Notes from the Field: Cruise Ship Norovirus Outbreak Associated with Person-to-Person Transmission — United States Jurisdiction, January 2023
Weekly / July 28, 2023 / 72(30);833–834
Carolyn A. Crisp, PhD1,2; Keisha A. Jenkins, DrPH2; Ian Dunn, MPH3; Andrew Kupper, MPH2; Jona Johnson, PhD2; Stefanie White, MPH2; Erin D. Moritz, PhD2; Luis O. Rodriguez, MS2 (View author affiliations)
View suggested citationAltmetric:
CDC’s Vessel Sanitation Program (VSP) monitors cases of acute gastroenteritis (AGE) on board cruise ships traveling to a U.S. port (1). Persons who have ≥3 loose stools (or more than normal for that person) within a 24-hour period or vomiting plus one other sign or symptom (e.g., fever, diarrhea, bloody stool, myalgia, abdominal cramps, or headache) meet the case definition for reportable AGE (2). When the percentage of passengers or crew members with AGE is ≥2% and the ship is due to arrive at a U.S. port within 15 days, the Maritime Illness Disease Reporting System alerts VSP and activates an investigation (1). During the first week of January 2023, VSP was notified of cases of AGE affecting >2% of passengers on board a ship that had completed three voyages in Europe and was within 15 days of arriving at a U.S. port (voyage 4)* (Figure). Ship medical crew members submitted stool samples from ill travelers for testing. All samples tested positive for norovirus genotype II. While the ship was sailing to a U.S. port, VSP monitored AGE cases on board and reviewed case data. By mid-January, passenger AGE prevalence reached 3.4%.
Investigation and Outcomes
During mid-January 2023, VSP’s outbreak team boarded the ship to conduct an epidemiological and environmental investigation. The investigation focused on exposure sources and routes of transmission. Occupational and social behaviors of crew members were evaluated because the epidemic curve (Figure) suggested that the index case occurred in a crew member during voyage 1 who developed symptoms after embarking, likely leading to transmission among other crew members (voyage 2), and then to passengers (voyage 3). After the investigation, VSP continued to monitor the ship (voyage 5) until it left U.S. jurisdiction. This activity was reviewed by CDC and was conducted consistent with applicable federal law and CDC policy.†
Among 410 reported cases during November 2022–January 2023 (voyages 1–5), 356 (87%) occurred in passengers and 54 (13%) in crew members. The index case likely occurred in a food and beverage crew member sailing on a crew-only voyage (voyage 1). In general, crew members with AGE reported to onboard medical personnel in a timely manner and were isolated until 48 hours after symptoms subsided. Crew member transmission was followed by passenger transmission on voyages 3, 4, and 5. Vomiting and diarrhea were the predominant symptoms among cases. Approximately 70% of crew members with AGE interacted with passengers (i.e., housekeeping and food and beverage services). VSP partnered with the United States Agency for Toxic Substances and Disease Registry’s Geospatial Research, Analysis, and Services Program to create four-dimensional visual models of the ship. These models helped visualize continued norovirus transmission and sources of potential exposure (e.g., contaminated surfaces in cabins of persons with AGE and high-touch surfaces in common areas).
Preliminary Conclusions and Actions
During this investigation, VSP used surveillance data and environmental and spatial analyses to improve field responses and quickly identify sources of norovirus exposure and transmission. Public health response to maritime AGE outbreaks involves robust and timely monitoring of AGE cases and collaborations with cruise companies. To prevent illness transmission across voyages, cruise ship personnel and travelers should always maintain proper hand hygiene and sanitation practices, and passengers and crew members should immediately isolate and report illness symptoms to the ship medical center (3). Cruise companies are encouraged to conduct frequent norovirus trainings for crew members, especially those with limited experience working with the cruise company (e.g., those who have served fewer than three contract terms).
Acknowledgments
Crew members and representatives of the cruise line; CDC’s National Calicivirus Laboratory; the Agency for Toxic Substances and Disease Registry’s Geospatial Research, Analysis, and Services Program; Vessel Sanitation Program members; Water, Food, and Environmental Health Services Branch, National Center for Environmental Health, CDC.
Corresponding author: Carolyn A. Crisp, tqy2@cdc.gov.
