Poisoning Associated with Consumption of a Homemade Medicinal Liquor — Chongqing, China, 2018
Weekly / April 22, 2022 / 71(16);569–573
Chunbei Zhou, MPH1,2; Shuquan Luo1; Jiang Tang3; Linda Quick, MD4; Huihui Liu, MD5; Yinan Zhao, MPH1,2 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
Aconite tubers and roots are used to prepare traditional Chinese medicinal drinks; fatal aconite poisonings resulting from improper production occur every year.
What is added by this report?
Fifteen cases of poisoning, including five deaths, were reported among persons who attended a birthday luncheon at a Chongqing hotel. Intoxication was most likely caused by homemade unlabeled medicinal liquor containing extremely high concentrations of toxic Aconitum alkaloids.
What are the implications for public health practice?
Public prevention strategies should focus on increasing recognition of the toxicity of Aconitum species and the identification of similar plants, strengthening existing regulation of the sale of raw materials containing Aconitum alkaloids, and appropriately labeling homemade products.
Altmetric:
On May 3, 2018, Chongqing Center for Disease Control and Prevention (CQCDC) received a report of 15 persons with numbness of the tongue or limbs and vomiting of unknown etiology; all ill persons had attended an adult birthday luncheon in Bishan District, Chongqing municipality, in southwest China. Initial reports indicated that one person had died. Within 2 hours, CQCDC and Western Chinese Field Epidemiology Training Program staff members launched an investigation that included identification of cases, laboratory testing of drinks, and patient interviews to identify the cause of what appeared to be a poisoning. Among the 15 cases, five persons died. The investigation of this apparent mass intoxication implicated a homemade alcoholic beverage produced from a highly toxic flowering plant in the genus Aconitum used in traditional Chinese medicine. Although the risk of aconite toxicity is known, approximately 5,000 cases of aconite poisoning incidents were reported in China, Germany, Japan, and other countries during 1993–2005; most cases of fatal poisoning occurred in China (1). This event highlights the importance of enforcing and complying with existing regulations regarding sale and purchase of Aconitum species (also known as wolfbane), and of dissemination of critical public health messages.
Investigation and Findings
Fifty-three persons had attended the birthday luncheon at a hotel restaurant. A case was defined as the onset of numbness of the tongue or limbs, vomiting, heart palpitations, or sudden death in any person who participated in the birthday luncheon. Demographic information and a list of food and beverages consumed by all attendees were obtained through in-person interviews using a structured questionnaire. Medical records for inpatients were requested and abstracted from hospital and outpatient providers.
A total of 15 cases were identified among the 53 attendees (attack rate = 28%). One patient died on the way to the hospital. Among the remaining 14 patients, all were hospitalized, including nine who were admitted to an intensive care unit; four hospitalized patients died. All patients were men with a median age of 52 years (range = 44–65 years). Among the 15 patients, all reported numbness of the tongue or extremities, 14 reported vomiting, nine reported heart palpitations, and nine reported dizziness (Table). Among the 14 hospitalized patients, 11 had cardiac involvement, including ventricular tachycardia (six patients) and premature ventricular contractions (five patients). Myoglobin was elevated in eight patients, and α-hydroxybutyrate dehydrogenase, a marker of myocardial damage, was elevated in five patients. Other indicators of myocardial damage included abnormal levels of creatine kinase isoenzyme and phosphocreatine kinase. The four patients who died in the hospital all had cardiogenic shock and severe lactic acidosis; two also experienced gastrointestinal bleeding.
On the day of the luncheon, the hotel entertained no other guests; all the attendees had eaten breakfast at home. The luncheon started at 12:10 p.m. The 53 attendees sat around five round tables and were all served identical dishes. Beverages consisted of beer, orange juice, soy milk purchased from a local store, and “baijiu,” a Chinese liquor generally distilled from fermented sorghum or other whole grains, provided by the hotel. Some attendees also drank a homemade medicinal liquor prepared by the host. All the drinks except for the homemade medicinal liquor were sealed before being opened and consumed. The hotel tap water was from the local water plant.
Symptom onset in the first case was reported at 12:20 p.m., and the last symptom onset occurred 70 minutes later at 1:30 p.m. (Figure 1). The first death occurred at 2:10 p.m. and the last at 10:10 p.m. The median interval from the beginning of the luncheon until symptom onset was 18 minutes among the five fatal cases and 40 minutes among the 10 surviving persons.
