HIV Preexposure Prophylaxis, by Race and Ethnicity — United States, 2014–2016
Weekly / October 19, 2018 / 67(41);1147–1150
Ya-lin A. Huang, PhD1; Weiming Zhu, PhD2; Dawn K. Smith, MD1; Norma Harris, PhD1; Karen W. Hoover, MD1 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
In 2015, approximately 1.1 million adults were at risk for acquiring human immunodeficiency virus infection and had indications for preexposure prophylaxis (PrEP); 26.3%, 43.7%, and 24.7% were white, black, and Hispanic, respectively.
What is added by this report?
In 2016, among 78,360 persons who filled prescriptions for PrEP in the United States, women accounted for only 4.7%. Among PrEP users with available race/ethnicity data, 68.7%, 11.2%, 13.1%, and 4.5% were white, black, Hispanic, and Asian, respectively.
What are the implications for public health practice?
The gap between numbers of persons with PrEP indications and those prescribed PrEP was substantial, especially among persons in female, black, and Hispanic populations. Focused efforts are needed to increase the impact of PrEP in the United States.
Preexposure prophylaxis (PrEP) with a daily, oral pill containing antiretroviral drugs is highly effective in preventing acquisition of human immunodeficiency virus (HIV) infection (1–4). The combination of tenofovir disoproxil fumarate (TDF) and emtricitabine (FTC) is the only medication approved by the Food and Drug Administration (FDA) for PrEP. PrEP is indicated for men and women with sexual or injection drug use behaviors that increase their risk for acquiring HIV (5). CDC analyzed 2014–2016 data from the IQVIA Real World Data — Longitudinal Prescriptions (IQVIA database) to estimate the number of persons prescribed PrEP (users) in the United States and to describe their demographic characteristics, including sex and race/ethnicity. From 2014 to 2016, the annual number of PrEP users aged ≥16 years increased by 470%, from 13,748 to 78,360. In 2016, among 32,853 (41.9%) PrEP users for whom race/ethnicity data were available, 68.7% were white, 11.2% were African American or black (black), 13.1% were Hispanic, and 4.5% were Asian. Approximately 7% of the estimated 1.1 million persons who had indications for PrEP were prescribed PrEP in 2016, including 2.1% of women with PrEP indications (6). Although black men and women accounted for approximately 40% of persons with PrEP indications (6), this study found that nearly six times as many white men and women were prescribed PrEP as were black men and women. The findings of this study highlight gaps in effective PrEP implementation efforts in the United States.
In 2012, FDA approved TDF/FTC for use as PrEP (7), and CDC published clinical practice guidelines for use of PrEP (5). A previous study estimated PrEP uptake among U.S. commercially insured populations and found that PrEP use increased among men during 2010–2014, but was very low among women (8). It is important to monitor PrEP uptake both among persons with private and public insurance. Because racial and ethnic disparities in HIV diagnoses exist in the United States (9), it is also important to better understand PrEP use by race/ethnicity. Monitoring trends in PrEP use can inform the development of interventions to ensure that PrEP is provided for persons who need it most to reduce racial and ethnic disparities in PrEP use and new HIV infections.
