Respiratory Virus Surveillance Among Children with Acute Respiratory Illnesses — New Vaccine Surveillance Network, United States, 2016–2021
Weekly / October 7, 2022 / 71(40);1253–1259
Ariana Perez, MPH1,2; Joana Y. Lively, MPH1; Aaron Curns, MPH1; Geoffrey A. Weinberg, MD3; Natasha B. Halasa, MD4; Mary Allen Staat, MD5,6; Peter G. Szilagyi, MD3,7; Laura S. Stewart, PhD4; Monica M. McNeal, MS5,6; Benjamin Clopper, MPH1; Yingtao Zhou, MS, MPH1; Brett L. Whitaker, MS1; Elizabeth LeMasters, MSc1,2; Elizabeth Harker, MPH1,8; Janet A. Englund, MD9; Eileen J. Klein, MD9; Rangaraj Selvarangan, PhD10; Christopher J. Harrison, MD10; Julie A. Boom, MD11; Leila C. Sahni, PhD11,12; Marian G. Michaels, MD13,14; John V. Williams, MD13,14; Gayle E. Langley, MD1; Susan I. Gerber, MD1; Angela Campbell, MD1; Aron J. Hall, DVM1; Brian Rha, MD1; Meredith McMorrow, MD1; New Vaccine Surveillance Network Collaborators (View author affiliations)
View suggested citationSummary
What is already known about this topic?
Acute respiratory illness (ARI) caused by viruses including respiratory syncytial virus (RSV) and SARS-CoV-2 (the virus that causes COVID-19) results in pediatric morbidity.
What is added by this report?
Rhinovirus and enterovirus and RSV were the most frequently detected viruses among children enrolled in the New Vaccine Surveillance Network during 2016–2021 through inpatient, outpatient, and emergency department settings. Throughout the COVID-19 pandemic, respiratory viruses exhibited uncharacteristic seasonality, with lower-than-expected circulation during April 2020–May 2021, and atypical RSV circulation and inpatient enrollment in summer 2021.
What are the implications for public health practice?
Continued ARI surveillance is critical as vaccines and therapeutics are introduced to protect children from SARS-CoV-2 and RSV to elucidate risk factors, health disparities, and to guide prevention policies.
Altmetric:
The New Vaccine Surveillance Network (NVSN) is a prospective, active, population-based surveillance platform that enrolls children with acute respiratory illnesses (ARIs) at seven pediatric medical centers. ARIs are caused by respiratory viruses including influenza virus, respiratory syncytial virus (RSV), human metapneumovirus (HMPV), human parainfluenza viruses (HPIVs), and most recently SARS-CoV-2 (the virus that causes COVID-19), which result in morbidity among infants and young children (1–6). NVSN estimates the incidence of pathogen-specific pediatric ARIs and collects clinical data (e.g., underlying medical conditions and vaccination status) to assess risk factors for severe disease and calculate influenza and COVID-19 vaccine effectiveness. Current NVSN inpatient (i.e., hospital) surveillance began in 2015, expanded to emergency departments (EDs) in 2016, and to outpatient clinics in 2018. This report describes demographic characteristics of enrolled children who received care in these settings, and yearly circulation of influenza, RSV, HMPV, HPIV1–3, adenovirus, human rhinovirus and enterovirus (RV/EV),* and SARS-CoV-2 during December 2016–August 2021. Among 90,085 eligible infants, children, and adolescents (children) aged <18 years† with ARI, 51,441 (57%) were enrolled, nearly 75% of whom were aged <5 years; 43% were hospitalized. Infants aged <1 year accounted for the largest proportion (38%) of those hospitalized. The most common pathogens detected were RV/EV and RSV. Before the emergence of SARS-CoV-2, detected respiratory viruses followed previously described seasonal trends, with annual peaks of influenza and RSV in late fall and winter (7,8). After the emergence of SARS-CoV-2 and implementation of associated pandemic nonpharmaceutical interventions and community mitigation measures, many respiratory viruses circulated at lower-than-expected levels during April 2020–May 2021. Beginning in summer 2021, NVSN detected higher than anticipated enrollment of hospitalized children as well as atypical interseasonal circulation of RSV. Further analyses of NVSN data and continued surveillance are vital in highlighting risk factors for severe disease and health disparities, measuring the effectiveness of vaccines and monoclonal antibody–based prophylactics, and guiding policies to protect young children from pathogens such as SARS-CoV-2, influenza, and RSV.
