Progress Toward Achieving and Sustaining Maternal and Neonatal Tetanus Elimination — Worldwide, 2000–2020
Weekly / March 18, 2022 / 71(11);406–411
Florence A. Kanu, PhD1; Nasir Yusuf, MD2; Modibo Kassogue, MD3; Bilal Ahmed, MBBS3; Rania A. Tohme, MD1 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
In 1999, the maternal and neonatal tetanus (MNT) initiative was relaunched to focus on 59 priority countries still at risk for maternal and neonatal tetanus.
What is added by this report?
During 2000–2020, 47 countries achieved elimination of MNT, reported neonatal tetanus cases decreased 88%, and estimated deaths declined 92%. Despite progress, 12 countries have not achieved elimination and are challenged by conflict, insecurity, and competing priorities. Other countries are struggling to maintain elimination.
What are the implications for public health practice?
To achieve MNT elimination in remaining priority countries and to maintain it globally, efforts are needed to enhance routine vaccination, integrate tetanus activities with other health activities, and promote a life-course vaccination approach for tetanus protection.
Altmetric:
Maternal and neonatal tetanus (MNT)* remains a major cause of neonatal mortality with an 80%–100% case-fatality rate among insufficiently vaccinated mothers after unhygienic deliveries, especially in low-income countries (1). In 1989, the World Health Assembly endorsed elimination† of neonatal tetanus; the activity was relaunched in 1999 as the MNT elimination (MNTE)§ initiative, targeting 59¶ priority countries. MNTE strategies include 1) achieving ≥80% coverage with ≥2 doses of tetanus toxoid–containing vaccine (TTCV2+)** among women of reproductive age through routine and supplementary immunization activities (SIAs)†† in high-risk districts,§§ 2) achieving ≥70% of deliveries by a skilled birth attendant,¶¶ and 3) implementing neonatal tetanus case-based surveillance (2). This report summarizes progress toward achieving and sustaining MNTE during 2000–2020 and updates a previous report (3). By December 2020, 52 (88%) of 59 priority countries had conducted TTCV SIAs. Globally, infants protected at birth*** against tetanus increased from 74% (2000) to 86% (2020), and deliveries assisted by a skilled birth attendant increased from 64% (2000–2006) to 83% (2014–2020). Reported neonatal tetanus cases worldwide decreased by 88%, from 17,935 (2000) to 2,229 (2020), and estimated deaths decreased by 92%, from 170,829 (2000) to 14,230 (2019).††† By December 2020, 47 (80%) of 59 priority countries were validated to have achieved MNTE, five of which conducted postvalidation assessments.§§§ To achieve elimination in the 12 remaining countries and sustain elimination, innovation is needed, including integrating SIAs to cover multiple vaccine preventable diseases and implementing TTCV life course vaccination.
Immunization Activities
To estimate TTCV vaccination coverage delivered through routine immunization services and the number of neonates protected at birth from tetanus, World Health Organization (WHO) and UNICEF use data from administrative records and vaccination coverage surveys reported annually by member countries (4). WHO and UNICEF receive summaries of the number of women of reproductive age receiving TTCV during SIAs (5). In 2020, 16 (27%) of 59 priority countries achieved ≥80% TTCV2+ coverage, with 34 countries increasing coverage since 2000 (Table). In 2020, among 58 priority countries with available data, 46 (79%) reported ≥80% of infants protected at birth. The global proportion of infants protected at birth increased from 74% (2000) to 86% (2020) (Table).
During 2000–2020, 52 priority countries conducted TTCV SIAs, and 168 million (67%) of the targeted 250 million women of reproductive age received TTCV2+ (Table) (Figure 1). In 2020, 59 million women targeted for protection by TTCV SIAs remained unreached, and TTCV SIA activities aiming to target an estimated 16 million women of reproductive age in five countries were postponed because of COVID-19–related disruptions in immunization services (Figure 1) (6).
Deliveries Assisted by Skilled Birth Attendants
WHO and UNICEF estimate the percentage of births assisted by a skilled birth attendant from health care facility reports and coverage survey estimates shared by countries (7). During 2000–2020, the percentage of deliveries assisted by a skilled birth attendant increased 30%, from 64% (2000–2006) to 83% (2014–2020) (7). In 2020, among 50 priority countries with available data, ≥70% of deliveries were assisted by a skilled birth attendant in 28 (58%) countries (Table).
