Neisseria gonorrhoeae Antimicrobial Susceptibility Surveillance — The Gonococcal Isolate Surveillance Project, 27 Sites, United States, 2014
Surveillance Summaries / July 15, 2016 / 65(7);1–19
Robert D. Kirkcaldy, MD1; Alesia Harvey1; John R. Papp, PhD1; Carlos del Rio, MD2; Olusegun O. Soge, PhD3; King K. Holmes, MD3; Edward W. Hook III, MD4; Grace Kubin, PhD5; Stefan Riedel, MD6,7; Jonathan Zenilman, MD7; Kevin Pettus1; Tremeka Sanders1; Samera Sharpe1; Elizabeth Torrone, PhD1 (View author affiliations)
View suggested citationAbstract
Problem/Condition: Gonorrhea is the second most commonly reported notifiable disease in the United States; 350,062 gonorrhea cases were reported in 2014. Sexually transmitted infections caused by Neisseria gonorrhoeae are a cause of pelvic inflammatory disease in women, which can lead to serious reproductive complications including tubal infertility, ectopic pregnancy, and chronic pelvic pain. Prevention of sequelae and of transmission to sexual partners relies largely on prompt detection and effective antimicrobial treatment. However, treatment has been compromised by the absence of routine antimicrobial susceptibility testing in clinical care and evolution of antimicrobial resistance to the antibiotics used to treat gonorrhea.
Period Covered: 2014.
Description of the System: The Gonococcal Isolate Surveillance Project (GISP) was established in 1986 as a sentinel surveillance system to monitor trends in antimicrobial susceptibilities of N. gonorrhoeae strains in the United States. Each month, N. gonorrhoeae isolates are collected from up to the first 25 men with gonococcal urethritis attending each of the participating sexually transmitted disease (STD) clinics at 27 sites. The number of participating sites has varied over time (21–30 per year). Selected demographic and clinical data are abstracted from medical records. Isolates are tested for antimicrobial susceptibility using agar dilution at one of five regional laboratories.
Results: A total of 5,093 isolates were collected in 2014. Of these, 25.3% were resistant to tetracycline, 19.2% to ciprofloxacin, and 16.2% to penicillin (plasmid-based, chromosomal, or both). Reduced azithromycin susceptibility (Azi-RS) (defined as minimum inhibitory concentration [MIC] ≥2.0 µg/mL) increased from 0.6% in 2013 to 2.5% in 2014. The increase occurred in all geographic regions, but was greatest in the Midwest, and among all categories of sex of sex partners (men who have sex with men [MSM], men who have sex with men and women [MSMW], and men who have sex with women [MSW]). No Azi-RS isolates exhibited reduced cefixime or ceftriaxone susceptibility (Cfx-RS and Cro-RS, respectively). The prevalence of Cfx-RS (MIC ≥0.25 µg/mL) increased from 0.1% in 2006 to 1.4% in both 2010 and 2011, decreased to 0.4% in 2013, and increased to 0.8% in 2014. Cro-RS (MIC ≥0.125 µg/mL) increased following a similar pattern but at lesser percentages (increased from 0.1% in 2008 to 0.4% in 2011 and decreased to 0.1% in 2013 and 2014). The percentage of isolates resistant to tetracycline, ciprofloxacin, penicillin, or all three antimicrobials, was greater in isolates from MSM than from MSW.
Interpretation: This is the first report to present comprehensive surveillance data from GISP and summarize gonococcal susceptibility over time, as well as underscore the history and public health implications of emerging cephalosporin resistance. Antimicrobial susceptibility patterns vary by geographic region within the United States and by sex of sex partner. Because dual therapy with ceftriaxone plus azithromycin is the only recommended gonorrhea treatment, increases in azithromycin and cephalosporin MICs are cause for concern that resistance to these antimicrobial agents might be emerging. It is unclear whether increases in the percentage of isolates with Azi-RS mark the beginning of a trend. The percentage of isolates with elevated cefixime MICs increased during 2009–2010, then decreased during 2012–2013 after treatment recommendations were changed in 2010 to recommend dual therapy (with a cephalosporin and a second antibiotic) and a higher dosage of ceftriaxone. Subsequently, the treatment recommendations were changed again in 2012 to no longer recommend cefixime as part of first-line therapy (leaving ceftriaxone-based dual therapy as the only recommended therapy). Despite the MIC decrease (i.e., trend of improved cefixime susceptibility) during 2012–2013, the increase in the number of strains with Cfx-RS in 2014 underscores the potential threat of cephalosporin-resistant N. gonorrhoeae.
Public Health Action: The National Strategy for Combating Antibiotic-Resistant Bacteria identifies prevention, rapid detection, and control of outbreaks of ceftriaxone-resistant N. gonorrhoeae infection as a priority for U.S. public health action. Antimicrobial susceptibility surveillance is conducted to guide development of treatment recommendations for effective therapy and prevention of complications from and transmission of gonorrhea. Federal agencies can use GISP data to develop national treatment recommendations and set research and prevention priorities. Local and state health departments can use GISP data to determine allocation of STD prevention services and resources, guide prevention planning, and communicate best treatment practices to health care providers. Continued surveillance, appropriate treatment, development of new antibiotics, and prevention of transmission remain the best strategies to reduce gonorrhea incidence and morbidity.
Introduction
Neisseria gonorrhoeae is a sexually transmitted pathogen that causes infections at the anatomic site of exposure (e.g., urethra, cervix, pharynx, and rectum) and in the eyes of newborn infants. Urethral infections often cause discharge and pain with urination; cervical, pharyngeal, and rectal infections are frequently asymptomatic. In women, gonorrhea is a major cause of pelvic inflammatory disease, which can lead to severe reproductive health complications (e.g., infertility, ectopic pregnancy, and chronic pelvic pain). Infrequently, disseminated gonococcal infection can result in localized septic arthritis, endocarditis, and meningitis (1). Infants born to infected mothers can acquire eye infections that lead to blindness. Gonococcal infections can increase the risk for sexual transmission of human immunodeficiency virus (HIV) (2).
Gonorrhea has been a nationally notifiable disease since 1944 (3). From 1968 to 1994, gonorrhea was consistently the most commonly reported notifiable disease in the United States (4,5). Since 1995 (and with the advent of chlamydia as a nationally notifiable disease), gonorrhea has been consistently the second most commonly reported notifiable disease in the United States; 350,062 cases were reported in 2014 (6,7). In 2014, the rate of reported gonorrhea cases was higher in the southern United States (131.4 per 100,000 population) than in the midwestern, western, and northeastern regions of the country. The reported case rate among women decreased from 108.0 per 100,000 population to 101.3 per 100,000 population during 2011–2014 but beginning in 2009, the reported case rate among men increased from 91.0 per 100,000 population to 120.1 per 100,000 population in 2014.
Prevention of gonorrhea sequelae and of transmission to sexual partners relies largely on prompt detection of infection and effective antimicrobial treatment. However, gonorrhea treatment has been compromised by the evolution of antimicrobial resistance to each recommended antibiotic agent (8).
Surveillance is critical for detecting and monitoring gonococcal resistance. N. gonorrhoeae culture is not routinely available in most clinical settings (because of the shift in clinical practice in the United States and other developed countries toward the use of nonculture-based diagnostic tests, such as nucleic acid amplification tests [NAATs]) and antimicrobial susceptibility testing is not routinely conducted in most laboratories (9). Thus, gonorrhea case reporting does not include reporting of antimicrobial susceptibility and supplemental surveillance approaches have been used. In the 1970s, CDC monitored antimicrobial susceptibility of N. gonorrhoeae through periodic large-scale surveys such as the National Gonorrhea Therapy Monitoring Study (10). In the late 1970s and 1980s, surveillance of penicillinase-producing N. gonorrhoeae (PPNG) was conducted by health departments and incorporated into case reporting to CDC (11). However, the approach lacked consistent sampling frames and methodologies (11).
This report presents data from the Gonococcal Isolate Surveillance Project (GISP) to describe trends in gonococcal antimicrobial susceptibility in the United States during 2000–2014 and summarizes data collected during January–December 2014. Federal agencies can use the findings in this report to develop national treatment recommendations and set research and prevention priorities. Local and state health departments can use GISP data to determine allocation of STD prevention services and resources, guide prevention planning, and communicate best treatment practices to health care providers.
Methods
Data Source
In 1986, CDC established the Gonococcal Isolate Surveillance Project (GISP) as a sentinel surveillance system to monitor trends in N. gonorrhoeae antimicrobial susceptibilities in the United States (12). Data collection began in 1987. Each sentinel site has used a standard protocol, sampling frames, and culture-based laboratory methods consistently since GISP’s inception. Urethral N. gonorrhoeae isolates and demographic and clinical data are collected each month from up to the first 25 men with gonococcal urethritis in participating STD clinics (i.e., maximum 300 isolates per site annually) (http://www.cdc.gov/std/gisp/gisp-protocol-may-2016.pdf). In 2014, 27 sites participated in GISP (range: 21–30 sites during 1987–2014) (Table 1).
Participating Clinical Sites
State and local health departments competitively applied to participate in GISP as part of the funding opportunity Improving STD Programs Through Assessment, Assurance, Policy Development, and Prevention Strategies (CDC-RFA-PS14-1402) (http://www.cdc.gov/std/foa/aapps/ps14-1402-foa-amendment-i-final_07-08-13.pdf). In 2014, 24 state and city health departments (comprising 27 clinic sites) participated in GISP (Table 2). GISP is not designed to be nationally representative, but rather to detect changes in antimicrobial susceptibility across a wide sample of geographic settings. Because resistant gonococcal strains, such as PPNG, are thought to have been imported into the United States from elsewhere, most often East Asia, GISP has preferentially included sites that are major ports of entry and transit hubs (12).
Participating Regional Laboratories
Regional laboratories applied to participate in GISP through a competitive application process as part of a separate funding opportunity (STD Laboratory-Based Surveillance and Gonococcal Isolate Surveillance Project [CDC-RFA-PS14-1401]) (http://www.cdc.gov/std/gisp/foa-8-2013.htm). In 2014, the five participating laboratories were located at Emory University (Atlanta, Georgia); Johns Hopkins University (Baltimore, Maryland); Texas Department of State Health Services (Austin, Texas); University of Alabama at Birmingham; and University of Washington (Seattle, Washington).