During December 1, 2016–August 31, 2021, NVSN enrolled children aged <18 years in inpatient and ED settings at seven surveillance sites (Supplementary Table 1, https://stacks.cdc.gov/view/cdc/121550). Children were eligible for enrollment if they had an illness duration of <14 days, were enrolled within 48 hours of admission (inpatient only), had at least one qualifying ARI sign or symptom (e.g., apnea, cough, earache, fever, myalgia, nasal congestion, runny nose, sore throat, vomiting after coughing, shortness of breath [rapid or shallow breathing], wheezing, or apparent life-threatening event or brief resolved unexplained event), and resided in a surveillance site area.§ Children were excluded if they had a known nonrespiratory cause for hospitalization, had fever and neutropenia from chemotherapy, were admitted <5 days after a previous hospitalization, were transferred from another hospital after an admission of >48 hours, were a newborn who had never been discharged home from the hospital, or had previously enrolled in this study <14 days before their current visit or hospitalization. Children could be enrolled in inpatient units ≥5 days per week and in the ED ≥4 days per week for ≥6 hours per day.
Outpatient clinic enrollment began in November 2018, with enrollment limited to children aged <2 years and testing for RSV only. Enrollment and testing were later expanded to include children aged <18 years and multipathogen testing.¶ Outpatient enrollment was paused during May–October 2019, and weekly enrollment targets of approximately 150 patients were required before July 2020. Outpatient eligibility and exclusion criteria differed slightly from that of other clinical settings.** Beginning in April 2020, outpatient surveillance was expanded in Houston, Texas to include drive-through testing for SARS-CoV-2 (9). Data in this report are summarized by highest level of care received by each child, irrespective of the child’s enrollment setting.
Midturbinate (MT) nasal or oropharyngeal (OP) specimens were obtained using flocked swabs; if both nasal and OP swabs were collected, they were combined and placed in universal transport medium. A tracheal aspirate was accepted as an alternative specimen for patients who were intubated. Among patients from whom research MT nasal and OP or tracheal aspirate specimens could not be obtained, clinically obtained respiratory specimens were salvaged.†† Specimens were transported to the laboratory at each site and stored at a temperature of 35.6°F–46.4°F (2°C–8°C) until they were processed (within 72 hours). Specimen aliquots were subsequently frozen at −94°F (−70°C) or lower. Specimens underwent molecular testing at each study site for respiratory pathogens including RSV, influenza, HMPV, HPIV1–3, RV/EV, and adenovirus. SARS-CoV-2 surveillance and associated testing methodologies§§ began in 2020.¶¶ Molecular diagnostic assay methods used for respiratory pathogens varied by site (Supplementary Table 2, https://stacks.cdc.gov/view/cdc/121551) (Supplementary Table 3, https://stacks.cdc.gov/view/cdc/121552). All assays met CDC-sponsored proficiency testing standards.
Pearson’s chi-square tests compared the percentage of positive results during the 2020–2021 season against previous seasons combined, among inpatients and those treated in the ED. All analyses were performed using SAS software (version 9.4; SAS Institute). Informed consent was obtained from a parent or legal guardian of eligible children before conducting a standardized parent or guardian interview; medical chart review; and collection, testing, and storage of respiratory specimens. Assent from eligible children was obtained at each site, according to local regulations. This study was reviewed and approved by the institutional review boards at each of the seven study sites.***
During December 2016–August 2021, a total of 90,085 eligible children with ARI were identified and 51,441 (57%) were enrolled. Within the highest clinical care setting received, enrolled children included 22,093 (43%) inpatients, 23,145 (45%) patients evaluated in the ED, and 6,203 (12%) evaluated in outpatient clinics (Table 1). Among all enrolled children, 38,267 (74%) were aged <5 years, 15,986 (42%) of whom were aged <1 year. The majority of enrolled children (55%) were male; 32% were non-Hispanic Black or African American (Black), 31% were non-Hispanic White (White) and 27% were Hispanic or Latino (Hispanic) children. Among hospitalized children, 8,280 (38%) were aged <1 year, 12,623 (57%) were male, and 9,042 (41%) were White.