Surveillance Activities
WHO recommends nationwide, case-based surveillance for neonatal tetanus, including zero-case reporting (submission of reports even if no neonatal tetanus cases are observed) and active surveillance through regular site visits (8). The number of reported neonatal tetanus cases worldwide decreased by 88% from 17,935 (2000) to 2,229 (2020).¶¶¶ In 2020, among all 59 priority countries, 10 (17%) reported zero cases, whereas seven countries (Angola, Central African Republic, Chad, Congo, Ethiopia, Madagascar, and Mozambique) reported more cases in 2020 than in 2000 (Table).
Most neonatal tetanus deaths occur in remote communities, which leads to underreporting. Hence, mathematical models are used to better estimate the number of neonatal tetanus deaths (9). The estimated number of neonatal tetanus deaths decreased by 92% from 170,829 (2000) to 14,230 (2019) (Figure 2). In 2019, tetanus accounted for 0.4% of all neonatal deaths, a decrease from 7% in 2000.
Validation of Maternal and Neonatal Tetanus Elimination
When a country believes it has eliminated MNT, validation activities are implemented, consisting of review of district-level core indicators, including reported neonatal tetanus cases per 1,000 live births and review of the surveillance system, percentage of clean deliveries assisted by a skilled birth attendant, and TTCV2+ coverage among pregnant women (6); the country also uses supplementary indicators, including TTCV SIA coverage, antenatal care coverage,**** infant coverage with 3 doses of the diphtheria, tetanus, and pertussis (DTP) vaccine, socioeconomic indices, urban versus rural status, field visits to assess the performance of the health system, validation surveys of poorly performing districts, and assessment of long-term plans for sustaining elimination.†††† During 2000–2020, 47 (80%) of 59 priority countries were validated to have achieved MNTE, and 12 remain to be validated (Table) (Figure 1). In addition, by 2020, three countries were partially validated to have achieved elimination in some regions: Mali (Southern regions), Nigeria (Southeast and Southwest zones), and Pakistan (Punjab province).§§§§
Sustainability of Maternal and Neonatal Tetanus Elimination
Once countries are validated for MNTE, WHO recommends four strategies to sustain elimination: 1) providing 3 primary doses of DTP during infancy and 3 TTCV booster doses at ages 12–23 months, 4–7 years, and 9–15 years; 2) checking maternal tetanus vaccination status during antenatal care and providing TTCV2+ to pregnant women, if needed, to ensure that ≥70% of infants are protected at birth; 3) promoting ≥60% clean deliveries through increased access to a skilled birth attendant ; and 4) maintaining strong neonatal tetanus surveillance (6). After validation, WHO recommends that countries conduct annual neonatal tetanus risk analyses as part of an immunization desk review and complete postvalidation assessments every 5 years to identify whether elimination status is maintained and take corrective actions as needed (6). In 2020, 14 (30%) of the 47 priority countries validated for MNTE achieved ≥90%¶¶¶¶ coverage with 3 doses of DTP; TTCV booster doses***** were provided to children aged 12–23 months in 11 (23%) of those countries, to children aged 4–7 years in 12 (26%) countries, and to children aged 9–15 years in nine (19%) countries. In 45 (96%) countries, ≥70% of infants were protected at birth against tetanus; and in 34 (72%), ≥60% of births were assisted by a skilled birth attendant.
Five countries (Algeria, Cameroon, Djibouti, Indonesia, and Timor-Leste) implemented postvalidation assessments for corrective actions and have met the sustainability indicators for infants protected at birth and the percentage of births with access to a skilled birth attendant. In addition, Cameroon conducted annual neonatal tetanus risk analyses and used assessment outcomes for corrective action by targeting women of reproductive age in high-risk districts with two rounds of TTCV SIAs to sustain MNTE.
Discussion
Substantial progress has been made toward global MNTE; 80% of the 59 priority countries were validated to have achieved MNTE by the end of 2020. Progress can be attributed to increases in TTCV2+ coverage among women of reproductive age in 34 (58%) of 59 priority countries, implementation of intensive SIAs in high-risk districts, and a 30% increase in deliveries with a skilled birth attendant. These efforts contributed to a 16% increase in infants protected against tetanus at birth and a 92% decline in estimated neonatal tetanus mortality since 2000.