Data and Specimen Collection
Sampling of urethral N. gonorrhoeae isolates with a paired clinical and sexual history allows for monitoring of antimicrobial susceptibility in isolates from men stratified by sex of sex partners and other epidemiological characteristics. Urethral infections in men are more likely to be symptomatic (prompting health care–seeking behavior) than genitourinary infections in women and also might be presumptively identified by Gram stain;* thus, sampling of urethral infections in men is an efficient means to detect gonococcal infection. Consistent sampling minimizes potential confounding by changes in screening or testing practices, as well as changes in population samples, thus allowing for reliable analyses of antimicrobial susceptibility trends.
Antimicrobial susceptibility in isolates from MSW has been demonstrated to be a reliable proxy of antimicrobial susceptibility in isolates from women (13,14). In addition, population-level susceptibility of urethral isolates from MSM has been similar to that of population-level susceptibility of pharyngeal and rectal isolates from this population (15).
Presumptive identification of N. gonorrhoeae is based on growth of typical-appearing colonies on Thayer-Martin selective media incubated at 95.0°–97.7°F (35.0°–36.5°C) in 5.0% CO2, a positive oxidase test, and the observation of gram-negative diplococci in stained smears. Gonococcal isolates are then subcultured from the selective primary medium to chocolate agar with 1.0% IsoVitaleX (Becton, Dickinson and Company, Franklin Lakes, New Jersey) noninhibitory media to obtain a pure colony of the isolate. After 18–20 hours of incubation, growth from the pure culture is suspended heavily in trypticase soy broth containing 20.0% (v/v) glycerol and immediately frozen to -94.0°F (-70.0°C). Frozen isolates are shipped monthly to the assigned regional laboratory for antimicrobial susceptibility testing.
Demographic and clinical data are abstracted from medical records by clinic staff and submitted each month using an electronic web-based application provided by CDC. Abstracted information includes age, race/ethnicity, sex, sex of sex partners,† previous diagnoses of gonorrhea, HIV status, selected behavioral characteristics including recent travel, and antimicrobial gonorrhea treatment. Clinical outcome is not collected for GISP. De-identified data are transmitted to CDC through a secure data portal. Jurisdictions are required to uphold the CDC National Center for HIV/AIDS, Viral Hepatitis, STD and TB Prevention’s Data Security and Confidentiality Guidelines for HIV, Viral Hepatitis, Sexually Transmitted Diseases, and Tuberculosis Programs (http://www.cdc.gov/nchhstp/programintegration/docs/pcsidatasecurityguidelines.pdf). The GISP protocol has been reviewed periodically by the CDC Human Research Protection Office and deemed to be a surveillance and disease control activity and not to be human subject research (most recent review, May 2016).
Antimicrobial Susceptibility Testing
Regional laboratories determine β-lactamase production and antimicrobial susceptibilities of all submitted isolates according to a standard protocol. In 2014, antimicrobial susceptibilities to azithromycin, cefixime, ceftriaxone, ciprofloxacin, penicillin, spectinomycin, and tetracycline were determined for each isolate by the agar dilution procedure on Difco GC medium base (Becton, Dickinson and Company, Franklin Lakes, New Jersey) inoculated with 104 colony forming units. Antimicrobial susceptibility results are reported as minimum inhibitory concentrations (MICs), which are the lowest antimicrobial concentrations that inhibit visible growth of a microorganism in the laboratory. As MICs increase, organisms can grow at higher antimicrobial concentrations, providing early warning of potentially impending resistance. Results are transmitted monthly to CDC through a secure data portal. Results are also transmitted to the clinical sites, but because the results are available approximately 2–3 months after specimen collection, results are not used to guide clinical management. For quality assurance, regional laboratories include a set of seven control strains with each testing run. Control strain results must be within a defined MIC range for the run to be valid. In addition, the regional laboratories participate in biannual external quality assessments that include antimicrobial susceptibility testing of a set of 15 blinded cultures provided by CDC.
Specimen Repository
GISP isolates that meet specific criteria are archived in the CDC/ATSDR Specimen Packaging, Inventory and Repository (CASPIR). Isolates from GISP are included in the Food and Drug Administration–CDC Antimicrobial Resistance Isolate Bank (http://www.cdc.gov/drugresistance/resistance-bank/index.html), which is available to support and advance development of diagnostic devices and antimicrobials.
Data Analysis
To minimize overrepresentation of individual geographic sites, the number of isolates in the analytic data set is limited to 300 per site each year. All numbers and percentages in this report are unweighted. Men were categorized by sex of sex partner as MSM, MSMW, or MSW. Antimicrobial susceptibility trends are stratified by census region and sex of sex partners and are interpreted according to criteria established by the Clinical and Laboratory Standards Institute (CLSI) when such criteria are available (16). For antibiotics for which CLSI has not established resistance criteria (i.e., cefixime, ceftriaxone, and azithromycin), the surveillance definition of reduced susceptibility is applied to isolates with MICs higher than wild-type distributions (cefixime MIC ≥0.25 µg/mL [Cfx-RS], ceftriaxone MIC ≥0.125 µg/mL [Cro-RS], and azithromycin MIC ≥2.0 µg/mL [Azi-RS]). Treatment outcome data from Toronto, Canada, suggest that the surveillance case definition for Cfx-RS correlates well with unsuccessful treatment (17). In 2005, the GISP surveillance definition of Azi-RS was changed from ≥1.0 µg/mL to ≥2.0 µg/mL. Descriptive analyses of annual prevalences of resistance phenotypes were performed. Bivariate analyses of epidemiological characteristics were conducted by chi-square, with significance level of p<0.05.
Results
During 2000–2014, a total of 86,517 isolates were submitted (annual median: 5,693; range: 5,093–6,552), including 5,093 isolates submitted from 27 sites in 2014 (Table 1). In 2014, 40.0% of the isolates were collected in the West and approximately 25.0% in the Midwest (Table 2). The West is relatively overrepresented when accounting for the proportion of reported gonorrhea cases among men (40.0% of GISP isolates and 24.9% of reported cases in 2014), and the South is relatively underrepresented (23.4% of GISP isolates versus 41.4% of reported cases in 2014) (7). The median age of men from whom isolates were collected was 28 years (interquartile range [Q3–Q1]: 23–36 years). The majority of participants were black; the racial/ethnic composition of GISP participants approximates that of reported gonorrhea cases among men in the United States (7).
In 2014, associated sex of sex partner data were available for 5,032 isolates (98.8% of total); 37.1% of the isolates were from MSM or MSMW. This percentage increased from 13.9% in 2000 and coincides with increases during 2009–2014 in reported gonorrhea case rates among men in the United States (7). In 2014, HIV co-infection and recent antibiotic use were more often reported by MSM and MSMW than MSW (Table 3).
During 2000–2014, treatment data were submitted for 98.6% of isolates; data were submitted for 99.3% in 2014. The percentage of isolates with treatment recorded as ceftriaxone 250 mg as a single intramuscular dose increased from 10.0% in 2005 to 94.3% in 2014 (Figure 1) and coincided with changes in STD treatment guidelines (18,19). Among men treated with ceftriaxone 250 mg in 2014, 96.4% were co-treated with azithromycin, 2.2% with doxycycline, and 0.1% with another antibiotic; 1.3% were not treated with a second antibiotic. Those not treated with ceftriaxone 250 mg were treated with ceftriaxone 125 mg, ciprofloxacin, cefixime, or levofloxacin (all <1.0%), or a study drug as part of a clinical trial (2.9%). The percentage of men treated with azithromycin 2 g as monotherapy ranged from 0.04% in 2000 to 3.2% in 2012; in 2014 the percentage was 1.9%.
Antimicrobial Susceptibility
Azithromycin
The percentage of Azi-RS isolates remained low during 2000–2013 but increased from 0.6% in 2013 to 2.5% in 2014 (Figure 2), representing the highest prevalence since 1992 when GISP began testing for azithromycin susceptibility. The increase during 2013–2014 was observed in all regions, but was greatest in the Midwest (Figure 3). In 2014, the largest numbers of Azi-RS isolates per site were collected in Minnesota (23), Texas (11), and Arizona (10). When stratified by sex of sex partner, the percentage of Azi-RS isolates increased in 2004 among MSM and MSMW, then decreased (Figure 4). During 2013–2014, the percentage increased among all categories of sex of sex partner: MSW (0.4% to 1.4%), MSM (0.9% to 4.3%), and MSMW (1.4% to 3.2%) (Figure 4). Of men whose isolates exhibited Azi-RS in 2014 and for whom data were available (96.8%), 45.2% were white, 35.5% were black, 57.4% were MSM, 36.1% were MSW, and 6.6% were MSMW. In addition, 21.0% reported recent travel (versus 8.7% of men with azithromycin-susceptible isolates, p<0.001) and 14.4% reported recent antibiotic use (versus 7.8% of men with azithromycin-susceptible isolates, p = 0.016).
Ten isolates with high-level azithromycin MIC (≥16.0 µg/mL) were identified in 2014. Of the seven with MIC = 16.0 µg/mL, three were from the West (two MSM and one MSMW), three were from the Northeast (all MSW), and one was from the Midwest (MSM). One isolate had MIC = 32.0 µg/mL (MSM in the West), one had MIC = 64.0 µg/mL (MSM in the West), and one had MIC ≥256.0 µg/mL (MSM in the West).
Cefixime and Ceftriaxone
The percentage of Cfx-RS isolates (MIC ≥0.25 µg/mL) increased from 2006 (0.1%) to 2010 (1.4%), decreased to 0.4% in 2013, and then increased to 0.8% in 2014 (Figure 2). Isolates from the West were more likely to be Cfx-RS than those from the Northeast, Midwest, or South (Figure 5). The majority of Cfx-RS isolates collected in 2014 were identified in Arizona (eight), California (seven), and Hawaii (six); however, at least three Cfx-RS isolates were identified in each U.S. region. Since 2004, Cfx-RS prevalence has been higher in isolates from MSM or MSMW than MSW (Figure 6). In 2014, the percentage of Cfx-RS isolates was 1.3% among MSM, 1.2% among MSMW, and 0.4% among MSW (Figure 6).
In combination with azithromycin, ceftriaxone is recommended for gonorrhea treatment (20). During 2000–2014, the percentage of Cro-RS (MIC ≥0.125 µg/mL) isolates ranged from 0.1% to 0.4% (Figure 7). In 2014, the percentage was 0.4% in the Northeast, 0.2% in the West, and 0.1% in the South and Midwest (Figure 7). Since 2002, Cro-RS prevalence has been higher in isolates from MSM or MSMW than from MSW (Figure 8). In 2014, the percentage of Cro-RS isolates was 0.4% among MSMW, 0.2% among MSM, and 0.1% among MSW. Among the seven Cro-RS isolates in 2014, five had MIC = 0.125 µg/mL (three MSW in the South, Midwest, and Northeast and two MSM in the West) and two had MIC = 0.25 µg/mL (one MSM in the Northeast and one MSMW in the West).