1Epidemic Intelligence Service, CDC; 2Division of Environmental Health Science and Practice, National Center for Environmental Health, CDC; 3Geospatial Research, Analysis, and Services Program, Agency for Toxic Substances and Disease Registry, Atlanta, Georgia.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
* Voyage 1 (early November 2022), voyage 2 (early November–mid-December 2022), voyage 3 (mid-to-late December 2022), voyage 4 (late December 2022–early January 2023) and voyage 5 (began in early January and lasted ≥30 days); the ship made port in the United States during mid-January 2023.
† 45 C.F.R. part 46, 21 C.F.R. part 56; 42 U.S.C. Sect. 241(d); 5 U.S.C. Sect. 552a; 44 U.S.C. Sect. 3501 et seq.
References
- Jenkins KA, Vaughan GH Jr, Rodriguez LO, Freeland A. Acute gastroenteritis on cruise ships—Maritime Illness Database and Reporting System, United States, 2006–2019. MMWR Surveill Summ 2021;70(No. SS-6):1–19. https://doi.org/10.15585/mmwr.ss7006a1 PMID:34555008
- Freeland AL, Vaughan GH Jr, Banerjee SN. Acute gastroenteritis on cruise ships—United States, 2008–2014. MMWR Morb Mortal Wkly Rep 2016;65:1–5. https://doi.org/10.15585/mmwr.mm6501a1 PMID:26766396
- CDC. Vessel sanitation program: illness prevention resources. Atlanta, GA: US Department of Health and Human Services, CDC; 2022. https://www.cdc.gov/nceh/vsp/healthy.htm
 FIGURE. Cases of acute gastroenteritis (N = 410),* by illness onset date† — Cruise ship A, five voyages, November 2022–January 2023§
FIGURE. Cases of acute gastroenteritis (N = 410),* by illness onset date† — Cruise ship A, five voyages, November 2022–January 2023§
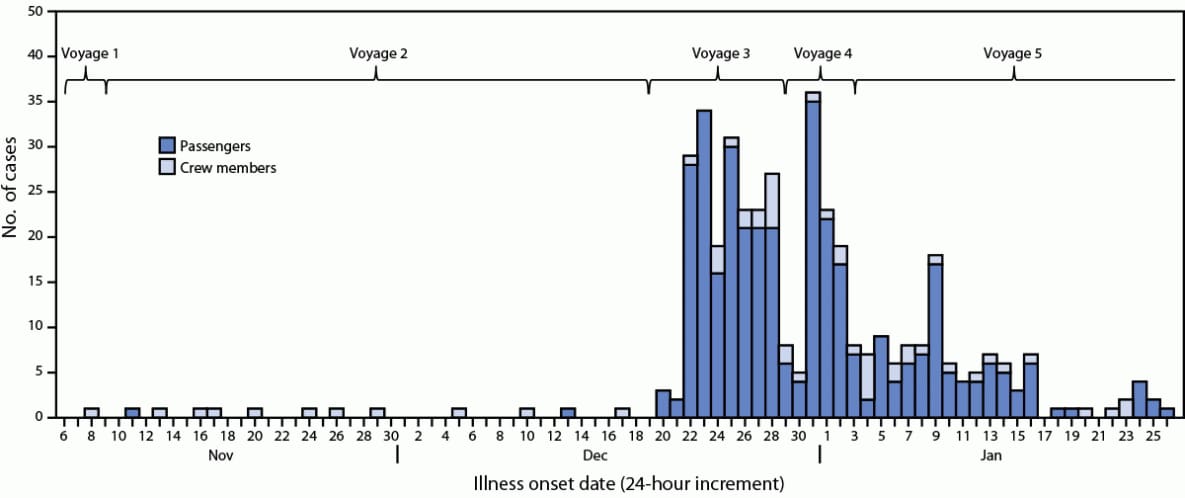
* Cases occurred among 356 passengers and 54 crew members.
† Index case likely occurred on November 8, 2022.
§ Voyage 5 was a world voyage that lasted ≥30 days.
Suggested citation for this article: Crisp CA, Jenkins KA, Dunn I, et al. Notes from the Field: Cruise Ship Norovirus Outbreak Associated with Person-to-Person Transmission — United States Jurisdiction, January 2023. MMWR Morb Mortal Wkly Rep 2023;72:833–834. DOI: http://dx.doi.org/10.15585/mmwr.mm7230a5.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.