Although progress has been made, countries that have not achieved MNTE still face several challenges. First, suboptimal health systems, evidenced by low vaccination coverage and low proportions of safe and clean deliveries assisted by a skilled birth attendant, make it difficult to adequately implement MNTE strategies. Second, conflict and political instability in some countries contribute to districts remaining inaccessible and at high risk for the incidence of maternal and neonatal tetanus. Lastly, country immunization programs might have competing priorities in addressing the overall incidence of vaccine preventable diseases (e.g., measles and polio) or responding to outbreaks (e.g., Ebola and COVID-19) that hinder their ability to achieve MNTE. During 2020, the COVID-19 pandemic affected TTCV SIAs planned in five countries.
Complete eradication of tetanus is not possible because tetanus spores are ubiquitous in the environment. Therefore, countries need to implement strategies to sustain MNTE. Only five of 47 countries validated for MNTE have conducted the recommended postvalidation assessments, and only 12 have introduced ≥1 TTCV booster doses in their routine immunization schedule. This low uptake could be attributed to competing priorities and the deprioritizing of MNTE once countries are validated, which put countries at risk for reemergence of MNT (6). Combining MNTE postvalidation assessments with review of immunization programs and integrating childhood and adolescent tetanus vaccination with other immunization activities (e.g., measles vaccination during second year of life, school vaccination programs, or human papillomavirus vaccination) promote better efficiency and use of resources and help sustain MNTE. Neonatal tetanus case-based surveillance could also be integrated into polio and measles case-based surveillance; community engagement might help raise awareness of neonatal tetanus and serve to strengthen community-based vaccine preventable disease surveillance systems (8).
The findings in this report are subject to at least three limitations. First, TTCV coverage among pregnant women can underestimate true tetanus protection because it excludes women who were unvaccinated during current pregnancy but protected through previous vaccination or those missing documentation of previous doses (6). Second, the percentage of infants protected at birth could be underestimated because of doses provided outside routine services (6). Finally, <10% of neonatal tetanus cases and deaths are estimated to be reported (2); although neonatal deaths are projected using mathematical models, cases and deaths might still be underestimated, especially in communities with suboptimal health systems.
The Immunization Agenda 2030,††††† the global immunization strategy for the next decade, includes MNTE as an endorsed vaccine-preventable disease elimination target. To achieve and sustain MNTE, strong national commitment and integration are needed, including integrating MNTE activities with polio, measles, cholera, yellow fever, or other vaccine-preventable disease SIAs, using MNTE to promote equitable access to health services, such as clean deliveries, and promoting a life course approach to tetanus vaccination by integrating TTCV booster doses in school health programs and other life course immunization platforms (10).
Acknowledgments
UNICEF country offices in Central African Republic, Nigeria, South Sudan; Jose Chivale; Mohammed Farid; Quamrul Hasan; Javid Iqbal; Julien Hyacinte Kabore; Mouctar Kande; Emmaculate Lebo; Richard Luce; Osama Mere; Terna Nomhwange; Constance Razaiarimanga; Abdoul Karim Sidibe; Maleghemi Sylvester; Patricia Tanifum.
Corresponding author: Florence A. Kanu, fkanu@cdc.gov.
1Global Immunization Division, Center for Global Health, CDC; 2Immunization, Vaccines and Biologicals, World Health Organization, Geneva, Switzerland; 3Maternal, Newborn, and Adolescent Health Program Division, UNICEF, New York, New York.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
* Tetanus occurring during pregnancy or within 6 weeks of the end of pregnancy; maternal tetanus infection occurs during abortion, miscarriages, or birth with unhygienic delivery. Neonatal tetanus occurs during the first 28 days of life, either following the cutting of the umbilical cord under nonsterile conditions or applying nonsterile traditional remedies to the umbilical stump in an infant without passively (transplacentally) acquired maternal antibodies.
† The occurrence of less than one neonatal tetanus case per 1,000 live births per year in every district in every country.
§ Neonatal tetanus elimination is considered a proxy for maternal tetanus elimination; the same strategies for elimination are shared.
¶ Initially, the total number of priority countries was 57. The creation of Timor-Leste in 2002 and South Sudan in 2011 increased the number of priority countries to 59.
** Tetanus toxoid (TT2+) or tetanus-diphtheria toxoid (Td2+).
†† Mass vaccination campaigns that aim to administer doses of tetanus toxoid–containing vaccines to women of reproductive age.
§§ Districts considered at high risk because the estimated neonatal tetanus case rate exceeds one per 1,000 live births, clean delivery coverage is <70%, and coverage with 3 tetanus toxoid–containing vaccine doses among pregnant women is <80% during the past 5 years.