Ciprofloxacin
GISP measures ciprofloxacin susceptibility to monitor fluoroquinolone-resistant N. gonorrhoeae (QRNG). During the 2000s, prevalence of QRNG strains increased in the United States (Figure 2), emerging in the western United States (Hawaii and then California), soon after among MSM, and then among MSW. In response, CDC adjusted treatment guidelines and no longer recommended use of fluoroquinolones in each affected location or population (21–23). In 2007, CDC no longer recommended use of fluoroquinolones for gonorrhea treatment for any patient in the United States (24). The percentage of QRNG isolates decreased from 14.8% in 2007 to 9.6% in 2009 (Figure 2) and then increased; by 2014, 19.2% of isolates exhibited QRNG, the highest prevalence observed in GISP since testing for ciprofloxacin susceptibility began in 1990. In 2014, QRNG strains were identified in 27.3% of isolates from the West, 21.4% from the Northeast, 14.3% from the South, and 10.2% from the Midwest (Figure 9); QRNG strains were identified in approximately 30.0% of isolates from MSM and MSMW and 12.7% of isolates from MSW (Figure 10).
Penicillin
Since 1989, penicillin has not been recommended for gonorrhea treatment because of high prevalence of resistance; penicillin resistance remains common (25). In 2014, a total of 826 (16.2%) isolates exhibited penicillin resistance alone or in combination with other resistance phenotypes (Figure 2). Of these isolates, 477 (57.8%) exhibited chromosomal resistance (MIC ≥2.0 µg/mL without β-lactamase) and 349 (42.2%) exhibited plasmid-based resistance (presence of β-lactamase). The percentage of isolates with penicillin resistance was high in all regions (Northeast [25.6%], West [17.4%], Midwest [13.3%], and Southeast [13.2%]). Penicillin resistance was common in isolates from MSM (22.1%), MSMW (21.9%), and MSW (12.5%).
Tetracycline
In 2014, a total of 1,287 (25.3%) isolates exhibited tetracycline resistance, either alone or in combination with other resistance phenotypes (Figure 2). The prevalence of tetracycline resistance was high in all regions (West [33.5%], Northeast [25.8%], South [19.6%], and Midwest [17.6%]). Tetracycline resistance was common in isolates from MSM (38.0%), MSMW (31.5%), and MSW (18.2%).
Multidrug Resistance
In 2014, a total of 1,946 (38.2%) of all isolates collected in GISP exhibited resistance (to penicillin, tetracycline, or ciprofloxacin) or reduced susceptibility (to cefixime, ceftriaxone, or azithromycin, for which CLSI has not yet established resistance breakpoints) (16) (Figure 11). Resistance or reduced susceptibility to one antibiotic was identified in 20.8% of isolates, to two in 9.6%, to three in 7.2%, to four in 0.5%, and to five in 0.1%.
Among 125 Azi-RS isolates, 48.8% were resistant to tetracycline, 22.4% to penicillin, and 8.8% to ciprofloxacin; none exhibited Cfx-RS or Cro-RS. Among 38 Cfx-RS isolates, 92.1% were resistant to ciprofloxacin, 76.3% to tetracycline, and 71.1% to penicillin. All seven Cro-RS isolates were resistant to penicillin; 85.7% were resistant to tetracycline and ciprofloxacin. No Cfx-RS or Cro-RS isolates exhibited Azi-RS.
Spectinomycin
Since 2000, no isolates collected in GISP have exhibited spectinomycin resistance. U.S. distribution of spectinomycin was discontinued in 2005 (26).
Discussion
This is the first report to present comprehensive surveillance data from GISP and summarize gonococcal susceptibility over time, as well as underscore the history and public health implications of emerging cephalosporin resistance. Before GISP, monitoring of gonococcal antimicrobial susceptibility was conducted periodically through large-scale surveys and later was limited primarily to detection of PPNG, identified by simple and inexpensive assays for β-lactamase (10,11). However, many public health laboratories lacked the resources to conduct susceptibility testing other than β-lactamase testing and to detect chromosomal resistance; such testing is more expensive and technically demanding (27). GISP was established in 1986 to address those challenges and to provide a scientific rationale for changes in gonorrhea treatment guidelines.
Data from GISP repeatedly have led to revisions to national guidelines for gonorrhea treatment, including the 2015 STD treatment guidelines (20). GISP has allowed CDC to continue to monitor gonococcal antimicrobial susceptibility despite decreasing use of cultures in clinical practice. Following the emergence of N. gonorrhoeae resistance to numerous antibiotics, and with decreasing susceptibility of the pathogen to cefixime (an oral cephalosporin), the primary gonorrhea treatment option recommended by CDC is dual therapy combining ceftriaxone (an injectable cephalosporin) and an oral dose of azithromycin (a macrolide antibiotic) (20). Monitoring of cephalosporin and azithromycin susceptibility is particularly important to ensure the effectiveness of the recommended treatment option. Data from GISP will remain important for monitoring effectiveness of potential treatment options and for success of the National Strategy for Combating Antibiotic-Resistant Bacteria (28).
Azithromycin Susceptibility Trends
Dual therapy with ceftriaxone, an injectable cephalosporin, and azithromycin, an oral macrolide, is the only recommended gonorrhea treatment in the United States (20). The recent increase in Azi-RS prevalence, just 5 years after cephalosporin MICs began to increase among isolates collected in GISP, highlights the potential for development of resistance to the only currently recommended antimicrobial agents. The increase in Azi-RS prevalence was greatest in the Midwest but occurred in all geographic regions. Azi-RS prevalence increased among MSM, who traditionally have experienced a disproportionate number of resistant N. gonorrhoeae infections (23,29,30), but also among MSW.
N. gonorrhoeae Azi-RS is associated with mutations in the mtrR coding region, conferring overexpression of an efflux pump, and mutations in at least one of the four (but usually two or greater) alleles encoding the 23S ribosomal subunit, causing decreased antimicrobial affinity (31–35). It is unclear whether circulating gonococcal strains in the United States acquired these mutations through domestic selection pressure or whether Azi-RS strains were introduced through importation from other countries, as might have occurred with PPNG, QRNG, and Cfx-RS, and spread clonally (11,36,37).
The epidemiology of Azi-RS differs from other resistance phenotypes and might suggest a potential role for domestic selection pressure. Other resistance phenotypes emerged in Hawaii and then California (likely because of relative proximity to East Asia) before spreading eastward. In contrast, azithromycin MICs have tended to be higher in isolates from the Midwest than from other regions (38); therefore, it is not surprising that in 2014, the Midwest had the greatest proportion of Azi-RS isolates. Selection pressure might be exerted by the high frequency with which azithromycin is prescribed in the United States for nonsexually transmitted illnesses, such as respiratory infections (39,40). The recommended dual-therapy regimen for gonorrhea treatment is not a likely contributor to increases in Azi-RS. Azithromycin has been recommended for use in gonorrhea treatment (to treat chlamydia co-infection) since 1998 (41) without increases in Azi-RS prevalence. Recent studies have not indicated antagonist effects between ceftriaxone and azithromycin (42,43). However, gonorrhea treatment with azithromycin monotherapy could conceivably select for Azi-RS; in a recently described case, higher azithromycin MIC was exhibited after unsuccessful treatment with azithromycin monotherapy (34). During 2000–2012, a small increase in use of azithromycin alone for gonorrhea treatment was observed in STD clinics participating in GISP. Data on gonorrhea treatment outside of STD clinics are limited.
Several countries have reported isolates with Azi-RS and increasing azithromycin MICs, which supports the idea of importation of Azi-RS strains into the United States and subsequent clonal spread (44–47). Such reports heighten concerns about whether a trend toward increased Azi-RS is beginning in the United States, which could compromise successful gonorrhea treatment. Not only is azithromycin part of currently recommended dual therapy, it is also a component of each of the alternative treatment regimens for gonorrhea (20). Azithromycin monotherapy is not recommended for gonorrhea treatment; clinicians are strongly encouraged to avoid its use to reduce potential for selection pressure and ensure treatment effectiveness.
Cephalosporin Susceptibility Trends
Concern is growing about potential for cephalosporin resistance. Higher oral cephalosporin (e.g., cefixime) MICs and cases of unsuccessful treatment with oral cephalosporins have been identified in multiple countries. Since 2009, ceftriaxone-resistant strains have been identified in Japan, Spain, and France (48–50). In response to these developments, CDC revised its gonorrhea treatment guidelines in 2010 to recommend 1) dual therapy with a cephalosporin (ceftriaxone [preferred] or cefixime) plus either azithromycin or doxycycline for gonorrhea treatment and 2) a higher dosage of ceftriaxone (18). Subsequently, increasing cefixime MICs were observed in GISP (19). Anticipating that increasing cefixime MICs would result in declining effectiveness of cefixime and that in turn, continued use of cefixime might hasten the development of resistance to ceftriaxone, CDC revised its guidelines again in 2012 to no longer recommend cefixime as part of dual therapy; ceftriaxone-based dual therapy was the only recommended therapy (19). Subsequent decreases in the proportion of Cfx-RS isolates suggested an association between changes in treatment guidelines and circulation of fewer clinical strains with reduced susceptibility. Although other countries observed similar trends, only some had changed treatment guidance to ceftriaxone-based dual therapy. Other factors (e.g., reduced transmission of a clonal lineage) might have contributed to decreasing prevalence (35,51–59). On the basis of historical precedent, such as patterns observed with QRNG strains, recent cephalosporin susceptibility data from Australia and Europe, and 2014 GISP data, improvement in cefixime susceptibility might be short lived (24,54,60).
N. gonorrhoeae strains in the United States remain highly susceptible to ceftriaxone, perhaps because additional mutations (beyond those required for Cfx-RS) seem to be required to confer Cro-RS (48–50). Although not yet documented, it is also conceivable that such mutations confer fitness costs (i.e., acquired mutations that confer resistance might impair the virulence, transmissibility, or infectivity of the bacteria), as the spread of the ceftriaxone-resistant strains appears to be limited (61,62).
