Notes from the Field: Cardiac Dysrhythmias After Loperamide Abuse — New York, 2008–2016
Weekly / November 18, 2016 / 65(45);1276–1277
William Eggleston, PharmD1,2; Jeanna M Marraffa, PharmD1,2; Christine M Stork, PharmD1,2; Maria Mercurio-Zappala, MS3; Mark K Su, MD3; Rachel S Wightman, MD3; Karen R Cummings, MPH4; Joshua G Schier, MD5 (View author affiliations)
View suggested citationLoperamide is an over-the-counter antidiarrheal with opioid-receptor agonist properties. Recommended over-the-counter doses (range = 2–8 mg daily) do not produce opioid effects in the central nervous system because of poor oral bioavailability and P-glycoprotein efflux* of the medication (1); recent reports suggest that large doses (50–300 mg) of loperamide produce euphoria, central nervous system depression, and cardiotoxicity (2–4). Abuse of loperamide for its euphoric effect or for self-treatment of opioid withdrawal is increasing (5). Cases of loperamide abuse reported to the Upstate New York Poison Center and New York City Poison Control Center were analyzed for demographic, exposure, clinical, and laboratory characteristics. Cases of intentional loperamide abuse reported to the National Poison Database System (NPDS) also were analyzed for demographic, dose, formulation, and outcome information.
A New York case of loperamide abuse was defined as any call to the Upstate New York Poison Center or New York City Poison Control Center reporting intentional loperamide abuse during January 1, 2008–March 31, 2016. An NPDS loperamide abuse case was defined as any exposure reported to NPDS citing intentional loperamide abuse during January 1, 2008–March 31, 2016.
Among the 22 New York loperamide abuse cases identified, 18 patients were male. Median patient age was 30 years (range = 19–48 years). Electrocardiogram abnormalities included QTc prolongation in 15 (68%) patients, QRS prolongation in nine (41%) patients, and ventricular dysrhythmia in eight (36%) patients. The average reported daily dose was 358 mg (range = 34–1,200 mg). Previous opioid abuse was reported by 15 patients, previous treatment with methadone or buprenorphine (a partial opioid agonist used to treat opioid addiction) by eight patients, and chronic abuse by 13 patients. Opioid withdrawal symptoms after cessation of loperamide were reported by nine patients. Serum loperamide concentrations were obtained from four patients and ranged from 77–210 ng/mL, representing 25–875 times the therapeutic range of 0.24–3.1 ng/mL (6).
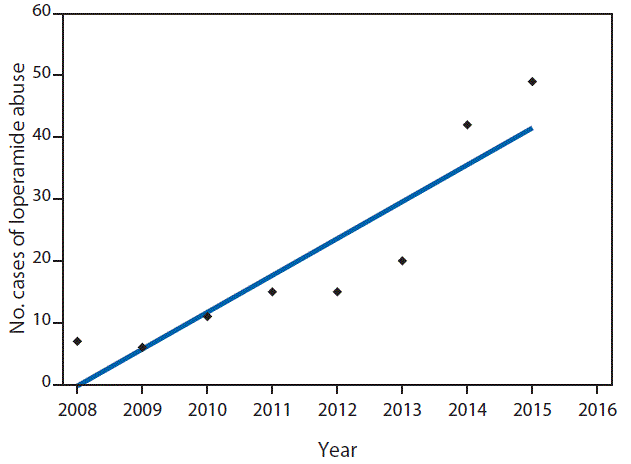
A total of 179 NPDS cases were identified, approximately half of which were reported after January 1, 2014 (Figure). Among the 179 cases, 137 (77%) of the patients were male, the median patient age was 26 years (range = 7–87 years), and 154 (86%) were admitted to or treated in a health care facility. The average loperamide dose was 196.5 mg (range = 2–1,200 mg), and nearly all cases (95%) involved tablets or capsules. Cardiac conduction disturbances were reported in 24 (13%) patients, ventricular tachycardia or fibrillation in 16 (9%) patients, other dysrhythmias in 10 (6%) patients, and other electrocardiogram changes in five (3%) patients. Outcome was reported for 132 patients and was coded as moderate (pronounced or prolonged systemic symptoms, non–life threatening) or major (life-threatening symptoms or residual disability) for 66 patients (50%). Four (3%) deaths were reported.
These cases support the reported association between loperamide abuse and cardiac toxicity. Although laboratory confirmation was only available in four cases, clinical features were consistent with previously reported cases (2–4). Optimal management of loperamide-associated cardiac toxicity is not yet clear; multiple interventions have been reported (2,4). Health care providers and public health officials should remain vigilant for loperamide abuse and report adverse events to the U.S. Food and Drug Administration’s MedWatch reporting system (http://www.fda.gov/Safety/MedWatch). Education of the public and health care providers regarding the dangers of loperamide abuse is an important component of combating opioid addiction in the United States.
Corresponding author: William Eggleston, williamdeggleston@gmail.com, 315-464-8906.
1Upstate New York Poison Center, Syracuse, New York; 2SUNY Upstate Medical University Hospital, Department of Emergency Medicine, Syracuse, New York; 3New York City Poison Control Center; 4New York State Department of Health; 5National Center for Environmental Health, Division of Environmental Hazards and Health Effects, CDC.
* P-glycoprotein acts as a cell membrane pump and increases the elimination of loperamide from the CNS and systemic circulation.
References
- Baker DE. Loperamide: a pharmacological review. Rev Gastroenterol Disord 2007;7(Suppl 3):S11–8. PubMed
- Wightman RS, Hoffman RS, Howland MA, Rice B, Biary R, Lugassy D. Not your regular high: cardiac dysrhythmias caused by loperamide. Clin Toxicol (Phila) 2016;54:454–8. CrossRef PubMed
- Enakpene EO, Riaz IB, Shirazi FM, Raz Y, Indik JH. The long QT teaser: loperamide abuse. Am J Med 2015;128:1083–6. CrossRef PubMed
- Marraffa JM, Holland MG, Sullivan RW, et al. Cardiac conduction disturbance after loperamide abuse. Clin Toxicol (Phila) 2014;52:952–7. CrossRef PubMed
- Daniulaityte R, Carlson R, Falck R, et al. “I just wanted to tell you that loperamide WILL WORK”: a web-based study of extra-medical use of loperamide. Drug Alcohol Depend 2013;130:241–4. CrossRef PubMed
- Eggleston W, Nacca N, Marraffa JM. Loperamide toxicokinetics: serum concentrations in the overdose setting. Clin Toxicol (Phila) 2015;53:495–6. CrossRef PubMed
 FIGURE. Number of reported cases of intentional loperamide abuse, by year — National Poison Database System, January 1, 2008–March 31, 2016
FIGURE. Number of reported cases of intentional loperamide abuse, by year — National Poison Database System, January 1, 2008–March 31, 2016
Suggested citation for this article: Eggleston W, Marraffa JM, Stork CM, et al. Notes from the Field: Cardiac Dysrhythmias After Loperamide Abuse — New York, 2008–2016. MMWR Morb Mortal Wkly Rep 2016;65:1276–1277. DOI: http://dx.doi.org/10.15585/mmwr.mm6545a7.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.