¶¶ A doctor, nurse, midwife, or health worker trained in providing lifesaving obstetric care, including giving necessary supervision, care, and advice to women during pregnancy, childbirth, and the postpartum period.
*** The status of an infant born to a mother who received 2 doses of TTCV during the last birth, ≥2 doses with the last dose received ≤3 years before the last delivery, ≥3 doses with the last dose received ≤5 years earlier, ≥4 doses with the last dose received ≤10 years earlier, or receipt of ≥5 previous doses.
††† Neonatal mortality data were unavailable for 2020. http://ghdx.healthdata.org/gbd-results-tool
§§§ A postvalidation assessment comprises a review of data to determine whether MNTE indicator standards are being maintained and to identify districts potentially at risk of not sustaining MNTE. Postvalidation assessments include field visits and interviews at both the facility and community level, cross checking the reported coverage of tetanus toxoid–containing vaccines, antenatal care, and skilled birth delivery. The assessment also includes bottleneck analysis and development of a work plan and time frame for implementing corrective actions, if needed.
**** Antenatal care coverage is the percentage of females aged 15–49 years with a live birth who received antenatal care provided by skilled health personnel (doctor, nurse, or midwife) at least once during pregnancy.
†††† https://www.who.int/immunization/documents/MNTE_Validation_survey_WHO_IVB_18.15.pdf
§§§§ https://www.who.int/initiatives/maternal-and-neonatal-tetanus-elimination-(mnte)/the-partnership
***** https://immunizationdata.who.int/pages/schedule-by-disease/tetanus.html?TARGETPOP_GENERAL = GENERAL
††††† https://www.who.int/publications/m/item/implementing-the-immunization-agenda-2030
References
- World Health Organization. Tetanus vaccines: WHO position paper–February 2017. Wkly Epidemiol Rec 2017;92:53–76. PMID:28185446
- World Health Organization. Maternal and neonatal tetanus elimination: the strategies. Geneva, Switzerland: World Health Organization; 2021. https://www.who.int/initiatives/maternal-and-neonatal-tetanus-elimination-(mnte)/the-strategies
- Njuguna HN, Yusuf N, Raza AA, Ahmed B, Tohme RA. Progress toward maternal and neonatal tetanus elimination—worldwide, 2000–2018. MMWR Morb Mortal Wkly Rep 2020;69:515–20. https://doi.org/10.15585/mmwr.mm6917a2 PMID:32352953
- World Health Organization. Protection at birth (PAB) against neonatal tetanus and tetanus toxoid-containing vaccine (TT2+/Td2+) vaccination coverage. Geneva, Switzerland: World Health Organization; 2021. https://immunizationdata.who.int/pages/coverage/tt2plus.html?CODE=Global&ANTIGEN=PAB+TT2PLUS&YEAR=
- World Health Organization. Maternal and neonatal tetanus elimination: programmatic update. Geneva, Switzerland: World Health Organization; 2021. https://www.who.int/initiatives/maternal-and-neonatal-tetanus-elimination-(mnte)/programmatic-update
- World Health Organization. Protecting all against tetanus: guide to sustaining maternal and neonatal tetanus elimination (MNTE) and broadening tetanus protection for all populations. Geneva, Switzerland: World Health Organization; 2019. https://www.who.int/publications/i/item/protecting-all-against-tetanus
- UNICEF; World Health Organization. UNICEF/WHO joint database: delivery at care. New York, NY: UNICEF; 2019. https://data.unicef.org/topic/maternal-health/delivery-care/
- World Health Organization. Surveillance standards for vaccine-preventable diseases. Geneva, Switzerland: World Health Organization; 2018. https://www.who.int/publications/i/item/surveillance-standards-for-vaccine-preventable-diseases-2nd-edition
- Yen LM, Thwaites CL. Tetanus. Lancet 2019;393:1657–68. https://doi.org/10.1016/S0140-6736(18)33131-3 PMID:30935736
- Yusuf N, Raza AA, Chang-Blanc D, et al. Progress and barriers towards maternal and neonatal tetanus elimination in the remaining 12 countries: a systematic review. Lancet Glob Health 2021;9:e1610–7. https://doi.org/10.1016/S2214-109X(21)00338-7 PMID:34678200
Sources: Neonatal tetanus data: WHO Global Health Observatory Data Repository (2000–2020), Protected at birth data: WHO/UNICEF Joint Reporting Form on Immunization (2000–2020), Skilled birth attendant data: WHO Global Health Observatory Data Repository (2000–2020), SIA data: WHO/UNICEF Maternal and Neonatal Tetanus Elimination Database, as of January 2022, TTCV data: WHO Global Health Observatory Data Repository (2000–2020).
