Hospitalizations of Children Aged 5–11 Years with Laboratory-Confirmed COVID-19 — COVID-NET, 14 States, March 2020–February 2022
Weekly / April 22, 2022 / 71(16);574-581
On April 19, 2022, this report was posted online as an MMWR Early Release.
Dallas S. Shi, MD, PhD1,2; Michael Whitaker, MPH1; Kristin J. Marks, PhD1,2; Onika Anglin, MPH1,3; Jennifer Milucky, MSPH1; Kadam Patel, MPH1,3; Huong Pham, MPH1; Shua J. Chai, MD4,5; Breanna Kawasaki, MPH6; James Meek, MPH7; Evan J. Anderson, MD8,9,10; Andy Weigel, MSW11; Justin Henderson, MPH12; Ruth Lynfield, MD13; Susan L. Ropp, PhD14; Alison Muse, MPH15; Sophrena Bushey, MHS16; Laurie M. Billing, MPH17; Melissa Sutton, MD18; H. Keipp Talbot, MD19; Andrea Price20; Christopher A. Taylor, PhD1; Fiona P. Havers, MD1; COVID-NET Surveillance Team (View author affiliations)
View suggested citationSummary
What is already known about this topic?
COVID-19 can cause severe illness in children. Children aged 5–11 years became eligible for COVID-19 vaccination on November 2, 2021.
What is added by this report?
During the period of Omicron predominance (December 19, 2021–February 28, 2022), COVID-19–associated hospitalization rates in children aged 5–11 years were approximately twice as high among unvaccinated as among vaccinated children. Non-Hispanic Black children represented the largest group of unvaccinated children. Thirty percent of hospitalized children had no underlying medical conditions, and 19% were admitted to an intensive care unit. Children with diabetes and obesity were more likely to experience severe COVID-19.
What are the implications for public health practice?
Increasing COVID-19 vaccination coverage among children aged 5–11 years, particularly among racial and ethnic minority groups disproportionately affected by COVID-19, can prevent COVID-19–associated hospitalization and severe outcomes.
Altmetric:
On October 29, 2021, the Food and Drug Administration expanded the Emergency Use Authorization for Pfizer-BioNTech COVID-19 vaccine to children aged 5–11 years; CDC’s Advisory Committee on Immunization Practices’ recommendation followed on November 2, 2021.* In late December 2021, the B.1.1.529 (Omicron) variant of SARS-CoV-2 (the virus that causes COVID-19) became the predominant strain in the United States,† coinciding with a rapid increase in COVID-19–associated hospitalizations among all age groups, including children aged 5–11 years (1). COVID-19–Associated Hospitalization Surveillance Network (COVID-NET)§ data were analyzed to describe characteristics of COVID-19–associated hospitalizations among 1,475 U.S. children aged 5–11 years throughout the pandemic, focusing on the period of early Omicron predominance (December 19, 2021–February 28, 2022). Among 397 children hospitalized during the Omicron-predominant period, 87% were unvaccinated, 30% had no underlying medical conditions, and 19% were admitted to an intensive care unit (ICU). The cumulative hospitalization rate during the Omicron-predominant period was 2.1 times as high among unvaccinated children (19.1 per 100,000 population) as among vaccinated¶ children (9.2).** Non-Hispanic Black (Black) children accounted for the largest proportion of unvaccinated children (34%) and represented approximately one third of COVID-19–associated hospitalizations in this age group. Children with diabetes and obesity were more likely to experience severe COVID-19. The potential for serious illness among children aged 5–11 years, including those with no underlying health conditions, highlights the importance of vaccination among this age group. Increasing vaccination coverage among children, particularly among racial and ethnic minority groups disproportionately affected by COVID-19, is critical to preventing COVID-19-associated hospitalization and severe outcomes.
COVID-NET conducts population-based surveillance for laboratory-confirmed COVID-19–associated hospitalizations in 99 counties across 14 U.S. states.†† COVID-19–associated hospitalizations are defined as receipt of a positive SARS-CoV-2 nucleic acid amplification tests or rapid antigen detection test result during hospitalization or during the 14 days preceding admission. This analysis describes hospitalization rates among children aged 5–11 years during March 1, 2020–February 28, 2022. Clinical data from the Omicron-predominant period were compared with those from the Delta-predominant (June 27–December 18, 2021) and pre-Delta (March 1, 2020–June 26, 2021) periods; a variant that accounted for >50% of sequenced isolates was considered predominant. Unadjusted weekly COVID-19–associated hospitalization rates (COVID-19–related hospitalizations per 100,000 children) were calculated by dividing the total number of COVID-19–associated hospitalizations by the population estimates for the counties included in the surveillance area.§§ ICU admission rates were calculated using 2-week periods. Population-based hospitalization rates and data for hospitalized children were compared by COVID-19 vaccination status for the Omicron-predominant period using linkage to state immunization information systems data.¶¶
Trained surveillance officers abstracted medical charts for hospitalized pediatric patients using standardized case report forms through November 2021. Because of the surge in hospitalizations during December 2021–February 2022, some sites examined clinical data on a representative sample of hospitalized children during this period.*** The representative sample included 1,252 of 1,475 (84.9%) children with positive SARS-CoV-2 test results; complete clinical data were available for 595 of 596 (99.8%), 438 of 468 (93.6%), and 219 of 225 (97.3%) sampled children aged 5–11 years during the pre-Delta period, Delta-predominant period, and Omicron-predominant period.