Gonorrhea Treatment Recommendations
CDC recommends that gonococcal infections be treated with ceftriaxone 250 mg as a single intramuscular dose and azithromycin 1 g orally (20). The combination regimen is thought to have a synergistic effect that enhances killing of N. gonorrhoeae resistant to one of the antimicrobials and minimizes potential for transmission of resistant strains. Furthermore, Cfx-RS and Cro-RS isolates are susceptible to azithromycin and vice versa. If a pathogen harbors reduced susceptibility to one antibiotic, the second antibiotic is expected to cure the infection. Patients with cephalosporin allergies should be treated with azithromycin 2 g plus either gentamicin or gemifloxacin (20).
Monitoring Antimicrobial Susceptibility
GISP is the only source of national and regional gonococcal antimicrobial susceptibility data in the United States. Since 1986, GISP has provided data to guide gonorrhea treatment recommendations and address the threat of antimicrobial resistance while monitoring antimicrobial susceptibility. Culture-based methods are necessary for N. gonorrhoeae antimicrobial susceptibility testing, which exposes live organisms to varying concentrations of antimicrobial agents. However, laboratories’ widespread adoption of molecular-based diagnostic tests that do not yield live organisms, such as NAATs, has led to a marked decrease in use of and capacity to perform culture testing and N. gonorrhoeae antimicrobial susceptibility testing in the United States (9). In comparison to culture, NAATs are more sensitive and specific, can be performed on less invasively collected specimens (e.g., urine and self-collected vaginal swabs), and have far less burdensome specimen handling requirements (63).
The limited amount of antimicrobial susceptibility testing conducted outside of GISP also precludes use of electronic health records as a data source for surveillance of gonococcal antimicrobial susceptibility. The potential for cephalosporin and azithromycin resistance with limited antimicrobial treatment options underscores the importance of GISP data (64). Beyond the recommended dual-therapy regimen, no other antibiotics are available to treat gonorrhea effectively with a high level of efficacy at all anatomic sites of infection, a favorable side-effect profile, low cost, and ease of administration.
The strengths of GISP surveillance include nonrandom, systematic sampling and consistent methodology. Systematic sampling of consecutive symptomatic men minimizes selection bias and provides a robust estimate of gonococcal susceptibility at each sentinel clinic. Consistent methodology minimizes potential confounding by changes in screening or testing practices, population samples, or laboratory practice over time, thus allowing for reliable analyses of antimicrobial susceptibility trends.
By design, GISP does not collect a representative sample of gonococcal strains from men throughout the United States. Rather, by relatively oversampling from the western United States (where resistance tends to emerge), GISP functions as a truly sentinel system and is able to more rapidly detect trends. STD specialty care clinics, particularly those that participate in GISP, maintain culture capacity and thus are able to provide a sufficient number of isolates for surveillance. Most gonorrhea cases in the United States are identified in settings other than STD clinics (7), but gonococcal infections usually are asymptomatic and often are distributed across multiple health care settings. Compared with other health care facilities, STD clinics still tend to diagnose a disproportionate number of gonococcal infections among men.
Data from GISP repeatedly have been used to develop CDC’s STD treatment guidelines for gonorrhea (18–20,23,24,41,65–68). These guidelines have a direct influence on health care–provider prescribing practices (69,70). GISP data indicate that health care providers in state and locally funded STD clinics tend to adopt changes in treatment guidelines quickly. For example, the percentage of patients treated with ceftriaxone 250 mg increased substantially following release of 2010 STD treatment guidelines. CDC’s guidelines also influence gonorrhea treatment in non–STD clinic settings, although adoption of new recommendations in these facilities tends to be slower (69).
GISP monitors N. gonorrhoeae antimicrobial susceptibility trends in the United States. The World Health Organization (WHO) conducts global surveillance of gonococcal antimicrobial susceptibility through the Gonococcal Antimicrobial Surveillance Programme (GASP), which is operated by regional coordinating centers in nearly all WHO regions. GASP collaborates with GISP and other national surveillance programs, such as United Kingdom’s Gonococcal Resistance to Antimicrobials Surveillance System (GRASP) and Australia’s Gonococcal Surveillance Programme (51–54). Although interpretation of data sometimes is limited by convenience sampling designs and lack of associated epidemiologic data in some countries, GASP and other international surveillance programs provide critical data on global transmission of resistant strains.
Limitations
The findings provided in this report are subject to at least four limitations. First, only men with urethral gonorrhea diagnosed in participating STD clinics are included, so epidemiologic characteristics of men in GISP are not expected to reflect those of all persons with gonorrhea diagnosed in the United States. Second, reported treatment data reflect prescribing practices in specialty STD clinics and are not expected to reflect gonorrhea treatment practices in private clinical settings and other non-STD clinic health care settings, where most gonorrhea is diagnosed in the United States (7). Third, although relative oversampling of the western United States might allow for more rapid identification of emerging resistance trends, such sampling might contribute to overestimates of national-level point prevalence of resistance phenotypes. Finally, although clear differences in susceptibility are evident by sex of sex partner even after stratification by geographic region and adjustment for other potential confounders (30), the magnitude of difference in prevalence of resistance between MSM and MSW might be influenced by differences in proportions of MSM, MSMW, and MSW in GISP by geographic region.
Future Directions
One goal of the United States National Strategy for Combating Antibiotic-Resistant Bacteria is to maintain ceftriaxone-resistant N. gonorrhoeae prevalence at <2.0% through 2020 (28). This effort will require a multipronged approach and new tools to prevent emergence of resistant gonorrhea strains. Strengthening gonorrhea control should be the foundation, possibly with a focus on the western United States and other regions where resistant strains tend to emerge. Optimal prevention measures include screening sexually active females aged <25 years, as well as women aged >25 years at increased risk for infection§ (20); screening sexually active MSM at anatomic sites of possible exposure at least annually (20); and appropriately treating patients with gonorrhea and their recent sex partners, including retesting 3 months after treatment. Gonorrhea control options for patients who cannot link their partners to evaluation and treatment include partner notification by health department staff or expedited partner therapy. Clinicians should use the dual-therapy regimen of ceftriaxone 250 mg intramuscularly and azithromycin 1 g orally (20) to treat patients with gonorrhea and stay abreast of changes to CDC’s treatment guidelines. Sexually active persons can be encouraged to use condoms consistently and correctly to prevent transmission of gonorrhea.
In 2016, the U.S. Congress approved $160 million in additional funding to CDC to implement its role in the National Strategy for Combating Antibiotic-Resistant Bacteria. The national strategy outlines interventions to reduce the emergence and spread of antibiotic-resistant pathogens, which include N. gonorrhoeae (http://www.cdc.gov/drugresistance/solutions-initiative/index.html). As part of this initiative, CDC plans to build capacity in up to nine U.S. counties to rapidly detect and respond to antibiotic-resistant gonococcal infections. Rapid identification of resistant infections could allow local public health authorities to slow transmission through rapid investigations, contact tracing, and network analyses. New diagnostic tools are needed. Development of well-validated molecular assays to quickly identify presence or absence of resistance determinants (absence suggests antimicrobial susceptibility) could guide more timely and tailored treatment and improve reach and speed of surveillance. Through the Advanced Molecular Detection initiative, CDC is supporting identification of N. gonorrhoeae resistance determinants, an important first step in the development of molecular assays (http://www.cdc.gov/amd/project-summaries/treating-gonorrhea-threat.html). Phenotypic culture-based antimicrobial susceptibility testing will remain critical, however; molecular tests alone will not detect new mutations conferring resistance or new resistance phenotypes. New antibiotics and new prevention tools, including a vaccine, are urgent needs. Surveillance is a core public health function that guides prevention and control efforts as new tools or approaches are developed and implemented.
Conclusion
Since its inception in 1986, GISP has monitored N. gonorrhoeae antimicrobial susceptibility. Data from GISP have advanced knowledge about gonococcal resistance, influenced policy and prevention decisions, and repeatedly provided evidence on which to base national treatment recommendations. In 2014, data from GISP demonstrated increases in prevalence of Azi-RS and to a lesser degree, of Cfx-RS. It is unclear whether these increases mark the beginning of trends, but emergence of cephalosporin and azithromycin resistance would complicate gonorrhea treatment substantially. Local and state health departments can use GISP data to determine allocation of STD prevention services and resources, guide prevention and control planning, and inform health care providers. Federal agencies can use GISP data to monitor susceptibility trends, identify novel resistances, guide national treatment recommendations, set research and prevention priorities, and monitor success of the National Strategy for Combating Antibiotic-Resistant Bacteria. Specimens collected through surveillance can be used to guide development of diagnostic methods, therapeutic agents, and vaccines. Continued surveillance, appropriate treatment, development of new antibiotics, and prevention of transmission remain the best strategies to reduce gonorrhea incidence and morbidity.
Acknowledgments
Regional laboratories and participating sites contributed to this report. Baderinwa Offutt and Tamayo Barnes (Emory University), Kar Mun Neoh (Johns Hopkins University), Tamara Baldwin and Carol Rodriguez (Texas), and Paula Dixon (University of Alabama at Birmingham) conducted antimicrobial susceptibility testing for GISP. The following are principal collaborators and project coordinators: Anthony Merriweather, Edward Khan, MD, Tracy Burkett (Alabama); Roxanne Ereth, Renuka Khurana, MD, Tom Mickey, Lovina Ogden (Arizona); Heidi Aiem, Heidi Bauer, MD, Karen Galliher, Loreto Negado, Christopher Ried, MD (California); Nanette Benbow, Roman Golash, Irina Tabidze, MD (Chicago, Illinois); Michelle Allen, Richard Hedenquist, Ruby Lewis-Hardy, Praful Bhatti (Georgia); Glenn Wasserman, MD, G. Luke Hasty, PhD, Chris Whelen, PhD (Hawaii); Andrea Radford, Janet Arno, MD, David Nguyen, Bonny Lewis Van, PhD (Indiana); Ryan Murphy, PhD, Marisol Mejia, Nicole Green, PhD (Los Angeles, California); DeAnn Gruber, PhD, Stephanie Taylor, MD (Louisiana); Katherine Hsu, MD, Donna Felsenstein, MD, Tracy Stiles (Massachusetts); Karen Krzanowski, Kristine Judd-Tuinier, Barbara Weberman (Michigan); Krissie Guerard, Dawn Ginzl, Anna Strain, PhD (Minnesota); David Bamberger, MD, Ron Griffin, Lesha Dennis, Tiffany Wilkinson (Missouri); Janine Waters, Cheryl Champlin, Lisa Onischuk (New Mexico); Alison Muse, Jackie Andula, Shirley Keenan (New York [Erie County]); Susan Blank, MD, Preeti Pathela, PhD, Inessa Rubinstein (New York, New York); Richard Whitley, Patricia Armour (Nevada); Laura Bachmann, MD, Ramon Cook, Cindy Toler (North Carolina); Amanda Dennison, Jose Bazan, DO, Jamie Blair, Neil Conway, PhD, Melissa Ervin, Marylou Zima (Ohio); Veda Latin, Kim Toevs, Sean Schafer, MD, Kathy Venator, Chris Nytko (Oregon); Caroline Johnson, MD, Lenore Asbel, MD, Greta Anschuetz (Philadelphia, Pennsylvania); Tomas Aragon, MD, Susan Philip, MD, Stephanie Cohen, MD, Robert Kohn, MPH, Jonathan Carlson (San Francisco, California); Felipe Rocha, Lynne Davis, Edward Bannister, PhD (Texas); Stephen Yamada, MS (Tripler Army Medical Center, Hawaii); and Matt Golden, MD, David Katz, PhD, Paul Swenson, PhD (Washington).