Abbreviations: NA = not available; SIA = supplementary immunization activity; TT2+/Td2+ = ≥2 doses of tetanus toxoid/tetanus-diphtheria toxoid; TTCV = tetanus toxoid–containing vaccine; WHO = World Health Organization.
* Includes first-year SIA conducted in Bangladesh in 1999 and first- and second-year SIAs conducted in Ethiopia in 1999.
† Includes skilled birth attendant surveys conducted within 5 years for year 2000 and year 2020.
§ Administrative data of TTCV coverage with ≥2 doses among women of reproductive age were used when official data were unavailable for select country.
¶ The increase in neonatal tetanus cases seen from 2000 to 2020 might be the result of improvement in surveillance.
 FIGURE 1. Number of women of reproductive age protected by tetanus toxoid–containing vaccine* received during supplementary immunization activities, number targeted but not yet vaccinated, number not yet targeted, and number of priority countries achieving maternal and neonatal tetanus elimination — worldwide, 2000–2020
FIGURE 1. Number of women of reproductive age protected by tetanus toxoid–containing vaccine* received during supplementary immunization activities, number targeted but not yet vaccinated, number not yet targeted, and number of priority countries achieving maternal and neonatal tetanus elimination — worldwide, 2000–2020
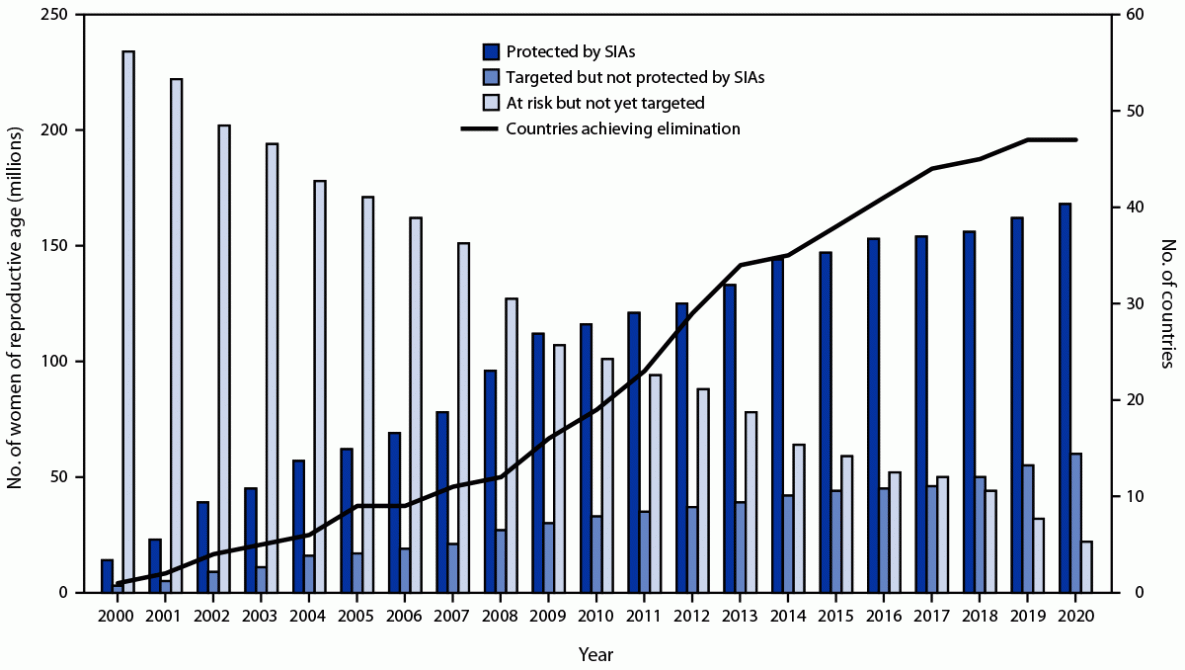
Source: WHO/UNICEF Maternal and Neonatal Tetanus Elimination Database, as of January 2022.
Abbreviations: SIA = supplementary immunization activities; WHO = World Health Organization.
* Protected with 2 doses of tetanus toxoid or 2 doses of tetanus and diphtheria toxoids.