Data on antiretroviral drug prescriptions dispensed during 2014–2016 were extracted from the IQVIA database,* which captured prescriptions from all payers and represented approximately 92% of all prescriptions dispensed from retail pharmacies and 60%–86% dispensed from mail order outlets in the United States. The database included antiretroviral drugs dispensed, demographic variables of persons to whom the drugs were dispensed, and medical claims for these persons. IQVIA acquired medical claims and race/ethnicity data from various sources, including ambulatory, hospital, and consumer databases, and linked these data to persons in the prescription database. Among persons with any antiretroviral drug prescription (1,418,621), approximately 69% had medical claims data available, and race/ethnicity information was available for about 32%. CDC estimated the annual number of PrEP users based on a previously developed algorithm that discerns whether TDF/FTC was prescribed for PrEP or for HIV treatment, hepatitis B treatment, or HIV postexposure prophylaxis (8). For each year of the study, records of persons aged ≥16 years who had at least one TDF/FTC prescription were selected. Persons were then excluded if they had any diagnostic codes for HIV or hepatitis B infection that preceded their initial TDF/FTC prescription. In addition, persons prescribed TDF/FTC for ≤30 days were defined as postexposure prophylaxis users and excluded; the remaining persons with TDF/FTC prescribed for >30 days were considered PrEP users. Postexposure prophylaxis is recommended for 28 days; however, it is often prescribed for 30 days. The 30-day definition of postexposure prophylaxis was chosen to produce conservative estimates of TDF/FTC for PrEP. PrEP use among persons prescribed TDF/FTC for >28 days was also estimated, to assess the impact of different duration of drug use on the estimates. PrEP use estimates were reported by age group, sex, geographic region, payer type, and race/ethnicity. Payer type was estimated for each person prescribed PrEP using a payer hierarchy of Medicaid, Medicare, commercial insurance, cash, and other payers. The number of PrEP users who received medication assistance program benefits from the manufacturer of PrEP also was estimated.
The annual number of PrEP users aged ≥16 years increased by 470%, from 13,748 in 2014 to 78,360 in 2016 (Table 1). In 2016, 65.0% of PrEP users were aged 25–44 years, and 0.1% were aged 16–17 years. Males accounted for 95.3% of all PrEP users. The percentage of PrEP users was highest in the Western U.S. Census Region (29.7%), followed by the Southern (27.2%) and Northeastern Regions (26.7%) and was lowest in the Midwestern Region (16.3%). Commercial health insurance was the payer for 81.0% of PrEP users’ medications and Medicaid for 12.2%. The number of PrEP users who received medication assistance program benefits from the manufacturer increased significantly, from 435 in 2014 to 5,437 in 2016.
When length of TDF/FTC prescription drug use for PrEP was defined as >28 days rather than >30 days, the total number of PrEP users in 2016 increased 26%, from 78,360 to 98,599. Demographic and payer type distributions were similar using both algorithms (Table 2).
Among the 78,360 PrEP users identified in 2016, information on race/ethnicity was available for 32,853 (41.9%), including 22,574 (68.7%) who were white, 3,687 (11.2%) who were black, 4,317 (13.1%) who were Hispanic, and 1,486 (4.5%) who were Asian. When stratified by sex, among the 1,146 female PrEP users with race/ethnicity data, 554 (48.3%) were white, 297 (25.9%) were black, and 201 (17.5%) were Hispanic (Figure).
Discussion
Compared with recently published estimates based on an analysis of the MarketScan database with commercial health insurance billing claims, the estimated number of PrEP users was higher using this IQVIA database (8). This is because the IQVIA database contains all third party payers, including Medicaid, and prescriptions claims paid by medication assistance programs. The number of PrEP users with commercial insurance was similar in both analyses. In 2014, a total of 7,792 PrEP users with commercial insurance were identified in the MarketScan database, compared with 9,980 users with commercial insurance in the IQVIA database (8); in 2015, a total of 33,273 PrEP users with commercial insurance were identified in MarketScan,† compared with 31,993 users with commercial insurance in IQVIA. The algorithm used in this study and in the MarketScan analysis defined postexposure prophylaxis as a TDF/FTC prescription for ≤30 days, resulting in a conservative estimate of PrEP use that might underestimate the number of PrEP users because persons might have been prescribed a 30-day supply of TDF/FTC for PrEP or postexposure prophylaxis. Persons prescribed TFD/FTC for ≤30 days might also have been using on-demand PrEP that is not taken daily. When a definition of postexposure prophylaxis as a TDF/FTC prescription for ≤28 days was used, the estimated number of PrEP users was higher. The true estimate of PrEP use likely falls between the estimate that defines PrEP use as a TDF/FTC prescription for >30 days and the one that defines it as >28 days. A validation study that compares estimates of PrEP use based on various algorithm definitions with a review of medical records will be helpful for future research.