Corresponding author: Robert Kirkcaldy, Division of STD Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC. Telephone: 404-639-8659; E-mail: hlg8@cdc.gov.
1Division of STD Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD and TB Prevention, CDC, Atlanta, Georgia; 2Rollins School of Public Health and Department of Medicine, Division of Infectious Diseases, Emory University School of Medicine, Emory University, Atlanta, Georgia; 3University of Washington, Seattle, Washington; 4University of Alabama at Birmingham, Birmingham, Alabama; 5Texas Department of State Health Services, Austin, Texas; 6Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, Massachusetts; 7Johns Hopkins University, Baltimore, Maryland
References
- Workowski K. Chlamydia and gonorrhea. Ann Intern Med 2013;158:ITC2–1. Erratum in: Ann Intern Med 2013;158:504. CrossRef PubMed
- Braunstein SL, Ingabire CM, Kestelyn E, et al. High human immunodeficiency virus incidence in a cohort of Rwandan female sex workers. Sex Transm Dis 2011;38:385–94. CrossRef PubMed
- CDC. National Notifiable Diseases Surveillance System (NNDSS). https://wwwn.cdc.gov/nndss/conditions/gonorrhea/
- CDC. Reported incidence of notifiable diseases in the United States, 1968. MMWR Morb Mortal Wkly Rep 1968;17:1–59.
- Adams DA, Anderson WJ, Dean AG, et al. Summary of notifiable diseases, United States 1994. MMWR Morb Mortal Wkly Rep 1994;43:1–80. PubMed
- Adams DA, Anderson WJ, Copeland TM, et al. Summary of notifiable diseases, United States, 1995. MMWR Morb Mortal Wkly Rep 1995;44:1–87. PubMed
- CDC. Sexually transmitted disease surveillance 2014. Atlanta: U.S. Department of Health and Human Services; 2015.
- Kidd S, Kirkcaldy RD, Burstein G. Antimicrobial resistance in Neisseria gonorrhoeae. In: Holland-Hall C, Braverman PK, editors. AM:STARs: hot topics in adolescent health. Elk Grove Village, IL: American Academy of Pediatrics; 2014.
- Association of Public Health Laboratories STD Steering Committee. Issues in brief: the role of public health laboratories in STD testing. May 2011. http://www.aphl.org/AboutAPHL/publications/Documents/ID_2011May_STDIssueBrief.pdf
- Jaffe HW, Biddle JW, Thornsberry C, et al. National gonorrhea therapy monitoring study: in vitro antibiotic susceptibility and its correlation with treatment results. N Engl J Med 1976;294:5–9. CrossRef PubMed
- Jaffe HW, Biddle JW, Johnson SR, Wiesner PJ. Infections due to penicillinase-producing Neisseria gonorrhoeae in the United States: 1976–1980. J Infect Dis 1981;144:191–7. CrossRef PubMed
- Schwarcz SK, Zenilman JM, Schnell D, et al. National surveillance of antimicrobial resistance in Neisseria gonorrhoeae. The Gonococcal Isolate Surveillance Project. JAMA 1990;264:1413–7. CrossRef PubMed
- Kidd S, Lee MV, Maningas E, et al. Gonococcal susceptibility to cephalosporins—Hawaii, 2003 to 2011. Sex Transm Dis 2013;40:756–9. CrossRef PubMed
- Kidd S, Moore PC, Kirkcaldy RD, et al. Comparison of antimicrobial susceptibility of urogenital Neisseria gonorrhoeae isolates obtained from women and men. Sex Transm Dis 2015;42:434–9. CrossRef PubMed
- Kidd S, Zaidi A, Asbel L, et al. Comparison of antimicrobial susceptibilities of pharyngeal, rectal, and urethral Neisseria gonorrhoeae isolates among men who have sex with men. Antimicrob Agents Chemother 2015;59:2588–95. CrossRef PubMed
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. Twenty-fourth informational supplement. CLSI document M100-S24. Wayne, PA: Clinical and Laboratory Standards Institute; 2014.
- Allen VG, Mitterni L, Seah C, et al. Neisseria gonorrhoeae treatment failure and susceptibility to cefixime in Toronto, Canada. JAMA 2013;309:163–70. CrossRef PubMed
- Workowski KA, Berman S. Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep 2010;59(No. RR-12). PubMed
- CDC. Update to CDC’s sexually transmitted diseases treatment guidelines, 2010: oral cephalosporins no longer a recommended treatment for gonococcal infections. MMWR Morb Mortal Wkly Rep 2012;61:590–4. PubMed
- Workowski KA, Bolan GA. CDC. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep 2015;64(No. RR-3). PubMed
- CDC. Fluoroquinolone-resistance in Neisseria gonorrhoeae, Hawaii, 1999, and decreased susceptibility to azithromycin in N. gonorrhoeae, Missouri, 1999. MMWR Morb Mortal Wkly Rep 2000;49:833–7. PubMed
- CDC. Increases in fluoroquinolone-resistant Neisseria gonorrhoeae—Hawaii and California, 2001. MMWR Morb Mortal Wkly Rep 2002;51:1041–4. PubMed
- CDC. Increases in fluoroquinolone-resistant Neisseria gonorrhoeae among men who have sex with men—United States, 2003, and revised recommendations for gonorrhea treatment, 2004. MMWR Morb Mortal Wkly Rep 2004;53:335–8. PubMed
- CDC. Update to CDC’s sexually transmitted diseases treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR Morb Mortal Wkly Rep 2007;56:332–6. PubMed
- CDC. 1989 sexually transmitted diseases treatment guidelines. MMWR Suppl 1989;38(No. Suppl 8):1–43. Erratum in: MMWR Morb Mortal Wkly Rep 1989 29;38:664. PubMed
- CDC. Notice to readers: discontinuation of spectinomycin. MMWR Morb Mortal Wkly Rep 2006; 55:370.
- Gorwitz RJ, Nakashima AK, Moran JS, Knapp JS; The Gonococcal Isolate Surveillance Project Study Group. Sentinel surveillance for antimicrobial resistance in Neisseria gonorrhoeae—United States, 1988–1991. MMWR CDC Surveill Summ 1993;42:29–39. PubMed
- National Strategy for Combating Antibiotic Resistant Bacteria. https://www.whitehouse.gov/sites/default/files/docs/carb_national_strategy.pdf
- CDC. Cephalosporin susceptibility among Neisseria gonorrhoeae isolates—United States, 2000–2010. MMWR Morb Mortal Wkly Rep 2011;60:873–7. PubMed
- Kirkcaldy RD, Zaidi A, Hook EW, et al. Neisseria gonorrhoeae antimicrobial resistance among men who have sex with men and men who have sex exclusively with women: the Gonococcal Isolate Surveillance Project, 2005–2010. Ann Intern Med 2013;158:321–8. CrossRef PubMed
- McLean CA, Wang SA, Hoff GL, et al. The emergence of Neisseria gonorrhoeae with decreased susceptibility to azithromycin in Kansas City, Missouri, 1999 to 2000. Sex Transm Dis 2004;31:73–8. CrossRef PubMed
- Zarantonelli L, Borthagaray G, Lee E-H, Shafer WM. Decreased azithromycin susceptibility of Neisseria gonorrhoeae due to mtrR mutations. Antimicrob Agents Chemother 1999;43:2468–72. PubMed
- Johnson SR, Sandul AL, Parekh M, Wang SA, Knapp JS, Trees DL. Mutations causing in vitro resistance to azithromycin in Neisseria gonorrhoeae. Int J Antimicrob Agents 2003;21:414–9. CrossRef PubMed
- Soge OO, Harger D, Schafer S, et al. Emergence of increased azithromycin resistance during unsuccessful treatment of Neisseria gonorrhoeae infection with azithromycin (Portland, OR, 2011). Sex Transm Dis 2012;39:877–9. CrossRef PubMed
- Grad YH, Kirkcaldy RD, Trees D, et al. Genomic epidemiology of Neisseria gonorrhoeae with reduced susceptibility to cefixime in the USA: a retrospective observational study. Lancet Infect Dis 2014;14:220–6. CrossRef PubMed
- Iverson CJ, Wang SA, Lee MV, et al. Fluoroquinolone resistance among Neisseria gonorrhoeae isolates in Hawaii, 1990–2000: role of foreign importation and increasing endemic spread. Sex Transm Dis 2004;31:702–8. CrossRef PubMed
- Unemo M, Nicholas RA. Emergence of multidrug-resistant, extensively drug-resistant and untreatable gonorrhea. Future Microbiol 2012;7:1401–22. CrossRef PubMed
- Kirkcaldy RD, Soge O, Papp JR, et al. Analysis of Neisseria gonorrhoeae azithromycin susceptibility in the United States by the Gonococcal Isolate Surveillance Project, 2005 to 2013. Antimicrob Agents Chemother 2015;59:998–1003. CrossRef PubMed
- Hicks LA, Taylor TH, Hunkler RJ. U.S. outpatient antibiotic prescribing, 2010. N Engl J Med 2013;368:1461–2. CrossRef PubMed
- Hicks LA, Chien Y-W, Taylor TH, Haber M, Klugman KP; Active Bacterial Core Surveillance (ABCs) Team. Outpatient antibiotic prescribing and nonsusceptible Streptococcus pneumoniae in the United States, 1996–2003. Clin Infect Dis 2011;53:631–9. CrossRef PubMed
- CDC. 1998 guidelines for treatment of sexually transmitted diseases. MMWR Recomm Rep 1998;47(No. RR-1):1–111. PubMed
- Pereira R, Cole MJ, Ison CA. Combination therapy for gonorrhoea: in vitro synergy testing. J Antimicrob Chemother 2013;68:640–3. CrossRef PubMed
- Barbee LA, Soge OO, Holmes KK, Golden MR. In vitro synergy testing of novel antimicrobial combination therapies against Neisseria gonorrhoeae. J Antimicrob Chemother 2014;69:1572–8. CrossRef PubMed
- Ni C, Xue J, Zhang C, Zhou H, van der Veen S. High prevalence of Neisseria gonorrhoeae with high-level resistance to azithromycin in Hangzhou, China. J Antimicrob Chemother 2016. Epub ahead of print. CrossRef PubMed
- Tanaka M, Furuya R, Irie S, Kanayama A, Kobayashi I. High prevalence of azithromycin-resistant Neisseria gonorrhoeae isolates with a multidrug resistance phenotype in Fukuoka, Japan. Sex Transm Dis 2015;42:337–41. CrossRef PubMed
- Martin I, Sawatzky P, Liu G, et al. Decline in decreased cephalosporin susceptibility and increase in azithromycin resistance in Neisseria gonorrhoeae, Canada. Emerg Infect Dis 2016;22:65–7. CrossRef PubMed