The 15 cases occurred among persons seated at three tables (tables C [nine cases among 10 guests and the host], D [two of 10], and E [four of 10]); no cases occurred among the 11 guests seated at table A or the 11 at table B (Figure 2). All 15 attendees who became ill drank the medicinal liquor (100%), including nine who drank only the medicinal liquor (five also consumed the baijiu, and one also drank beer). Five of the nine patients who drank only the medicinal liquor died. All attendees who drank the medicinal liquor became ill. None of the 10 hotel employees serving became ill, nor were any similar cases reported in the surrounding area.
The host of the luncheon had made the liquor, and also died after drinking it; therefore, details of how it was prepared were not available. His wife recalled that aconite roots were given to her husband by a friend several years earlier. The medicinal liquor was prepared several years before, distilled spirits were added to the roots for soaking, and the mixture was kept at the host’s home until the day of the birthday luncheon. The host’s wife speculated that aconite roots were mistaken by her husband for Lepidium meyenii, a nontoxic plant root also reported to be used as medicine.
Examination of the plant roots remaining in the bottle of medicinal liquor were morphologically similar to aconite roots used in traditional Chinese medicine. Samples were sent to the CQCDC Physicochemical and Toxicology Testing Laboratory. The alkaloid database was built by Thermo Trace Finder software (https://www.thermofisher.com/us/en/home.html), and rapidly screened for 42 alkaloids by ultra-performance liquid chromatography-quadrupole/electrostatic field orbitrap high-resolution mass spectrometry. Bullatine A, bullatine B, aconitine, and mesaconitine, which are aconite byproducts, were detected in the medicinal liquor, and all four of these Aconitum alkaloids are known to be toxic. Bullatine A and aconitine concentrations were 81.9 μg/mL (a highly toxic dose) and 0.393 μg/mL, respectively. The concentrations of the other two components were extremely low. On the basis of the evidence, the medicinal liquor was suspected to be the cause of the poisoning, most likely the result of bullatine A ingestion. No toxicology testing was performed on any of the persons who were poisoned.
Public Health Response
The local government immediately responded to this public health emergency. The response included patient treatment and joint investigation by public security (police), health departments, and regulatory authorities. At the conclusion of the investigation, public health safety messages were disseminated through the media to ensure public awareness of the poisonous qualities of Aconitum species.
Discussion
This investigation provides compelling evidence that this mass intoxication was caused by homemade medicinal liquor containing Aconitum alkaloids. Aconitum lycoctonum is included in the Aconitum genus of >250 species of flowering plants belonging to the Ranunculaceae family. These herbaceous perennial plants are native to the mountainous parts of the Northern Hemisphere, growing in the moisture-retentive but well-draining soils of mountain meadows, mainly in southwestern China. Among medicinal herbs, Aconitum species are unique: while reportedly considered a beneficial herb root used historically and extensively for medicinal purposes in China, Aconitum species, also known as wolfbane, are highly toxic, and if not prepared correctly, consumption can be rapidly fatal.
Aconite tubers and roots are traditionally used in medications to treat pain and a range of diverse ailments, including diarrhea, edema, asthma, various tumors, rheumatism, rheumatoid arthritis, and other inflammatory disorders (2,3). They are frequently used in traditional Chinese medicine, and, although the sale of unprocessed Aconitum species is prohibited, they can be illicitly acquired in open markets in China, especially in Yunnan and Guizhou provinces. These sales of unprocessed roots occur, even though the activity is forbidden by drug safety regulation stipulated by the State Administration for Market Regulation, and even though the pharmaceutical use of drugs containing Aconitum alkaloids is strictly monitored in China.
The medicinal use of Aconitum species requires strict adherence to processing and cooking guidance, which is based on the prescribed dosage (4). Only licensed manufacturers or authorized practitioners are allowed to prepare Aconitum species for medical use. Traditional Chinese methods for processing herbs (páo zhì), including heating, soaking, and boiling for several hours, are reported to enhance the properties or reduce or eliminate toxicity, and in the case of Aconitum species, convert the alkaloids to comparatively less toxic or nontoxic derivatives (5), so that medicinal liquors containing aconite roots may be safely drunk for health promotion (3). If the products are misused or not prepared correctly, aconite ingestion can result in rapid death. Aconite roots and leaves, as well as honey made from aconite nectar, are all highly toxic. If they are not properly decocted for oral consumption, Aconitum plant products can be safely used only when applied topically. As little as 0.2 mg (200 μg) of Aconitum alkaloids can cause poisoning, and 2.0 mg (2,000 μg) is sufficient to result in death.