Across all settings, 32,259 (63%) specimens had at least one viral pathogen detected, 4,492 (9%) had more than one viral pathogen detected, and 19,182 (37%) had no viral pathogen detected. The pathogens most frequently detected were RV/EV (14,906; 31%) and RSV (8,461; 17%) (Table 2). Total proportions for each virus varied by setting; RSV was detected most frequently in inpatient settings (24%), influenza in EDs (11%), and RV/EV in outpatient clinics (39%). During the COVID-19 pandemic period (March 2020–August 31, 2021), 1,171 (7%) children received a positive SARS-CoV-2 test result, 411 (35%) of whom were outpatients. During the 2020–2021 season (September 15, 2020–August 31, 2021), lower total proportions of test results were positive for seasonal viruses compared with previous seasons combined among inpatient and ED settings, except for HPIV1–3 (8%) and RV/EV (36%) (p<0.001). Enrollment during December 2016–February 2020, peaked in inpatient and ED settings, with concurrent peaks in RSV and influenza detections. Other viruses such as adenovirus and HMPV circulated throughout this period, but smaller peaks occurred later in winter and early spring (Figure). After onset of the COVID-19 pandemic in March 2020, inpatient and ED enrollment did not follow previously observed seasonal patterns; enrollment and virus circulation during winter months of 2020 was lower than expected and a distinct peak in RSV circulation and overall enrollment occurred during summer months of 2021.
Discussion
During 2016–2021, approximately 51,000 children with ARI were prospectively enrolled in NVSN. Nearly 75% of enrolled children were aged <5 years, and children aged <1 year accounted for approximately one third of those hospitalized, consistent with previous studies among this age group (1–5). NVSN enrollees were racially and ethnically diverse, with nearly one third being Black children followed by slightly lower percentages of White and Hispanic children. Before the COVID-19 pandemic, seasonal patterns of respiratory virus circulation followed previously described trends, including annual peaks of influenza and RSV during late fall and winter months (7,8). RV/EV and RSV were the most frequently detected viruses in children in all settings; however, by setting, RSV was more commonly detected among hospitalized children than it was in ED or outpatient clinics. During the 2020–2021 season, the total proportion of seasonal respiratory viruses was lower than that during previous seasons for all except HPIV1–3 and RV/EV. These declines support previous studies, which postulated that community mitigation measures (e.g., school and child care facility closures) during the COVID-19 pandemic had contributed to decreased circulation of respiratory viruses such as influenza and RSV (10). Pandemic period enrollment did not follow seasonal trends, with a notable increase in inpatient and ED enrollments during summer months of 2021. This increase was largely associated with the return of RSV after nearly a year without community circulation.
The findings in this report are subject to at least four limitations. First, NVSN data are limited to enrolled and consented participants who might not be representative of all children seeking care at a healthcare facility. Second, although NVSN surveillance sites are located across the United States, they might not be representative of the entire country. Third, outpatient clinic surveillance differed from the more consistent inpatient and ED surveillance in several ways, including a later start date, prioritized RSV testing during first year of enrollment, paused enrollment during May 2019–October 2019, and age restrictions in several sites, making it difficult to establish trends during the surveillance period. Finally, new approaches to outpatient surveillance (e.g., drive-through clinics) were implemented during the COVID-19 pandemic, which affected enrollment and proportion of positive SARS-CoV-2 test results in this setting.
Prospective ARI surveillance in NVSN measured seasonal trends in respiratory virus circulation before and during the COVID-19 pandemic. These data have the potential to estimate population-based rates of SARS-CoV-2, RSV, and other respiratory virus hospitalizations, ED, and outpatient visits. Further analyses of NVSN data and continued surveillance are vital in highlighting risk factors for severe disease and health disparities, measuring the effectiveness of vaccines and monoclonal antibody–based prophylactics, and guiding policies to protect young children from pathogens such as SARS-CoV-2, influenza, and RSV.