Data regarding likely primary reason for hospital admission,††† symptoms at admission,§§§ underlying medical conditions, ¶¶¶ vaccination status (complete versus incomplete), and indicators of severe disease (e.g., length of stay, ICU admission, receipt of invasive mechanical ventilation [IMV],**** and in-hospital death) were collected (2). Children who completed their primary COVID-19 vaccination series were defined as those who had received the second dose of a 2-dose series ≥14 days before receipt of a positive SARS-CoV-2 test result associated with their hospitalization. Wilcoxon rank-sum tests and chi-square tests were used to compare medians and proportions, respectively; p<0.05 was considered statistically significant. Percentages were weighted to account for probability of selection for sampled cases and adjusted to account for nonresponse. Association of underlying medical conditions with severe COVID-19 (defined as requiring ICU admission or IMV, or in-hospital death) was modeled using multivariable generalized estimating equations (2). Multivariable models were limited to children whose primary reason for admission was likely COVID-19–related. Unadjusted risk ratios (RRs), adjusted RRs (aRRs), and 95% CIs were calculated for the association of demographic characteristics, underlying medical conditions, and variant periods with severe COVID-19. Data were analyzed using SAS (version 9.4; SAS Institute). This activity was reviewed by CDC and conducted consistent with applicable federal law and CDC policy. ††††
During the Delta- and Omicron-predominant periods, weekly hospitalization rates of children aged 5–11 years peaked during the weeks ending September 25, 2021 and January 22, 2022, respectively; the Omicron-predominant peak (2.8 per 100,000 children) was 2.3 times the Delta-predominant peak (1.2).§§§§ Peak ICU admission rates were 1.7 times as high during Omicron predominance (2-week period ending January 25, 2022 [1.2]) than during Delta predominance (2-week period ending October 2, 2021 [0.7]).
During the Omicron-predominant period, cumulative hospitalization rates among unvaccinated children aged 5–11 years were 2.1 times as high (19.1) as those among vaccinated children (9.2) (Figure). Most (87%) children aged 5–11 years hospitalized during the Omicron-predominant period were unvaccinated (Supplemental Table, https://stacks.cdc.gov/view/cdc/116353). Among unvaccinated children, the largest proportion were Black (34%), followed by White (31%), and Hispanic (19%). There were no significant differences for severe outcomes by vaccination status. However, the number of vaccinated children was small. No vaccinated children required higher level O2 support (e.g., bilevel positive airway pressure/continuous positive airway pressure [BiPAP/CPAP], high flow nasal canula, or IMV).
COVID-19–related illness was the primary reason for admission among a lower proportion of hospitalized children aged 5–11 years during the Omicron period (73%) compared with the Delta period (84%) (p<0.01); across all periods, a majority (78%) of children were hospitalized with COVID-19 as the likely primary reason for admission (Table 1). Of the hospitalized children, 67% had one or more underlying medical conditions. During the period of Omicron predominance, a larger proportion of children hospitalized with COVID-19 had neurologic disorders (33%) compared with those hospitalized during the pre-Delta period (21%) (p<0.01), and a lower proportion had obesity (33% and 21%, respectively; p = 0.01). Similar trends were observed when comparing the Omicron- and Delta-predominant periods. Among children hospitalized during the Omicron-predominant period, 19% required ICU admission, including 15% with no underlying medical conditions; 5% received IMV; none died.
Across periods, 32% of hospitalized children aged 5–11 years had severe COVID-19; 44% of Black children and 26% of Hispanic children experienced severe disease, compared with 22% of White children, but the association between severe COVID-19 and race or Hispanic ethnicity was not statistically significant (Table 2). The risk for severe COVID-19 among hospitalized children was significantly higher among those with diabetes (aRR = 2.5) and obesity (aRR = 1.2). Risk for severe disease was lower among children with asthma (aRR = 0.8), immunocompromising conditions (aRR = 0.7), and those hospitalized during the Delta-predominant (aRR = 0.8) and Omicron-predominant periods (aRR = 0.6). Other conditions were not significantly associated with severe COVID-19 among hospitalized children.
Discussion
Peak weekly COVID-19–associated hospitalization rates among children aged 5–11 years were higher during the Omicron-predominant period than during the Delta-predominant period. During Omicron predominance, shortly after the Food and Drug Administration authorized COVID-19 vaccination for this age group, population-based hospitalization rates among unvaccinated children were twice as high as were those among vaccinated children. Most hospitalized children were unvaccinated, and nearly one in three were Black. Approximately one third had no underlying medical conditions, and nearly one fifth required ICU admission. The potential for serious illness among children aged 5–11 years, including those with no underlying health conditions, highlights the importance of vaccination among this age group.