Although the risk of aconite toxicity is known, in some areas of China, Aconitum plant products are still sought for their purported health benefits. Approximately 5,000 cases of aconite poisoning were reported in China, Germany, Japan, and other countries during 1993–2005; most cases of fatal poisoning occurred in China (1). Aconite roots contain various chemical constituents, such as aconitine, mesaconitine, and bullatine A, which have significant pharmacologic activity and are also toxic. The Aconitum alkaloids primarily affect the central nervous system, heart, and muscle cells (6–8), with cardiac damage the most serious consequence. As was the case with most of the victims of this intoxication, severe cases of cardiac toxicity from consumption of aconitine-containing herbal preparations typically manifest clinically as ventricular tachycardia and fibrillation, frequently resulting in death. Aconitum alkaloids bind to cardiac muscle cell receptors that regulate sodium-ion channels, preventing repolarization of cells, and resulting in paralysis and death (9,10).
The findings in this report are subject to at least three limitations. First, no toxicology testing was done on the victims. However, the hypothesis that the toxicant was a homemade medicinal liquor containing products from Aconitum species is supported by the fact that 100% of persons who drank it and none of those who did not drink it became ill. Second, it was not possible to ascertain the precise volume of medicinal liquor consumed by each person, but the amount consumed by each person ranged from 5–125 mL (approximately 1–25 tsp). On the basis of the concentration of residue identified by toxicology testing (81.9 μg/mL), it can be inferred that as little as 25 mL (approximately 5 tsp) contained sufficient Aconitum alkaloids to be lethal. Finally, the source of Aconitum species could not be ascertained. In a retrospective analysis of case reports of aconite poisoning in China during 2004–2015, the most commonly reported route of exposure was drinking medicinal liquors containing Aconitum alkaloids; concurrent ethanol ingestion could rapidly increase the absorption of Aconitum alkaloids into the blood (3), which might have contributed to the rapid onset and five fatal cases in persons who drank only the medicinal liquor in this event. In addition, the absence of labeling of the root might have contributed to its mistaken inclusion in the recipe.
Although Aconitum species can be used medicinally if properly prepared by a licensed manufacturer or authorized practitioner, ingestion can also result in fatal intoxication; therefore, regulations and public health messaging to increase awareness of the toxicity of Aconitum species can help prevent inadvertent aconite poisoning. Emphasizing the danger of drinking medicinal liquors containing Aconitum alkaloids and the identification of plants that are easily confused are critical to ensure the safe use of Aconitum species. Conspicuous labeling of bottles containing homemade brews might prevent misuse, especially oral consumption of products that are only appropriate for topical use. In addition, existing safety monitoring of materials containing Aconitum alkaloids and surveillance of the poison control network are critical.
Acknowledgments
Huilai Ma; local health departments; U.S. CDC; Field Epidemiology Training Program; China Center for Disease Control and Prevention (CDC); Chinese Field Epidemiology Training Program; Chongqing CDC; Bishan District CDC; Bishan District People’s Hospital.
Corresponding author: Yinan Zhao, 651563120@qq.com, +86-23-63259380.