Acknowledgments
Daniel C. Payne, Division of Foodborne, Waterborne, and Environmental Diseases, National Center for Emerging and Zoonotic Infectious Diseases, CDC; Jenelle Putzig, Lynne Shelley, School of Medicine & Dentistry, University of Rochester; Flor M. Muñoz, Department of Pediatrics, Baylor College of Medicine; Laura L. Loftis, Texas Children’s Hospital.
New Vaccine Surveillance Network Collaborators
Bonnie Strelitz, Seattle Children’s Hospital, Seattle, Washington; Kirsten Lacombe, Seattle Children’s Hospital, Seattle, Washington; Mary Moffatt, Children’s Mercy, Kansas City, Missouri; Jennifer Schuster, Children’s Mercy, Kansas City, Missouri; Chelsea Rohlfs, College of Medicine, University of Cincinnati, Cincinnati, Ohio; Miranda Howard, College of Medicine, University of Cincinnati, Cincinnati, Ohio; Yesenia Romero, Vanderbilt University Medical Center, Nashville, Tennessee; James Chappell, Vanderbilt University Medical Center, Nashville, Tennessee; Pedro A. Piedra, Baylor College of Medicine, Houston, Texas; Vasanthi Avadhanula, Baylor College of Medicine, Houston, Texas; Wende Fregoe, School of Medicine & Dentistry, University of Rochester, Rochester, New York; Christina Albertin, School of Medicine & Dentistry, University of Rochester, Rochester, New York; Robert H. Hickey, UPMC Children’s Hospital of Pittsburgh, Pittsburgh, Pennsylvania; Judith M Martin, UPMC Children’s Hospital of Pittsburgh, Pittsburgh, Pennsylvania
Corresponding author: Ariana Perez, oss0@cdc.gov, 404-718-7378.
1Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, CDC; 2General Dynamics Information Technology, Inc., Falls Church, Virginia; 3Department of Pediatrics, University of Rochester School of Medicine & Dentistry, Rochester, New York; 4Department of Pediatrics, Vanderbilt University Medical Center, Nashville, Tennessee; 5Department of Pediatrics, University of Cincinnati College of Medicine, Cincinnati, Ohio; 6Division of Infectious Diseases, Cincinnati Children’s Hospital, Cincinnati, Ohio; 7University of California at Los Angeles, Los Angeles, California; 8IHRC Inc, Atlanta, Georgia; 9Department of Pediatrics, Seattle Children’s Hospital, Seattle, Washington; 10Department of Pathology and Laboratory Medicine, Children’s Mercy, Kansas City, Missouri; 11Texas Children’s Hospital, Houston, Texas; 12Baylor College of Medicine, Houston, Texas; 13Department of Pediatrics, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania; 14UPMC Children’s Hospital of Pittsburgh, Pittsburgh, Pennsylvania.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. Janet A. Englund reports support from AstraZeneca, GSK (GlaxoSmithKline), and Pfizer, Inc., and consulting fees from Sanofi Pasteur, Meissa Vaccines, and AstraZeneca. Natasha B. Halasa reports grant support from Sanofi Pasteur and Quidel and an education grant from Genetech. Christopher J. Harrison reports institutional support from GSK, Merck, and Pfizer, Inc., and honoraria from Pediatric News. Rangaraj Selvarangan reports grants from Hologic, BioFire Diagnostics, Becton Dickinson, Luminex, and Cepheid and serves on the GSK advisory board. Geoffrey A. Weinberg reports consulting fees from ReViral and honoraria from Merck for writing textbook chapters in the Merck Manual. John V. Williams reports grant support from the National Institutes of Health (for work unrelated to the report), consulting fees from Quidel’s scientific advisory board, and honorarium from the Infectious Disease of Children for a conference presentation, participation on a GSK independent data monitoring committee and on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases IMPAACT Study. No other potential conflicts of interest were disclosed.
* Diagnostic assays used for RV/EV might detect only rhinovirus, only enterovirus, or rhinovirus and enterovirus combined.