Vaccination eligibility was expanded to include children aged 5–11 years on November 2, 2021. As of March 5, 2022, 32% of children in this age group had completed a COVID-19 primary vaccination series. ¶¶¶¶ In this study, approximately one half (53%) of unvaccinated hospitalized children were Black or Hispanic, two groups known to have lower vaccination rates (3). Implementing strategies that result in equitable receipt of COVID-19 vaccine among children is a public health priority.
The finding that hospitalization rates in unvaccinated children were double those of vaccinated children suggests that vaccines are effective in preventing COVID-19–associated morbidities. This is consistent with recent studies, which suggest that vaccination reduces the risk for Omicron infection, protects against COVID-19–associated illness among children aged 5–11 years and prevents multisystem inflammatory syndrome in children, a severe postinfectious hyperinflammatory condition with a higher incidence in this age group than in other age groups (4–7).
Consistent with other studies, this analysis demonstrated that the Omicron-predominant period was associated with less severe disease among hospitalized children (8). However, both population-based peak hospitalization and ICU admission rates were higher during the Omicron-predominant period compared with those during the Delta-predominant period, likely because of the high transmissibility of the Omicron variant and greater number of persons infected. Although a higher proportion of children hospitalized with laboratory-confirmed SARS-CoV-2 infection were admitted for reasons that were not likely primarily COVID-19–related during the Omicron period compared with the Delta period, most children admitted during both periods were hospitalized primarily for COVID-19. These findings suggest that incidental admissions do not account for the increase in hospitalization rates observed during the Omicron period and reinforce that children continued to experience serious COVID-19 illness.
As in previous investigations, diabetes and obesity were associated with increased risk for severe COVID-19 in children (2). One third of hospitalized children aged 5–11 years had underlying neurologic disorders during the Omicron-predominant period, an increase from previous periods. Neurologic disorders have been shown to increase risk for severe illness in other respiratory diseases such as influenza (9). Consistent with findings from influenza-associated hospitalizations, this study found that some underlying medical conditions, including asthma and immunocompromising conditions, were not associated with increased risk for severe COVID-19, which might be explained by a lower threshold for hospital admission in children with these conditions (10).
The findings in this report are subject to at least five limitations. First, COVID-19–associated hospitalizations might have been missed because of testing practices and availability. Second, stratification of hospitalization rate by vaccination status is subject to error if misclassification of vaccination status occurred. Third, analyses based on vaccination status are biased toward the null because partially vaccinated children were grouped with unvaccinated children. Fourth, primary reason for admission was not always clear, and medical charts might not completely capture underlying conditions, potentially resulting in misclassification. Finally, COVID-NET catchment areas include approximately 10% of the U.S. population; thus, these findings might not be generalizable to the rest of the United States.
Potential for serious disease requiring hospitalization, ICU admission, or IMV among children aged 5–11 years reinforces the importance of increasing vaccination coverage among this population. Black children accounted for the highest percentage of unvaccinated children in this analysis and represented one third of COVID-19–associated hospitalizations in this age group. Increasing COVID-19 vaccination coverage among children aged 5–11 years, with particular attention to racial and ethnic minority groups disproportionately affected by COVID-19, is critical to reducing COVID-19–associated morbidity.*****
Acknowledgments
Brooke Heidenga, Joelle Nadle, Monica Napoles, Sherry Quach, Gretchen Rothrock, California Emerging Infections Program; Nisha Alden, Madelyn Lensing, Sarah McLafferty, Jordan Surgnier, Millen Tsegaye, Colorado Department of Public Health and Environment; Ann Basting, Tessa Carter, Maria Correa, Daewi Kim, Carol Lyons, Amber Maslar, Julie Plano, Hazhia Sorosindi, Connecticut Emerging Infections Program, Yale School of Public Health, New Haven, Connecticut; Marina Bruck, Rayna Ceaser, Taylor Eisenstein, Emily Fawcett, Asmith Joseph, Sabrina Hendrick, Johanna Hernandez, Grayson Kallas, Stephanie Lehman, Jana Manning, Annabel Patterson, Allison Roebling, Suzanne Segler, Chandler Surell, Katelyn Ward, Hope Wilson, School of Medicine, Emory University, Atlanta, Georgia; Georgia Emerging Infections Program, Georgia Department of Public