- Chisholm SA, Wilson J, Alexander S, et al. An outbreak of high-level azithromycin resistant Neisseria gonorrhoeae in England. Sex Transm Infect 2015;sextrans-2015-052312. Epub ahead of print. CrossRef PubMed
- Ohnishi M, Saika T, Hoshina S, et al. Ceftriaxone-resistant Neisseria gonorrhoeae, Japan. Emerg Infect Dis 2011;17:148–9. CrossRef PubMed
- Cámara J, Serra J, Ayats J, et al. Molecular characterization of two high-level ceftriaxone-resistant Neisseria gonorrhoeae isolates detected in Catalonia, Spain. J Antimicrob Chemother 2012;67:1858–60. CrossRef PubMed
- Unemo M, Golparian D, Nicholas R, Ohnishi M, Gallay A, Sednaoui P. High-level cefixime- and ceftriaxone-resistant Neisseria gonorrhoeae in France: novel penA mosaic allele in a successful international clone causes treatment failure. Antimicrob Agents Chemother 2012;56:1273–80. CrossRef PubMed
- Ison CA, Town K, Obi C, et al. GRASP Collaborative Group. Decreased susceptibility to cephalosporins among gonococci: data from the Gonococcal Resistance to Antimicrobials Surveillance Programme (GRASP) in England and Wales, 2007–2011. Lancet Infect Dis 2013;13:762–8. CrossRef PubMed
- Public Health England. GRASP 2013 report: the Gonococcal Resistance to Antimicrobials Surveillance Programme (England and Wales). October 2014. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/368477/GRASP_Report_2013.pdf
- Lahra MM, Donovan B, Whiley DM. Decreased susceptibility to cephalosporins among gonococci [letter]? Lancet Infect Dis 2014;14:186. CrossRef PubMed
- Lahra MM; Australian Gonococcal Surveillance Programme. Australian Gonococcal Surveillance Programme annual report, 2013. Commun Dis Intell Q Rep 2015;39:E137–45. PubMed
- Bala M, Kakran M, Singh V, et al. ; Members of WHO GASP SEAR Network. Monitoring antimicrobial resistance in Neisseria gonorrhoeae in selected countries of the WHO South-East Asia Region between 2009 and 2012: a retrospective analysis. Sex Transm Infect 2013;89(Suppl 4):iv28–35. CrossRef PubMed
- Kubanova A, Kubanov A, Frigo N, et al. Russian gonococcal antimicrobial susceptibility programme (RU-GASP)—resistance in Neisseria gonorrhoeae during 2009–2012 and NG-MAST genotypes in 2011 and 2012. BMC Infect Dis 2014;14:342–8. CrossRef PubMed
- Jeverica S, Golparian D, Matičič M, et al. Phenotypic and molecular characterization of Neisseria gonorrhoeae isolates from Slovenia, 2006–12: rise and fall of the multidrug-resistant NG-MAST genogroup 1407 clone? J Antimicrob Chemother 2014;69:1517–25. CrossRef PubMed
- European Centre for Disease Prevention and Control. Gonococcal antimicrobial susceptibility surveillance in Europe, 2011. March 2013. http://ecdc.europa.eu/en/publications/Publications/gonococcal-antimicrobial-susceptibility-surveillance-27-mar-2013.pdf
- Bignell C, Fitzgerald M; Guideline Development Group, British Association for Sexual Health and HIV UK. UK national guideline for the management of gonorrhoea in adults, 2011. Int J STD AIDS 2011;22:541–7. CrossRef PubMed
- Cole MJ, Spiteri G, Jacobsson S, Pitt R, Grigorjev V, Unemo M; Euro-GASP Network. Is the tide turning again for cephalosporin resistance in Neisseria gonorrhoeae in Europe? Results from the 2013 European surveillance. BMC Infect Dis 2015;15:321–8. CrossRef PubMed
- Melnyk AH, Wong A, Kassen R. The fitness costs of antibiotic resistance mutations. Evol Appl 2015;8:273–83. CrossRef PubMed
- Shimuta K, Unemo M, Nakayama S, et al. ; Antibiotic-Resistant Gonorrhea Study Group. Antimicrobial resistance and molecular typing of Neisseria gonorrhoeae isolates in Kyoto and Osaka, Japan, 2010 to 2012: intensified surveillance after identification of the first strain (H041) with high-level ceftriaxone resistance. Antimicrob Agents Chemother 2013;57:5225–32. CrossRef PubMed
- Papp JR, Schachter J, Gaydos CA, Van Der Pol B. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae—2014. MMWR Recomm Rep 2014;63(No. RR-2). PubMed
- Shlaes DM, Sahm D, Opiela C, Spellberg B. The FDA reboot of antibiotic development. Antimicrob Agents Chemother 2013;57:4605–7. CrossRef PubMed
- CDC. 1989 sexually transmitted diseases treatment guidelines. MMWR Morb Mortal Wkly Rep 1989;38(No. Suppl 8).
- CDC. 1993 sexually transmitted diseases treatment guidelines. MMWR Recomm Rep 1993;42(No. RR-14). PubMed
- CDC. Sexually transmitted diseases treatment guidelines 2002. MMWR Recomm Rep 2002;51(No. RR-6). PubMed
- Workowski KA, Berman SM; CDC. Sexually transmitted diseases treatment guidelines, 2006. MMWR Recomm Rep 2006;55(No. RR-11). PubMed
- Dowell D, Tian LH, Stover JA, et al. Changes in fluoroquinolone use for gonorrhea following publication of revised treatment guidelines. Am J Public Health 2012;102:148–55. CrossRef PubMed
- Kerani RP, Stenger MR, Weinstock H, et al. Gonorrhea treatment practices in the STD Surveillance Network, 2010–2012. Sex Transm Dis 2015;42:6–12. CrossRef PubMed
* Per CDC’s 2014 STD laboratory guidelines, Gram stains of endocervical specimens or pharyngeal or rectal specimens are not considered sufficient to detect gonococcal infection and therefore are not recommended.
† Sex of sex partner data collected in accordance with local clinic practice and can include chart notation from previous visits, patient self-report of sexual orientation, or both, or sex of sex partner (from prior 3, 6, or 12 months).
§ Those who have a new sex partner, more than one sex partner, a sex partner with concurrent partners, or a sex partner who has a sexually transmitted infection.
 TABLE 1. Sentinel clinic sites, by region and years of participation — Gonococcal Isolate Surveillance Project, 1987–2014
TABLE 1. Sentinel clinic sites, by region and years of participation — Gonococcal Isolate Surveillance Project, 1987–2014
| Region (site) | Years of participation |
|---|---|
| West | |
| Alaska (Anchorage) | 1987–2003 |
| Arizona (Maricopa County) | 1987–2014 |
| California (Long Beach) | 1987–2007 |
| California (Los Angeles) | 2003–2014 |
| California (Orange County) | 1991–2014 |
| California (San Diego County) | 1987–2014 |
| California (San Francisco County) | 1987–2014 |
| Colorado (Denver) | 1987–2013 |
| Hawaii (Honolulu) | 1987–2014 |
| Hawaii (Tripler Army Medical Center) | 2001–2006, 2009–2014 |
| New Mexico (Albuquerque) | 1987–2014 |
| Nevada (Southern Nevada) | 2002–2014 |
| Oregon (Multnomah County) | 1987–2014 |
| Utah (Salt Lake City) | 2003 |
| Washington (Seattle/King County) | 1987–2014 |
| Midwest | |
| Illinois (Chicago) | 1996–2014 |
| Indiana (Marion County) | 2013–2014 |
| Michigan (Detroit) | 2003–2011 |
| Michigan (Oakland County) | 2012–2014 |
| Minnesota (Hennepin County) | 1992–2014 |
| Missouri (Kansas City) | 1991–2001, 2007–2014 |
| Missouri (St. Louis) | 1987–2004 |
| Ohio (Cincinnati) | 1987–2011 |
| Ohio (Cleveland) | 1991–2014 |
| Ohio (Columbus) | 2012–2014 |
| Northeast | |
| Massachusetts (Boston) | 1987–1992, 2014 |
| New Jersey (Nassau County) | 1991–1999 |
| New York (Erie County) | 2013–2014 |
| New York (New York) | 2006–2014 |
| Pennsylvania (Philadelphia) | 1987–2014 |
| South | |
| Alabama (Jefferson County) | 1987–2014 |
| Florida (Miami) | 1998–2013 |
| Florida (West Palm Beach) | 1987–1998 |
| Georgia (Fulton County) | 1987–2014 |
| Louisiana (New Orleans) | 1987–2014 |
| Maryland (Baltimore) | 1987–2013 |
| North Carolina (Fort Bragg) | 1987–2002 |
| North Carolina (Guilford County) | 2002–2014 |
| Oklahoma (Oklahoma City) | 2003–2013 |
| Texas (Dallas) | 2000–2014 |
| Virginia (Richmond) | 2007–2013 |
 TABLE 2. Number and percentage of participants, by region, state, and site* — Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014
TABLE 2. Number and percentage of participants, by region, state, and site* — Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014
| Region and state (site) | No. (%) |
|---|---|
| West | 2,028 (39.8) |
| Arizona (Maricopa County) | 300 (5.9) |
| California (Orange County, San Diego County) | 389 (7.6) |
| Hawaii (Honolulu, Tripler Army Medical Center) | 132 (2.6) |
| Los Angeles County, California | 171 (3.4) |
| New Mexico (Albuquerque) | 114 (2.2) |
| Nevada (Southern Nevada) | 300 (5.9) |
| Oregon (Multnomah County) | 128 (2.5) |
| San Francisco, California | 300 (5.9) |
| Washington (Seattle/King County) | 194 (3.8) |
| Midwest | 1,312 (25.8) |
| Chicago, Illinois | 129 (2.5) |
| Indiana (Marion County) | 300 (5.9) |
| Michigan (Oakland County) | 135 (2.7) |
| Minnesota (Hennepin County) | 103 (2.0) |
| Missouri (Kansas City) | 232 (4.6) |
| Ohio (Cleveland, Columbus) | 413 (8.1) |
| South | 1,207 (23.8) |
| Alabama (Jefferson County) | 223 (4.4) |
| Georgia (Fulton County) | 300 (5.9) |
| Louisiana (New Orleans) | 171 (3.4) |
| North Carolina (Guilford County) | 213 (4.2) |
| Texas (Dallas) | 300 (5.9) |
| Northeast | 546 (10.7) |
| Massachusetts (Boston) | 27 (0.5) |
| New York (Erie County) | 87 (1.7) |
| New York, New York | 132 (2.6) |
| Philadelphia, Pennsylvania | 300 (5.9) |
| Total | 5,093 (100) |
* Directly funded cities and counties (e.g., Chicago and Los Angeles County) are listed as separate sites.