† Surveillance sites in Kansas City, Pittsburgh, and Seattle restricted ED enrollment primarily to children aged <5 years annually during winter–spring (2016–2019).
§ The seven U.S. pediatric medical center NVSN surveillance sites were in Cincinnati, Ohio; Houston, Texas; Kansas City, Missouri; Nashville, Tennessee; Pittsburgh, Pennsylvania; Rochester, New York; and Seattle, Washington.
¶ Outpatient enrollment of children aged <2 years began in November 2018 and expanded to enroll children aged <18 years during July–August 2020; testing in outpatient settings expanded from RSV alone to multipanel testing in November 2019.
** In general, children were eligible for enrollment if they met some of the same criteria as inpatient and ED patients, which included apnea, myalgia, vomiting after coughing, or apparent life-threatening event or brief resolved unexplained event. Outpatient exclusion criteria also differed; children were excluded if they were seen at an outpatient, inpatient, or ED setting <5 days after an acute respiratory illness, or had been enrolled as outpatients within the previous 4 days.
†† Surveillance sites were provided with instructions on how to properly obtain respiratory specimens and a list of specimen types that might be acceptable to use when a clinical salvage was the only option (i.e., tracheal aspirate, MT, OP, bronchoalveolar lavage, sputum, or nasal wash). Investigators were asked to consult with CDC to determine acceptability of clinically salvaged specimens.
§§ SARS-CoV-2 research testing began during March–April 2020 and was not implemented systematically during the onset of the pandemic because of suspension of enrollment and surveillance activities for 1–3 weeks across sites.
¶¶ Each site performed retrospective SARS-CoV-2 testing on respiratory specimens from children who were enrolled and had specimens collected beginning either January 1 or February 1, 2020.
*** 45 C.F.R. part 46; 21 C.F.R. part 56.
References
- Iwane MK, Edwards KM, Szilagyi PG, et al.; New Vaccine Surveillance Network. Population-based surveillance for hospitalizations associated with respiratory syncytial virus, influenza virus, and parainfluenza viruses among young children. Pediatrics 2004;113:1758–64. https://doi.org/10.1542/peds.113.6.1758 PMID:15173503
- Poehling KA, Edwards KM, Griffin MR, et al. The burden of influenza in young children, 2004–2009. Pediatrics 2013;131:207–16. https://doi.org/10.1542/peds.2012-1255 PMID:23296444
- Hall CB, Weinberg GA, Blumkin AK, et al. Respiratory syncytial virus–associated hospitalizations among children less than 24 months of age. Pediatrics 2013;132:e341–8. https://doi.org/10.1542/peds.2013-0303 PMID:23878043
- Edwards KM, Zhu Y, Griffin MR, et al.; New Vaccine Surveillance Network. Burden of human metapneumovirus infection in young children. N Engl J Med 2013;368:633–43. https://doi.org/10.1056/NEJMoa1204630 PMID:23406028
- Weinberg GA, Hall CB, Iwane MK, et al.; New Vaccine Surveillance Network. Parainfluenza virus infection of young children: estimates of the population-based burden of hospitalization. J Pediatr 2009;154:694–9. https://doi.org/10.1016/j.jpeds.2008.11.034 PMID:19159905
- Rha B, Lively JY, Englund JA, et al. Severe acute respiratory syndrome coronavirus 2 infections in children: multicenter surveillance, United States, January–March 2020. J Pediatric Infect Dis Soc 2020;9:609–12. https://doi.org/10.1093/jpids/piaa075 PMID:32556327
- CDC. Update: influenza activity—United States, 1999–2000 season. MMWR Morb Mortal Wkly Rep 2000;49:173–7. PMID:11795497
- Rose EB, Wheatley A, Langley G, Gerber S, Haynes A. Respiratory syncytial virus seasonality—United States, 2014–2017. MMWR Morb Mortal Wkly Rep 2018;67:71–6. https://doi.org/10.15585/mmwr.mm6702a4 PMID:29346336
- Sahni LC, Avadhanula V, Ortiz CS, et al. Comparison of mid-turbinate and nasopharyngeal specimens for molecular detection of SARS-CoV-2 among symptomatic outpatients at a pediatric drive-through testing site. J Pediatric Infect Dis Soc 2021;10:872–9. https://doi.org/10.1093/jpids/piab046 PMID:34173660
- Haddadin Z, Schuster JE, Spieker AJ, et al. Acute respiratory illnesses in children in the SARS-CoV-2 pandemic: prospective multicenter study. Pediatrics 2021;148:e2021051462. https://doi.org/10.1542/peds.2021-051462 PMID:33986150
Abbreviations: ED = emergency department; RSV = respiratory syncytial virus.