1Chongqing Center for Disease Control and Prevention, Chongqing, China; 2Western Chinese Field Epidemiology Training Program, Beijing, China; 3Bishan District Center for Disease Control and Prevention, Chongqing, China; 4Division of Global Health Protection, Center for Global Health, CDC; 5Chinese Field Epidemiology Training Program, CDC China, Beijing, China.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
References
- Lu G, Dong Z, Wang Q, et al. Toxicity assessment of nine types of decoction pieces from the daughter root of Aconitum carmichaeli (Fuzi) based on the chemical analysis of their diester diterpenoid alkaloids. Planta Med 2010;76:825–30. https://doi.org/10.1055/s-0029-1240688 PMID:20013637
- Sun X-J, Liu T-T, Zhao Y-L, et al. Toxicity of five herbs in Aconitum L. on Tetrahymena thermophila based on spectrum-effect relationship. Chin Herb Med 2014;6:29–35. https://doi.org/10.1016/S1674-6384(14)60003-4
- Li H, Liu L, Zhu S, Liu Q. Case reports of aconite poisoning in mainland China from 2004 to 2015: a retrospective analysis. J Forensic Leg Med 2016;42:68–73. https://doi.org/10.1016/j.jflm.2016.05.016 PMID:27266651
- Chan TY. Aconitum alkaloid poisoning related to the culinary uses of aconite roots. Toxins (Basel) 2014;6:2605–11. https://doi.org/10.3390/toxins6092605 PMID:25184557
- Singhuber J, Zhu M, Prinz S, Kopp B. Aconitum in traditional Chinese medicine: a valuable drug or an unpredictable risk? J Ethnopharmacol 2009;126:18–30. https://doi.org/10.1016/j.jep.2009.07.031 PMID:19651200
- Fu M, Wu M, Wang J-F, Qiao Y-J, Wang Z. Disruption of the intracellular Ca2+ homeostasis in the cardiac excitation-contraction coupling is a crucial mechanism of arrhythmic toxicity in aconitine-induced cardiomyocytes. Biochem Biophys Res Commun 2007;354:929–36. https://doi.org/10.1016/j.bbrc.2007.01.082 PMID:17276394
- Zhang S-W, Liu Y, Huang G-Z, Liu L. Aconitine alters connexin43 phosphorylation status and [Ca2+] oscillation patterns in cultured ventricular myocytes of neonatal rats. Toxicol In Vitro 2007;21:1476–85. https://doi.org/10.1016/j.tiv.2007.06.013 PMID:17728094
- Peng C, Wang L, Wang Y-H, Li Y-X, Pan Y. The toxicity of aconitine, emodin on ICC cell and the antagonist effect of the compatibility. Eur J Drug Metab Pharmacokinet 2009;34:213–20. https://doi.org/10.1007/BF03191176 PMID:20166441
- Lin C-C, Phua D-H, Deng J-F, Yang C-C. Aconitine intoxication mimicking acute myocardial infarction. Hum Exp Toxicol 2011;30:782–5. https://doi.org/10.1177/0960327110385960 PMID:20937638
- Dhesi P, Ng R, Shehata MM, Shah PK. Ventricular tachycardia after ingestion of ayurveda herbal antidiarrheal medication containing aconitum. Arch Intern Med 2010;170:303–5. https://doi.org/10.1001/archinternmed.2009.518 PMID:20142579
Abbreviations: IU = international units; NA = not available; U = units.
* Normal: <72 μg/L.
† Normal: <182 U/L.
§ Normal: <25 IU/L.
¶ Normal: <200 IU/L.
** Admitted to an intensive care unit.
 FIGURE 1. Time line of symptom onset and outcomes among patients with illness and sudden death associated with aconite poisoning from homemade medicinal liquor (N = 15) — Bishan District, Chongqing, China, May 3, 2018
FIGURE 1. Time line of symptom onset and outcomes among patients with illness and sudden death associated with aconite poisoning from homemade medicinal liquor (N = 15) — Bishan District, Chongqing, China, May 3, 2018
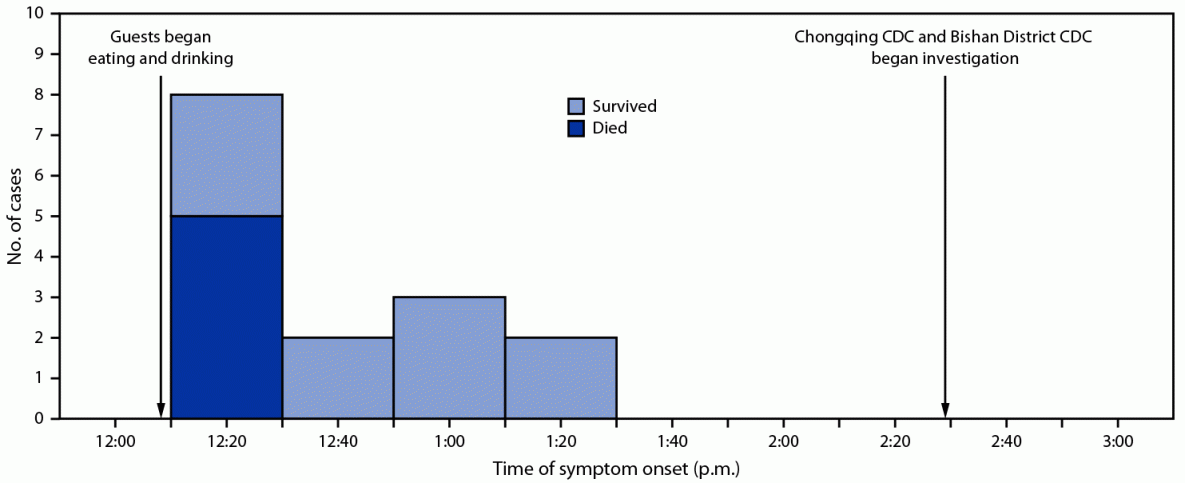
Abbreviation: CDC = Center for Disease Control and Prevention.