 TABLE 3. Number and percentage of men from whom isolates were collected, by selected characteristics and sex of sex partner — Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014
TABLE 3. Number and percentage of men from whom isolates were collected, by selected characteristics and sex of sex partner — Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014
| Characteristic | Sex of sex partner | |||
|---|---|---|---|---|
| MSM | MSMW | MSW | Total* | |
| No. (%) | No. (%) | No. (%) | No. (%) | |
| Total | 1,616 (100) | 251 (100) | 3,165 (100) | 5,093 (100) |
| Race/Ethnicity | ||||
| Black | 446 (27.6) | 105 (41.8) | 2,381 (75.2) | 2,953 (58.0) |
| White | 726 (44.9) | 78 (31.1) | 309 (9.8) | 1,122 (22.0) |
| Hispanic/Latino | 299 (18.5) | 39 (15.5) | 316 (10.0) | 655 (12.9) |
| Asian | 48 (3.0) | 10 (4.0) | 46 (1.5) | 104 (2.0) |
| American Indian/Alaska Native | 11 (0.7) | 0 (0) | 8 (0.3) | 19 (0.4) |
| Native Hawaiian/Other Pacific Islander | 11 (0.7) | 2 (0.8) | 33 (1.0) | 47 (0.9) |
| Other/Multiracial | 55 (3.4) | 11 (4.4) | 61 (1.9) | 127 (2.5) |
| Missing/Unknown | 20 (1.2) | 6 (2.4) | 11 (0.4) | 66 (1.3) |
| Age at time of clinic visit (yrs) | ||||
| 10–14 | 0 (0.0) | 0 (0.0) | 2 (0.1) | 2 (0.04) |
| 15-19 | 45 (2.8) | 20 (8.0) | 315 (10.0) | 382 (7.5) |
| 20–24 | 368 (22.8) | 53 (21.1) | 888 (28.1) | 1,312 (25.8) |
| 25-29 | 448 (27.7) | 51 (20.3) | 696 (22.0) | 1,203 (23.6) |
| 30–34 | 291 (18.0) | 42 (16.7) | 446 (14.1) | 785 (15.4) |
| 35-39 | 150 (9.3) | 34 (13.6) | 258 (8.2) | 447 (8.8) |
| 40–49 | 201(12.4) | 27 (10.8) | 317 (10.0) | 550 (10.8) |
| 50–59 | 95 (5.9) | 19 (7.6) | 182 (5.8) | 302 (5.9) |
| ≥60 | 18 (1.1) | 5 (2.0) | 60 (1.9) | 85 (1.7) |
| Missing/Unknown | 0 (0) | 0 (0) | 1 (<0.1) | 25 (0.5) |
| Region | ||||
| West | 958 (59.3) | 132 (52.6) | 905 (29.0) | 2,028 (39.8) |
| Midwest | 291 (18.0) | 48 (19.1) | 956 (30.2) | 1,312 (25.8) |
| Northeast | 165 (10.2) | 32 (12.8) | 343 (10.8) | 546 (10.7) |
| South | 202 (12.5) | 39 (15.5) | 961 (30.4) | 1,207 (23.7) |
| Previous history of gonorrhea | ||||
| Yes | 972 (60.2) | 140 (55.8) | 1,358 (42.9) | 2,478 (48.7) |
| No | 594 (36.8) | 103 (41.0) | 1,752 (55.4) | 2,462 (48.3) |
| Missing/Unknown | 50 (3.1) | 8 (3.2) | 55 (1.7) | 153 (3) |
| No. episodes of gonococcal infection in previous 12 mos | ||||
| 0 | 1,124 (69.6) | 174 (69.3) | 2,426 (76.7) | 3,748 (73.6) |
| 1 | 348 (21.5) | 54 (21.5) | 481 (15.2) | 886 (17.4) |
| 2 | 88 (5.5) | 11 (4.4) | 67 (2.1) | 167 (3.3) |
| >3 | 22 (1.4) | 3 (1.2) | 17 (0.5) | 45 (0.9) |
| Missing/Unknown | 34 (2.1) | 9 (3.6) | 174 (5.5) | 247 (4.9) |
| HIV infected | ||||
| Yes | 389 (24.1) | 44 (17.5) | 45 (1.4) | 479 (9.4) |
| No | 1,113 (68.9) | 192 (76.5) | 2,816 (89.0) | 4,149 (81.5) |
| Indeterminate | 3 (0.2) | 0 (0) | 17 (0.5) | 20 (0.4) |
| Missing/Unknown | 111 (6.9) | 15 (6.0) | 287 (9.1) | 445 (8.7) |
| Took antibiotics for any reason during previous 60 days | ||||
| Yes | 142 (8.8) | 13 (5.2) | 142 (4.5) | 299 (5.9) |
| No | 924 (57.2) | 150 (59.8) | 2,388 (75.5) | 3,472 (68.2) |
| Missing/Unknown | 550 (34.0) | 88 (35.1) | 635 (20.1) | 1,322 (26.0) |
| Traveled outside of the United States during the past 60 days | ||||
| Yes | 131 (8.1) | 20 (8.0) | 149 (4.7) | 304 (6.0) |
| No | 857 (53.0) | 142 (56.6) | 2,069 (65.4) | 3,079 (60.5) |
| Missing/Unknown | 628 (38.9) | 89 (35.5) | 947 (29.9) | 1,710 (33.6) |
| Exchanged drugs or money for sex in previous 12 mos | ||||
| Yes | 22 (1.4) | 9 (3.6) | 70 (2.2) | 101 (2.0) |
| No | 1,207 (74.7) | 176 (70.0) | 2,561 (80.9) | 3,958 (77.7) |
| Missing/Unknown | 387 (24.0) | 66 (26.3) | 534 (16.9) | 1,034 (20.3) |
| Injection recreational drug use during previous 12 mos | ||||
| Yes | 42 (2.6) | 10 (4.0) | 35 (1.1) | 90 (1.8) |
| No | 1,365 (84.5) | 207 (82.5) | 2,833 (89.5) | 4,420 (86.8) |
| Missing/Unknown | 209 (12.9) | 34 (13.6) | 297 (9.4) | 583 (11.5) |
| Noninjection recreational drug use during previous 12 mos | ||||
| Yes | 292 (18.1) | 55 (21.9) | 669 (21.1) | 1,020 (20.0) |
| No | 1,049 (64.9) | 149 (59.4) | 2,117 (66.9) | 3,326 (65.3) |
| Missing/Unknown | 275 (17.0) | 47 (18.7) | 379 (12.0) | 747 (14.7) |
Abbreviations: MSM = men who have sex with men; MSMW = men who have sex with men and women; MSW = men who have sex with women.
* Total = 5,093. Total includes 61 men for whom sex of sex partner is unknown; this accounts for discrepancies in totals for each characteristic.
 FIGURE 1. Percentage of ceftriaxone, oral cephalosporins,* oral fluoroquinolones,† and other antibiotics§ used for gonorrhea treatment,¶ by year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 1. Percentage of ceftriaxone, oral cephalosporins,* oral fluoroquinolones,† and other antibiotics§ used for gonorrhea treatment,¶ by year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
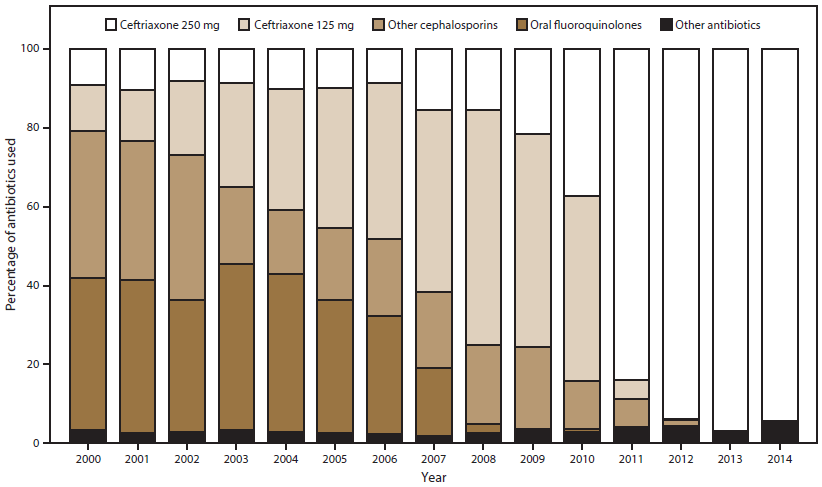
* Oral cephalosporins = cefdinir, cefixime, cefotaxime, cefoxitin, cefpodoxime, ceftibuten, or ceftizoxime.
† Oral fluoroquinolones = ciprofloxacin, levofloxacin, or ofloxacin.
§ Other antibiotics = azithromycin, doxycycline, spectinomycin, clinical trial drugs, other, or none.
