, Minneapolis, Minnesota; 7Massachusetts General Hospital, Boston, Massachusetts; 8Texas Department of State Health Services; 9Dallas County Health and Human Services, Dallas, Texas; 10District of Columbia Department of Health, Washington, D.C.; 11Maricopa County Department of Health, Phoenix, Arizona; 12Massachusetts Eye and Ear Infirmary, Boston, Massachusetts; 13University of Texas Southwestern Medical Center, Dallas, Texas; 14The George Washington University School of Medicine and Health Sciences, Washington, D.C.; 15Banner University Medical Center, University of Arizona, Phoenix, Arizona; 16Mayo Clinic Hospital, Phoenix, Arizona; 17Pueblo Family Physicians, Phoenix, Arizona.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. James Chodosh is a consultant to the Food and Drug Administration, where he chairs an advisory committee for new ophthalmic medications. He also receives grant support from the National Institutes of Health (NIH) to study adenovirus keratitis. Caitlin M. Dugdale reports institutional support from the National Institute for Child Health and Human Development, NIH; Harvard University Center for AIDS Research; the Massachusetts General Hospital Executive Committee on Research; the International AIDS Vaccine Initiative; and the International Maternal, Pediatric, Adolescent AIDS Clinical Trials (IMPAACT) Network, NIH. Aaron R. Kaufman reports support by a Heed Fellowship awarded by the Heed Ophthalmic Foundation. Roberto Pineda II reports royalties from Elsevier and consulting fees from Sanofi-Genzyme. No other potential conflicts of interest were disclosed.
* Case counts included confirmed and probable monkeypox cases. https://www.cdc.gov/poxvirus/monkeypox/response/2022/us-map.html (Accessed October 11, 2022).
† Tecovirimat, an FDA-approved treatment for smallpox, is an antiviral drug that has demonstrated effectiveness in animal MPXV models. Interim CDC guidance currently recommends that tecovirimat be considered in patients with severe monkeypox, those at high risk for severe disease, or those whose infection involves accidental implantation in the eyes or other sensitive anatomic areas where monkeypox might constitute a special hazard. https://www.cdc.gov/poxvirus/monkeypox/clinicians/Tecovirimat.html
§ Trifluridine, an antiviral drug used to treat herpes simplex keratitis, can be considered to treat conjunctivitis and keratitis caused by MPXV, in consultation with an ophthalmologist. Trifluridine is a preferred treatment for ocular infection with vaccinia virus, which can occur as a complication of autoinoculation after vaccination with ACAM 2000. https://www.cdc.gov/poxvirus/monkeypox/clinicians/ocular-infection.html
¶ CDC is offering a monkeypox clinical consultation service during the ongoing monkeypox outbreak. Health care providers seeking additional clinical guidance can contact CDC Emergency Operations by phone (770-488-7100) or by email (eocevent482@cdc.gov).
** Case definitions for use in the 2022 monkeypox response. https://www.cdc.gov/poxvirus/monkeypox/clinicians/case-definition.html
†† 45 C.F.R. part 46.102(l)(2), 21 C.F.R. part 56; 42 U.S.C. Sect. 241(d); 5 U.S.C. Sect. 552a; 44 U.S.C. Sect. 3501 et seq.
§§ Clinical recognition, monkeypox. https://www.cdc.gov/poxvirus/monkeypox/clinicians/clinical-recognition.html
¶¶ Interim clinical considerations for management of ocular monkeypox virus infection. https://www.cdc.gov/poxvirus/monkeypox/clinicians/ocular-infection.html