 FIGURE 2. Estimated number of neonatal tetanus deaths* and estimated proportion of children protected at birth† against tetanus — worldwide, 2000–2020§
FIGURE 2. Estimated number of neonatal tetanus deaths* and estimated proportion of children protected at birth† against tetanus — worldwide, 2000–2020§
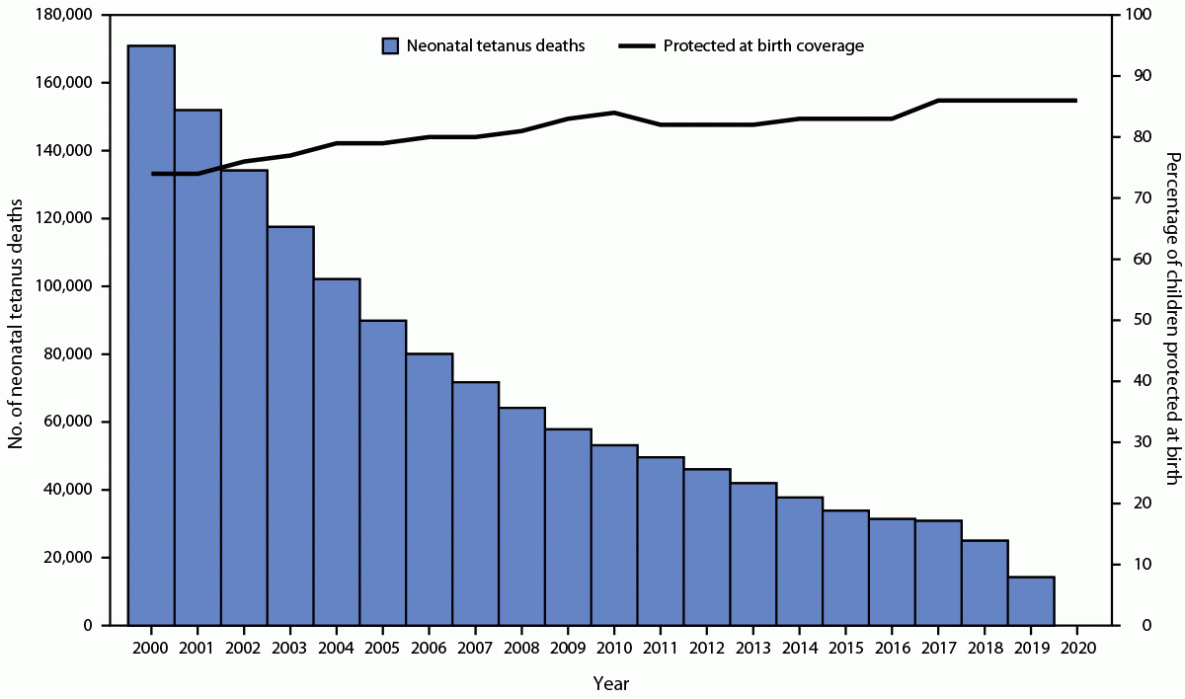
Sources: Neonatal tetanus data: WHO Global Health Observatory Data Repository (2000–2018) and the Global Health Data Exchange (2019), Protected at birth data: WHO/UNICEF Joint Reporting Form on Immunization (2000–2020).
Abbreviations: TTCV = tetanus toxoid–containing vaccine; WHO = World Health Organization.
* The number of deaths is estimated from mathematical models that compute the yearly incidence and mortality for each country using the baseline rate of neonatal tetanus before introduction of TTCVs and promotion of clean deliveries, with adjustment for the estimated proportion of women vaccinated with TTCV and deliveries assisted by trained personnel.
† The status of an infant born to a mother who received 2 doses of TTCV during the last birth, ≥2 doses with the last dose received ≤3 years before the last delivery, ≥3 doses with the last dose received ≤5 years earlier, ≥4 doses with the last dose received ≤10 years earlier, or receipt of ≥5 previous doses.
§ Data on deaths for 2020 were not available.
Suggested citation for this article: Kanu FA, Yusuf N, Kassogue M, Ahmed B, Tohme RA. Progress Toward Achieving and Sustaining Maternal and Neonatal Tetanus Elimination — Worldwide, 2000–2020. MMWR Morb Mortal Wkly Rep 2022;71:406–411. DOI: http://dx.doi.org/10.15585/mmwr.mm7111a2.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.