* Among ED surveillance sites, enrollment was restricted to children aged <5 years during the following periods: Seattle during December 2016–June 2017, November 2017–June 2018, November 2018–June 2019, and December 2019–March 2020; Pittsburgh during December 2016–June 2018, November 2018–June 2019, and December 2019–March 2020; Kansas City during December 2016–June 2017, November 2017–June 2018, and November 2018–June 2019.
† Outpatient enrollment began in November 2018, paused during May–October 2019, and resumed with enrolled children aged <2 years during November 2018–July 2020; RSV testing was prioritized during November 2018–April 2019.
Abbreviations: ED = emergency department; HMPV = human metapneumovirus; HPIV1–3 = human parainfluenza virus types 1–3; NA = not applicable; RSV = respiratory syncytial virus; RV/EV = rhinovirus and enterovirus.
* Respiratory virus detection results are from research swab specimens that underwent molecular testing, except for SARS-CoV-2, which included both research and clinical specimens to most accurately represent viral detections across surveillance years. Denominators for positivity rates are pathogen-specific.
† Surveillance seasons during 2016–2017 were December 1, 2016–November 30, 2017; 2017–2018: December 1, 2017–October 31, 2018; 2018–2019: November 1, 2018–October 31, 2019; 2019–2020: November 1, 2019–September 14, 2020; 2020–2021: September 15, 2020–August 31, 2021.
§ SARS-CoV-2 was first detected in 2020, test results for SARS-CoV-2 reported in this table are from the pandemic period (March 2020–August 2021); surveillance years 2016–2017 through 2018–2019 were not applicable.
¶ Outpatient data were not included for seasons 2016–2017 and 2017–2018 because outpatient enrollment did not begin until November 2018.