*** Interim clinical guidance for the treatment of monkeypox. https://www.cdc.gov/poxvirus/monkeypox/clinicians/treatment.html#anchor_1655488233196
††† This frequency is lower than that reported from the Democratic Republic of the Congo, where 23% of patients with monkeypox had conjunctivitis, a difference that might be related to the virulence of the MPXV clade causing infection during the ongoing outbreak, or to epidemiologic or clinical factors. https://www.cdc.gov/poxvirus/monkeypox/clinicians/ocular-infection.html
References
- Abdelaal A, Serhan HA, Mahmoud MA, Rodriguez-Morales AJ, Sah R. Ophthalmic manifestations of monkeypox virus. Eye (Lond) 2022. https://doi.org/10.1038/s41433-022-02195-z PMID:35896700
- start highlightFoos W, Wroblewski K, Ittoop S. Subconjunctival nodule in a patient with acute monkeypox. JAMA Ophthalmol 2022;140:e223742. https://doi.org/10.1001/jamaophthalmol.2022.3742 PMID:36069930end highlight
- start highlightCDC. Monkeypox. Isolation and infection control at home. Atlanta, GA: US Department of Health and Human Services, CDC; 2022. Accessed October 12, 2022. https://www.cdc.gov/poxvirus/monkeypox/clinicians/infection-control-home.htmlend highlight
- start highlightCDC. Emergency preparedness and response. Severe manifestations of monkeypox among people who are immunocompromised due to HIV or other conditions. Atlanta, GA: US Department of Health and Human Services, CDC; 2022. Accessed October 6, 2022. https://emergency.cdc.gov/han/2022/han00475.aspend highlight
- start highlightCDC. Monkeypox. Technical report 3: multi-national monkeypox outbreak, United States, 2022. Atlanta, GA: US Department of Health and Human Services, CDC; 2022. Accessed October 12, 2022. https://www.cdc.gov/poxvirus/monkeypox/cases-data/technical-report/report-3.htmlend highlight
- start highlightHughes C, McCollum A, Pukuta E, et al. Ocular complications associated with acute monkeypox virus infection, DRC. Int J Infect Dis 2014;21:276–7 https://doi.org/10.1016/j.ijid.2014.03.994end highlight
- start highlightMazzotta V, Mondi A, Carletti F, et al. Ocular involvement in monkeypox: description of an unusual presentation during the current outbreak. J Infect 2022. Epub August 18, 2022. https://doi.org/10.1016/j.jinf.2022.08.011end highlight
 start highlightFIGURE 1. Timeline of testing, symptom onset, and initiation of medical countermeasures for patients with ocular monkeypox — United States, July–September 2022
start highlightFIGURE 1. Timeline of testing, symptom onset, and initiation of medical countermeasures for patients with ocular monkeypox — United States, July–September 2022
end highlight
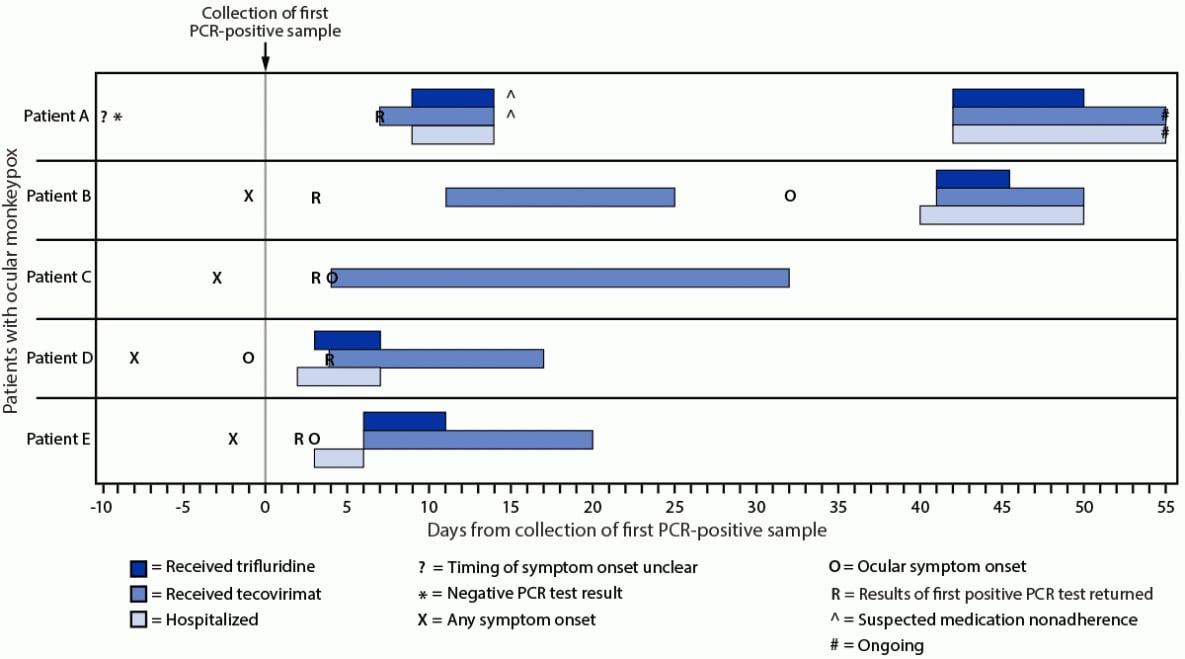
Abbreviation: PCR = polymerase chain reaction.
 FIGURE 2. Left eye in a patient* with HIV-associated immunocompromise and ocular monkeypox, with conjunctivitis and conjunctival lesion earlier in the course of monkeypox illness (A), and with conjunctival ulcer and peripheral keratitis later in the course of monkeypox illness (B) — United States, August–September 2022
FIGURE 2. Left eye in a patient* with HIV-associated immunocompromise and ocular monkeypox, with conjunctivitis and conjunctival lesion earlier in the course of monkeypox illness (A), and with conjunctival ulcer and peripheral keratitis later in the course of monkeypox illness (B) — United States, August–September 2022

Photo A/Nathanael Adjei-Kyeremeh
Photo B/Dharmendra R. Patel
* Patient has consented to the publication of these photographs.
Suggested citation for this article: Cash-Goldwasser S, Labuda SM, McCormick DW, et al. Ocular Monkeypox — United States, July–September 2022. MMWR Morb Mortal Wkly Rep 2022;71. DOI: http://dx.doi.org/10.15585/mmwr.mm7142e1.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.