¶ In 2010, CDC revised its gonorrhea treatment guidelines to recommend dual therapy with a cephalosporin (either ceftriaxone [preferred] or cefixime) and a second antimicrobial (azithromycin or doxycycline) and to increase the recommended dosage of ceftriaxone to 250 mg. In 2012, CDC again revised its gonorrhea treatment guidelines to no longer recommend cefixime and to recommend only ceftriaxone-based dual therapy for gonorrhea treatment.
 FIGURE 2. Prevalence of tetracycline, penicillin, or fluoroquinolone resistance* or reduced cefixime or azithromycin susceptibility,† by year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 2. Prevalence of tetracycline, penicillin, or fluoroquinolone resistance* or reduced cefixime or azithromycin susceptibility,† by year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
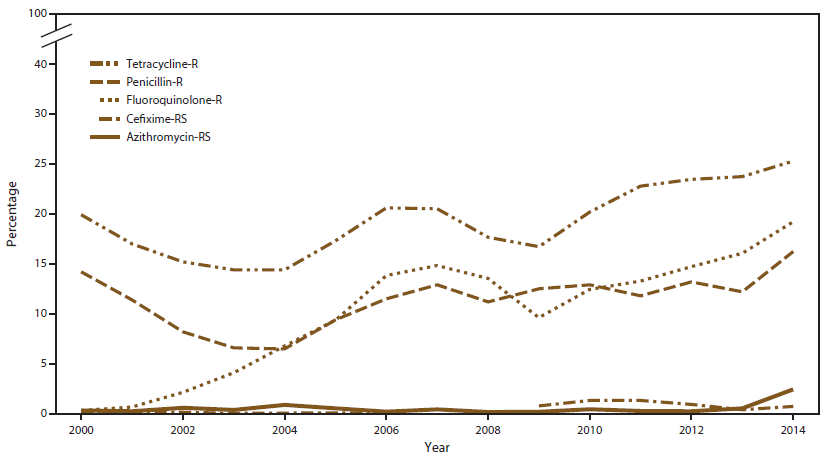
* Fluoroquinolone resistance (R) = ciprofloxacin minimum inhibitory concentration (MIC) =1.0 µg/mL; penicillin-R = MIC =2.0 µg/mL or ß-lactamase positive; tetracycline-R = MIC =2.0 µg/mL.
† Azithromycin reduced susceptibility (RS) = MIC =1.0 µg/mL (2000–2004); =2.0 µg/mL (2005–2014); cefixime-RS = MIC =0.25 µg/mL. Cefixime susceptibility was not tested in 2007 and 2008.
 FIGURE 3. Percentage of urethral Neisseria gonorrhoeae isolates with reduced azithromycin susceptibility,* by region and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 3. Percentage of urethral Neisseria gonorrhoeae isolates with reduced azithromycin susceptibility,* by region and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014

* Minimum inhibitory concentration (MIC) =1.0 µg/mL (2000–2004); MIC =2.0 µg/mL (2005–2014).
 FIGURE 4. Percentage of urethral Neisseria gonorrhoeae isolates with reduced azithromycin susceptibility,* by sex of sex partner and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 4. Percentage of urethral Neisseria gonorrhoeae isolates with reduced azithromycin susceptibility,* by sex of sex partner and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014

Abbreviations: MSM = men who have sex with men; MSMW = men who have sex with men and women; MSW = men who have sex with women.
* Minimum inhibitory concentration (MIC) =1.0 µg/mL (2000–2004); MIC =2.0 µg/mL (2005–2014).
 FIGURE 5. Percentage of Neisseria gonorrhoeae isolates with reduced cefixime susceptibility,* by region and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 5. Percentage of Neisseria gonorrhoeae isolates with reduced cefixime susceptibility,* by region and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014

* Minimum inhibitory concentration (MIC) =0.25 µg/mL. Cefixime susceptibility was not tested in 2007 and 2008.
 FIGURE 6. Percentage of Neisseria gonorrhoeae isolates with reduced cefixime susceptibility,* by sex of sex partner and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 6. Percentage of Neisseria gonorrhoeae isolates with reduced cefixime susceptibility,* by sex of sex partner and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014

Abbreviations: MSM = men who have sex with men; MSMW = men who have sex with men and women; MSW = men who have sex with women.
* Minimum inhibitory concentration (MIC) =0.25 µg/mL. Cefixime susceptibility was not tested in 2007 and 2008.
 FIGURE 7. Percentage of Neisseria gonorrhoeae isolates with reduced ceftriaxone susceptibility,* by region, total, and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 7. Percentage of Neisseria gonorrhoeae isolates with reduced ceftriaxone susceptibility,* by region, total, and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014

* Minimum inhibitory concentration (MIC) =0.125 µg/mL.
 FIGURE 8. Percentage of Neisseria gonorrhoeae isolates with reduced ceftriaxone susceptibility,* by sex of sex partner, and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 8. Percentage of Neisseria gonorrhoeae isolates with reduced ceftriaxone susceptibility,* by sex of sex partner, and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
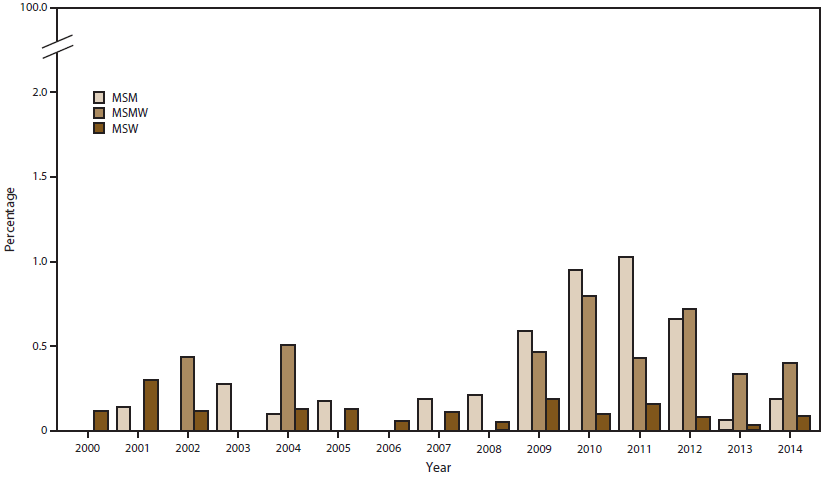
Abbreviations: MSM = men who have sex with men; MSMW = men who have sex with men and women; MSW = men who have sex with women.
* Minimum inhibitory concentration (MIC) =0.125 µg/mL.
 FIGURE 9. Percentage of Neisseria gonorrhoeae isolates with ciprofloxacin resistance,* by region and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 9. Percentage of Neisseria gonorrhoeae isolates with ciprofloxacin resistance,* by region and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
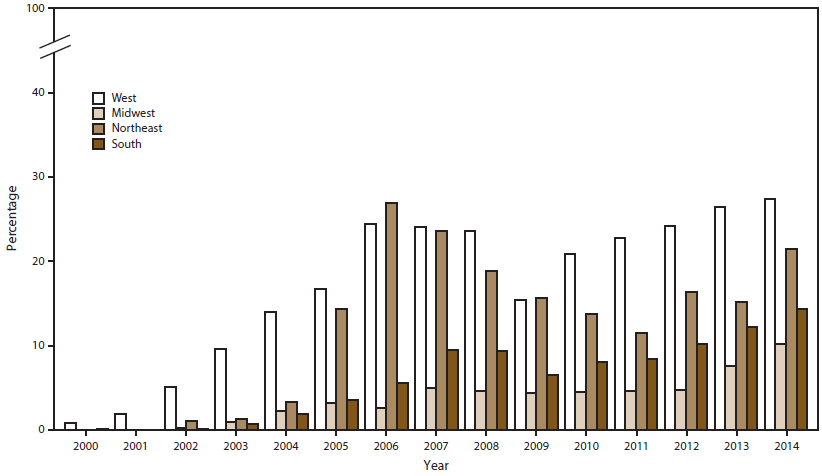
* Minimum inhibitory concentration (MIC) =1.0 µg/mL.
 FIGURE 10. Percentage of Neisseria gonorrhoeae isolates with ciprofloxacin resistance* by sex of sex partner, and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014
FIGURE 10. Percentage of Neisseria gonorrhoeae isolates with ciprofloxacin resistance* by sex of sex partner, and year — Gonococcal Isolate Surveillance Project, United States, 2000–2014

Abbreviations: MSM = men who have sex with men; MSMW = men who have sex with men and women; MSW = men who have sex with women.
* Minimum inhibitory concentration (MIC) =1.0 µg/mL.
 FIGURE 11. Percentage of urethral Neisseria gonorrhoeae isolates* susceptible and resistant to penicillin, tetracycline, ciprofloxacin, cefixime, ceftriaxone, or azithromycin, and other combinations † — Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014
FIGURE 11. Percentage of urethral Neisseria gonorrhoeae isolates* susceptible and resistant to penicillin, tetracycline, ciprofloxacin, cefixime, ceftriaxone, or azithromycin, and other combinations † — Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014

Abbreviations: Azi-RS = reduced azithromycin susceptibility; Cip-R = ciprofloxacin resistance, Cfx-RS = reduced cefixime susceptibility; Cro-RS = reduced ceftriaxone susceptibility; Pen-R = penicillin resistance; Tet-R = tetracycline resistance.
* N = 5,093.
† Other combinations: Azi-RS (1.1%); Pen-R and Tet-R (2.1%); Pen-R and Cip-R (1.6%). Each =1.0%: Cfx-RS; Pen-R and Azi-RS; Tet-R and Azi-RS; Cip-R and Cfx-RS; Pen-R, Tet-R, and Azi-RS; Pen-R, Cfx-RS, and Cro-RS; Tet-R, Cip-R, and Azi-RS; Tet-R, Cip-R, and Cfx-RS; Pen-R, Tet-R, Cip-R, and Azi-RS; Pen-R, Tet-R, Cip-R, and Cfx-RS; Pen-R, Tet-R, Cip-R, Cfx-RS, and Cro-RS.
Suggested citation for this article: Kirkcaldy RD, Harvey A, Papp JR, et al. Neisseria gonorrhoeae Antimicrobial Susceptibility Surveillance — The Gonococcal Isolate Surveillance Project, 27 Sites, United States, 2014. MMWR Surveill Summ 2016;65(No. SS-7):1–19. DOI: http://dx.doi.org/10.15585/mmwr.ss6507a1.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.