Women accounted for 3,678 (4.7%) of the 78,360 PrEP users and 2.1% of the estimated 176,670 heterosexual women for whom PrEP is indicated (6). Among the estimated 1.1 million adults for whom PrEP is indicated, 303,230 (26.3%) were white, 500,340 (43.7%) were black, and 282,260 (24.7%) were Hispanic (6). However, among PrEP users with available race/ethnicity data in this study, 68.7% were white, 11.2% were black, and 13.1% were Hispanic. The large gap between the numbers of persons with indications for PrEP and those who were prescribed PrEP, and the low proportions of women and racial/ethnic minorities prescribed PrEP, suggests that more equitable implementation of PrEP recommendations for women and persons in racial/ethnic minority populations is needed. In addition, whereas men and women in the South had 52% of HIV diagnoses in the United States in 2016 (8), this study found that only 27% of the PrEP users were in the South.
The findings in this report are subject to at least four limitations. First, 58% of PrEP users identified in the IQVIA database did not have race/ethnicity information available. Race/ethnicity data were obtained from a convenience sample of a consumer database, in which persons who were older and had a credit history were more likely to be included. Although race/ethnicity data were not available for many PrEP users, this study suggests a substantial unmet prevention need for black and Hispanic populations who might benefit from PrEP. Second, PrEP users were identified using an algorithm that might be subject to misclassification bias. However, a similar algorithm was validated based on a review of electronic medical records (10). Third, the estimates were based on prescriptions dispensed rather than actual use. Finally, the IQVIA database did not include diagnosis data for 31% of persons, which might result in an overestimate of PrEP users by including persons potentially using TDF/FTC for treatment of HIV or hepatitis B infection. However, most persons (99%) with HIV in the IQVIA database had other antiretroviral medications in addition to TDF/FTC and were excluded.
Barriers to the provision of PrEP for persons in populations with the highest rates of annual HIV diagnoses, such as black and Hispanic men and women, need to be better understood to help guide the development of interventions to increase access to and utilization of PrEP. Focused public health efforts to support increasing PrEP prescriptions for persons in populations who might benefit from its use could increase the impact of PrEP on HIV incidence in the United States.
Corresponding author: Ya-lin A. Huang, yhuang@cdc.gov, 404-639-2992.
1Division of HIV/AIDS Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC; 2Prosphere Tek, Inc., Alexandria, Virginia.
All authors have completed and submitted the ICMJE form for disclosure of potential conflicts of interest. No individual potential conflicts of interest were disclosed; however, CDC and other CDC staff are named in US government patents and patent applications related to methods for HIV prophylaxis.
† MarketScan data came from Table 11, Monitoring selected national HIV prevention and care objectives by using HIV surveillance data—United States and 6 dependent areas, 2015. HIV Surveillance Supplemental Report 2017; vol. 22, no. 2. https://www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-supplemental-report-vol-22-2.pdf.
References
- Grant RM, Lama JR, Anderson PL, et al. ; iPrEx Study Team. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med 2010;363:2587–99. CrossRef PubMed
- Baeten JM, Donnell D, Ndase P, et al. ; Partners PrEP Study Team. Antiretroviral prophylaxis for HIV prevention in heterosexual men and women. N Engl J Med 2012;367:399–410. CrossRef PubMed