Health, Veterans Affairs Medical Center, Foundation for Atlanta Veterans Education and Research, Atlanta, Georgia; Chloe Brown, Jim Collins, Shannon Johnson, Alexander Kohrman, Lauren Leegwater, Sierra Peguies-Khan, Libby Reeg, Michigan Department of Health and Human Services; Alison Babb, Kayla Bilski, Kathy Como-Sabetti, Emma Contestabile, Jennifer Gilbertson, Claire Henrichsen, Grace Hernandez, Erica Mumm, Lisa Nguyen, Kieu My Phi, Minnesota Department of Health; Cory Cline, Melissa Judson, Sunshine Martinez, Mark Montoya, Florent Nkouaga, Kelly Plymesser, Jasmyn Sanchez, Chad Smelser, Daniel Sosin, New Mexico Department of Health; Kathy M. Angeles, Molly Bleecker, Emily B. Hancock, Sarah A. Khanlian, Sarah Lathrop, Wickliffe Omondi, Francesca Pacheco, Mayvilynne Poblete, Dominic Rudin, Yadira Salazar-Sanchez, Sarah Shrum Davis, New Mexico Emerging Infections Program; Jennifer Akpo, Celina Chavez, Murtada Khalifa, Alesia Reed, Yassir Talha, CDC Foundation, Atlanta, Georgia; Kerianne Engesser, Adam Rowe, Nancy Spina, New York State Department of Health; Virginia Cafferky, Liz Cej, Maria Gaitan, Christine Long, Thomas Peer, Kevin Popham, University of Rochester School of Medicine and Dentistry, Rochester, New York; Julie Freshwater, Denise Ingabire-Smith, Nancy Moran, Ann Salvator, Rebekah Sutter, Ohio Department of Health; Kathy Billings, Katie Dyer, Anise Elie, Gail Hughett, Karen Leib, Tiffanie Markus, Terri McMinn, Danielle Ndi, Manideepthi Pemmaraju, Emmanuel Sackey, Vanderbilt University Medical Center, Nashville, Tennessee; Amanda Carter, Ryan Chatelain, Melanie Crossland, Andrea George, Andrew Haraghey, Mary Hill, Laine McCullough, Emily Roberts, Ashley Swain, Salt Lake County Health Department, Salt Lake City, Utah; Rainy Henry, Sonja Mali Nti-Berko, Robert W. Pinner, Alvin Shultz, Rebecca C. Woodruff, CDC; Elizabeth Daly, Council of State and Territorial Epidemiologists.
COVID-NET Surveillance Team
Pam Daily Kirley, California Emerging Infections Program, Oakland, California; Isaac Armistead, Colorado Department of Public Health and Environment; Kimberly Yousey-Hindes, Connecticut Emerging Infections Program, Yale School of Public Health, New Haven, Connecticut; Kyle P. Openo, Georgia Emerging Infections Program, Georgia Department of Public Health, Division of Infectious Diseases, School of Medicine, Emory University, Atlanta, Georgia and Atlanta Veterans Affairs Medical Center, Atlanta, Georgia; Kenzie Teno, Iowa Department of Public Health; Val Tellez Nunez, Michigan Department of Health and Human Services; Erica Bye, Minnesota Department of Health; Nancy L. Eisenberg, New Mexico Emerging Infections Program, University of New Mexico, Albuquerque, New Mexico; Grant R. Barney, New York State Department of Health; Christina B. Felsen, University of Rochester School of Medicine and Dentistry, Rochester, New York; Eli Shiltz, Ohio Department of Health; Nasreen Abdullah, Public Health Division, Oregon Health Authority; William Schaffner, Vanderbilt University Medical Center, Nashville, Tennessee; Keegan McCaffrey, Utah Department of Health.
Corresponding author: Dallas S. Shi, Dshi@cdc.gov.
1CDC COVID-19 Emergency Response Team; 2Epidemic Intelligence Service, CDC; 3General Dynamics Information Technology, Atlanta, Georgia; 4California Emerging Infections Program, Oakland, California; 5Career Epidemiology Field Officer Program, CDC; 6Colorado Department of Public Health and Environment; 7Connecticut Emerging Infections Program, Yale School of Public Health, New Haven, Connecticut; 8Emory University School of Medicine, Atlanta, Georgia; 9Georgia Emerging Infections Program, Georgia Department of Health; 10Atlanta Veterans Affairs Medical Center, Atlanta, Georgia; 11Iowa Department of Public Health; 12Michigan Department of Health and Human Services; 13Minnesota Department of Health; 14New Mexico Department of Health; 15New York State Department of Health; 16University of Rochester School of Medicine and Dentistry, Rochester, New York; 17Ohio Department of Health; 18Public Health Division, Oregon Health Authority; 19Vanderbilt University Medical Center, Nashville, Tennessee; 20Salt Lake County Health Department, Salt Lake City, Utah.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. Evan J. Anderson reports grants from Pfizer, Merck, PaxVax, Micron, Sanofi-Pasteur, Janssen, MedImmune, and GlaxoSmithKline; personal fees from Pfizer, Medscape, Kentucky Bioprocessing, Inc., Sanofi-Pasteur, Janssen, and GlaxoSmithKline outside the submitted work; and institutional funding from the National Institutes of Health to conduct clinical trials of Moderna and Janssen COVID-19 vaccines. Laurie M. Billing, Andy Weigel, Justin Henderson, and Andrea Price report grants from the Council of State and Territorial Epidemiologists during the conduct of the study. Ruth Lynfield reports editorial payments from the American Academy of Pediatrics Red Book (Committee on Infectious Diseases), which were donated to the Minnesota Department of Health. No other potential conflicts of interest were disclosed.