 FIGURE. Respiratory virus detections among enrolled children and adolescents aged <18 years with research tested specimens, by highest level of care in inpatient (A), emergency department (B), and outpatient (C) settings — New Vaccine Surveillance Network, United States, December 2016–August 2021 *,†,§
FIGURE. Respiratory virus detections among enrolled children and adolescents aged <18 years with research tested specimens, by highest level of care in inpatient (A), emergency department (B), and outpatient (C) settings — New Vaccine Surveillance Network, United States, December 2016–August 2021 *,†,§
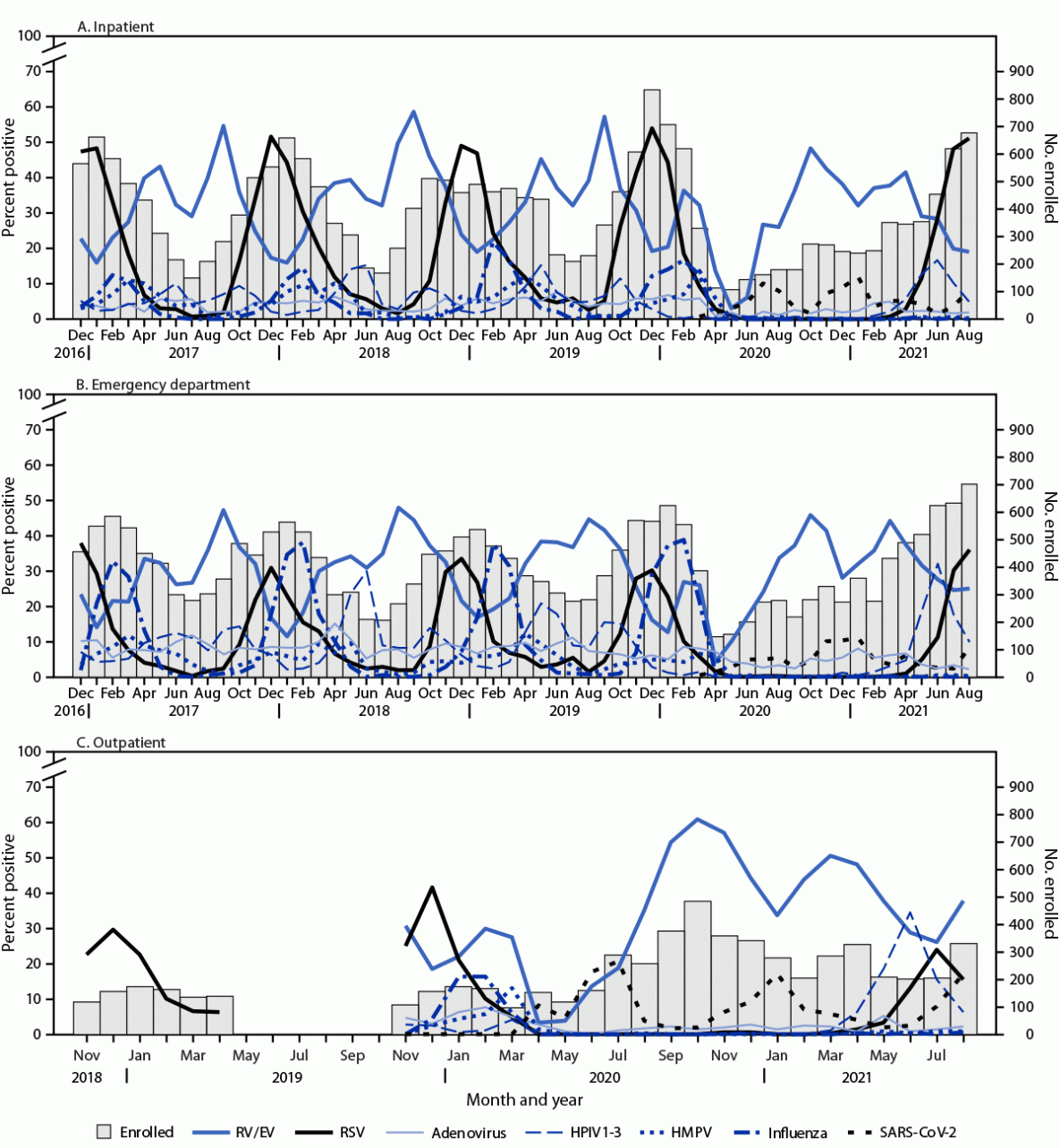
Abbreviations: ED = emergency department; HPMV = human metapneumovirus; HPIV1–3 = human parainfluenza virus types 1–3; RSV = respiratory syncytial virus; RV/EV = rhinoviruses and enteroviruses.
* Outpatient enrollment began in November 2018, paused during May–October 2019, and resumed with enrolled children aged <2 years during November 2018–July 2020; RSV testing was prioritized during November 2018–April 2019.
† SARS-CoV-2 detections only included research positive test results for consistency across pathogens; therefore, total detections are underrepresented.
§ Surveillance was paused at these sites during the COVID-19 pandemic: Cincinnati (inpatient: March 25–30, 2020; ED: March 24–30, 2020; and outpatient: March 25, 2020); Seattle (outpatient: March 2–12, 2020 and March 13–31, 2020); Houston (inpatient, ED, and outpatient: March 23–31, 2020); Kansas City (inpatient: March 18–29, 2020; ED: March 18–28, 2020; outpatient: March 18–31, 2020); and Pittsburgh (inpatient and ED: March 22–29, 2020 and outpatient: March 13–31, 2020).
Suggested citation for this article: Perez A, Lively JY, Curns A, et al. Respiratory Virus Surveillance Among Children with Acute Respiratory Illnesses — New Vaccine Surveillance Network, United States, 2016–2021. MMWR Morb Mortal Wkly Rep 2022;71:1253–1259. DOI: http://dx.doi.org/10.15585/mmwr.mm7140a1.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.