- Thigpen MC, Kebaabetswe PM, Paxton LA, et al. ; TDF2 Study Group. Antiretroviral preexposure prophylaxis for heterosexual HIV transmission in Botswana. N Engl J Med 2012;367:423–34. CrossRef PubMed
- Choopanya K, Martin M, Suntharasamai P, et al. ; Bangkok Tenofovir Study Group. Antiretroviral prophylaxis for HIV infection in injecting drug users in Bangkok, Thailand (the Bangkok Tenofovir Study): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2013;381:2083–90. CrossRef PubMed
- CDC. Preexposure prophylaxis for the prevention of HIV infection in the United States—2017 update. Atlanta, GA: US Department of Health and Human Services, CDC; 2018. https://www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2017.pdf
- Smith DK, Van Handel M, Grey J. Estimates of adults with indications for HIV pre-exposure prophylaxis by jurisdiction, transmission risk group, and race/ethnicity, United States, 2015. Ann Epidemiol 2018;S1047-2797(17)31069–4. CrossRef PubMed
- Food and Drug Administration. FDA approves first drug for reducing the risk of sexually acquired HIV infection [news release]. Silver Spring, MD: US Department of Health and Human Services, Food and Drug Administration; 2012. https://aidsinfo.nih.gov/news/1254/fda-approves-first-drug-for-reducing-the-risk-of-sexually-acquired-hiv-infection
- Wu H, Mendoza MC, Huang YA, Hayes T, Smith DK, Hoover KW. Uptake of HIV preexposure prophylaxis among commercially insured persons—United States, 2010–2014. Clin Infect Dis 2017;64:144–9. CrossRef PubMed
- CDC. Diagnoses of HIV infection in the United States and dependent areas, 2016. HIV surveillance report; vol. 28. Atlanta, GA: US Department of Health and Human Services, CDC; 2017. https://www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-report-2016-vol-28.pdf
- Mera-Giler R, MacCannell T, Magnuson D, et al. Validation of a Truvada for PrEP algorithm through chart reviews from an electronic medical record. Presented at the National HIV Prevention Conference, Atlanta, GA; December 6–9, 2015.
Abbreviation: CHIP = Children’s Health Insurance Program.
* https://www.iqvia.com/.
† Other region included U.S. territories.
§ Payer type is a calculated hierarchical variable, thus numbers of each category are mutually exclusive. Before 2014, payer type information was not available for some of the specialty mail order suppliers.
¶ Other payer types included coupon/voucher programs, discount card programs, and federal or state assistance programs.
Abbreviation: CHIP = Children’s Health Insurance Program.
* Percentages calculated among 32,853 (41.9%) >30-day users and 39,654 (40.2%) >28-day users with information on race/ethnicity available.
† Payer type is a calculated hierarchical variable, thus numbers of each category are mutually exclusive. Persons who identified their race as white, black, Asian, or unspecified were all non-Hispanic. Persons who identified as Hispanic might be of any race.
§ Other payer type included coupon and voucher programs, discount card programs, and federal or state assistance programs.
 FIGURE. Number of PrEP users by sex and race/ethnicity*— IQVIA Longitudinal Prescription Database, United States, 2016
FIGURE. Number of PrEP users by sex and race/ethnicity*— IQVIA Longitudinal Prescription Database, United States, 2016
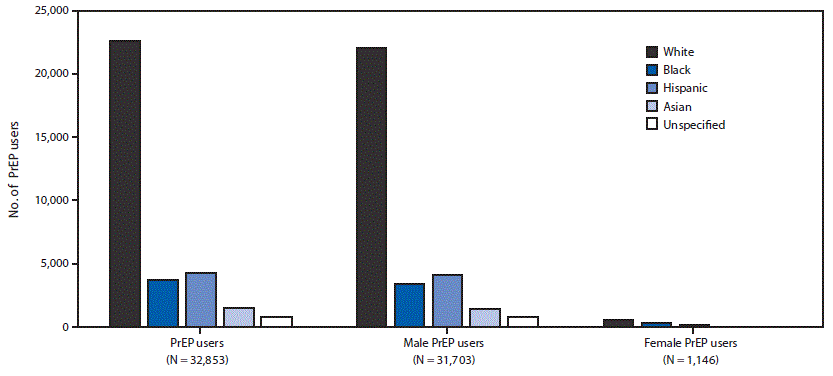
Abbreviation: PrEP = preexposure prophylaxis.
* Among 32,853 (42%) persons with race/ethnicity data available, among all 78,360 PrEP users identified in 2016; information on sex was missing/unknown for four of these 32,853 persons.
Suggested citation for this article: Huang YA, Zhu W, Smith DK, Harris N, Hoover KW. HIV Preexposure Prophylaxis, by Race and Ethnicity — United States, 2014–2016. MMWR Morb Mortal Wkly Rep 2018;67:1147–1150. DOI: http://dx.doi.org/10.15585/mmwr.mm6741a3.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.