* https://www.fda.gov/news-events/press-announcements/fda-authorizes-pfizer-biontech-covid-19-vaccine-emergency-use-children-5-through-11-years-age; https://www.cdc.gov/media/releases/2021/s1102-PediatricCOVID-19Vaccine.html
† Omicron became the predominant variant during the week ending December 25, 2021 at 74% of sequenced isolates. https://covid.cdc.gov/covid-data-tracker/#variant-proportions
§ https://www.cdc.gov/coronavirus/2019-ncov/covid-data/covid-net/purpose-methods.html
¶ Vaccinated children aged 5–11 years were defined as those who had received the final dose in their primary series ≥14 days before receiving a positive SARS-CoV-2 test result associated with their hospitalization. Children who had received only 1 vaccine dose ≥14 days before the SARS-CoV-2 test date or had received a single dose of vaccine <14 days before the positive SARS-CoV-2 test results were considered partially vaccinated; these children were not included in rates and were grouped with unvaccinated children in other analyses.
** https://covid.cdc.gov/covid-data-tracker/#covidnet-hospitalizations-vaccination
†† California, Colorado, Connecticut, Georgia, Iowa, Maryland, Michigan, Minnesota, New Mexico, New York, Ohio, Oregon, Tennessee, and Utah.
§§ Rates are calculated using the National Center for Health Statistics’ vintage 2020 bridged-race postcensal population estimates for the counties included in surveillance. https://www.cdc.gov/nchs/nvss/bridged_race.htm
¶¶ COVID-NET sites, through agreements with state health departments and other partners, collect COVID-19 vaccination information on COVID-19–associated hospitalizations through state-based vaccination registries. When possible, sites collect COVID-19 vaccination status on all persons with COVID-19 cases who are hospitalized, including the number of vaccine doses received, the vaccine product, and dates of vaccine administration. Vaccination information was not available for Iowa, Maryland, and Michigan and only available for sampled cases in Minnesota.
*** During December 2021–February 2022, sites sampled pediatric patients at rates of 12%–100%. Random numbers (1–100) were automatically generated and assigned to each patient on entry into the surveillance database to produce random samples of hospitalized patients for medical record abstraction. Percentages were weighted to account for the probability of selection for sampled patients.
††† Among sampled cases, COVID-NET collects data on the primary reason for admission to differentiate hospitalizations of patients with laboratory-confirmed SARS-CoV-2 infection who are likely admitted primarily for COVID-19 illness from those admitted for other reasons, including inpatient surgery or trauma. During chart review, if the surveillance officer finds that the chief complaint or history of present illness mentions fever/respiratory illness, COVID-19–like illness, or a suspicion for COVID-19, then the case is categorized with COVID-19–related illness as the primary reason for admission.
§§§ COVID-19–related symptoms included respiratory symptoms (congestion/runny nose, cough, hemoptysis/bloody sputum, shortness of breath/respiratory distress, sore throat, upper respiratory infection, influenza-like illness, and wheezing) and nonrespiratory symptoms (abdominal pain, altered mental status/confusion, anosmia/decreased smell, chest pain, conjunctivitis, diarrhea, dysgeusia/decreased taste, fatigue, fever/chills, headache, muscle aches/myalgias, nausea/vomiting, rash, and seizures). Symptoms were abstracted from the medical chart and might be incomplete.
¶¶¶ Thirteen underlying conditions were considered, including airway abnormality, asthma, blood disorders, cardiovascular disease, developmental delay, diabetes mellitus (type 1 or 2), feeding tube dependence, immunocompromising conditions, obesity (body mass index [kg/m2] ≥95th percentile for age and sex based on CDC growth charts; International Classification of Diseases, Tenth Revision, Clinical Modification codes for obesity; or obesity selected on the case report form), nonasthma chronic lung disease, nondiabetes chronic metabolic disease, nondevelopmental delay neurologic disorders, or other conditions (gastrointestinal or liver disease; renal disease; or rheumatologic, autoimmune, or inflammatory disease).
**** ICU admission and need for mechanical ventilation are not mutually exclusive categories, and patients could have received both.
†††† 45 C.F.R. part 46.102(l)(2), 21 C.F.R. part 56; 42 U.S.C. Sect. 241(d); 5 U.S.C. Sect. 552a; 44 U.S.C. Sect. 3501 et seq.