 FIGURE 2. Seat locations of 53 lunch attendees, drinks consumed, and outcomes among patients with illness and sudden death associated with aconite poisoning from homemade medicinal liquor — Bishan District, Chongqing, China, May 3, 2018
FIGURE 2. Seat locations of 53 lunch attendees, drinks consumed, and outcomes among patients with illness and sudden death associated with aconite poisoning from homemade medicinal liquor — Bishan District, Chongqing, China, May 3, 2018
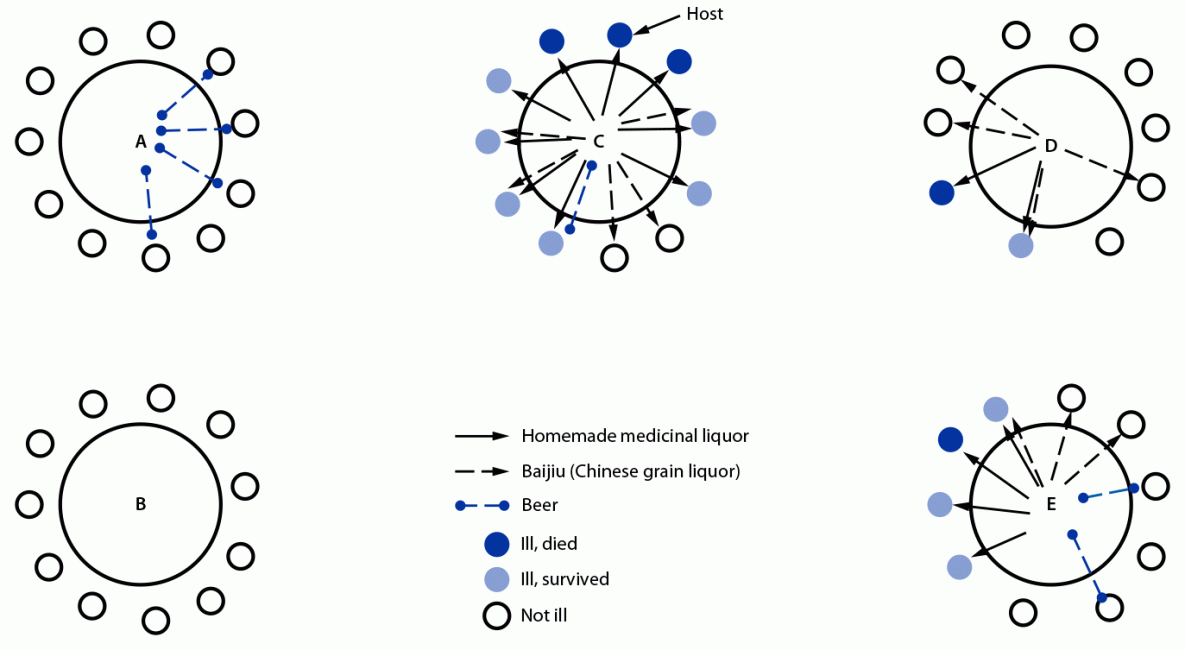
Suggested citation for this article: Zhou C, Luo S, Tang J, Quick L, Liu H, Zhao Y. Poisoning Associated with Consumption of a Homemade Medicinal Liquor — Chongqing, China, 2018. MMWR Morb Mortal Wkly Rep 2022;71:569–573. DOI: http://dx.doi.org/10.15585/mmwr.mm7116a2.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.