Notes from the Field: Mumps Outbreak — Alaska, May 2017–July 2018
Weekly / August 24, 2018 / 67(33);940–941
Amanda Tiffany, PhD1,2; Drew Shannon3; Willy Mamtchueng3; Louisa Castrodale, DVM2; Joseph McLaughlin, MD2 (View author affiliations)
View suggested citationIn May 2017, the Alaska Section of Epidemiology (SOE) was notified of an Anchorage resident with laboratory-confirmed mumps who reported exposure to an out-of-state visitor with mumps-like symptoms. Another seven laboratory-confirmed cases were reported in late July and August; all were in Anchorage residents, mostly in persons who self-identified as Native Hawaiian or other Pacific Islander (NH/PI). In response, SOE disseminated educational materials and recommended that all Alaskans ensure that they were up to date on their measles-mumps-rubella (MMR) vaccinations. Cases were classified as suspected, probable, or confirmed according to the Council of State and Territorial Epidemiologists case definition (1).
On November 15, with 56 confirmed and probable mumps cases identified (including 82% among NH/PI, who represent 4.8% of the Anchorage population), SOE recommended a third dose of MMR vaccine (MMR3) for persons at increased risk for acquiring mumps, such as persons participating in group settings (e.g., school, daycare, church) where mumps cases were identified or any persons who self-identified as NH/PI, if at least 5 years had passed since their second MMR dose (2). Despite this recommendation, cases continued to occur among persons at increased risk and among persons without documented epidemiologic links to other cases. Consequently, on December 28, 2017, when 138 cases had been reported, the MMR3 recommendation was expanded to all Anchorage residents. On February 22, 2018, with 247 cases reported, and cases continuing and occurring statewide among persons with indeterminate epidemiologic links and no history of in-state or out-of-state travel, the recommendation was extended to all 737,080 Alaska residents.
Concurrently, SOE and the Anchorage Department of Health and Human Services coordinated community outreach in collaboration with local partners to offer targeted vaccination clinics, presentations, and media campaigns to raise awareness about the outbreak and the importance of vaccination. Since November 15, 2017, when the first MMR3 recommendation was made, through July 31, 2018, the average number of MMR doses administered in Anchorage (where most of the outreach was focused) increased by 136% to 461 per month, from 195 per month before the recommendation (November 1, 2016–November 15, 2017) (p = 0.001) (Figure).
As of July 31, 2018, the outbreak is ongoing, with 391 confirmed and probable cases reported; 94% of cases have been in Anchorage residents. The median age of patients was 25 years (range = 3 months–79 years) and 193 (49%) self-identified as NH/PI. Overall, 162 (41%) patients had received ≥2 MMR doses before symptom onset, 51 (13%) received 1 dose, and 15 (4%) had not received MMR; vaccination status was unknown for 163 (42%) patients.
Compared with mumps outbreaks in discrete populations such as universities where the population at risk is well defined, community outbreaks pose unique challenges. Following updated Advisory Committee on Immunization Practice recommendations (3), a third MMR dose was recommended for persons at increased risk for acquiring mumps as defined by the epidemiologic data. However, as the outbreak evolved, it became more difficult to determine who was at increased risk. Group-specific MMR3 recommendations were challenging for clinicians to implement when faced with uncertainty about whether their patients participated in group settings where mumps was circulating. In response, SOE implemented a stepwise expansion of its MMR3 recommendation that eventually included all Alaskans. Evaluation of Alaska’s response to the mumps outbreak, including the impact of MMR3 recommendations on MMR uptake, is ongoing. Disseminating information through social media, working with community groups, and vaccination clinics have been important in raising awareness and increasing vaccine uptake.
Acknowledgments
Alaska Section of Public Health Nursing staff members; Bruce Chandler; staff members of the Municipality of Anchorage Disease Prevention and Control; Alaska State Public Health Laboratories (Anchorage and Fairbanks); Alaska Section of Epidemiology Infectious Disease and Immunization Programs staff members; Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, CDC; Viral and Rickettsial Disease Laboratory at the California Department of Public Health.
Corresponding author: Amanda Tiffany, atiffany@cdc.gov, 907-269-8891.
1Epidemic Intelligence Service, CDC; 2Alaska Division of Public Health, Section of Epidemiology, Anchorage, Alaska; 3Department of Health and Social Services, Municipality of Anchorage, Anchorage, Alaska.
All authors have completed and submitted the ICMJE form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
References
- Council of State and Territorial Epidemiologists. CSTE position statement, 11-ID-18: mumps 2012 case definition. Atlanta, GA: Council of State and Territorial Epidemiologists; 2012. https://wwwn.cdc.gov/nndss/conditions/mumps/case-definition/2012/
- Tiffany A. State of Alaska epidemiology bulletin. Mumps outbreak update and recommendations for a third dose of vaccine. Anchorage, AK: Alaska Division of Public Health, Section of Epidemiology; 2017. http://www.epi.alaska.gov/bulletins/docs/b2017_28.pdf
- Marin M, Marlow M, Moore KL, Patel M. Recommendation of the Advisory Committee on Immunization Practices for use of a third dose of mumps virus-containing vaccines in persons at increased risk for mumps during an outbreak. MMWR Morb Mortal Wkly Rep 2018;67:33–8. CrossRef PubMed
 FIGURE. Cumulative number of confirmed and probable mumps cases and MMR vaccine doses administered,* by dose number and month of vaccination — Anchorage, Alaska, November 2016–July 2018
FIGURE. Cumulative number of confirmed and probable mumps cases and MMR vaccine doses administered,* by dose number and month of vaccination — Anchorage, Alaska, November 2016–July 2018
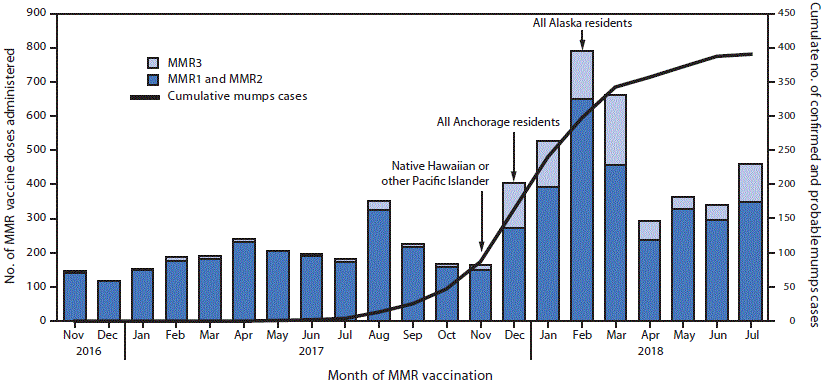
Abbreviations: MMR = measles-mumps-rubella vaccine; MMR1 = first MMR dose; MMR2 = second MMR dose; MMR3 = third MMR dose.
* Arrows indicate the month during which MMR3 recommendation was made for specific populations.
The figure above is a bar chart showing the cumulative number of confirmed and probable mumps cases and MMR vaccine doses administered, by dose number and month of vaccination, in Anchorage, Alaska during November 2016–July 2018.
Suggested citation for this article: Tiffany A, Shannon D, Mamtchueng W, Castrodale L, McLaughlin J. Notes from the Field: Mumps Outbreak — Alaska, May 2017–July 2018. MMWR Morb Mortal Wkly Rep 2018;67:940–941. DOI: http://dx.doi.org/10.15585/mmwr.mm6733a6.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.