§§§§ https://covid.cdc.gov/covid-data-tracker/#covidnet-hospitalization-network
¶¶¶¶ https://covid.cdc.gov/covid-data-tracker/#vaccination-demographics-trends
***** https://www.cdc.gov/coronavirus/2019-ncov/vaccines/stay-up-to-date.html
References
- Marks KJ, Whitaker M, Anglin O, et al.; COVID-NET Surveillance Team. Hospitalizations of children and adolescents with laboratory-confirmed COVID-19—COVID-NET, 14 states, July 2021–January 2022. MMWR Morb Mortal Wkly Rep 2022;71:271–8. https://doi.org/10.15585/mmwr.mm7107e4 PMID:35176003
- Woodruff RC, Campbell AP, Taylor CA, et al. Risk factors for severe COVID-19 in children. Pediatrics 2022;149:e2021053418. https://doi.org/10.1542/peds.2021-053418
- Murthy NC, Zell E, Fast HE, et al. Disparities in first dose COVID-19 vaccination coverage among children 5–11 years of age, United States. Emerg Infect Dis 2022;. https://doi.org/10.3201/eid2805.220166 PMID:35226801
- Fowlkes AL, Yoon SK, Lutrick K, et al. Effectiveness of 2-dose BNT162b2 (Pfizer BioNTech) mRNA vaccine in preventing SARS-CoV-2 Infection among children aged 5–11 years and adolescents aged 12–15 years—PROTECT Cohort, July 2021–February 2022. MMWR Morb Mortal Wkly Rep 2022;71:422–8. https://doi.org/10.15585/mmwr.mm7111e1 PMID:35298453
- Klein NP, Stockwell MS, Demarco M, et al. Effectiveness of COVID-19 Pfizer-BioNTech BNT162b2 mRNA vaccination in preventing COVID-19–associated emergency department and urgent care encounters and hospitalizations among nonimmunocompromised children and adolescents aged 5–17 Years—VISION Network, 10 States, April 2021–January 2022. MMWR Morb Mortal Wkly Rep 2022;71:352–8. https://doi.org/10.15585/mmwr.mm7109e3 PMID:35239634
- Zambrano LD, Newhams MM, Olson SM, et al.; Overcoming COVID-19 Investigators. Effectiveness of BNT162b2 (Pfizer-BioNTech) mRNA vaccination against Multisystem Inflammatory Syndrome in Children among persons aged 12–18 years—United States, July–December 2021. MMWR Morb Mortal Wkly Rep 2022;71:52–8. https://doi.org/10.15585/mmwr.mm7102e1 PMID:35025852
- Payne AB, Gilani Z, Godfred-Cato S, et al.; MIS-C Incidence Authorship Group. Incidence of multisystem inflammatory syndrome in children among US persons infected with SARS-CoV-2. JAMA Netw Open 2021;4:e2116420. https://doi.org/10.1001/jamanetworkopen.2021.16420 PMID:34110391
- Iuliano AD, Brunkard JM, Boehmer TK, et al. Trends in disease severity and health care utilization during the early Omicron variant period compared with previous SARS-CoV-2 high transmission periods—United States, December 2020–January 2022. MMWR Morb Mortal Wkly Rep 2022;71:146–52. https://doi.org/10.15585/mmwr.mm7104e4 PMID:35085225
- Havers F, Fry AM, Chen J, et al. Hospitalizations attributable to respiratory infections among children with neurologic disorders. J Pediatr 2016;170:135–41.e15. https://doi.org/10.1016/j.jpeds.2015.11.030 PMID:26687576
- Collins JP, Campbell AP, Openo K, et al. Clinical features and outcomes of immunocompromised children hospitalized with laboratory-confirmed influenza in the United States, 2011–2015. J Pediatric Infect Dis Soc 2019;8:539–49. https://doi.org/10.1093/jpids/piy101 PMID:30358877
 FIGURE. Weekly COVID-19–associated hospitalization rates* among children aged 5–11 years, by vaccination status† during the Omicron-predominant period — COVID-NET,§ 11 states, December 25, 2021– February 26, 2022
FIGURE. Weekly COVID-19–associated hospitalization rates* among children aged 5–11 years, by vaccination status† during the Omicron-predominant period — COVID-NET,§ 11 states, December 25, 2021– February 26, 2022
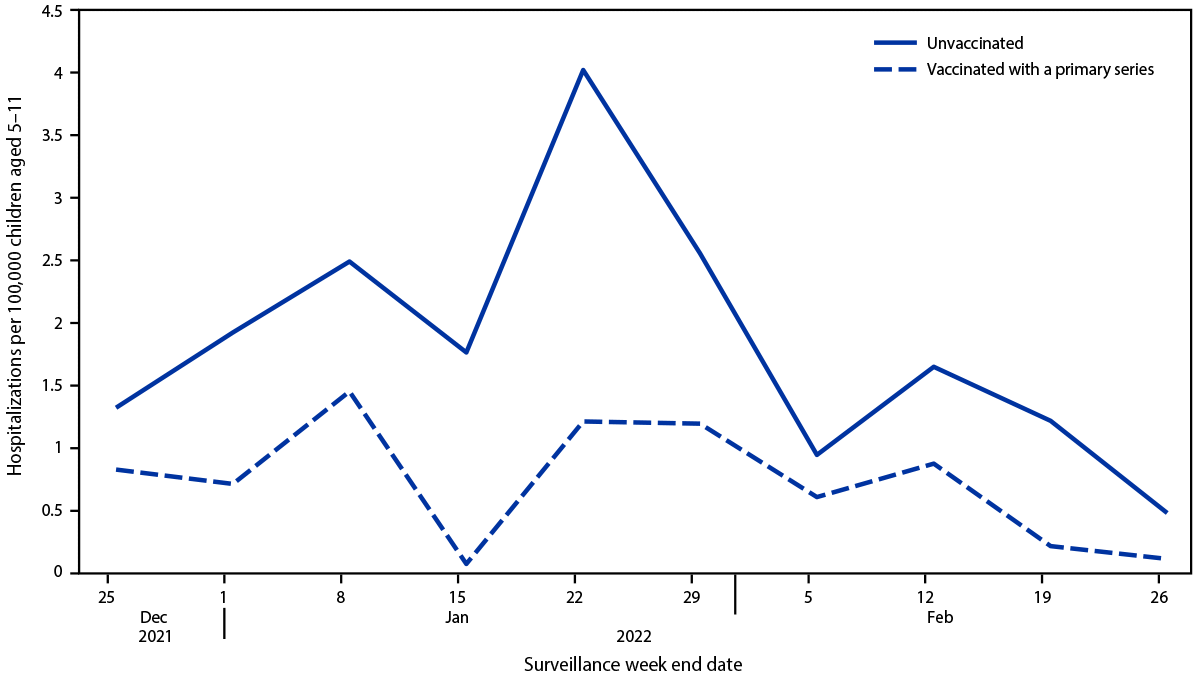
Abbreviation: COVID-NET = COVID-19–Associated Hospitalization Surveillance Network.
* Number of children aged 5–11 years with laboratory-confirmed COVID-19–associated hospitalizations per 100,000 population; rates are subject to change as additional data are reported.
† Children who completed their primary COVID-19 vaccination series were defined as those who had received the second dose of a 2-dose series ≥14 days before receipt of a positive SARS-CoV-2 test result associated with their hospitalization.
§ COVID-NET sites during the period shown are in the following 11 states: California, Colorado, Connecticut, Georgia, Minnesota, New Mexico, New York, Ohio, Oregon, Tennessee, and Utah.
Abbreviations: COVID-NET = COVID-19–Associated Hospitalization Surveillance Network; GI = gastrointestinal; ICU = intensive care unit; NA = not applicable.
* Includes persons admitted to a hospital during March 1, 2020–February 28, 2022. Maryland contributed data through November 26, 2021. Counties included in COVID-NET surveillance during this period: California (Alameda, Contra Costa, and San Francisco counties); Colorado (Adams, Arapahoe, Denver, Douglas, and Jefferson counties); Connecticut (Middlesex and New Haven counties); Georgia (Clayton, Cobb, DeKalb, Douglas, Fulton, Gwinnett, Newton, and Rockdale counties); Iowa (one county represented); Maryland (Allegany, Anne Arundel, Baltimore, Baltimore City, Calvert, Caroline, Carroll, Cecil, Charles, Dorchester, Frederick, Garrett, Harford, Howard, Kent, Montgomery, Prince George’s, Queen Anne’s, St. Mary’s, Somerset, Talbot, Washington, Wicomico, and Worcester counties); Michigan (Clinton, Eaton, Genesee, Ingham, and Washtenaw counties); Minnesota (Anoka, Carver, Dakota, Hennepin, Ramsey, Scott, and Washington counties); New Mexico (Bernalillo, Chaves, Doña Ana, Grant, Luna, San Juan, and Santa Fe counties); New York (Albany, Columbia, Genesee, Greene, Livingston, Monroe, Montgomery, Ontario, Orleans, Rensselaer, Saratoga, Schenectady, Schoharie, Wayne, and Yates counties); Ohio (Delaware, Fairfield, Franklin, Hocking, Licking, Madison, Morrow, Perry, Pickaway and Union counties); Oregon (Clackamas, Multnomah, and Washington counties); Tennessee (Cheatham, Davidson, Dickson, Robertson, Rutherford, Sumner, Williamson, and Wilson counties); and Utah (Salt Lake County).
† Data are from a weighted sample of hospitalized children with completed medical record abstractions. Sample sizes presented are unweighted with weighted percentages.
§ Proportions between the Omicron and Delta- and Omicron-predominant and pre-Delta periods were compared using chi-square tests, and medians were compared using Wilcoxon rank-sum tests; p<0.05 was considered statistically significant.
¶ Data are missing for <3% of observations for all variables.
** If ethnicity was unknown, non-Hispanic ethnicity was assumed.
†† Includes non-Hispanic persons reported as other or multiple races.
§§ Primary reason for admission was collected beginning June 1, 2020; hospitalizations before June 1, 2020 (42) are excluded. Among sampled patients, COVID-NET collects data on the primary reason for admission to differentiate hospitalizations of patients with laboratory-confirmed SARS-CoV-2 infection who are likely admitted primarily for COVID-19 illness rather than for other reasons. During chart review, if the surveillance officer finds that the chief complaint or history of present illness mentions fever or respiratory illness, COVID-19–like illness, or suspected COVID-19, then the case is categorized with COVID-19–related illness as the primary reason for admission. Reasons for admission that are likely primarily not related to COVID-19 include the following categories: inpatient surgery or procedures, psychiatric admission requiring acute medical care, trauma, other, or unknown. Reasons categorized as “other” are reviewed by two physicians to determine whether the admission is likely COVID-19–related.
¶¶ Defined as one or more of the following: chronic lung disease, chronic metabolic disease, blood disorder/hemoglobinopathy, cardiovascular disease, neurologic disorder, immunocompromising condition, renal disease, gastrointestinal/liver disease, rheumatologic/autoimmune/inflammatory condition, obesity, feeding tube dependency, and wheelchair dependency.
*** Includes children with development delay (211), seizure disorders (139), cerebral palsy (62), and other neurologic disorders such as Down Syndrome, neural tube defect, neuropathy, paralysis, and mitochondrial disorders.
††† Includes children with obstructive sleep apnea (74), oxygen dependency (18), bronchopulmonary dysplasia (22), and other chronic lung conditions such as airway abnormality, tracheostomy dependency, restrictive lung disease, pulmonary fibrosis, chronic obstructive pulmonary disease, idiopathic lung disease, chronic bronchitis, bronchiolitis obliterans, and bronchiectasis.
§§§ Includes children with congenital heart disease (55), aortic regurgitation (45), aortic stenosis (30) and other cardiological disorders such as cardiomyopathy and dysrhythmias.
¶¶¶ Includes children with sickle cell anemia (81), asplenia (20), thrombocytopenia (11), and other blood disorders such as thalassemia, coagulopathy, and myelodysplastic syndromes.
**** Includes children with immunosuppressive therapy (70), leukemia (40), immunoglobulin deficiency (13), and other immunocompromising conditions including lymphoma and solid organ malignancies.
†††† Includes children with thyroid dysfunction (20), adrenal disorders (13), and other metabolic conditions such as pituitary dysfunction, inborn errors of metabolism, parathyroid dysfunction, and glycogen or other storage diseases.
§§§§ Includes children with rheumatoid arthritis (32), lupus erythematosus (four), systemic sclerosis (four), and other autoimmune or inflammatory disorders such as Kawasaki disease and juvenile idiopathic arthritis.
¶¶¶¶ Includes children with ulcerative colitis (six), Crohn’s disease (two), chronic liver disease (two), and other GI/liver diseases such as nonalcoholic fatty liver disease, hepatitis B, and esophageal strictures.
***** Includes children with renal insufficiency (13), nephrotic syndrome (five), and other renal diseases, such as glomerulonephritis, polycystic kidney disease, and end stage renal disease.
††††† Excludes genetic diseases listed above.
§§§§§ Across periods, the number of children aged 5–11 years tested for additional viral pathogens was 654 (55%); 85 (12%) had received a positive test result. Positive test results include those for respiratory syncytial virus (13), influenza (four), rhinovirus/enterovirus (52), and other viruses (19).
¶¶¶¶¶ Hospitalization outcomes are not mutually exclusive; patients can be included in more than one category.
Abbreviations: aRR = adjusted risk ratio; COVID-NET = COVID-19–Associated Hospitalization Surveillance Network; ICU = intensive care unit; Ref = referent group; RR = risk ratio.
* Defined as requiring ICU admission or invasive mechanical ventilation, or in-hospital death.
† Among sampled patients, COVID-NET collects data on the primary reason for admission to differentiate hospitalizations of patients with laboratory-confirmed SARS-CoV-2 infection who are likely admitted primarily for COVID-19 illness rather than for other reasons. During chart review, if the surveillance officer finds that the chief complaint or history of present illness mentions fever or respiratory illness, COVID-19–like illness, or suspected COVID-19, then the case is categorized with COVID-19–related illness as the primary reason for admission. Reasons for admission that are likely primarily not related to COVID-19 include the following categories: inpatient surgery or procedures, psychiatric admission requiring acute medical care, trauma, other, or unknown. Reasons categorized as “other” are reviewed by two physicians to determine whether the admission is likely COVID-19–related.
§ Data are from a weighted sample of hospitalized children with completed medical record abstractions. Sample sizes presented are unweighted with weighted percentages.
¶ Age was modeled as a continuous variable and presented as the median and IQR.
** Includes non-Hispanic persons reported as other, multiple races, and unknown race or ethnicity.
†† Includes type 1 and type 2 diabetes mellitus.
§§ Chronic lung disease excludes asthma and chronic metabolic disease excludes diabetes mellitus.
¶¶ Includes liver disease; renal disease; rheumatologic, autoimmune, and inflammatory conditions; and other conditions specified on the case report form.
Suggested citation for this article: Shi DS, Whitaker M, Marks KJ, et al. Hospitalizations of Children Aged 5–11 Years with Laboratory-Confirmed COVID-19 — COVID-NET, 14 States, March 2020–February 2022. MMWR Morb Mortal Wkly Rep 2022;71:574-581. DOI: http://dx.doi.org/10.15585/mmwr.mm7116e1.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.
